Pollution is a major threat for freshwater ecosystems; particularly, environmental degradation is depleting water resources, whether because the surface waters contain higher amounts of pollutants, or because pollutants through the soil attain even the underground water reserves. Assessing the effects of pollution is recently considered among the most relevant aims for subterranean biology. However, the impact of pollution on stygofauna varies according to pollutants typology and abundance, and assessing its effects may not be trivial. Even in cases of relatively small concentrations of pollutants in soils and epikarst, their release may be prolonged and determine a chronic groundwater pollution with unexpected detrimental effects. As stygobionts usually have long life cycles, the comprehension of exposure to certain compounds is far from being understood and may have implications on survival, fitness, and fertility rates. Stygofauna is likely intolerant to even a small alteration of such chemical concentrations.
- invertebrates
- macrobenthos
- sewage
- mining
- detritus
- minerals
- stygobite Niphargus
- flatworm
- aqueduct
- salamander
- freshwater benthos
1. Pollutants Affecting Groundwater Animals
Numerous pollutants may act as anthropogenic stressors for stygofauna, as recently highlighted by an extensive review on the subject [1]. These pollutants include pesticides, fertilizers, metal, salt water, different volatile organic compounds (including aromatic compounds such as benzene), and likely also different emerging pollutants such as micro-plastics and flame retardant tris (1-chloro-2-propyl) phosphate (TCPP) [1][2]. The recent review of Castano-Sanchez, Hose and Reboleira [1] was quite exhaustive, thus it is preferable for readers to refer to it. For the purposes of this perspective, it is important to underline how the effects of these pollutants were observed in both stygophile and stygobiont species by generally measuring in experimental conditions the mortality of field collected individuals [1][3]. Most studies deal with single species, and the effects on the whole groundwater community remain to be assessed; additionally, the choice of the organisms is not mainly based on their functional role they play in the community but on a few abundant and easy-to-sample model species [2][4][5].
2. Effects and Research Perspectives at Community Level
While a relatively rich literature exists on the effects of pollution at the single species level of typical stygobiont species (see the recent review of [1]), few conservation biology and sustainability studies embrace a large part of the groundwater community [6][7][8][9][10]. A whole perspective is important to perform remediation actions and management activities that could be effective at the scale of whole groundwater systems impacted by pollution.
Multiple factors are reported to shape species composition of communities occurring in a groundwater system [11], with no single factor providing a complete explanation for the observed patterns, even in the relatively simple conditions offered by subterranean environments [12]. The complex interactions between species’ physiological constraints, evolutionary processes, and selective pressures currently acting in both surface and subterranean adjacent environments shape the proportion between species stygobionts and species typical of epigean freshwaters that compose the groundwater community (Figure 1). In natural conditions (Figure 1B), species composition of a local groundwater community is a consequence of multiple factors interacting in a hierarchical fashion. Along with the evolutionary and the historical events that shaped stygobionts’ adaptations, chemo-physiological constraints limit the interchange between epigean and subterranean species. Pollution may influence contemporarily more of these factors, disrupting the community features at different levels. In particular, when pollution occurs (Figure 1C), it may alter interspecific interactions, epigean species’ dispersal ability, and habitat selection with deep consequences at the whole community level. We can recognize two main different typologies of effects. First, we can distinguish the effects that act at the core level of groundwater communities represented by stygobionts [13]. Pollution may affect stygobiont keystone species and alter stygobiont functional roles in the trophic webs. Secondly, it is possible to recognize effects that act at the border level between groundwaters and surface freshwater, potentially affecting groundwater colonization by surface species. We discuss challenges and implications of these two effects in the next paragraphs.
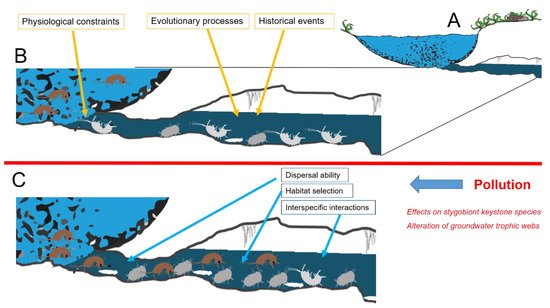
Figure 1. Diagram of the terminal stretch of a groundwater system (A). In natural conditions (B), species composition of a local groundwater community is a consequence of multiple factors interacting in a hierarchical fashion. Along the evolutionary and the historical events that shaped stygobionts’ adaptations, chemo-physiological constraints limit the interchange between epigean and subterranean species. When pollution occurs (C), effects on stygobiont keystone species and trophic webs may alter interspecific interactions, epigean species’ dispersal ability, and habitat selection with deep consequences at the whole community level. Brown figures exemplify epigean animals (amphipods), while grey and white ones represent stygobionts (amphipods, isopods, and planarians).
2.1. Effects on Stygobiont Keystone Species
Stygobiont vertebrates usually are the top predators of the groundwaters they inhabit. As top predators, they likely play keystone roles for the groundwaters communities [14], even if it is difficult to find studies dealing with top-down effects played by top predator stygobiont vertebrates in groundwaters. Among vertebrates, two groups that successfully entered and adapted to subterranean aquatic environments are cave salamanders and cavefishes; direct and indirect information on the possible effects of pollution occur for some of them.
In Europe, the olm (Proteus anguinus) is the most well-known species of stygobiont vertebrate and one of the most outstanding cave dwellers of the karst. Some past studies measured metal concentrations in the olms’ tissues, providing comparisons with the concentrations occurring in the surrounding groundwaters [15][16]. Although groundwaters were not polluted with metals, olms showed higher concentrations of mercury, zinc, and copper, especially in the liver; individuals of the subspecies P. anguinus parkelj, that are locally linked to some spring habitats showed also higher levels of arsenic, especially in the integument [15]. Considering the long life span of the species [17], these data suggest that olms can accumulate metals and toxic substances with potential negative consequences on their fertility, fitness, and survival [16]. Considering that even animals with short lifespan can accumulate contaminants, the effect can be enhanced in stygobionts with long life cycles. Bioaccumlation may be particularly relevant for top predators, even if pollution is likely to affect all the levels of the trophic web. Assessments of the role played by olms’ occurrence and density on whole groundwaters’ communities could be particularly important to highlight the key role that stygobiont salamanders may play and to understand how pollution may disrupt it.
When predator vertebrates are not present, other stygobiont species may occupy top predator level and be affected by pollution with potential cascade effects for the whole community. This could be the case of stygobiont planarians; they are predator flatworms with specialized structures and behaviors that allow them to prey upon all the other aquatic invertebrates [18]. Thus, stygobiont planarians often occupy the highest position of the trophic webs of small subterranean river courses and groundwater interstices [19][20]. Even if there are no direct studies on the effect of pollution for stygobiont planarians, recent research underlined that 30% of the known sites of all the stygobiont planarians of Northern Italy shows signs of pollution, which likely determines the disappearances of species locally [21]. Most genera of epigean planarians are particularly sensitive to organic matter and chemical pollution (Reynoldson and Young, 2000), therefore, it is possible that groundwater pollution has similar detrimental effects on the underground species of the same genera.
The effects of planarians disappearance in polluted groundwaters still need to be assessed and understood, but it is likely that cave-dwelling triclads play control roles for other stygobiont and stygophile invertebrates, mainly crustaceans, bivalves, and annelids, which are their main prey [22].
2.2. Alteration of Groundwater Trophic Webs
Several metals and contaminants were reported to cause mortality in a number of stygobiont species that do not necessarily occupy key positions in the groundwater trophic web [1]. The effects of such contaminants can vary according to the duration and the intensity of the pollution events. In the case of chronical events with extensive releases of toxic contaminants, the effects on the groundwater trophic webs are likely to resemble those observed in epigean freshwaters, where strongly unbalanced communities are characterized by a few dominant, tolerant species, usually detritivores [23][24]. Unfortunately, despite several anecdotal reports of subterranean rivers and streams being chronically or occasionally polluted by heavy metals and xenobiotic compounds, no studies detail the features of their stygofauna; as an example, hearsay affirms that, because of chemical pollution, the stygofauna of one of the most important Italian subterranean rivers, the Timavo, was extremely impoverished during the 19th century, with the disappearance of both top predators such as the olm and typical detrtivore species such as the shrimps of the genus Troglocaris. Studies depicting such effects and providing management perspectives in terms of recovery of the stygofauna communities can be particularly important, especially for the karst areas occurring in emerging countries where pollution effects can be overlooked by current policies.
Something much more challenging is addressing the effects of organic pollution. In temperate regions, groundwaters are usually oligotrophic environments, especially if compared to the surface water bodies [25]. When organic pollution occurs, major changes in the trophic web structure are likely [26]. The first strong effect is on biofilms, which strongly increases their shape and biomass [27]. Thus, signs of organic enrichment in subterranean streams and pools can be easily detected when the sites are accessible [21]. Both organic compounds and biofilms may constitute a resource for stygobionts but may also be detrimental. Indeed, in a review of the few studies documenting the effects of organic pollution on cave macrobenthos, Wood et al. [28] showed that responses at the community level may be quite variable. Particularly, not only reductions in abundance or local extinctions of stygobionts were reported but also cases of increase in the abundance of certain species and trophic groups. Among taxa that could be favored by nutrient enrichment, there are stygobiont isopods. They usually occupy the role of primary consumers and feed on decaying organic material [29] and biofilms [26]. Different past studies evidenced high abundances of isopods in polluted subterranean streams. For example, Holsinger [30], in a cave polluted by sewage, observed in the same sampling occasions both the stygobiont isopod Caecidotea recurvata and its predator Phagocata subterranea. Maximum density of isopods was particularly high (60.9 individuals/m2) compared to that that of planarians (25 individuals/m2) [30]. More than 30 years after, a study performed on the same site that considered only crustaceans revealed that the maximum density of C. recurvata was even higher in moderately and slightly polluted pools (74.6 individuals/m2), while the stygobiont C. recurvata was only present in unpolluted water bodies of the cave [31]. Even though there is no information on the situation prior to pollution, organic enrichment seems to have privileged only one species of primary consumer that became dominant. Moreover, the main predator of this primary consumer was apparently favored, while its predator other species with different tolerance and possibly trophic niches disappeared [28][31]. A similar situation with dearth of amphipods and richness of isopods following organic pollution was recorded by Graening and Brown [32]. In that case, the alteration of the groundwater trophic web apparently provided advantage to a cavefish predator, Amblyopsis rosae, and diminished the occurrence of a salamander predator, Typhlotriton spelaeus [32].
A natural way of organic enrichment for groundwaters is represented by bat guano that may strongly affect groundwater’s community structure [28]. Bat guano not only may supply sufficient resources to decrease the selective pressure posed by oligotrophy, but when it is particularly abundant, it may also generate situations similar to anthropogenic organic inputs for stygofauna. A case of natural organic input suggested to be detrimental is represented by Paradactylodon gorganensis, a salamander that is considered as a valid species as an ecotype of Paradactylodon persicus. P. gorganensis breeds in the terminal trait of an Iranian subterranean stream, which seems polluted by the guano of the bat Myotis blythii [33], posing questions of whether to limit bat occurrence in that site. In this case, further studies are necessary to assess if the organic enrichment could be a threat or, instead, provides resources for the prey of the salamanders.
Moreover, as guano abundance reflects bats’ populations trends, it may be a variable resource. Currently, bat populations are declining, and this decline may be detrimental also for groundwaters communities that rely on guano for their survival [34]. Assessing how bat populations trends result in changes in groundwaters’ community structure could be a promising field of research as the hypothesis that minor organic enrichment/pollution may be advantageous to stygofauna under some circumstances [9].
2.3. Effects on Interspecific Competition between Stygobionts and Epigean Animals
One aspect that often strongly limits the advantages of groundwaters’ organic pollution for stygofauna is that it often allows epigean species to successfully invade subterranean habitats [28]. The enrichment of trophic resources is likely to reduce the constraints that limit epigean taxa dispersion in subterranean environments. The reduced metabolism that characterizes cave organisms and their relatively low rates of activity if compared with surface species are considered advantageous in oligotrophic waters [35][36]. However, when trophic resources increase, the higher metabolic rates in stygophile species may allow them to outcompete stygobionts [36]. We recorded a similar situation in the cave “Grotta di Bocca Lupara” in Liguria (North-western Italy) during a survey performed from December 2017 to March 2018. The subterranean stream flowing through the cave showed strong cover of biofilm/periphyton and evident signs of pollution linked to bat guano and likely to uncollected sewages. Along the stream, we recorded, over different transects, high abundances of the epigean gammarid Echinogammarus sp. gr. veneris (maximum abundance: 8.64 individuals/m2), most of which were depigmented, suggesting a possible ongoing adaptation to the subterranean environment (maximum observed abundance of depigmented individuals: 6.57 individuals/m2). Only few individuals of the stygobiont Niphargus speziae, for which the cave is the type locality, were observed (maximum abundance 0.36 individuals/m2). Other cases of epigean gammarids colonizing groundwaters after being polluted were reported for the Peak Speedwell Cavern system (Derbyshire, UK), where Gammarus pulex invaded subterranean habitats where it was not previously recorded [28]. When groundwaters reach high levels of pollution, other epigean taxa that are tolerant in surface waters may prevail; especially Tubificidae and other annellids are reported to successfully colonize highly polluted subterranean streams [10][37].
This entry is adapted from the peer-reviewed paper 10.3390/su13137030
References
- Castano-Sanchez, A.; Hose, G.C.; Reboleira, A. Ecotoxicological effects of anthropogenic stressors in subterranean organisms: A review. Chemosphere 2020, 244, 125422.
- Mercurio, S.; Messinetti, S.; Manenti, R.; Ficetola, G.F.; Pennati, R. Embryotoxicity characterization of the flame retardant tris(1-chloro-2-propyl)phosphate (TCPP) in the invertebrate chordate Ciona intestinalis. J. Exp. Zool. Part A Ecol. Integr. Physiol. 2021, 335, 339–347.
- Canivet, V.; Gibert, J. Sensitivity of epigean and hypogean freshwater macroinvertebrates to complex mixtures. Part 1: Laboratory experiments. Chemosphere 2002, 46, 999–1009.
- Page, M.J.; Moher, D. Evaluations of the uptake and impact of the Preferred Reporting Items for Systematic reviews and Meta-Analyses (PRISMA) Statement and extensions: A scoping review. Syst. Rev. 2017, 6, 1–14.
- Bolker, B.M.; Brooks, M.E.; Clark, C.J.; Geange, S.W.; Poulsen, J.R.; Stevens, M.H.H.; White, J.S. Generalized linear mixed models: A practical guide for ecology and evolution. Trends Ecol. Evol. 2008, 24, 127–135.
- Asmyhr, M.G.; Hose, G.; Graham, P.; Stow, A.J. Fine-scale genetics of subterranean syncarids. Freshw. Biol. 2014, 59, 1–11.
- Di Lorenzo, T.; Galassi, D.M.P. Agricultural impact on Mediterranean alluvial aquifers: Do groundwater communities respond? Fund. Appl. Limnol. 2013, 182, 271–282.
- Reboleira, A.; Abrantes, N.; Oromi, P.; Goncalves, F. Acute Toxicity of Copper Sulfate and Potassium Dichromate on Stygobiont Proasellus: General Aspects of Groundwater Ecotoxicology and Future Perspectives. Water Air Soil Pollut. 2013, 224, 1–9.
- Sket, B. High biodiversity in hypogean waters and its endangerment—The situation in Slovenia, the Dinaric Karst, and Europe. Crustaceana 1999, 72, 767–779.
- Sket, B. The nature of biodiversity in hypogean waters and how it is endangered. Biodivers. Conserv. 1999, 8, 1319–1338.
- Romero, A. Hypogean Communities as Cybernetic Systems. Diversity 2020, 12, 413.
- Culver, D.C.; Pipan, T. Shallow Subterranean Habitats: Ecology, Evolution, and Conservation; Oxford University Press: New York, NY, USA, 2014.
- Dole-Olivier, M.J.; Malard, F.; Ferreira, D.; Gibert, J. Groundwater biodiversity. Houille Blanche 2005, 39–44.
- Mancinelli, G.; Costantini, M.L.; Rossi, L. Top-down control of reed detritus processing in a lake littoral zone: Experimental evidence of a seasonal compensation between fish and invertebrate predation. Int. Rev. Hydrobiol. 2007, 92, 117–134.
- Bulog, B.; Mihajl, K.; Jeran, Z.; Toman, M.J. Trace element concentrations in the tissues of Proteus anguinus (Amphibia, Caudata) and the surrounding environment. Water Air Soil Pollut. 2002, 136, 147–163.
- Bulog, B. Analyses of some microelements in the tissues of Proteus anguinus (Amphibia, Caudata) and in its habitat. Acta Carsol. 1996, 25, 309–321.
- Voituron, Y.; de Fraipont, M.; Issartel, J.; Guillaume, O.; Clobert, J. Extreme lifespan of the human fish (Proteus anguinus): A challenge for ageing mechanisms. Biol. Lett. 2011, 7, 105–107.
- Gourbault, N. Recherches sur les Triclades Paludicoles hypogés. Mémoires Muséum Natl. d’Histoire Nat. Ser. A 1972, 73, 1–249.
- Barzaghi, B.; De Giorgi, D.; Pennati, R.; Manenti, R. Planarians, a neglected component of biodiversity in groundwaters. Diversity 2021, 13, 178.
- Manenti, R.; Lunghi, E.; Barzaghi, B.; Melotto, A.; Falaschi, M.; Ficetola, G.F. Do salamanders limit the abundance of groundwater invertebrates in subterranean habitats? Diversity 2020, 12, 161.
- Manenti, R.; Barzaghi, B.; Lana, E.; Stocchino, G.A.; Manconi, R.; Lunghi, E. The stenoendemic cave-dwelling planarians (Platyhelminthes, Tricladida) of the Italian Alps and Apennines: Conservation issues. J. Nat. Conserv. 2018, 45, 90–97.
- De Beauchamp, P. Biospeleologica. Turbellariés, Hirudinées, Branchiobdellidés (Deuxième série). In Archives de Zoologie Expérimentale et Générale: Histoire Naturelle, Morphologie, Histologie, Evolution des Animaux; Centre National de la Recherche Scientifique: Paris, France, 1932; Volume 73, pp. 113–380.
- Bosnir, J.; Puntaric, D.; Skes, I.; Klaric, M.; Simic, S.; Zoric, I.; Galic, R. Toxic metals in freshwater fish from the Zagreb area as indicators of environmental pollution. Coll. Antropol. 2003, 27, 31–39.
- Nikanorov, A.M.; Stradomskaya, A.G. Chronic pollution of freshwater bodies: Data on accumulation of pesticides, oil products, and other toxic substances in bottom deposits. Water Resour. 2007, 34, 314–320.
- Culver, D.C.; Pipan, T. Redefining the extent of the aquatic subterranean biotope-shallow subterranean habitats. Ecohydrology 2011, 4, 721–730.
- Francois, C.M.; Mermillod-Blondin, F.; Malard, F.; Fourel, F.; Lecuyer, C.; Douady, C.J.; Simon, L. Trophic ecology of groundwater species reveals specialization in a low-productivity environment. Funct. Ecol. 2016, 30, 262–273.
- Williamson, W.M.; Close, M.E.; Leonard, M.M.; Webber, J.B.; Lin, S. Groundwater biofilm dynamics grown in situ along a nutrient gradient. GroundWater 2012, 50, 690–703.
- Wood, P.J.; Gunn, J.; Rundle, S.D. Response of benthic cave invertebrates to organic pollution events. Aquat. Conserv. 2008, 18, 909–922.
- Ercoli, F.; Lefebvre, F.; Delangle, M.; Gode, N.; Caillon, M.; Raimond, R.; Souty-Grosset, C. Differing trophic niches of three French stygobionts and their implications for conservation of endemic stygofauna. Aquat. Conserv. 2019, 29, 2193–2203.
- Holsinger, J.R. A preliminary study of the effects of organic pollution of Banners Corner Cave, Virginia. Int. J. Speleol. 1966, 2, 75–89.
- Simon, K.S.; Buikema, A.L. Effects of organic pollution on an Appalachian cave: Changes in macroinvertebrate populations and food supplies. Am. Midl. Nat. 1997, 138, 387–401.
- Graening, G.O.; Brown, A.V. Ecosystem dynamics and pollution effects in an Ozark cave stream. J. Am. Water Resour. Assoc. 2003, 39, 1497–1507.
- Raffaëlli, J. Les Urodeles du Monde; Penclen Edition: Condé-sur-Noireau, France, 2007.
- Romero, A. Cave Biology; Cambridge University Press: New York, NY, USA, 2009.
- Hervant, F.; Mathieu, J.; Durand, J.P. Metabolism and circadian rhythms of the European blind cave salamander Proteus anguinus and a facultative cave dweller, the Pyrenean newt (Euproctus asper). Can. J. Zool. 2000, 78, 1427–1432.
- Wilhelm, F.M.; Taylor, S.J.; Adams, G.L. Comparison of routine metabolic rates of the stygobite, Gammarus acherondytes (Amphipoda: Gammaridae) and the stygophile, Gammarus troglophilus. Freshw. Biol. 2006, 51, 1162–1174.
- Wood, P.J.; Gunn, J.; Perkins, J. The impact of pollution on aquatic invertebrates within a subterranean ecosystem—Out of sight out of mind. Arch. Hydrobiol. 2002, 155, 223–237.
