Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is an old version of this entry, which may differ significantly from the current revision.
Subjects:
Cell Biology
Treg/Th17 Imbalance means Treg and Th17 cells are dynamically balanced under healthy conditions, but imbalance occurs in inflammatory and pathological states, such as obesity. Some studies demonstrated that peripheral Treg and Th17 cells exhibit increased imbalance with worsening of glucose metabolic dysfunction, already in children with obesity.
- childhood obesity
- adipose tissue-associated inflammation
- Th17
- Treg
- glucose metabolism disorders
1. Introduction
In the last few decades, obesity has dramatically increased in pediatric patients and the link between obesity-induced inflammation and its complications has been described in numerous studies [1][2]. As reported by World Health Organization, the global prevalence of overweight and obesity in children and adolescents aged 5–19 has risen from 4% in 1975 to 18% in 2016 [3]. In 2016 more than 340 million children and adolescents worldwide were in a condition of excess body weight [3].
Obesity is a chronic disease correlated with various factors such as environment, heredity, lifestyle and others [4]. The underlying process is triggered by imbalanced energy intake and consumption [4]. It is well know that systemic inflammation correlates to obesity, characterized by the presence of CD4 and CD8 T cell infiltration and modified immune response, which contributes to the development of obesity related diseases and metabolic disorders like dyslipidemia, type 2 diabetes (T2DM), and cardiovascular pathologies already in pediatric age [5][6].
Adipose tissue (AT) furnishes the organism with a storage of nutrients that is drained during starvation. It produces signals which limit immune cell amount and activity under conditions of nutrient deficiency, allowing proper immune system activity when food sources are available [7]. When excessive AT deposition occurs, it becomes the site of pathological immune system activation, leading to chronic low-grade systemic inflammation. It becomes clear hence that obesity is associated with various disorders in which the immune system plays a key role [8][9]. Naïve T cells are normally quiescent and metabolically inactive, but after stimulation they proliferate and differentiate into various T helper cells (Th), including Th17, and T-regulatory (Treg) cells. Th17 cells primarily fight against extracellular microbial pathogens and mediate autoimmune disease, but they are also known to be involved in allograft rejection. Treg lymphocytes have an opposite function compared to Th17; they attend in modifying the immune response, in order to sustain immune self-tolerance, and prevent autoimmune disease [10][11][12]. Treg produce and secrete, among the others, IL-10 and TGF-β, which could regulate the differentiation and proliferation of lymphocytes and other immune cells [13] suppressing the activation of the immune system. A lack of Treg leads to autoimmune disorders, and a high ratio of Treg/Th17 is proved to be associated with cancer incidence [14][15]. Treg and Th17 cells are dynamically balanced under healthy conditions, but imbalance occurs in inflammatory and pathological states such as obesity [16].
Some studies demonstrated that peripheral Treg and Th17 cells exhibit increased imbalance with worsening of glucose metabolic dysfunction in obese adult and pediatric patients [16][17].
2. Treg/Th17 Dysregulation and Gluco-Metabolic Abnormalities
It is well known that metabolic reprogramming is critically important for lymphocytes. Manipulating metabolic pathways can shape the differentiation and function of these cells [18]. Metabolism furnishes T cells with energy and precursors for many biological processes. Some primary metabolic pathways, such as oxidative phosphorylation, fatty acid oxidation and glycolysis, are considered to play fundamental roles in T cell activation and differentiation.
Functional IL-6 and TGF-β signalling are the initial events needed to start Th17 differentiation. IL-23 and IL-21 play a fundamental role in the maintenance of the Th17 progeny by increasing the transcription of IL-17 and other cytokines. STAT3 (signal transducer and activator of transcription 3), which is critical for the effects of IL-6, IL-21, and IL-23 is required for Th17 differentiation while, on the other hand, the same cytokines are fundamental to initiate the signalling pathways [19][20]. Th17 were identified as a new progeny of CD4+ T helper cells after the finding that experimental autoimmune encephalomyelitis in animal models was caused by high levels of IL-23 rather than IL-12 and Th1 cells [21]. Consequently, it became evident that the function of IL-23 was to promote differentiation and proliferation of the IL-17 secreting cells, classified as Th17. Nevertheless, IL-23 alone has been found to be unable to make naïve T cells differentiate into Th17; some studies showed that polarization of Th17 can be appropriately induced by IL-6 and TGF-β1, which activate STAT3 and Smad family proteins, respectively [22][23]. Dormant naive T cells have relatively small energetic demands, generally supported by glucose oxidation via the Krebs cycle and the oxidation of lipids with low levels of glycolysis, in order to maintain cellular homeostasis [24]. After stimulation, they start to proliferate and differentiate into Th cells. This requires metabolic reprogramming to support their rapid expansion and further functions, such as synthesis of macromolecules, intracellular mediators and cytokines. First, glucose transporter (GLUT) and alanine–serine–cysteine transporter (ASCT2) become highly expressed, then glycolysis fatty acid metabolism, along with OXPHOS, PPP, hexosamine pathway all become active [25].
Aerobic glycolysis and glutamine catabolism become the main pathways, along with a down-regulation of the metabolic characteristic processes of resting cells. The T-cell receptor starts the signalling cascade, along with MAPK (mitogen-activated proteinkinase) ERK (extracellular signal-regulated kinase), PI3K (phosphoinositol-3 kinase), mTOR (mammalian target of rapamycin) and NfκB (nuclear factor-κB). These costimulatory molecules are necessary to induce the Myc and HIF-1α transcription factors, known to induce various gene expression implicated in glycolysis and glutaminolysis [26][27]. If this upregulation of glucose metabolism is not achieved, T-cell differentiation, both in vitro and in vivo, is inhibited [28].
Cellular crosstalk plays a critical role in regulating T-helper maturation and differentiation. As described above, specific cytokines, for the so-called antagonism effect, while drive the generation and function of the specific subset, work to reduce alternative pathways [29].
Treg/Th17 imbalance has been associated with metabolic dysregulation in diabetic patients. A study conducted in HDF-fed transgenic animal models revealed that Treg expansion determined a significant reduction in blood glucose, insulin resistance, and increase in glucose tolerance [30]. In addition, different researches showed a Treg decrease in VAT or in peripheral blood derived from obese and diabetic adults [31][32]. In a recent study, Wen et al. observed Treg/Th17 imbalance in obese and overweight subjects with or without metabolic dysfunctions. The authors reported a severe decrease in Treg/Th17 ratio in peripheral blood of overweight/obese patients with impaired glucose regulation or T2DM compared to healthy subjects or overweight/obese patients with normal glucose tolerance. Moreover, the authors observed that the degree of the imbalance was positively correlated with the exacerbation of metabolic alterations. Furthermore, the serum IL-6 level in patients with metabolic compliance was higher than in controls while the Treg/Th17 ratio was negatively correlated with HbA1c [17].
In the last decades, several studies showed that functional defects of Treg are correlated with the development of IR [33][34][35] (Figure 1). In fact, the IR is known to be linked to the promotion of T cell activation in obese subjects [36]. Recently, Gilleron et al. observed that adipocyte hypertrophy and IR in obese mice were driven by an increase in adipose Th17 and a decrease in adipose Treg. In particular, it has been also described that Treg/Th17 imbalance reduced adipogenesis [37]. In another study, the effects of OX40 has been associated to Th cell differentiation, proliferation and reduction of Treg regulatory activity. The authors underlined how Treg/Th17 balance was crucial for the development of AT inflammation and IR [38]. A lot of studies have shown that pro-inflammatory cytokines such as IL-6, IL-1β, TNF-α, NF-κB can lead to IR and consequently to the development of related diseases like metabolic syndrome and/or diabetes [39][40].
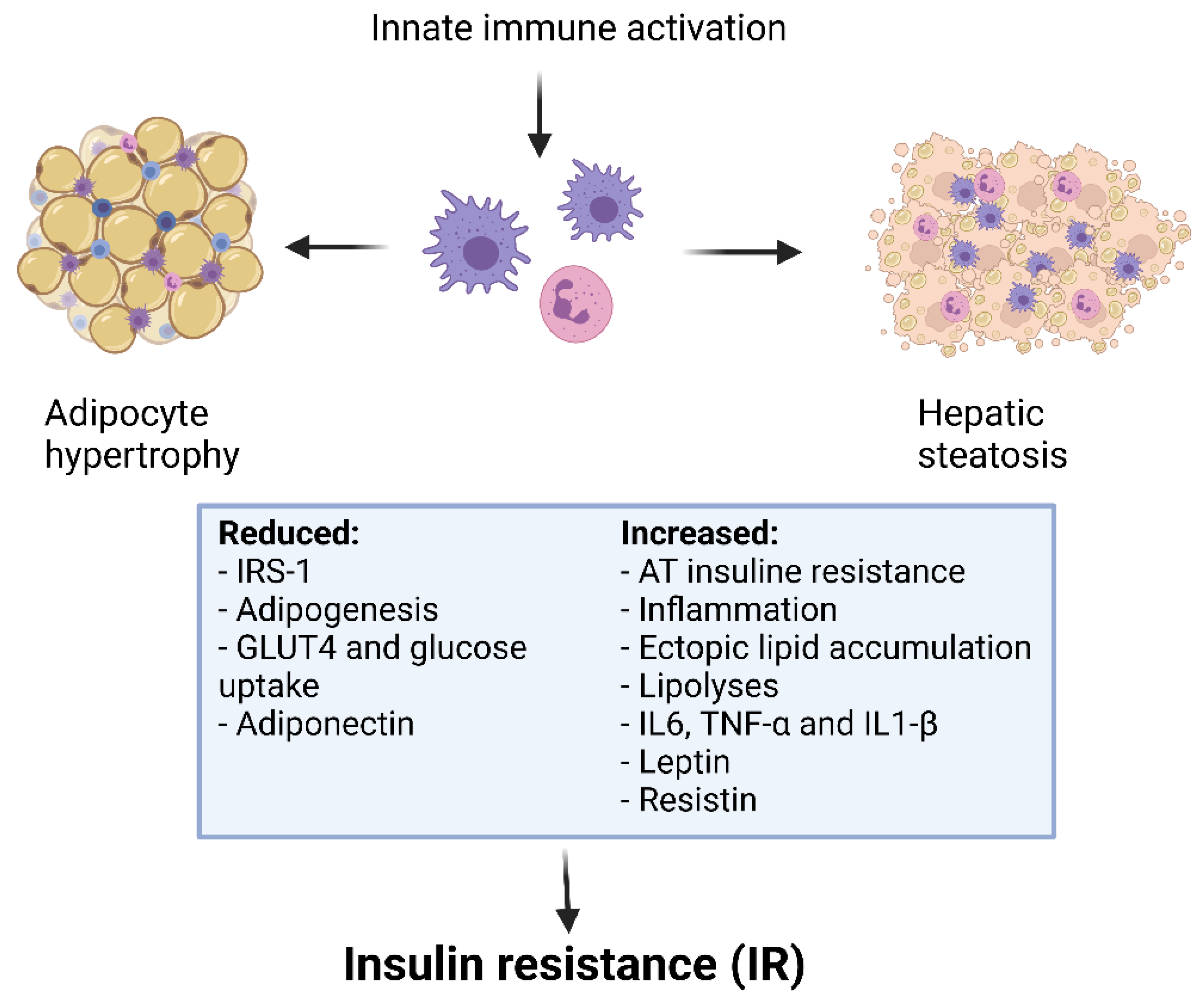 Figure 1. Obesity, inflammation and insulin resistance.
Figure 1. Obesity, inflammation and insulin resistance.Inflammation activates macrophages, along with the release and activation of inflammatory molecules. TNF-α also induces MCP-1 production, which ultimately activates the chemotactic migration of macrophages. These mechanisms result in the inhibition of insulin signalling and sensitivity. Inflammatory molecules also increase peripheral free fatty acid levels through lipolysis, which further aggravates insulin resistance. This determines a perpetual cycle, in which altered values of blood sugar and lipids lead to many related complications [41]. The specific therapeutic treatments for obesity and/or metabolic dysfunctions have been implicated in the regulation of Th17 and Treg balance. Of interest, Martinez-Sanchez et al. showed that high levels of insulin increased the differentiation toward Th17 and decreased Treg maturation in vitro, supporting the hypothesis that Treg/Th17 imbalance could be a mechanism for the onset of metabolic disorders in obesity [42]. Moreover, several data suggest that high insulin levels in obesity cause an inflammatory state by impairing Treg-induced suppression. It has been demonstrated that insulin affects Treg receptors, decreasing IL-10 release through activation of the AKT protein signalling pathway and mTOR [43].
Metformin is a recommended drug for T2DM treatment that improves insulin sensitivity and prevents hyperglycemia by reducing chronic inflammation. It has been reported that the treatment of T2DM patients with metformin induced a decrease in Th17 [44][45][46]. Similarly, in a recent study, Borzouei et al. investigated the expression of immune factors related to Th17 such as RORγt, STAT3, and IL-17, and Treg such as FoxP3, STAT5, and IL-10 in T2DM patients before and after empagliflozin plus metformin and gliclazide. After six months of treatment, a significantly reduction in RORγt and a significantly increase in FoxP3 and STAT5 were reported; IL-17 level was decreased while IL-10 level was enhanced compared to patients treated with only metformin and gliclazide. Empagliflozin showed anti-proliferative and anti-inflammatory effects reducing Th17 and increasing at the same time Treg levels [47].
Han et al. investigated the possible influence of IL-33 on Treg in VAT of four-week-old male mice. They observed that in HFD obese mice, Treg levels diminished but the treatment with IL-33 reversed this condition and counteracted VAT inflammation, leading to a reduction in hyperinsulinemia and IR [48].
Recently, the effect of different molecules on Treg/Th17 ratio has been investigated in animal models. For example, Wei et al. described a possible amelioration in obesity-dependent IR by acacetin in a mouse model. Acacetin seems to down-regulate IL-17 and up-regulate Foxp3 expression, promoting Treg/Th17 balance via targeting miR-23b-3p/NEU1 axis [49]. Finally, a therapeutic effect of epigallocatechin-3-gallate, has been observed in obese mice, showing a significant reduction in weight, LDL-cholesterol and triglyceride levels. Moreover, a higher Treg/Th17 ratio was reported [50].
Recently, data investigating the role of Th17 and Treg in metabolic derangement in pediatric patients are reported. Calcaterra et al. [16] evaluated the Treg/Th17 balance in obese children, in relation with their metabolic status. A correlation between Th17 and systolic hypertension, Treg/Th17 ratio and HOMA-IR was noted. The Treg/Th17 balance appeared to be involved in glycemic homeostasis and blood pressure control [16]. In pediatric patients with chronic inflammation associated-obesity, it has been reported a Th17 involvement, evaluating the frequency of this T cell subset in the peripheral blood. Children with central obesity were characterized by higher percentages of Th17 compared to normal weight children. Moreover, Treg/Th17 ratio positively correlated with total plasma cholesterol concentration [51]. Still referring to paediatric patients, Schindler et al. observed a correlation between overweight and elevated frequency of circulating Th17, IL-17A mRNA levels and RORC. Moreover, Th17 frequency positively correlated with BMI [52]. Of interest, in contrast to previous studies reporting elevated IL-17 levels in obese adults, Jung et al. observed a significant decrease in IL-17 levels in overweight adolescents compared to lean controls. The authors suggested as a possible explanation that the disease conditions associated with obesity such as hypertension and vascular pathologies were not yet present in overweight teenagers [53].
3. Conclusions
Children and adolescents with obesity have a high risk of developing impaired glucose metabolism. Adipose tissue appears to be involved in T cell regulation of tissue inflammatory and in Treg/Th17 imbalance influencing metabolic responses. In the patient care, immune monitoring could play an important role to define preventive strategies of pediatric metabolic disease treatments.
This entry is adapted from the peer-reviewed paper 10.3390/children8070554
References
- Lumeng, C.N.; Saltiel, A.R. Inflammatory links between obesity and metabolic disease. J. Clin. Investig. 2011, 121, 2111–2117.
- Fang, X.; Henao-Mejia, J.; Henrickson, S.E. Obesity and immune status in children. Curr. Opin. Pediatr. 2020, 32, 805–815.
- World Health Organization. WHO. Available online: (accessed on 18 May 2021).
- Kumar, S.; Kelly, A.S. Review of childhood obesity: From epidemiology, etiology, and comorbidities to clinical assessment and treatment. Mayo Clin. Proc. 2017, 92, 251–265.
- Umer, A.; Kelley, G.A.; Cottrell, L.E.; Giacobbi, P.; Innes, K.E.; Lilly, C.L. Childhood obesity and adult cardiovascular disease risk factors: A systematic review with meta-analysis. BMC Public Health 2017, 17, 683.
- Nehus, E.; Mitsnefes, M. Childhood obesity and the metabolic syndrome. Pediatr. Clin. N. Am. 2019, 66, 31–43.
- Childs, C.E.; Calder, P.C.; Miles, E.A. Diet and immune function. Nutrients 2019, 11, 1933.
- Lynch, L.; Nowak, M.; Varghese, B.; Clark, J.; Hogan, A.E.; Toxavidis, V.; Balk, S.P.; O’Shea, D.; O’Farrelly, C.; Exley, M.A. Adipose tissue invariant NKT cells protect against diet-induced obesity and metabolic disorder through regulatory cytokine production. Immunity 2012, 37, 574–587.
- Zhou, H.; Liu, F. Regulation, communication, and functional roles of adipose tissue-resident CD4+ T cells in the control of metabolic homeostasis. Front. Immunol. 2018, 9, 1961.
- Yoshida, S.; Haque, A.; Mizobuchi, T.; Iwata, T.; Chiyo, M.; Webb, T.J.; Baldridge, L.A.; Heidler, K.M.; Cummings, O.W.; Fujisawa, T.; et al. Anti-Type V collagen lymphocytes that express IL-17 and IL-23 induce rejection pathology in fresh and well-healed lung transplants. Am. J. Transplant. Off. J. Am. Soc. Transplant. Am. Soc. Transpl. Surg. 2006, 6, 724–735.
- Burrell, B.E.; Bishop, D.K. Th17 cells and transplant acceptance. Transplantation 2010, 90, 945–948.
- Sakaguchi, S. N aturally A rising CD4 + R egulatory T C ells for I mmunologic S elf -T olerance and N egative C ontrol of I mmune R esponses. Annu. Rev. Immunol. 2004, 22, 531–562.
- Li, M.O.; Sanjabi, S.; Flavell, R.A. Transforming growth factor-β controls development, homeostasis, and tolerance of T cells by regulatory t cell-dependent and -independent mechanisms. Immunity 2006, 25, 455–471.
- Gaur, P.; Qadir, G.A.; Upadhyay, S.; Singh, A.K.; Shukla, N.K.; Das, S.N. Skewed immunological balance between Th17 (CD4+IL17A+) and Treg (CD4+CD25+FOXP3+) cells in human oral squamous cell carcinoma. Cell. Oncol. 2012, 35, 335–343.
- Tosolini, M.; Kirilovsky, A.; Mlecnik, B.; Fredriksen, T.; Mauger, S.; Bindea, G.; Berger, A.; Bruneval, P.; Fridman, W.-H.; Pagès, F.; et al. Clinical impact of different classes of infiltrating t cytotoxic and helper cells (Th1, Th2, Treg, Th17) in patients with colorectal cancer. Cancer Res. 2011, 71, 1263–1271.
- Calcaterra, V.; Croce, S.; Vinci, F.; De Silvestri, A.; Cordaro, E.; Regalbuto, C.; Zuccotti, G.V.; Mameli, C.; Albertini, R.; Avanzini, M.A. Th17 and Treg balance in children with obesity and metabolically altered status. Front. Pediatr. 2020, 8, 591012.
- Wen, J.; Liu, Q.; Liu, M.; Wang, B.; Li, M.; Wang, M.; Shi, X.; Liu, H.; Wu, J. Increasing imbalance of Treg/Th17 indicates more severe glucose metabolism dysfunction in overweight/obese patients. Arch. Med. Res. 2021, 52, 339–347.
- Sun, L.; Fu, J.; Zhou, Y. Metabolism controls the balance of Th17/T-regulatory cells. Front. Immunol. 2017, 8, 1632.
- Zhou, L.; Lopes, J.E.; Chong, M.M.W.; Ivanov, I.I.; Min, R.; Victora, G.D.; Shen, Y.; Du, J.; Rubtsov, Y.P.; Rudensky, A.Y.; et al. TGF-β-induced Foxp3 inhibits TH17 cell differentiation by antagonizing RORγt function. Nature 2008, 453, 236–240.
- Harris, T.J.; Grosso, J.F.; Yen, H.-R.; Xin, H.; Kortylewski, M.; Albesiano, E.; Hipkiss, E.L.; Getnet, D.; Goldberg, M.V.; Maris, C.H.; et al. Cutting edge: An in vivo requirement for STAT3 signaling in T H 17 development and T H 17-dependent autoimmunity. J. Immunol. 2007, 179, 4313–4317.
- Becher, B.; Durell, B.G.; Noelle, R.J. IL-23 Produced by CNS-resident cells controls T cell encephalitogenicity during the effector phase of experimental autoimmune encephalomyelitis. J. Clin. Investig. 2003, 112, 1186–1191.
- Mangan, P.R.; Harrington, L.E.; O’Quinn, D.B.; Helms, W.S.; Bullard, D.C.; Elson, C.O.; Hatton, R.D.; Wahl, S.M.; Schoeb, T.R.; Weaver, C.T. Transforming growth factor-β induces development of the TH17 lineage. Nature 2006, 441, 231–234.
- Veldhoen, M.; Hocking, R.J.; Atkins, C.J.; Locksley, R.M.; Stockinger, B. TGFβ in the context of an inflammatory cytokine milieu supports de novo differentiation of IL-17-producing T cells. Immunity 2006, 24, 179–189.
- Pearce, E.L. Metabolism in T cell activation and differentiation. Curr. Opin. Immunol. 2010, 22, 314–320.
- Wang, R.; Dillon, C.P.; Shi, L.Z.; Milasta, S.; Carter, R.; Finkelstein, D.; McCormick, L.L.; Fitzgerald, P.; Chi, H.; Munger, J.; et al. The transcription factor Myc controls metabolic reprogramming upon T lymphocyte activation. Immunity 2011, 35, 871–882.
- Wang, R.; Green, D.R. The immune diet: Meeting the metabolic demands of lymphocyte activation. F1000 Biol. Rep. 2012, 4, 9.
- Wang, R.; Green, D.R. Metabolic reprogramming and metabolic dependency in T cells. Immunol. Rev. 2012, 249, 14–26.
- Gerriets, V.A.; Rathmell, J.C. Metabolic pathways in T cell fate and function. Trends Immunol. 2012, 33, 168–173.
- Yagi, R.; Junttila, I.S.; Wei, G.; Urban, J.F.; Zhao, K.; Paul, W.E.; Zhu, J. The transcription factor GATA3 actively represses RUNX3 protein-regulated production of interferon-γ. Immunity 2010, 32, 507–517.
- Wang, M.; Chen, F.; Wang, J.; Zeng, Z.; Yang, Q.; Shao, S. Th17 and Treg lymphocytes in obesity and type 2 diabetic patients. Clin. Immunol. 2018, 197, 77–85.
- Deiuliis, J.; Shah, Z.; Shah, N.; Needleman, B.; Mikami, D.; Narula, V.; Perry, K.; Hazey, J.; Kampfrath, T.; Kollengode, M.; et al. Visceral adipose inflammation in obesity is associated with critical alterations in tregulatory cell numbers. PLoS ONE 2011, 6, e16376.
- Wagner, N.-M.; Brandhorst, G.; Czepluch, F.; Lankeit, M.; Eberle, C.; Herzberg, S.; Faustin, V.; Riggert, J.; Oellerich, M.; Hasenfuss, G.; et al. Circulating regulatory T cells are reduced in obesity and may identify subjects at increased metabolic and cardiovascular risk: Regulatory T cells and obesity. Obesity 2013, 21, 461–468.
- Eller, K.; Kirsch, A.; Wolf, A.M.; Sopper, S.; Tagwerker, A.; Stanzl, U.; Wolf, D.; Patsch, W.; Rosenkranz, A.R.; Eller, P. Potential role of regulatory T cells in reversing obesity-linked insulin resistance and diabetic nephropathy. Diabetes 2011, 60, 2954–2962.
- Feuerer, M.; Herrero, L.; Cipolletta, D.; Naaz, A.; Wong, J.; Nayer, A.; Lee, J.; Goldfine, A.B.; Benoist, C.; Shoelson, S.; et al. Lean, but not obese, fat is enriched for a unique population of regulatory T cells that affect metabolic parameters. Nat. Med. 2009, 15, 930–939.
- Cipolletta, D.; Feuerer, M.; Li, A.; Kamei, N.; Lee, J.; Shoelson, S.E.; Benoist, C.; Mathis, D. PPAR-γ Is a major driver of the accumulation and phenotype of adipose tissue treg cells. Nature 2012, 486, 549–553.
- Tao, L.; Liu, H.; Gong, Y. Role and mechanism of the Th17/Treg cell balance in the development and progression of insulin resistance. Mol. Cell. Biochem. 2019, 459, 183–188.
- Gilleron, J.; Bouget, G.; Ivanov, S.; Meziat, C.; Ceppo, F.; Vergoni, B.; Djedaini, M.; Soprani, A.; Dumas, K.; Jacquel, A.; et al. Rab4b deficiency in T cells promotes adipose Treg/Th17 imbalance, adipose tissue dysfunction, and insulin resistance. Cell Rep. 2018, 25, 3329–3341.e5.
- Liu, B.; Yu, H.; Sun, G.; Sun, X.; Jin, H.; Zhang, C.; Shi, W.; Tian, D.; Liu, K.; Xu, H.; et al. OX40 Promotes obesity-induced adipose inflammation and insulin resistance. Cell. Mol. Life Sci. 2017, 74, 3827–3840.
- Kowalska, I.; Straczkowski, M.; Nikolajuk, A.; Adamska, A.; Karczewska-Kupczewska, M.; Otziomek, E.; Kinalska, I.; Gorska, M. Insulin resistance, serum adiponectin, and proinflammatory markers in young subjects with the metabolic syndrome. Metabolism 2008, 57, 1539–1544.
- Tilg, H.; Moschen, A.R. Inflammatory mechanisms in the regulation of insulin resistance. Mol. Med. 2008, 14, 222–231.
- Fuentes, L.; Rőszer, T.; Ricote, M. Inflammatory mediators and insulin resistance in obesity: Role of nuclear receptor signaling in macrophages. Mediators Inflamm. 2010, 2010, 1–10.
- Martinez-Sanchez, M.E.; Hiriart, M.; Alvarez-Buylla, E.R. The CD4+ T cell regulatory network mediates inflammatory responses during acute hyperinsulinemia: A simulation study. BMC Syst. Biol. 2017, 11, 64.
- Han, J.M.; Patterson, S.J.; Speck, M.; Ehses, J.A.; Levings, M.K. Insulin inhibits IL-10–mediated regulatory T cell function: Implications for obesity. J. Immunol. 2014, 192, 623–629.
- Sumarac-Dumanovic, M.; Jeremic, D.; Pantovic, A.; Janjetovic, K.; Stamenkovic-Pejkovic, D.; Cvijovic, G.; Stevanovic, D.; Micic, D.; Trajkovic, V. Therapeutic improvement of glucoregulation in newly diagnosed type 2 diabetes patients is associated with a reduction of IL-17 levels. Immunobiology 2013, 218, 1113–1118.
- Amoani, B.; Sakyi, S.A.; Mantey, R.; Laing, E.F.; Ephraim, R.D.; Sarfo-Katanka, O.; Koffie, S.; Obese, E.; Afranie, B.O. Increased metformin dosage suppresses pro-inflammatory cytokine levels in systemic circulation and might contribute to its beneficial effects. J. Immunoassay Immunochem. 2021, 42, 252–264.
- Lee, S.K.; Park, M.-J.; Jhun, J.Y.; Beak, J.-A.; Choi, J.W.; Rye, J.-Y.; Jang, J.W.; Bae, S.H.; Yoon, S.K.; Choi, H.J.; et al. Combination treatment with metformin and tacrolimus improves systemic immune cellular homeostasis by modulating Treg and Th17 imbalance. Front. Immunol. 2021, 11, 581728.
- Borzouei, S.; Moghimi, H.; Zamani, A.; Behzad, M. Changes in T helper cell-related factors in patients with type 2 diabetes mellitus after empagliflozin therapy. Hum. Immunol. 2021, 82, 422–428.
- Han, J.M.; Wu, D.; Denroche, H.C.; Yao, Y.; Verchere, C.B.; Levings, M.K. IL-33 reverses an obesity-induced deficit in visceral adipose tissue ST2 + T regulatory cells and ameliorates adipose tissue inflammation and insulin resistance. J. Immunol. 2015, 194, 4777–4783.
- Wei, Y.; Jing, J.; Peng, Z.; Liu, X.; Wang, X. Acacetin ameliorates insulin resistance in obesity mice through regulating Treg/Th17 balance via MiR-23b-3p/NEU1 axis. BMC Endocr. Disord. 2021, 21, 57.
- Byun, J.-K.; Yoon, B.-Y.; Jhun, J.-Y.; Oh, H.-J.; Kim, E.; Min, J.-K.; Cho, M.-L. Epigallocatechin-3-gallate ameliorates both obesity and autoinflammatory arthritis aggravated by obesity by altering the balance among CD4+ T-cell subsets. Immunol. Lett. 2014, 157, 51–59.
- Łuczyński, W.; Grubczak, K.; Moniuszko, M.; Głowińska-Olszewska, B.; Bossowski, A. Elevated levels of Th17 cells in children with central obesity. Scand. J. Clin. Lab. Investig. 2015, 75, 595–601.
- Schindler, T.I.; Wagner, J.-J.; Goedicke-Fritz, S.; Rogosch, T.; Coccejus, V.; Laudenbach, V.; Nikolaizik, W.; Härtel, C.; Maier, R.F.; Kerzel, S.; et al. TH17 cell frequency in peripheral blood is elevated in overweight children without chronic inflammatory diseases. Front. Immunol. 2017, 8, 1543.
- Jung, C.; Lichtenauer, M.; Strodthoff, D.; Winkels, H.; Wernly, B.; Bürger, C.; Kamchybekov, U.; Lutgens, E.; Figulla, H.-R.; Gerdes, N. Alterations in systemic levels of Th1, Th2, and Th17 cytokines in overweight adolescents and obese mice. Pediatr. Diabetes 2017, 18, 714–721.
This entry is offline, you can click here to edit this entry!
