Authors: Anna Koumarianou, Krystallenia I. Alexandraki, Göran Wallin, Gregory Kaltsas and Kosmas Daskalakis.
Objective: Mesenteric fibrosis (MF) constitutes an underrecognized sequela in patients with small intestinal neuroendocrine neoplasms (SI-NENs), often complicating the disease clinical course. The aim of the present systematic review was to provide an update in evolving aspects of MF pathogenesis and its clinical management in SI-NENs.
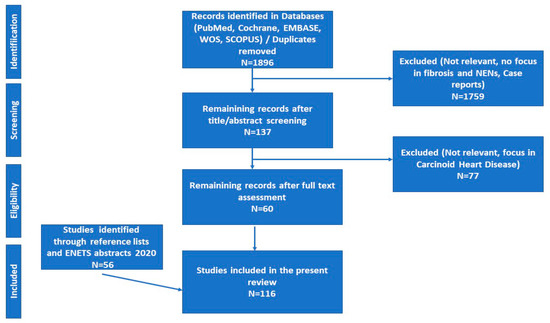
Search strategy: We followed the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) methodology.
Results: Complex and dynamic interactions are present in the microenvironment of tumor deposits in the mesentery. Serotonin, as well as the signaling pathways of certain growth factors play a pivotal, yet not fully elucidated role in the pathogenesis of MF. Clinically, MF often results in significant morbidity by causing either acute complications, such as intestinal obstruction and/or acute ischemia or more chronic conditions involving abdominal pain, venous stasis, malabsorption and malnutrition.
Conclusions: Surgical resection in patients with locoregional disease only or symptomatic distant stage disease, as well as palliative minimally invasive interventions in advanced inoperable cases seem clinically meaningful, whereas currently available systemic and/or targeted treatments do not unequivocally affect the development of MF in SI-NENs. Increased awareness and improved understanding of the molecular pathogenesis of MF in SI-NENs may provide better diagnostic and predictive tools for its timely recognition and intervention and also facilitates the development of agents targeting MF.
- neuroendocrine tumors
- small intestine
- mesenteric fibrosis
- serotonin
- TGF
- FGF
- PDGF
- VEGF
- CTGF
1. Introduction
2. Experimental Section
3. Results
3.1. Pathogenesis of Mesenteric Fibrosis (MF)
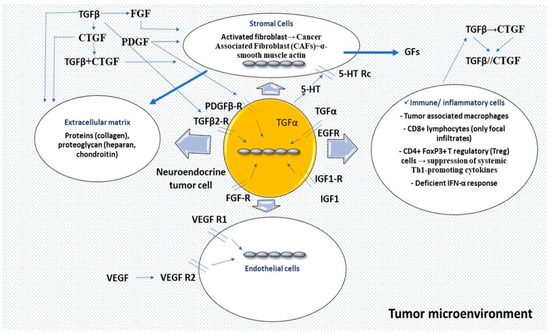
3.1.1. Serotonin
3.1.2. Growth Factors (GFs)
Transforming Growth Factor-β (TGF-β)
Connective Tissue Growth Factor (CTGF)
Platelet-Derived Growth Factor (PDGF)
Insulin-Like Growth Factor 1 (IGF-1)
Epidermal Growth Factor (EGF) and Transforming Growth Factor-a (TGF-α)
Other Growth Factors (GFs)
3.2. Clinical Presentation
Symptoms and Related Deficiencies
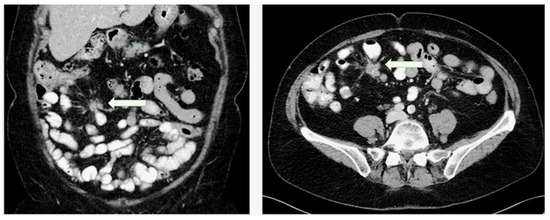
3.3. Diagnostic and Classification Systems
3.4. Clinical Management of Mesenteric Fibrosis
3.4.1. Surgical Management
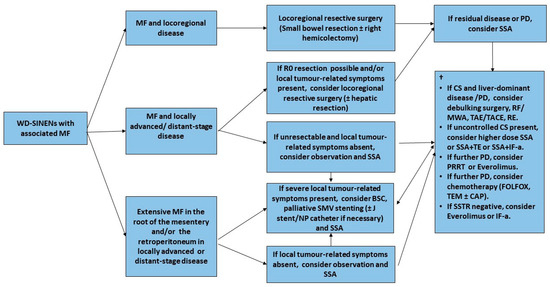
Minimal Invasive Management
3.4.2. Medical Treatment
Somatostatin Analogues (SSAs)
Serotonin Synthesis Inhibitors and 5-HT Receptor Antagonists
Molecular Targeted Therapies (mTOR and Tyrosine Kinase Inhibitors)
Peptide Receptor Radionuclide Therapy (PRRT)
Other Antifibrotic Agents
4. Conclusions
References
- Dasari, A.; Shen, C.; Halperin, D.; Zhao, B.; Zhou, S.; Xu, Y.; Shih, T.; Yao, J.C. Trends in the incidence, prevalence, and survival outcomes in patients with neuroendocrine tumors in the United States. JAMA Oncol. 2017, 3, 1335–1342. [Google Scholar] [CrossRef]
- Eriksson, J.; Norlen, O.; Ogren, M.; Garmo, H.; Ihre-Lundgren, C.; Hellman, P. Primary small intestinal neuroendocrine tumors are highly prevalent and often multiple before metastatic disease develops. Scand. J. Surg. 2019. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Makinen, N.; Kasai, Y.; Kim, G.E.; Diosdado, B.; Nakakura, E.; Meyerson, M. Patterns of chromosome 18 loss of heterozygosity in multifocal ileal neuroendocrine tumors. Genes Chromosomes Cancer 2020. [Google Scholar] [CrossRef] [PubMed]
- Modlin, I.M.; Oberg, K.; Chung, D.C.; Jensen, R.T.; de Herder, W.W.; Thakker, R.V.; Caplin, M.; Delle Fave, G.; Kaltsas, G.A.; Krenning, E.P.; et al. Gastroenteropancreatic neuroendocrine tumours. Lancet Oncol. 2008, 9, 61–72. [Google Scholar] [CrossRef]
- Daskalakis, K.; Karakatsanis, A.; Stalberg, P.; Norlen, O.; Hellman, P. Clinical signs of fibrosis in small intestinal neuroendocrine tumours. Br. J. Surg. 2017, 104, 69–75. [Google Scholar] [CrossRef] [PubMed]
- Cives, M.; Pelle, E.; Quaresmini, D.; Rizzo, F.M.; Tucci, M.; Silvestris, F. The tumor microenvironment in neuroendocrine tumors: Biology and therapeutic implications. Neuroendocrinology 2019, 109, 83–99. [Google Scholar] [CrossRef]
- Druce, M.R.; Bharwani, N.; Akker, S.A.; Drake, W.M.; Rockall, A.; Grossman, A.B. Intra-abdominal fibrosis in a recent cohort of patients with neuroendocrine (‘carcinoid’) tumours of the small bowel. QJM Mon. J. Assoc. Physicians 2010, 103, 177–185. [Google Scholar] [CrossRef]
- Ohrvall, U.; Eriksson, B.; Juhlin, C.; Karacagil, S.; Rastad, J.; Hellman, P.; Akerstrom, G. Method for dissection of mesenteric metastases in mid-gut carcinoid tumors. World J. Surg. 2000, 24, 1402–1408. [Google Scholar] [CrossRef]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G.; Prisma Group. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. Ann. Intern. Med. 2009, 151, 264–269. [Google Scholar] [CrossRef]
- Shapiro, M.; Kidd, M.; Lye, K.D.; Usinger, W.; Murren, J.; Modlin, I.M. Ileal carcinoids over-express the fibrosis-related peptide, CTGF. Gastroenterology 2004, 126, 505. [Google Scholar]
- Modlin, I.M.; Shapiro, M.D.; Kidd, M. Carcinoid tumors and fibrosis: An association with no explanation. Am. J. Gastroenterol. 2004, 99, 2466–2478. [Google Scholar] [CrossRef]
- Svejda, B.; Kidd, M.; Giovinazzo, F.; Eltawil, K.; Gustafsson, B.I.; Pfragner, R.; Modlin, I.M. The 5-HT(2B) receptor plays a key regulatory role in both neuroendocrine tumor cell proliferation and the modulation of the fibroblast component of the neoplastic microenvironment. Cancer 2010, 116, 2902–2912. [Google Scholar] [CrossRef] [PubMed]
- Funa, K.; Papanicolaou, V.; Juhlin, C.; Rastad, J.; Akerstrom, G.; Heldin, C.H.; Oberg, K. Expression of platelet-derived growth factor beta-receptors on stromal tissue cells in human carcinoid tumors. Cancer Res. 1990, 50, 748–753. [Google Scholar] [PubMed]
- Blazevic, A.; Iyer, A.; Van Velthuysen, M.L.F.; Feelders, R.A.; Franssen, G.J.H.; Zajec, M.; Hofland, J.; Luider, T.M.; De Herder, W.W.; Hofland, L.J. Proteomic analysis of small intestinal neuroendocrine tumours and associated mesenteric fibrosis reveals primarily differences in mesenteric stroma. In Proceedings of the ENETS 2019, Barcelona, Spain, 6–8 March 2019. [Google Scholar]
- Vikman, S.; Sommaggio, R.; De La Torre, M.; Oberg, K.; Essand, M.; Giandomenico, V.; Loskog, A.; Totterman, T.H. Midgut carcinoid patients display increased numbers of regulatory T cells in peripheral blood with infiltration into tumor tissue. Acta Oncol. 2009, 48, 391–400. [Google Scholar] [CrossRef] [PubMed]
- Lamarca, A.; Nonaka, D.; Breitwieser, W.; Ashton, G.; Barriuso, J.; McNamara, M.G.; Moghadam, S.; Rogan, J.; Mansoor, W.; Hubner, R.A.; et al. PD-L1 expression and presence of TILs in small intestinal neuroendocrine tumours. Oncotarget 2018, 9, 14922–14938. [Google Scholar] [CrossRef]
- Kim, S.T.; Ha, S.Y.; Lee, S.; Ahn, S.; Lee, J.; Park, S.H.; Park, J.O.; Lim, H.Y.; Kang, W.K.; Kim, K.M.; et al. The impact of PD-L1 expression in patients with metastatic GEP-NETs. J. Cancer 2016, 7, 484–489. [Google Scholar] [CrossRef]
- Sampedro-Nunez, M.; Serrano-Somavilla, A.; Adrados, M.; Cameselle-Teijeiro, J.M.; Blanco-Carrera, C.; Cabezas-Agricola, J.M.; Martinez-Hernandez, R.; Martin-Perez, E.; Munoz de Nova, J.L.; Diaz, J.A.; et al. Analysis of expression of the PD-1/PD-L1 immune checkpoint system and its prognostic impact in gastroenteropancreatic neuroendocrine tumors. Sci. Rep. 2018, 8, 17812. [Google Scholar] [CrossRef]
- Ear, P.H.; Li, G.; Wu, M.; Abusada, E.; Bellizzi, A.M.; Howe, J.R. Establishment and characterization of small bowel neuroendocrine tumor spheroids. J. Vis. Exp. 2019. [Google Scholar] [CrossRef]
- Bresciani, G.; Hofland, L.J.; Dogan, F.; Giamas, G.; Gagliano, T.; Zatelli, M.C. Evaluation of spheroid 3D culture methods to study a pancreatic neuroendocrine neoplasm cell line. Front. Endocrinol. (Lausanne) 2019, 10, 682. [Google Scholar] [CrossRef]
- Van Kempen, L.C.; Ruiter, D.J.; van Muijen, G.N.; Coussens, L.M. The tumor microenvironment: A critical determinant of neoplastic evolution. Eur. J. Cell Biol. 2003, 82, 539–548. [Google Scholar] [CrossRef]
- Seuwen, K.; Magnaldo, I.; Pouyssegur, J. Serotonin stimulates DNA synthesis in fibroblasts acting through 5-HT1B receptors coupled to a Gi-protein. Nature 1988, 335, 254–256. [Google Scholar] [CrossRef] [PubMed]
- Nebigil, C.G.; Launay, J.M.; Hickel, P.; Tournois, C.; Maroteaux, L. 5-hydroxytryptamine 2B receptor regulates cell-cycle progression: Cross-talk with tyrosine kinase pathways. Proc. Natl. Acad. Sci. USA 2000, 97, 2591–2596. [Google Scholar] [CrossRef] [PubMed]
- Nemecek, G.M.; Coughlin, S.R.; Handley, D.A.; Moskowitz, M.A. Stimulation of aortic smooth muscle cell mitogenesis by serotonin. Proc. Natl. Acad. Sci. USA 1986, 83, 674–678. [Google Scholar] [CrossRef] [PubMed]
- Pakala, R.; Pakala, R.; Radcliffe, J.D.; Benedict, C.R. Serotonin-induced endothelial cell proliferation is blocked by omega-3 fatty acids. Prostaglandins Leukot. Essent. Fatty Acids 1999, 60, 115–123. [Google Scholar] [CrossRef]
- Spatz, M. Pathogenetic studies of experimentally induced heart lesions and their relation to the carcinoid syndrome. Lab. Investig. 1964, 13, 288–300. [Google Scholar]
- Launay, J.M.; Birraux, G.; Bondoux, D.; Callebert, J.; Choi, D.S.; Loric, S.; Maroteaux, L. Ras involvement in signal transduction by the serotonin 5-HT2B receptor. J. Biol. Chem. 1996, 271, 3141–3147. [Google Scholar] [CrossRef]
- Beauchamp, R.D.; Coffey, R.J., Jr.; Lyons, R.M.; Perkett, E.A.; Townsend, C.M., Jr.; Moses, H.L. Human carcinoid cell production of paracrine growth factors that can stimulate fibroblast and endothelial cell growth. Cancer Res. 1991, 51, 5253–5260. [Google Scholar]
- Chaudhry, A.; Oberg, K.; Gobl, A.; Heldin, C.H.; Funa, K. Expression of transforming growth factors beta 1, beta 2, beta 3 in neuroendocrine tumors of the digestive system. Anticancer Res. 1994, 14, 2085–2091. [Google Scholar] [PubMed]
- Foltyn, W.; Zemczak, A.; Rosiek, V.; Kilian-Kita, A.; Kos-Kudła, B. Effect of treatment with prolonged-release somatostatin analogues on the concentration of serum fibrosis markers in patients with carcinoid syndrome. Neuroendocrinology 2017, 105, 197. [Google Scholar] [CrossRef]
- Wimmel, A.; Wiedenmann, B.; Rosewicz, S. Autocrine growth inhibition by transforming growth factor beta-1 (TGFbeta-1) in human neuroendocrine tumour cells. Gut 2003, 52, 1308–1316. [Google Scholar] [CrossRef] [PubMed]
- Fendrich, V.; Lopez, C.L.; Manoharan, J.; Maschuw, K.; Wichmann, S.; Baier, A.; Holler, J.P.; Ramaswamy, A.; Bartsch, D.K.; Waldmann, J. Enalapril and ASS inhibit tumor growth in a transgenic mouse model of islet cell tumors. Endocr. Relat. Cancer 2014, 21, 813–824. [Google Scholar] [CrossRef] [PubMed]
- Arganini, M.; Spinelli, C.; Cecchini, G.M.; Miccoli, P. Long term treatment with tamoxifen for metastatic carcinoid tumor. Acta Chir. Belg. 1989, 89, 209–211. [Google Scholar]
- Myers, C.F.; Ershler, W.B.; Tannenbaum, M.A.; Barth, R. Tamoxifen and carcinoid tumor. Ann. Intern. Med. 1982, 96, 383. [Google Scholar] [CrossRef] [PubMed]
- Stathopoulos, G.P.; Karvountzis, G.G.; Yiotis, J. Tamoxifen in carcinoid syndrome. N. Engl. J. Med. 1981, 305, 52. [Google Scholar] [CrossRef] [PubMed]
- Zhang, P.J.; Furth, E.E.; Cai, X.; Goldblum, J.R.; Pasha, T.L.; Min, K.W. The role of beta-catenin, TGF beta 3, NGF2, FGF2, IGFR2, and BMP4 in the pathogenesis of mesenteric sclerosis and angiopathy in midgut carcinoids. Hum. Pathol. 2004, 35, 670–674. [Google Scholar] [CrossRef] [PubMed]
- Kaltsas, G.A.; Cunningham, J.L.; Falkmer, S.E.; Grimelius, L.; Tsolakis, A.V. Expression of connective tissue growth factor and IGF1 in normal and neoplastic gastrointestinal neuroendocrine cells and their clinico-pathological significance. Endocr. Relat. Cancer 2011, 18, 61–71. [Google Scholar] [CrossRef] [PubMed]
- Cunningham, J.L.; Tsolakis, A.V.; Jacobson, A.; Janson, E.T. Connective tissue growth factor expression in endocrine tumors is associated with high stromal expression of alpha-smooth muscle actin. Eur. J. Endocrinol. 2010, 163, 691–697. [Google Scholar] [CrossRef] [PubMed]
- Leask, A.; Parapuram, S.K.; Shi-Wen, X.; Abraham, D.J. Connective tissue growth factor (CTGF, CCN2) gene regulation: A potent clinical bio-marker of fibroproliferative disease? J. Cell Commun. Signal. 2009, 3, 89–94. [Google Scholar] [CrossRef]
- Grotendorst, G.R.; Okochi, H.; Hayashi, N. A novel transforming growth factor beta response element controls the expression of the connective tissue growth factor gene. Cell Growth Differ. 1996, 7, 469–480. [Google Scholar]
- Moussad, E.E.; Brigstock, D.R. Connective tissue growth factor: What’s in a name? Mol. Genet. Metab. 2000, 71, 276–292. [Google Scholar] [CrossRef]
- Chaudhry, A.; Papanicolaou, V.; Oberg, K.; Heldin, C.H.; Funa, K. Expression of platelet-derived growth factor and its receptors in neuroendocrine tumors of the digestive system. Cancer Res. 1992, 52, 1006–1012. [Google Scholar] [PubMed]
- Chaudhry, A.; Funa, K.; Oberg, K. Expression of growth factor peptides and their receptors in neuroendocrine tumors of the digestive system. Acta Oncol. (Stockh. Swed.) 1993, 32, 107–114. [Google Scholar] [CrossRef] [PubMed]
- Yao, J.C.; Zhang, J.X.; Rashid, A.; Yeung, S.C.; Szklaruk, J.; Hess, K.; Xie, K.; Ellis, L.; Abbruzzese, J.L.; Ajani, J.A. Clinical and in vitro studies of imatinib in advanced carcinoid tumors. Clin. Cancer Res. 2007, 13, 234–240. [Google Scholar] [CrossRef] [PubMed]
- Alexandraki, K.I.; Philippou, A.; Boutzios, G.; Theohari, I.; Koutsilieris, M.; Delladetsima, I.K.; Kaltsas, G.A. IGF-IEc expression is increased in secondary compared to primary foci in neuroendocrine neoplasms. Oncotarget 2017, 8, 79003–79011. [Google Scholar] [CrossRef]
- Bowen, K.A.; Silva, S.R.; Riall, T.S.; Doan, H.Q.; Evers, B.M. Population-based analysis and growth factor receptor expression of GI carcinoid tumors. Gastroenterology 2009, 136, A880. [Google Scholar] [CrossRef]
- Nilsson, O.; Wangberg, B.; McRae, A.; Dahlstrom, A.; Ahlman, H. Growth factors and carcinoid tumours. Acta Oncol. 1993, 32, 115–124. [Google Scholar] [CrossRef]
- Svejda, B.; Kidd, M.; Timberlake, A.; Harry, K.; Kazberouk, A.; Schimmack, S.; Lawrence, B.; Pfragner, R.; Modlin, I.M. Serotonin and the 5-HT7 receptor: The link between hepatocytes, IGF-1 and small intestinal neuroendocrine tumors. Cancer Sci. 2013, 104, 844–855. [Google Scholar] [CrossRef]
- Chaudhry, A.; Oberg, K. Transforming growth-factor-alpha and epithelial growth-factor receptor expression in neuroendocrine tumors of the digestive-system. Diagn. Oncol. 1993, 3, 81–85. [Google Scholar]
- Haugen, M.; Dammen, R.; Svejda, B.; Gustafsson, B.I.; Pfragner, R.; Modlin, I.; Kidd, M. Differential signal pathway activation and 5-HT function: The role of gut enterochromaffin cells as oxygen sensors. Am. J. Physiol. Gastrointest. Liver Physiol. 2012, 303, G1164–G1173. [Google Scholar] [CrossRef]
- Sagar, V.M.; Neil, D.A.; Papakyriacou, P.; Shah, T.; Liu, B.; Hirschfield, G.; Steeds, R.P.; Shetty, S.; Weston, C.J. The proinflammatory molecule, VAP-1, is enriched in the stroma of midgut NETs and plaques of carcinoid heart disease valves. In Proceedings of the 17th Annual ENETS Conference for the Diagnosis and Treatment of Neuroendocrine Tumor Disease, Barcelona, Spain, 11–13 March 2020. [Google Scholar]
- Bligt-Linden, E.; Pihlavisto, M.; Szatmari, I.; Otwinowski, Z.; Smith, D.J.; Lazar, L.; Fulop, F.; Salminen, T.A. Novel pyridazinone inhibitors for vascular adhesion protein-1 (VAP-1): Old target-new inhibition mode. J. Med. Chem. 2013, 56, 9837–9848. [Google Scholar] [CrossRef]
- Sanvitale, C.E.; Kerr, G.; Chaikuad, A.; Ramel, M.C.; Mohedas, A.H.; Reichert, S.; Wang, Y.; Triffitt, J.T.; Cuny, G.D.; Yu, P.B.; et al. A new class of small molecule inhibitor of BMP signaling. PLoS ONE 2013, 8, e62721. [Google Scholar] [CrossRef] [PubMed]
- Kodach, L.L.; Bleuming, S.A.; Peppelenbosch, M.P.; Hommes, D.W.; van den Brink, G.R.; Hardwick, J.C. The effect of statins in colorectal cancer is mediated through the bone morphogenetic protein pathway. Gastroenterology 2007, 133, 1272–1281. [Google Scholar] [CrossRef] [PubMed]
- Chen, P.Y.; Sun, J.S.; Tsuang, Y.H.; Chen, M.H.; Weng, P.W.; Lin, F.H. Simvastatin promotes osteoblast viability and differentiation via Ras/Smad/Erk/BMP-2 signaling pathway. Nutr. Res. 2010, 30, 191–199. [Google Scholar] [CrossRef] [PubMed]
- Anthony, P.P.; Drury, R.A. Elastic vascular sclerosis of mesenteric blood vessels in argentaffin carcinoma. J. Clin. Pathol. 1970, 23, 110–118. [Google Scholar] [CrossRef]
- Denney, W.D.; Kemp, W.E., Jr.; Anthony, L.B.; Oates, J.A.; Byrd, B.F., 3rd. Echocardiographic and biochemical evaluation of the development and progression of carcinoid heart disease. J. Am. Coll. Cardiol. 1998, 32, 1017–1022. [Google Scholar] [CrossRef]
- Robiolio, P.A.; Rigolin, V.H.; Wilson, J.S.; Harrison, J.K.; Sanders, L.L.; Bashore, T.M.; Feldman, J.M. Carcinoid heart disease. Correlation of high serotonin levels with valvular abnormalities detected by cardiac catheterization and echocardiography. Circulation 1995, 92, 790–795. [Google Scholar] [CrossRef]
- Zuetenhorst, J.M.; Bonfrer, J.M.; Korse, C.M.; Bakker, R.; van Tinteren, H.; Taal, B.G. Carcinoid heart disease: The role of urinary 5-hydroxyindoleacetic acid excretion and plasma levels of atrial natriuretic peptide, transforming growth factor-beta and fibroblast growth factor. Cancer 2003, 97, 1609–1615. [Google Scholar] [CrossRef]
- Gustafsson, B.I.; Tommeras, K.; Nordrum, I.; Loennechen, J.P.; Brunsvik, A.; Solligard, E.; Fossmark, R.; Bakke, I.; Syversen, U.; Waldum, H. Long-term serotonin administration induces heart valve disease in rats. Circulation 2005, 111, 1517–1522. [Google Scholar] [CrossRef]
- Blazevic, A.; Lyer, A.; Van Velthuysen, M.L.F.; Hofland, J.; Franssen, G.J.H.; Feelders, R.A.; Zajec, M.; Luider, T.M.; De Herder, W.W.; Hofland, L.J. Aberrant tryptophan metabolism in stromal cells is associated with mesenteric fibrosis in small intestinal neuroendocrine tumours. In Proceedings of the 17th Annual ENETS Conference for the Diagnosis and Treatment of Neuroendocrine Tumor Disease, Barcelona, Spain, 11–13 March 2020; p. 56. [Google Scholar]
- Welford, R.W.D.; Vercauteren, M.; Trebaul, A.; Cattaneo, C.; Eckert, D.; Garzotti, M.; Sieber, P.; Segrestaa, J.; Studer, R.; Groenen, P.M.A.; et al. Serotonin biosynthesis as a predictive marker of serotonin pharmacodynamics and disease-induced dysregulation. Sci. Rep. 2016, 6, 30059. [Google Scholar] [CrossRef]
- Ignotz, R.A.; Massague, J. Transforming growth factor-beta stimulates the expression of fibronectin and collagen and their incorporation into the extracellular matrix. J. Biol. Chem. 1986, 261, 4337–4345. [Google Scholar]
- Schober, J.M.; Chen, N.; Grzeszkiewicz, T.M.; Jovanovic, I.; Emeson, E.E.; Ugarova, T.P.; Ye, R.D.; Lau, L.F.; Lam, S.C. Identification of integrin alpha(M)beta(2) as an adhesion receptor on peripheral blood monocytes for Cyr61 (CCN1) and connective tissue growth factor (CCN2): Immediate-early gene products expressed in atherosclerotic lesions. Blood 2002, 99, 4457–4465. [Google Scholar] [CrossRef]
- Blom, I.E.; Goldschmeding, R.; Leask, A. Gene regulation of connective tissue growth factor: New targets for antifibrotic therapy? Matrix Biol. 2002, 21, 473–482. [Google Scholar] [CrossRef]
- Brigstock, D.R. The connective tissue growth factor/cysteine-rich 61/nephroblastoma overexpressed (CCN) family. Endocr. Rev. 1999, 20, 189–206. [Google Scholar] [CrossRef] [PubMed]
- Shapiro, M.D.; Hanon, S. Carcinoid heart disease arising from an ovarian teratoma. Int. J. Cardiol. 2005, 104, 115–118. [Google Scholar] [CrossRef]
- Sato, S.; Nagaoka, T.; Hasegawa, M.; Tamatani, T.; Nakanishi, T.; Takigawa, M.; Takehara, K. Serum levels of connective tissue growth factor are elevated in patients with systemic sclerosis: Association with extent of skin sclerosis and severity of pulmonary fibrosis. J. Rheumatol. 2000, 27, 149–154. [Google Scholar] [PubMed]
- Kidd, M.; Modlin, I.M.; Shapiro, M.D.; Camp, R.L.; Mane, S.M.; Usinger, W.; Murren, J.R. CTGF, intestinal stellate cells and carcinoid fibrogenesis. World J. Gastroenterol. 2007, 13, 5208–5216. [Google Scholar] [CrossRef] [PubMed]
- Stoscheck, C.M.; King, L.E., Jr. Role of epidermal growth factor in carcinogenesis. Cancer Res. 1986, 46, 1030–1037. [Google Scholar] [PubMed]
- Druce, M.; Rockall, A.; Grossman, A.B. Fibrosis and carcinoid syndrome: From causation to future therapy. Nat. Rev. Endocrinol. 2009, 5, 276–283. [Google Scholar] [CrossRef]
- Sundin, A.; Arnold, R.; Baudin, E.; Cwikla, J.B.; Eriksson, B.; Fanti, S.; Fazio, N.; Giammarile, F.; Hicks, R.J.; Kjaer, A.; et al. ENETS consensus guidelines for the standards of care in neuroendocrine tumors: Radiological, nuclear medicine & hybrid imaging. Neuroendocrinology 2017, 105, 212–244. [Google Scholar] [CrossRef]
- Landau, M.; Wisniewski, S.; Davison, J. Jejunoileal Neuroendocrine tumors complicated by intestinal ischemic necrosis are associated with worse overall survival. Arch. Pathol. Lab. Med. 2016, 140, 461–466. [Google Scholar] [CrossRef]
- Taylor, J.K. Retroperitoneal fibrosis, regional enteritis, and carcinoid tumors. JAMA 1971, 217, 1864. [Google Scholar] [CrossRef]
- Clement, D.S.; Tesselaar, M.E.; van Leerdam, M.E.; Srirajaskanthan, R.; Ramage, J.K. Nutritional and vitamin status in patients with neuroendocrine neoplasms. World J. Gastroenterol. 2019, 25, 1171–1184. [Google Scholar] [CrossRef]
- Bouma, G.; van Faassen, M.; Kats-Ugurlu, G.; de Vries, E.G.; Kema, I.P.; Walenkamp, A.M. Niacin (Vitamin B3) supplementation in patients with serotonin-producing neuroendocrine tumor. Neuroendocrinology 2016, 103, 489–494. [Google Scholar] [CrossRef]
- Shah, G.M.; Shah, R.G.; Veillette, H.; Kirkland, J.B.; Pasieka, J.L.; Warner, R.R. Biochemical assessment of niacin deficiency among carcinoid cancer patients. Am. J. Gastroenterol. 2005, 100, 2307–2314. [Google Scholar] [CrossRef] [PubMed]
- Pantongrag-Brown, L.; Buetow, P.C.; Carr, N.J.; Lichtenstein, J.E.; Buck, J.L. Calcification and fibrosis in mesenteric carcinoid tumor: CT findings and pathologic correlation. AJR Am. J. Roentgenol. 1995, 164, 387–391. [Google Scholar] [CrossRef] [PubMed]
- Akerstrom, G.; Hellman, P. Surgery on neuroendocrine tumours. Best Pract. Res. Clin. Endocrinol. Metab. 2007, 21, 87–109. [Google Scholar] [CrossRef]
- Laskaratos, F.M.; Hall, A.; Alexander, S.; Stempel, C.V.; Bretherton, J.; Luong, T.V.; Watkins, J.; Ogunbiyi, O.; Toumpanakis, C.; Mandair, D.; et al. Is computed tomography an accurate diagnostic modality for the detection of mesenteric fibrosis in midgut neuroendocrine tumours? Gastroenterology 2019, 156, S-369. [Google Scholar] [CrossRef]
- Laskaratos, F.-M.; Mandair, D.; Hall, A.; Alexander, S.; von Stempel, C.; Bretherton, J.; Luong, T.; Watkins, J.; Ogunbiyi, O.; Rombouts, K.; et al. Clinicopathological correlations of mesenteric fibrosis and evaluation of a novel biomarker for fibrosis detection in small bowel neuroendocrine neoplasms. Endocrine 2020. [Google Scholar] [CrossRef]
- Norlen, O.; Montan, H.; Hellman, P.; Stalberg, P.; Sundin, A. Preoperative (68)Ga-DOTA-Somatostatin Analog-PET/CT hybrid imaging increases detection rate of intra-abdominal small intestinal neuroendocrine tumor lesions. World J. Surg. 2018, 42, 498–505. [Google Scholar] [CrossRef]
- Toumpanakis, C.; Kim, M.K.; Rinke, A.; Bergestuen, D.S.; Thirlwell, C.; Khan, M.S.; Salazar, R.; Oberg, K. Combination of cross-sectional and molecular imaging studies in the localization of gastroenteropancreatic neuroendocrine tumors. Neuroendocrinology 2014, 99, 63–74. [Google Scholar] [CrossRef]
- Shao, T.; Chen, Z.; Belov, V.; Wang, X.; Rwema, S.H.; Kumar, V.; Fu, H.; Deng, X.; Rong, J.; Yu, Q.; et al. [(18)F]-Alfatide PET imaging of integrin alphavbeta3 for the non-invasive quantification of liver fibrosis. J. Hepatol. 2020. [Google Scholar] [CrossRef] [PubMed]
- Laskaratos, F.-M.; Walker, M.; Wilkins, D.; Tuck, A.; Ramakrishnan, S.; Phillips, E.; Gertner, J.; Megapanou, M.; Papantoniou, D.; Shah, R.; et al. Evaluation of clinical prognostic factors and further delineation of the effect of mesenteric fibrosis on survival in advanced midgut neuroendocrine tumours. Neuroendocrinology 2018, 107, 292–304. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez Laval, V.; Pavel, M.; Steffen, I.G.; Baur, A.D.; Dilz, L.M.; Fischer, C.; Detjen, K.; Prasad, V.; Pascher, A.; Geisel, D.; et al. Mesenteric fibrosis in midgut neuroendocrine tumors: Functionality and radiological features. Neuroendocrinology 2018, 106, 139–147. [Google Scholar] [CrossRef] [PubMed]
- De Vries, H.; Wijffels, R.T.M.; Willemse, P.H.B.; Verschueren, R.C.J.; Kema, I.P.; Karrenbeld, A.; Prins, T.R.; de Vries, E.G.E. Abdominal angina in patients with a midgut carcinoid, a sign of severe pathology. World J. Surg. 2005, 29, 1139–1142. [Google Scholar] [CrossRef]
- Daskalakis, K.; Karakatsanis, A.; Hessman, O.; Stuart, H.C.; Welin, S.; Tiensuu Janson, E.; Oberg, K.; Hellman, P.; Norlen, O.; Stalberg, P. Association of a prophylactic surgical approach to stage IV small intestinal neuroendocrine tumors with survival. JAMA Oncol. 2018, 4, 183–189. [Google Scholar] [CrossRef]
- Blazevic, A.; Zandee, W.; Franssen, G.J.H.; Hofland, J.; Van Velthuysen, M.L.F.; Feelders, R.A.; De Herder, W. Palliative surgery in advanced small intestinal neuroendocrine tumors. Neuroendocrinology 2018, 106, 267. [Google Scholar] [CrossRef]
- Bosch, F.; Bruewer, K.; D’Anastasi, M.; Ilhan, H.; Knoesel, T.; Pratschke, S.; Thomas, M.; Rentsch, M.; Guba, M.; Werner, J.; et al. Neuroendocrine tumors of the small intestine causing a desmoplastic reaction of the mesentery are a more aggressive cohort. Surgery 2018, 164, 1093–1099. [Google Scholar] [CrossRef]
- Piccioli, A.N.; Funicelli, L.; Fazio, N.; Borin, S.; Petz, W.; Spinoglio, G.; Bertani, E. Predicting resectability of primary tumor and mesenteric lumps in patients with small intestine neuroendocrine tumors. Neuroendocrinology 2018, 106, 283. [Google Scholar] [CrossRef]
- Gonzalez, R.S.; Cates, J.M.M.; Shi, C. Number, not size, of mesenteric tumor deposits affects prognosis of small intestinal well-differentiated neuroendocrine tumors. Mod. Pathol. Off. J. U. S. Can. Acad. Pathol. 2018, 31, 1560–1566. [Google Scholar] [CrossRef]
- Wu, L.; Fu, J.; Wan, L.; Pan, J.; Lai, S.; Zhong, J.; Chung, D.C.; Wang, L. Survival outcomes and surgical intervention of small intestinal neuroendocrine tumors: A population based retrospective study. Oncotarget 2017, 8, 4935–4947. [Google Scholar] [CrossRef]
- Niederle, B.; Pape, U.F.; Costa, F.; Gross, D.; Kelestimur, F.; Knigge, U.; Oberg, K.; Pavel, M.; Perren, A.; Toumpanakis, C.; et al. ENETS consensus guidelines update for neuroendocrine neoplasms of the jejunum and ileum. Neuroendocrinology 2016, 103, 125–138. [Google Scholar] [CrossRef] [PubMed]
- Clift, A.K.; Frilling, A. Liver transplantation and multivisceral transplantation in the management of patients with advanced neuroendocrine tumours. World J. Gastroenterol. 2018, 24, 2152–2162. [Google Scholar] [CrossRef] [PubMed]
- Pavel, M.; Oberg, K.; Falconi, M.; Krenning, E.; Sundin, A.; Perren, A.; Berruti, A.; Committee, E.G. Gastroenteropancreatic neuroendocrine neoplasms: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann. Oncol. 2020. [Google Scholar] [CrossRef] [PubMed]
- Anthoney, A.; Patel, J.; Kamposioras, K. The use of endoluminal stents to overcome vascular obstruction arising from mesenteric and retroperitoneal neuroendocrine metastases. Neuroendocrinology 2014, 99, 292. [Google Scholar] [CrossRef]
- Caplin, M.E.; Pavel, M.; Ruszniewski, P. Lanreotide in metastatic enteropancreatic neuroendocrine tumors. N. Engl. J. Med. 2014, 371, 1556–1557. [Google Scholar] [CrossRef]
- Rinke, A.; Muller, H.H.; Schade-Brittinger, C.; Klose, K.J.; Barth, P.; Wied, M.; Mayer, C.; Aminossadati, B.; Pape, U.F.; Blaker, M.; et al. Placebo-controlled, double-blind, prospective, randomized study on the effect of octreotide LAR in the control of tumor growth in patients with metastatic neuroendocrine midgut tumors: A report from the PROMID Study Group. J. Clin. Oncol. 2009, 27, 4656–4663. [Google Scholar] [CrossRef]
- Davar, J.; Connolly, H.M.; Caplin, M.E.; Pavel, M.; Zacks, J.; Bhattacharyya, S.; Cuthbertson, D.J.; Dobson, R.; Grozinsky-Glasberg, S.; Steeds, R.P.; et al. Diagnosing and managing carcinoid heart disease in patients with neuroendocrine tumors: An expert statement. J. Am. Coll. Cardiol. 2017, 69, 1288–1304. [Google Scholar] [CrossRef]
- Ertilav, M.; Hur, E.; Bozkurt, D.; Sipahi, S.; Timur, O.; Sarsik, B.; Akcicek, F.; Duman, S. Octreotide lessens peritoneal injury in experimental encapsulated peritoneal sclerosis model. Nephrology (Carlton) 2011, 16, 552–557. [Google Scholar] [CrossRef]
- Lang, A.; Sakhnini, E.; Fidder, H.H.; Maor, Y.; Bar-Meir, S.; Chowers, Y. Somatostatin inhibits pro-inflammatory cytokine secretion from rat hepatic stellate cells. Liver Int. 2005, 25, 808–816. [Google Scholar] [CrossRef]
- Mota, J.M.; Sousa, L.G.; Riechelmann, R.P. Complications from carcinoid syndrome: Review of the current evidence. Ecancermedicalscience 2016, 10, 662. [Google Scholar] [CrossRef]
- Kulke, M.H.; Horsch, D.; Caplin, M.E.; Anthony, L.B.; Bergsland, E.; Oberg, K.; Welin, S.; Warner, R.R.; Lombard-Bohas, C.; Kunz, P.L.; et al. Telotristat ethyl, a tryptophan hydroxylase inhibitor for the treatment of carcinoid syndrome. J. Clin. Oncol. 2017, 35, 14–23. [Google Scholar] [CrossRef]
- Pavel, M.; Gross, D.J.; Benavent, M.; Perros, P.; Srirajaskanthan, R.; Warner, R.R.P.; Kulke, M.H.; Anthony, L.B.; Kunz, P.L.; Horsch, D.; et al. Telotristat ethyl in carcinoid syndrome: Safety and efficacy in the TELECAST phase 3 trial. Endocr. Relat. Cancer 2018, 25, 309–322. [Google Scholar] [CrossRef]
- Saavedra, C.; Barriuso, J.; McNamara, M.G.; Valle, J.W.; Lamarca, A. Spotlight on telotristat ethyl for the treatment of carcinoid syndrome diarrhea: Patient selection and reported outcomes. Cancer Manag. Res. 2019, 11, 7537–7556. [Google Scholar] [CrossRef]
- Zacks, J.; Lavine, R.; Ratner, L.; Warner, R. Telotristat etiprate appears to halt carcinoid heart disease. Neuroendocrinology 2016, 103, 90. [Google Scholar] [CrossRef]
- Robertson, J.I. Carcinoid syndrome and serotonin: Therapeutic effects of ketanserin. Cardiovasc. Drugs Ther. 1990, 4 (Suppl 1), 53–58. [Google Scholar] [CrossRef]
- Moertel, C.G.; Kvols, L.K.; Rubin, J. A study of cyproheptadine in the treatment of metastatic carcinoid tumor and the malignant carcinoid syndrome. Cancer 1991, 67, 33–36. [Google Scholar] [CrossRef]
- Chaturvedi, S.; Misra, D.P.; Prasad, N.; Rastogi, K.; Singh, H.; Rai, M.K.; Agarwal, V. 5-HT2 and 5-HT2B antagonists attenuate pro-fibrotic phenotype in human adult dermal fibroblasts by blocking TGF-beta1 induced non-canonical signaling pathways including STAT3: Implications for fibrotic diseases like scleroderma. Int. J. Rheum. Dis. 2018, 21, 2128–2138. [Google Scholar] [CrossRef] [PubMed]
- Yao, J.C.; Fazio, N.; Singh, S.; Buzzoni, R.; Carnaghi, C.; Wolin, E.; Tomasek, J.; Raderer, M.; Lahner, H.; Voi, M.; et al. Everolimus for the treatment of advanced, non-functional neuroendocrine tumours of the lung or gastrointestinal tract (RADIANT-4): A randomised, placebo-controlled, phase 3 study. Lancet 2016, 387, 968–977. [Google Scholar] [CrossRef]
- Orr-Asman, M.A.; Chu, Z.; Jiang, M.; Worley, M.; LaSance, K.; Koch, S.E.; Carreira, V.S.; Dahche, H.M.; Plas, D.R.; Komurov, K.; et al. mTOR kinase inhibition effectively decreases progression of a subset of neuroendocrine tumors that progress on rapalog therapy and delays cardiac impairment. Mol. Cancer Ther. 2017, 16, 2432–2441. [Google Scholar] [CrossRef]
- Lundin, L.; Norheim, I.; Landelius, J.; Oberg, K.; Theodorsson-Norheim, E. Carcinoid heart disease: Relationship of circulating vasoactive substances to ultrasound-detectable cardiac abnormalities. Circulation 1988, 77, 264–269. [Google Scholar] [CrossRef]
- Kidd, M.; Schimmack, S.; Lawrence, B.; Alaimo, D.; Modlin, I.M. EGFR/TGFalpha and TGFbeta/CTGF signaling in neuroendocrine neoplasia: Theoretical therapeutic targets. Neuroendocrinology 2013, 97, 35–44. [Google Scholar] [CrossRef] [PubMed]
- Rybinski, B.; Franco-Barraza, J.; Cukierman, E. The wound healing, chronic fibrosis, and cancer progression triad. Physiol. Genomics 2014, 46, 223–244. [Google Scholar] [CrossRef]
- Chan, J.A.; Mayer, R.J.; Jackson, N.; Malinowski, P.; Regan, E.; Kulke, M.H. Phase I study of sorafenib in combination with everolimus (RAD001) in patients with advanced neuroendocrine tumors. Cancer Chemother. Pharmacol. 2013, 71, 1241–1246. [Google Scholar] [CrossRef]
- Kulke, M.H.; Lenz, H.J.; Meropol, N.J.; Posey, J.; Ryan, D.P.; Picus, J.; Bergsland, E.; Stuart, K.; Tye, L.; Huang, X.; et al. Activity of sunitinib in patients with advanced neuroendocrine tumors. J. Clin. Oncol. 2008, 26, 3403–3410. [Google Scholar] [CrossRef] [PubMed]
- Pietras, K.; Hanahan, D. A multitargeted, metronomic, and maximum-tolerated dose “chemo-switch” regimen is antiangiogenic, producing objective responses and survival benefit in a mouse model of cancer. J. Clin. Oncol. 2005, 23, 939–952. [Google Scholar] [CrossRef]
- Distler, O.; Gay, S. Scleroderma. Internist (Berl.) 2010, 51, 30–38. [Google Scholar] [CrossRef]
- Strosberg, J.; El-Haddad, G.; Wolin, E.; Hendifar, A.; Yao, J.; Chasen, B.; Mittra, E.; Kunz, P.L.; Kulke, M.H.; Jacene, H.; et al. Phase 3 trial of (177)Lu-Dotatate for midgut neuroendocrine tumors. N. Engl. J. Med. 2017, 376, 125–135. [Google Scholar] [CrossRef]
- Laskaratos, F.; Cox, N.; Woo, W.L.; Khalifa, M.; Ewang, M.; Navalkissoor, S.; Quigley, A.M.; Mandair, D.; Caplin, M.; Toumpanakis, C. Assessment of changes in mesenteric fibrosis (MF) after peptide receptor radionuclide therapy (PRRT) in midgut neuroendocrine tumours (NETs). Neuroendocrinology 2019, 108, 217. [Google Scholar] [CrossRef]
- Ager, E.I.; Neo, J.; Christophi, C. The renin-angiotensin system and malignancy. Carcinogenesis 2008, 29, 1675–1684. [Google Scholar] [CrossRef] [PubMed]
- Moertel, C.G.; Engstrom, P.F.; Schutt, A.J. Tamoxifen therapy for metastatic carcinoid tumor: A negative study. Ann. Intern. Med. 1984, 100, 531–532. [Google Scholar] [CrossRef] [PubMed]
- Mikulec, A.A.; Hanasono, M.M.; Lum, J.; Kadleck, J.M.; Kita, M.; Koch, R.J. Effect of tamoxifen on transforming growth factor beta1 production by keloid and fetal fibroblasts. Arch. Facial Plast. Surg. 2001, 3, 111–114. [Google Scholar] [CrossRef]
- Dreger, N.M.; Degener, S.; Roth, S.; Ahmad-Nejad, P.; Kamper, L.; Muller, E.; von Rundstedt, F.C.; Brandt, A.S. Impact of CYP2D6 polymorphisms on tamoxifen treatment in patients with retroperitoneal fibrosis: A first step towards tailored therapy? Urology 2020, 137, 84–90. [Google Scholar] [CrossRef]
This entry is adapted from the peer-reviewed paper 10.3390/jcm9061777
