In the present work, the state of the art of the most common additive manufacturing (AM) technologies used for the manufacturing of complex shape structures of graphene-based ceramic nanocomposites, ceramic and graphene-based parts is explained. The most important works about the fabrication of composites using graphene-based ceramic pastes by Direct Ink Writing (DIW) are disclosed in detail and illustrated with representative examples. Various examples of the most relevant approaches for the manufacturing of graphene-based ceramic nanocomposites by DIW are provided. Furthermore, different feedstock formulations and their corresponding rheological behavior were explained.
- additive manufacturing
- graphene oxide
- graphene-based paste
- Direct Ink Writing
- ceramic nanocomposites
1. Introduction
| Category | Additive Manufacturing Technology Type | Abbreviation | Feedstock |
|---|---|---|---|
| Vat photopolymerization | Stereolithography | SLA | Liquid photopolymers, hybrid polymer-ceramic, hybrid polymer-graphene |
| Digital Light Processing | DLP | ||
| Two-Photon Polymerization | TPP | ||
| Continuous Liquid Interface Production | CLIP | Liquid photopolymers | |
| Powder bed fusion | Multi Jet Fusion | MJF | Thermoplastic polymers |
| Selective Laser Sintering | SLS | Plastics, composites | |
| Selective Laser Melting | SLM | Metals | |
| Electron Beam Melting | EBM | Metals | |
| Material jetting | Material Jetting | MJ | Photopolymers |
| NanoParticle Jetting | NPJ | Metals, ceramics | |
| Drop On Demand | DOD | Wax, ceramic-, graphene-inks | |
| Material Extrusion | Fused Deposition Modeling | FDM | Thermoplastic polymers, metal-, ceramic-, graphene-reinforced polymers |
| Direct Ink Writing | DIW | Ceramics | |
| Direct Energy Deposition | Electron Beam Additive Manufacturing | EBAM | Metals and alloys in the form of powder or wire |
| Laser Engineering Net Shape | LENS | ||
| Binder jetting | Binder Jetting | BJ | Ceramic, metal, gypsum, sand |
| Sheet Lamination | Laminated Object Manufacturing | LOM | Ceramic, metal-filled tapes, paper, polymer composites. |
-
In vat photopolymerization the 3D object is created layer by layer thanks to the curing of a liquid photopolymer resin under the influence of an ultraviolet (UV) light. The liquid photopolymer is held in a vat with a built support submerged near the surface of the resin. Then the UV light is directed to the resin surface following a determined path thus allowing a selective local polymerization of the liquid photopolymer. After that, the built support is re-submerged into the resin and the process is repeated until the 3D object is fully obtained;
-
The material jetting principle is used to create a solid 3D object layer by layer from droplets, which are mainly composed of liquid photopolymer resin, that are selectively sprayed by an inkjet-style printhead and immediately cured thanks to the expose of a UV light. Commonly, technologies that work under this principle are compared to the two-dimensional (2D) inkjet printing, which deposits only a single layer of ink droplets;
-
In the technologies that operate under the principles of binder jetting a liquid binder, which is selectively deposited by drops onto a powder-based material using an inkjet-style print head, is utilized in order to produce a solid 3D object layer by layer. During the process, alternate layers of powder material and binding material are depositing as follow: powder particles are spread over a built support using a roller while the print head deposits the liquid binder, which acts as a glue between powder particles and layers, on top of the powder bed; after that, the built support is lowered by the model’s layer thickness and then the process is repeated until the 3D object is formed;
-
The powder bed fusion category utilizes an energy source that allows the local sintering or melting between the particles of a powder material for the forming of a solid 3D object layer by layer. The energy sources can be lasers or electron beams depending on the using material powder. The electron beam is necessary for metals, while lasers are required for polymers. The forming part process is very similar to binder jetting: powder particles are spread over a built support using a roller while the energy source fuses the first layer; after that, the built support is lowered by the model’s layer thickness and a new layer of powder is spread across the previous layer repeating the process until the 3D object is formed;
-
In direct energy deposition the 3D object is created layer by layer thanks to the directly melting of build-material and deposing them on the workpiece using a focused thermal energy source such as laser, electron beam or plasma arc. This principle can be applied for a wide kind of materials such as polymers, ceramics, and metal framework composites; however, it is predominantly used for wire and powder metals, which explains why this technology is often called metal deposition. Direct energy deposition utilizes a nozzle mounted on a multi-axis arm can move freely in any direction of the x, y and z-axes that deposits melted material onto the predetermined workpiece surface, where it is automatically solidified;
-
In sheet lamination a 3D part is created by bonding together, layer-by-layer, thin sheets of material (usually supplied via a system of feed rollers), which is then cutting into a final 3D object. In the process, the sheet material is positioned on the cutting bed and then it is bonded over the previous layer using any suitable sticky method; after that, the required shape is cut by laser or knife and the process is repeated until the 3D object is formed. Laminated object manufacturing (LOM) and ultrasonic consolidation (UC) are both examples of sheet lamination techniques;
-
Material extrusion is a category of AM, in which the 3D object is formed by a layer by layer selective deposition of the extruded build-material through a nozzle in a continuous stream. In material extrusion, the layers are built when the nozzle deposits a viscoelastic material where it is required. The following layers are added on top of previous layers and bonded upon deposition as the material shows viscoelastic behavior. In the last past years, this technology became popular in the world for its use in 3D printers. Direct Ink Writing (DIW) and Fused deposition modeling (FDM) are the two common technologies that operate under the principles of material extrusion. However, in the last past years, a new AM technique named Pyro-EHD Tethered Electrospinning (TPES) that is based on electrohydrodynamic processes and can be related to material extrusion category became more and more popular[17][18][19].
2. Direct Ink Writing Technology of Graphene-Based Ceramic Pastes
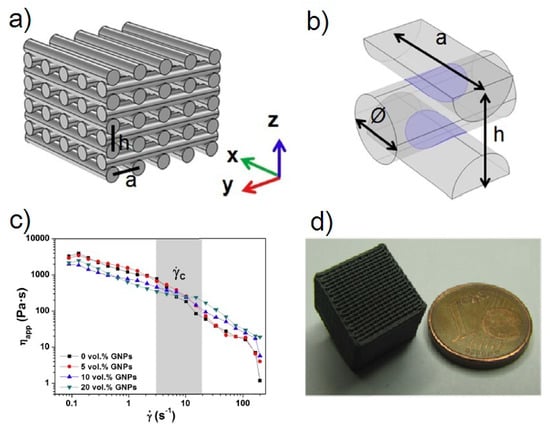
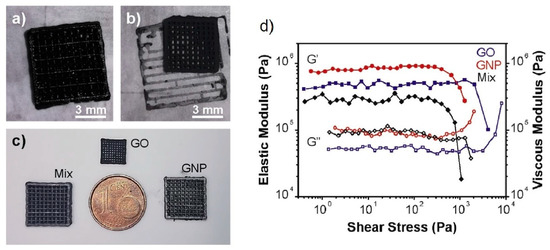
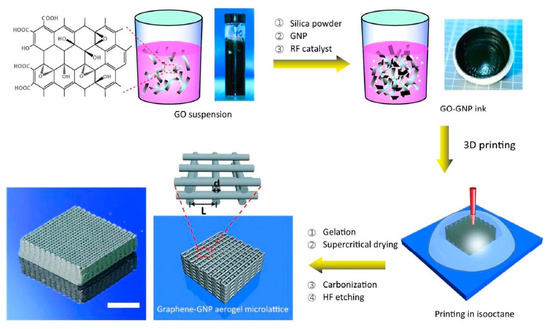
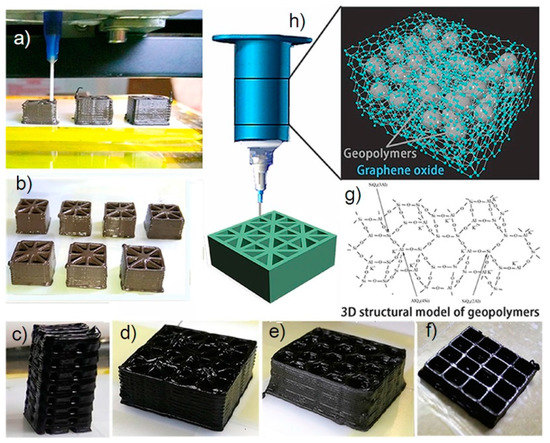


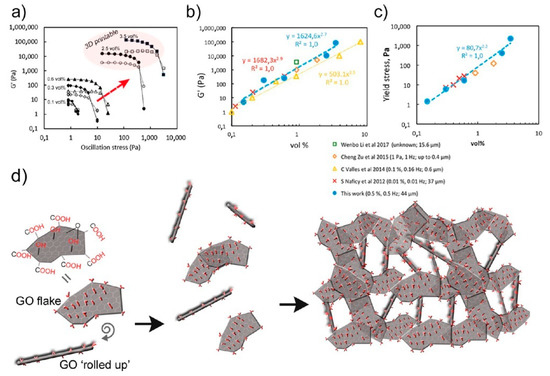


3. Summary
This entry is adapted from the peer-reviewed paper 10.3390/nano10071300
References
- Kingery, W.D.; Bowen, H.K.; Uhlmann, D.R. . Introduction to Ceramics, 2nd ed.; Wiley: New York, NY, USA, 1976; pp. 1–1056.
- Carter, C.B.; Norton, M.G. . Ceramic Materials: Science and Engineering; Springer: New York, NY, USA, 2013; pp. 1–766.
- Smirnov, A.; Kurland, H.-D.; Grabow, J.; Müller, F.A.; Bartolomé, J.F. Microstructure, mechanical properties and low temperature degradation resistance of 2Y-TZP ceramic materials derived from nanopowders prepared by laser vaporization. J. Eur. Ceram. Soc. 2015, 35, 2685–2691.
- Smirnov, A.; Beltrán, J.I.; Rodriguez-Suarez, T.; Pecharromán, C.; Muñoz, M.C.; Moya, J.S.; Bartolomé, J.F. Unprecedented simultaneous enhancement in flaw tolerance and fatigue resistance of zirconia–Ta composites. Sci. Rep. 2017, 7, 44922.
- Smirnov, A.; Bartolomé, J.F.; Kurland, H.-D.; Grabow, J.; Müller, F.A. Design of a new zirconia-alumina-Ta micro-nanocomposite with unique mechanical properties. J. Am. Ceram. Soc. 2016, 99, 3205–3209.
- Bartolomé, J.F.; Smirnov, A.; Kurland, H.-D.; Grabow, J.; Müller, F.A. New ZrO2/Al2O3 nanocomposites fabricated from hybrid nanoparticles prepared by CO2 laser Co-vaporisation. Sci. Rep. 2016, 6, 20589.
- Smirnov, A.; Bartolomé, J.F. Microstructure and mechanical properties of ZrO2 ceramics toughened by 5–20 vol% ta metallic particles fabricated by pressureless sintering. Ceram. Int. 2014, 40, 1829–1834.
- Gutierrez-Gonzalez, C.F.; Smirnov, A.; Bartolomé, J.F. Cyclic fatigue life- and crack-growth behavior of zirconia-niobium composites. J. Am. Ceram. Soc. 2013, 96, 1709–1712.
- Bengisu, M. Engineering Ceramics; Springer-Verlag: Berlin/Heidelberg, Germany, 2001; pp. 620.
- Richerson, D.W.; Lee, W.E. Modern Ceramic Engineering: Properties, Processing, and Use in Design, 4th ed.; CRC Press: Boca, FL, USA, 2018; pp. 1–791.
- Pristinskiy, Y.; Solis Pinargote, N.W.; Smirnov, A. The effect of MgO addition on the microstructure and mechanical properties of alumina ceramic obtained by spark plasma sintering. Mater. Today Proc. 2019, 19, 1990–1993.
- Chen, Z.; Li, Z.; Li, J.; Liu, C.; Lao, C.; Fu, Y.; Liu, C.; Li, Y.; Wang, P.; He, Y. 3D printing of ceramics: A review. J. Eur. Ceram. Soc. 2019, 39, 661–687.
- Abdulhameed, O.; Al-Ahmari, A.; Ameen, W.; Mian, S.H. Additive manufacturing: Challenges, trends, and applications. Adv. Mech. Eng. 2019, 11, 1–27.
- Gibson, I.; Rosen, D.; Stucker, B. Additive Manufacturing Technologies: 3D Printing, Rapid Prototyping, and Direct Digital Manufacturing, 2nd ed.; Springer: New York, NY, USA, 2015; pp. 1–498.
- International Organization for Standardization. ISO/ASTM 52900:2015 [ASTM F2792] Additive Manufacturing—General Principles—Terminology; ISO: Geneva, Switzerland, 2015.
- Diegel, O. A Practical Guide to Design for Additive Manufacturing; Series in Advanced Manufacturing; Springer: New York, NY, USA, 2020; pp. 226.
- Coppola, S.; Nasti, G.; Vespini, V.; Ferraro, P. Layered 3D printing by tethered pyro-electrospinning. Adv. Polym. Technol. 2020, 2, 1–9.
- Coppola, S.; Vespini, V.; Nasti, G.; Gennari, O.; Grilli, S.; Ventre, M.; Iannone, M.; Netti, P.A.; Ferraro, P. Tethered pyro-electrohydrodynamic spinning for patterning well-ordered structures at micro- and nanoscale. Chem. Mater. 2014, 26, 3357–3360.
- Coppola, S.; Nasti, G.; Todino, M.; Olivieri, F.; Vespini, V.; Ferraro, P. Direct writing of microfluidic footpaths by pyro-EHD printing. ACS Appl. Mater. Int. 2017, 9, 16488–16494.
- Lewis, J.A.; Gratson, G.M. Direct writing in three dimensions. Mater. Today 2004, 7, 32–39.
- Martínez-Vázquez, F.J.; Perera, F.H.; Miranda, P.; Pajares, A.; Guiberteau, F. Improving the compressive strength of bioceramic robocast scaffolds by polymer infiltration. Acta Biomater. 2010, 6, 4361–4368.
- Lewis, J.A. Direct ink writing of 3D functional materials. Adv. Funct. Mater. 2006, 16, 2193–2204.
- Revelo, C.F.; Colorado, H.A. 3D printing of kaolinite clay ceramics using the Direct Ink Writing (DIW) technique. Ceram. Int. 2018, 44, 5673–5682.
- Martínez-Vázquez, F.J.; Pajares, A.; Miranda, P. A simple graphite-based support material for robocasting of ceramic parts. J. Eur. Ceram. Soc. 2018, 38, 2247–2250.
- Ordoñez, E.; Gallego, J.M.; Colorado, H.A. 3D printing via the direct ink writing technique of ceramic pastes from typical formulations used in traditional ceramics industry. Appl. Clay Sci. 2019, 182, 105285.
- Ahn, B.Y.; Duoss, E.B.; Motala, M.J.; Guo, X.; Park, S.I.; Xiong, Y.; Yoon, J.; Nuzzo, R.G.; Rogers, J.A.; Lewis, J.A. Omnidirectional printing of flexible, stretchable, and spanning silver microelectrodes. Science 2009, 323, 1590−1593.
- Liu, D.-M. Influence of porosity and pore size on the compressive strength of porous hydroxyapatite ceramic. Ceram. Int. 1997, 23, 135–139.
- Yao, Y.; Fu, K.K.; Yan, C.; Dai, J.; Chen, Y.;Wang, Y.; Zhang, B.; Hitz, E.; Hu, L. Three-dimensional printable high-temperature and high-rate heaters. ACS Nano 2016, 10, 272–5279.
- García-Tuñón, E.; Feilden, E.; Zheng, H.; D’Elia, E.; Leong, A.; Saiz, E. Graphene oxide: An all-in-one processing additive for 3D printing. ACS Appl. Mater. Interfaces 2017, 9, 32977–32989.
- Lewis, J.A. Colloidal processing of ceramics. J. Am. Ceram. Soc. 2000, 83, 2341−2359.
- Xu, Z.; Gao, C. Aqueous liquid crystals of graphene oxide. ACS Nano 2011, 5, 2908−2915.
- Solis Pinargote, N.W.; Peretyagin, P.; Torrecillas, R.; Fernández, A.; Menéndez, J.L.; Mallada, C.; Díaz, L.A.; Moya, J.S. Electrically conductor black zirconia ceramic by SPS using graphene oxide. J. Electroceram 2017, 38, 119–124.
- Smirnov, A.; Peretyagin, P.; Bartolomé, J.F. Processing and mechanical properties of new hierarchical metal-graphene flakes reinforced ceramic matrix composites. J. Eur. Ceram. Soc. 2019, 39, 3491–3497.
- Gutierrez-Gonzalez, C.F.; Smirnov, A.; Centeno, A.; Fernández, A.; Alonso, B.; Rocha, V.G.; Torrecillas, R.; Zurutuza, A.; Bartolomé, J.F. Wear behavior of graphene/alumina nanocomposite. Ceram. Int. 2015, 41, 7434–7438.
- Cascales, A.; Tabares, N.; Bartolomé, J.F.; Cerpa, A.; Smirnov, A.; Moreno, R.; Nieto, M.I. Processing and mechanical properties of mullite and mullite–alumina composites reinforced with carbon nanofibers. J. Eur. Ceram. Soc. 2015, 35, 3613–3621.
- Huang, C.-T.; Kumar Shrestha, L.; Ariga, K.; Hsu, S.-H. A graphene-polyurethane composite hydrogel as a potential bioink for 3D bioprinting and differentiation of neural stem cells. J. Mater. Chem. B 2017, 5, 8854–8864.
- Wang, J.; Liu, Y.; Fan, Z.; Wang, W.; Wang, B.; Guo, Z. Ink-based 3D printing technologies for graphene-based materials a review. Adv. Compos. Hybrid Mater. 2019, 2, 1–33.
- Roman-Manso, B.; Figueiredo, F.M.; Achiaga, B.; Barea, R.; Perez-Coll, D.; Morelos-Gomez, A.; Terrones, M.; Osendi, M.I.; Belmonte, M.; Miranzo, P. Electrically functional 3D-architectured graphene-SiC composites. Carbon 2016, 100, 318–328.
- Zhong, J.; Zhou, G.-X.; He, P.-G.; Yang, Z.-H.; Jia, D.-C. 3D printing strong and conductive geo-polymer nanocomposite structures modified by graphene oxide. Carbon 2017, 117, 421–426.
- Tubio, C.R.; Rama, A.; Gomez, M.; del Rio, F.; Guitian, F.; Gil, A. 3D-printed graphene-Al2O3 composites with complex mesoscale architecture. Ceram. Int. 2018, 44, 5760–5767.
- Moyano, J.J.; Gomez-Gomez, A.; Perez-Coll, D.; Belmonte, M.; Miranzo, P.; Osendi, M.I. Filament printing of graphene-based inks into self-supported 3D architectures. Carbon 2019, 151, 94–102.
- de la Osa, G.; Perez-Coll, D.; Miranzo, P.; Osendi, M.I.; Belmonte, M. Printing of graphene nanoplatelets into highly electrically conductive three-dimensional porous macrostructures. Chem. Mater. 2016, 28, 6321–6328.
- Zhu, C.; Liu, T.; Qian, F.; Han, T.Y.-J.; Duoss, E.B.; Kuntz, J.D.; Spadaccini, C.M.; Worsley, M.A.; Li, Y. Supercapacitors based on three-dimensional hierarchical graphene aerogels with periodic macropores. Nano Lett. 2016, 16, 3448–3456.
- Shen, C.; Calderon, J.E.; Barrios, E.; Soliman, M.; Khater, A.; Jeyaranjan, A.; Tetard, L.; Gordon, A.; Seal, S.; Zhai, L. Anisotropic electrical conductivity in polymer derived ceramics induced by graphene aerogels. J. Mater.Chem. C 2017, 5, 11708–11716.
- Bernardo, E.; Fiocco, L.; Parcianello, G.; Storti, E.; Colombo, P. Advanced ceramics from preceramic polymers modified at the nano-scale-a review. Materials 2014, 7, 1927–1956.
- Pierin, G.; Grotta, C.; Colombo, P.; Mattevi, C. Direct Ink Writing of micrometric SiOC ceramic structures using a preceramic polymer. J. Eur. Ceram. Soc. 2016, 36, 1589–1594.
- Manso, B.R.; Moyano, J.J.; Perez-Coll, D.; Belmonte, M.; Miranzo, P.; Osendi, M.I. Polymer-derived ceramic-graphene oxide architected composite with high electrical conductivity and enhanced thermal resistance. J. Eur. Ceram. Soc. 2018, 38, 2265–2271.
- Moyano, J.J.; Mosa, J.; Aparicio, M.; Pérez-Coll, D.; Belmonte, M.; Miranzo, P.; Osendi, M.I. Strong and light cellular silicon carbonitride–Reduced graphene oxide material with enhanced electrical conductivity and capacitive response. Addit. Manuf. 2019, 30, 100849.
- You, X.; Yang, J.; Huang, K.; Wang, M.; Zhang, X.; Dong, S. Multifunctional silicon carbide matrix composites optimized by three-dimensional graphene scaffolds. Carbon 2019, 155, 215–222.
