The cyclic nucleotide cAMP (3′,5′-cyclic adenosine monophosphate) is nowadays recognised as an important signalling molecule in plants, involved in many molecular processes, including sensing and response to biotic and abiotic environmental stresses. The validation of a functional cAMP-dependent signalling system in higher plants has spurred a great scientific interest on the polyhedral role of cAMP, as it actively participates in plant adaptation to external stimuli, in addition to the regulation of physiological processes. The complex architecture of cAMP-dependent pathways is far from being fully understood, because the actors of these pathways and their downstream target proteins remain largely unidentified. Recently, a genetic strategy was effectively used to lower cAMP cytosolic levels and hence shed light on the consequences of cAMP deficiency in plant cells. This review aims to provide an integrated overview of the current state of knowledge on cAMP’s role in plant growth and response to environmental stress. Current knowledge of the molecular components and the mechanisms of cAMP signalling events is summarised.
- abiotic stress
- cAMP
- cyclic nucleotides-gated channels
- plant innate immunity
1. Introduction
2. cAMP Involvement in Plant Response to Abiotic Stress
| Stress | Mechanisms | Molecular Players | References |
|---|---|---|---|
| Salinity | Limitation of Na+ influx | VICs; CNGCs | [71,72] |
| Aluminium | K+ current permitting malate outflux | Cation channels | [73] |
| K+ deficiency | K+ homeostasis regulation | AtKUP5; AtKUP7; CNGCs. | [20,21] |
| Heat | Ca2+ influx and HSPs expression | CNGCs; HSPs. | [74] |
| Drought | Synthesis of protective polypeptides | ABA signalling | [75] |
| Wounding | Regulations of the phenylpropanoid pathway | PAL; 4CL; CHS. | [76] |
| ROS | Reduction of Ca2+ influx and K+ efflux | CNGCs | [72] |
3. Role of cAMP in Plant Innate Immunity
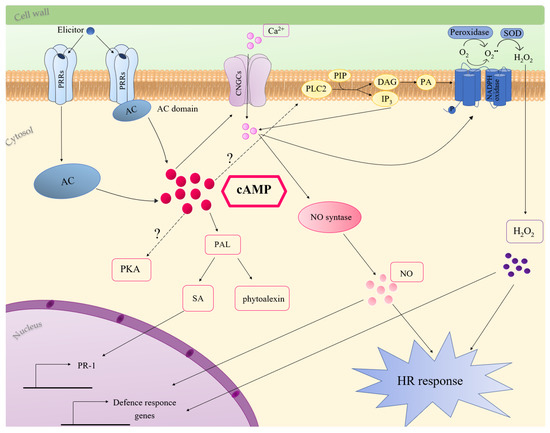
4. Conclusions
This entry is adapted from the peer-reviewed paper 10.3390/ijms21144862
References
- Rall, T.W.; Sutherland, E.W.; Berthet, J. The relation of epinephrine and glucagon to liver phosphorylase. IV. Effect of epinephrine and glucagon on the reactivation of phosphorylase in liver homogenates. J. Biol. Chem. 1957, 224, 1987–1995.
- Shabb, J.B.; Corbin, J.D. Cyclic nucleotide-binding domains in proteins having diverse functions. J. Biol. Chem. 1992, 267, 5723–57236.
- Newton, R.P.; Smith, C.J. Cyclic nucleotides. Phytochemistry 2004, 65, 2423–2437, doi:10.1016/j.phytochem.2004.07.026.
- Gancedo, J.M. Biological roles of cAMP: Variations on a theme in the different kingdoms of life. Biol. Rev. Camb. Philos. Soc. 2013, 88, 645–668, doi:10.1111/brv.12020.
- Arora, K.; Sinha, C.; Zhang, W.; Ren, A.; Moon, C.S.; Yarlagadda, S.; Naren, A.P. Compartmentalization of cyclic nucleotide signaling: A question of when, where, and why? Pflugers Arch. 2013, 465, 1397–1407, doi:10.1007/s00424-013-1280-6.
- Cheepala, S.; Hulot, J.S.; Morgan, J.A.; Sassi, Y.; Zhang, W.; Naren, A.P.; Schuetz, J.D. Cyclic nucleotide compartmentalization: Contributions of phosphodiesterases and ATP-binding cassette transporters. Annu. Rev. Pharmacol. Toxicol. 2013, 53, 231–235, doi:10.1146/annurev-pharmtox-010611-134609.
- Stork, P.J.S.; Schmitt, J.M. Crosstalk between cAMP and MAP kinase signaling in the regulation of cell proliferation. Trends Cell Biol. 2002, 12, 258–266, doi:10.1016/S0962-8924(02)02294-8.
- Amrhein, N. The current status of cyclic AMP in higher plants. Ann. Rev. Plant Physiol. 1977, 28, 123–132, doi:10.1146/annurev.pp.28.060177.001011.
- Brown, E.G., Newton, R.P. Cyclic AMP and higher plants. Phytochemistry 1981, 20, 2453–2463, doi:10.1016/0031-9422(81)83071-3.
- Witters, E.; Roef, L.; Newton, R.P.; VanDongen, W.; Esmans, E.L.; VanOnckelen, H.A. Quantitation of cyclic nucleotides in biological samples by negative electrospray tandem mass spectrometry coupled to ion suppression liquid chromatography. Rapid Commun. Mass Spectrom. 1996, 10, 225–231, doi:10.1002/(Sici)1097-0231(19960131)10:2<225::Aid-Rcm469>3.3.Co;2-A.
- Witters, E.; Vanhoutte, K.; Dewitte, W.; Machackova, I.; Benkova, E.; Van Dongen, W.; Esmans, E.L.; Van Onckelen, H.A. Analysis of cyclic nucleotides and cytokinins in minute plant samples using phase-system switching capillary electrospray-liquid chromatography-tandem mass spectrometry. Phytochem. Anal. 1999, 10, 143–151, doi:10.1002/(Sici)1099-1565(199905/06)10:3<143::Aid-Pca441>3.3.Co;2-7.
- Beavo, J.A.; Brunton, L.L. Cyclic nucleotide research—Still expanding after half a century. Nat. Rev. Mol. Cell Biol. 2002, 3, 710–718, doi:10.1038/nrm911.
- Craven, K.B.; Zagotta, W.N. CNG and HCN channels: Two peas, one pod. Annu. Rev. Physiol. 2006, 68, 375–401, doi:10.1146/annurev.physiol.68.040104.134728.
- Moutinho, A.; Hussey, P.J.; Trewavas, A.J.; Malho, R. cAMP acts as a second messenger in pollen tube growth and reorientation. Proc. Natl. Acad. Sci. USA 2001, 98, 10481–10486, doi:10.1073/pnas.171104598.
- Gehring, C. Adenyl cyclases and cAMP in plant signaling—Past and present. Cell Commun. Signal. 2010, 8, 15, doi:10.1186/1478-811x-8-15.
- Gehring, C.; Turek, I.S. Cyclic Nucleotide monophosphates and their cyclases in plant signaling. Front. Plant Sci. 2017, 8, 1704. 10.3389/Fpls.2017.01704.
- Wong, A.; Tian, X.C.; Gehring, C.; Marondedze, C. Discovery of novel functional centers with rationally designed amino acid motifs. Comput. Struct. Biotechnol. J. 2018, 16, 70–76, doi:10.1016/j.csbj.2018.02.007.
- Ruzvidzo, O.; Gehring, C.; Wong, A. New perspectives on plant adenylyl cyclases. Front. Mol. Biosci. 2019, 6, 136, doi:10.3389/Fmolb.2019.00136.
- Irving, H.R.; Cahill, D.M.; Gehring, C. Moonlighting proteins and their role in the control of signaling microenvironments, as exemplified by cGMP and Phytosulfokine Receptor 1 (PSKR1). Front. Plant Sci. 2018, 9, 415, doi:10.3389/Fpls.2018.00415.
- Al-Younis, I.; Wong, A.; Gehring, C. The Arabidopsis thaliana K+-uptake permease 7 (AtKUP7) contains a functional cytosolic adenylate cyclase catalytic centre. FEBS Lett. 2015, 589, 3848–3852, doi:10.1016/j.febslet.2015.11.038.
- Al-Younis, I.; Wong, A.; Lemtiri-Chlieh, F.; Schrnockel, S.; Tester, M.; Gehring, C.; Donaldson, L. The Arabidopsis thaliana K+-uptake permease 5 (AtKUP5) contains a functional cytosolic adenylate cyclase essential for K+ transport. Front. Plant Sci. 2018, 9, 1645, doi:10.3389/Fpls.2018.01645.
- Bianchet, C.; Wong, A.; Quaglia, M.; Alqurashi, M.; Gehring, C.; Ntoukakis, V.; Pasqualini, S. An Arabidopsis thaliana leucine-rich repeat protein harbors an adenylyl cyclase catalytic center and affects responses to pathogens. J. Plant Physiol. 2019, 232, 12–22, doi:10.1016/j.jplph.2018.10.025.
- Swiezawska, B.; Duszyn, M.; Jaworski, K.; Szmidt-Jaworska, A. Downstream targets of cyclic nucleotides in plants. Front. Plant Sci. 2018, 9, 1428, doi:10.3389/Fpls.2018.01428.
- Kasahara, M.; Suetsugu, N.; Urano, Y.; Yamamoto, C.; Ohmori, M.; Takada, Y.; Okuda, S.; Nishiyama, T.; Sakayama, H.; Kohchi, T.; et al. An adenylyl cyclase with a phosphodiesterase domain in basal plants with a motile sperm system. Sci. Rep. 2016, 6, 39232, doi:10.1038/Srep39232.
- Ma, Y.; He, K.; Berkowitz, G.A., Editorial: From structure to signalsomes: New perspectives about membrane receptors and channels. Front. Plant Sci. 2019, 10, 682, doi:10.3389/Fpls.2019.00682.
- Martinez-Atienza, J.; Van Ingelgem, C.; Roef, L.; Maathuis, F.J. Plant cyclic nucleotide signalling: Facts and fiction. Plant Signal Behav. 2007, 2, 540–543, doi:10.4161/psb.2.6.4789.
- Ma, W.; Smigel, A.; Walker, R.K.; Moeder, W.; Yoshioka, K.; Berkowitz, G.A. Leaf senescence signaling: The Ca2+-conducting Arabidopsis cyclic nucleotide gated channel2 acts through nitric oxide to repress senescence programming. Plant Physiol. 2010, 154, 733–43, doi:10.1104/pp.110.161356.
- Ma, W.; Berkowitz, G.A. Ca2+ conduction by plant cyclic nucleotide gated channels and associated signaling components in pathogen defense signal transduction cascades. New Phytol. 2011, 190, 566–572, doi:10.1111/j.1469-8137.2010.03577.x.
- Jha, S.K.; Sharma, M.; Pandey, G.K. Role of Cyclic nucleotide gated channels in stress management in plants. Curr. Genomics 2016, 17, 315–329, doi:10.2174/1389202917666160331202125.
- Duszyn, M.; Swiezawska, B.; Szmidt-Jaworska, A.; Jaworski, K. Cyclic nucleotide gated channels (CNGCs) in plant signalling-Current knowledge and perspectives. J. Plant Physiol. 2019, 241, 153035, doi:10.1016/J.Jplph.2019.153035.
- Lefkimmiatis, K.; Moyer, M.P.; Curci, S.; Hofer, A.M. “cAMP Sponge”: A buffer for cyclic adenosine 3 ‘, 5 ‘-monophosphate. PLoS ONE 2009, 4, e7649, doi:10.1371/journal.pone.0007649.
- Sabetta, W.; Vannini, C.; Sgobba, A.; Marsoni, M.; Paradiso, A.; Ortolani, F.; Bracale, M.; Viggiano, L.; Blanco, E.; de Pinto, M.C., Cyclic AMP deficiency negatively affects cell growth and enhances stress-related responses in tobacco Bright Yellow-2 cells. Plant Mol. Biol. 2016, 90, 467–483, doi:10.1007/s11103-016-0431-5.
- Sabetta, W.; Vandelle, E.; Locato, V.; Costa, A.; Cimini, S.; Bittencourt Moura, A.; Luoni, L.; Graf, A.; Viggiano, L.; De Gara, L.; et al.. Genetic buffering of cyclic AMP in Arabidopsis thaliana compromises the plant immune response triggered by an avirulent strain of Pseudomonas syringae pv. tomato. Plant J. 2019, 98, 590–606, doi:10.1111/tpj.14275.
- Anderson, J.A.; Huprikar, S.S.; Kochian, L.V.; Lucas, W.J.; Gaber, R.F., Functional expression of a probable Arabidopsis-thaliana potassium channel in Saccharomyces-cerevisiae. Proc. Natl. Acad. Sci. USA 1992, 89, 3736–3740, doi:10.1073/pnas.89.9.3736.
- Bolwell, G.P. Cyclic-AMP, the reluctant messenger in plants. Trends Biochem. Sci. 1995, 20, 492–495, doi:10.1016/S0968-0004(00)89114-8.
- Trewavas, A.J. Plant cyclic AMP comes in from the cold. Nature 1997, 390, 657–658, doi:10.1038/37720.
- Ehsan, H.; Reichheld, J.P.; Roef, L.; Witters, E.; Lardon, F.; Van Bockstaele, D.; Van Montagu, M.; Inze, D.; Van Onckelen, H. Effect of indomethacin on cell cycle dependent cyclic AMP fluxes in tobacco BY-2 cells. FEBS Lett. 1998, 422, 165–169, doi:10.1016/S0014-5793(97)01610-4.
- Ehsan, H.; Roef, L.; Witters, E.; Reichheld, J.P.; Van Bockstaele, D.; Inze, D.; Van Onckelen, H. Indomethacin-induced G1/S phase arrest of the plant cell cycle. FEBS Lett. 1999, 458, 349–353, doi:10.1016/S0014-5793(99)01152-7.
- Curvetto, N. Effect of two cAMP analogs on stomatal opening in Vicia faba: Possible relationship with cytosolic calcium concentration. Plant Physiol. Biochem. 1994, 32, 365–372.
- Jin, X.C.; Wu, W.H. Involvement of cyclic AMP in ABA- and Ca2+-mediated signal transduction of stomatal regulation in Vicia faba. Plant Cell Physiol. 1999, 40, 1127–1133, doi:10.1093/oxfordjournals.pcp.a029497.
- Thomas, L.; Marondedze, C.; Ederli, L.; Pasqualini, S.; Gehring, C. Proteomic signatures implicate cAMP in light and temperature responses in Arabidopsis thaliana. J. Proteom. 2013, 83, 47–59, doi:10.1016/j.jprot.2013.02.032.
- Alqurashi, M.; Gehring, C.; Marondedze, C. Changes in the Arabidopsis thaliana proteome implicate cAMP in biotic and abiotic stress responses and changes in energy metabolism. Int. J. Mol. Sci. 2016, 17, 852, doi:10.3390/Ijms17060852.
- Donaldson, L.; Meier, S.; Gehring, C. The arabidopsis cyclic nucleotide interactome. Cell Commun. Signal. 2016, 14, 10, doi:10.1186/s12964-016-0133-2.
- Kurosaki, F.; Tsurusawa, Y.; Nishi, A. The elicitation of phytoalexins by Ca2+ and cyclic AMP in carrot cells. Phytochemistry, 1987, 26, 1919–1923, doi:10.1016/S0031-9422(00)81729-X.
- Li, W.W.; Luan, S.; Schreiber, S.L.; Assmann, S.M. Cyclic-AMP stimulates K+ channel activity in mesophyll-cells of Vicia-faba-K. Plant Physiol. 1994, 106, 957–961, doi:10.1104/Pp.106.3.957.
- Uchiyama, T.; Yoshikawa, F.; Hishida, A.; Furuichi, T.; Mikoshiba, K. A novel recombinant hyperaffinity inositol 1,4,5-trisphosphate (IP3) absorbent traps IP3, resulting in specific inhibition of IP3-mediated calcium signaling. J. Biol. Chem. 2002, 277, 8106–8113, doi:10.1074/jbc.M108337200.
- Saraswat, L.D.; Ringheim, G.E.; Bubis, J.; Taylor, S.S. Deletion mutants as probes for localizing regions of subunit interaction in cAMP-dependent protein-kinase. J. Biol. Chem. 1988, 263, 18241–18246.
- Volotovski, I.D.; Sokolovsky, S.G.; Molchan, O.V.; Knight, M.R. Second messengers mediate increases in cytosolic calcium in tobacco protoplasts. Plant Physiol. 1998, 117, 1023–1030, doi:10.1104/pp.117.3.1023.
- Lemtiri-Chlieh, F.; Berkowitz, G.A. Cyclic adenosine monophosphate regulates calcium channels in the plasma membrane of Arabidopsis leaf guard and mesophyll cells. J. Biol. Chem. 2004, 279, 35306–35312, doi:10.1074/jbc.M400311200.
- Talke, I.N.; Blaudez, D.; Maathuis, F.J.M.; Sanders, D. CNGCs: Prime targets of plant cyclic nucleotide signalling? Trends Plant Sci. 2003, 8, 286–293, doi:10.1016/S1360-1385(03)00099-2.
- Ali, R.; Ma, W.; Lemtiri-Chlieh, F.; Tsaltas, D.; Leng, Q.; von Bodman, S.; Berkowitz, G.A. Death don’t have no mercy and neither does calcium: Arabidopsis CYCLIC NUCLEOTIDE GATED CHANNEL2 and innate immunity. Plant Cell 2007, 19, 1081–1095, doi:10.1105/tpc.106.045096.
- Munemasa, S.; Hossain, M.A.; Nakamura, Y.; Mori, I.C.; Murata, Y. The Arabidopsis calcium-dependent protein kinase, CPK6, functions as a positive regulator of methyl jasmonate signaling in guard cells. Plant Physiol. 2011, 155, 553–561, doi:10.1104/pp.110.162750.
- Lu, M.; Zhang, Y.Y.; Tang, S.K.; Pan, J.B.; Yu, Y.K.; Han, J.; Li, Y.Y.; Du, X.H.; Nan, Z.J.; Sun, Q.P. AtCNGC2 is involved in jasmonic acid-induced calcium mobilization. J. Exp. Bot. 2016, 67, 809–819, doi:10.1093/jxb/erv500.
- Malho, R.; Camacho, L.; Moutinho, A. Signalling pathways in pollen tube growth and reorientation. Ann. Bot. 2000, 85, 59–68, doi:10.1006/anbo.1999.0991.
- Wu, J.Y.; Qu, H.Y.; Jin, C.; Shang, Z.L.; Wu, J.; Xu, G.H.; Gao, Y.B.; Zhang, S.L. cAMP activates hyperpolarization-activated Ca2+ channels in the pollen of Pyrus pyrifolia. Plant Cell Rep. 2011, 30, 1193–1200, doi:10.1007/s00299-011-1027-9.
- Tezuka, T.; Hiratsuka, S.; Takahashi, S.Y. Promotion of the growth of self-incompatible pollen tubes in lily by cAMP. Plant Cell Physiol. 1993, 34, 955–958, doi:10.1093/oxfordjournals.pcp.a078508.
- Tsuruhara, A.; Tezuka, T. Relationship between the self-incompatibility and cAMP level in Lilium longiflorum. Plant Cell Physiol. 2001, 42, 1234–1238, doi:10.1093/Pcp/Pce159.
- Tezuka, T.; Akita, I.; Yoshino, N. Self-incompatibility involved in the level of acetylcholine and cAMP. Plant Signal Behav. 2007, 2, 475–476, doi:10.4161/psb.2.6.4483.
- Duffus, C.M.; Duffus, J.H. A possible role for cyclic AMP in gibberellic acid triggered release of alpha-amylase in barley endosperm slices. Experientia 1969, 25, 581, doi:10.1007/BF01896521.
- Hall, K.A.; Galsky, A.G. The action of cyclic-AMP on GA3 controlled responses IV. Characteristics of the promotion of seed germination in Lactuca sative variety ‘Spartan Lake’ by gibberellic acid and cyclic 3,5′-adenosine monophosphate. Plant Cell Physiol. 1973, 14, 565–571, doi:10.1093/oxfordjournals.pcp.a074892.
- Holm, R.; Miller, M. Hormonal control of weed seed germination. Weed Sci. 1972, 20, 209–212, doi:10.1017/S0043174500035426.
- Uematsu, K.; Nakajima, M.; Yamaguchi, I.; Yoneyama, K.; Fukui, Y. Role of cAMP in gibberellin promotion of seed germination in Orobanche minor smith. J. Plant Growth Regul. 2007, 26, 245–254, doi:10.1007/s00344-007-9012-9.
- Uematsu, K.; Fukui, Y. Role and regulation of cAMP in seed germination of Phacelia tanacetifolia. Plant Physiol. Bioch. 2008, 46, 768–774, doi:10.1016/j.plaphy.2007.10.015.
- Domanska, A.; Godlewski, M.; Aniol, P. Db-cAMP, forskolin or 2 ‘-d3 ‘-AMP influence on proliferation of Raphanus sativus root meristem cells. Caryologia 2009, 62, 267–275, doi:10.1080/00087114.2004.10589692.
- Moulding, D.A.; Blundell, M.P.; Spiller, D.G.; White, M.R.; Cory, G.O.; Calle, Y.; Kempski, H.; Sinclair, J.; Ancliff, P.J.; Kinnon, C.; et al. Unregulated actin polymerization by WASp causes defects of mitosis and cytokinesis in X-linked neutropenia. J. Exp. Med. 2007, 204, 2213–24, doi:10.1084/jem.20062324.
- Terakado, J.; Okamura, M.; Fujihara, S.; Ohmori, M.; Yoneyama, T. Cyclic AMP in rhizobia and symbiotic nodules. Ann. Bot-London 1997, 80, 499–503, doi:10.1006/anbo.1997.0477.
- Terakado, J.; Fujihara, S.; Yoneyama, T. Changes in cyclic nucleotides during nodule formation. Soil Sci. Plant Nutr. 2003, 49, 459–462, doi:10.1080/00380768.2003.10410032.
- He, M.; He, C.Q.; Ding, N.Z. Abiotic stresses: General defenses of land plants and chances for engineering multistress tolerance. Front. Plant Sci. 2018, 9, 1771, doi:10.3389/fpls.2018.01771.
- Kosova, K.; Vitamvas, P.; Prasil, I.T.; Renaut, J. Plant proteome changes under abiotic stress-contribution of proteomics studies to understanding plant stress response. J. Proteomics 2011, 74, 1301–1322, doi:10.1016/j.jprot.2011.02.006.
- Lamers, J.; van der Meer, T.; Testerink, C. How plants sense and respond to stressful environments. Plant Physiol. 2020, 182, 1624–1635, doi:10.1104/pp.19.01464.
- Maathuis, F.J.; Sanders, D. Sodium uptake in Arabidopsis roots is regulated by cyclic nucleotides. Plant Physiol. 2001, 127, 1617–1625, doi:10.1104/pp.010502.
- Ordonez, N.M.; Marondedze, C.; Thomas, L.; Pasqualini, S.; Shabala, L.; Shabala, S.; Gehring, C. Cyclic mononucleotides modulate potassium and calcium flux responses to H2O2 in Arabidopsis roots. FEBS Lett. 2014, 588, 1008–1015, doi:10.1016/j.febslet.2014.01.062.
- Zhang, W.H.; Ryan, P.R.; Tyerman, S.D. Malate-permeable channels and cation channels activated by aluminum in the apical cells of wheat roots. Plant Physiol. 2001, 125, 1459–1472, doi:10.1104/pp.125.3.1459.
- Gao, F.; Han, X.W.; Wu, J.H.; Zheng, S.Z.; Shang, Z.L.; Sun, D.Y.; Zhou, R.G.; Li, B. A heat-activated calcium-permeable channel—Arabidopsis cyclic nucleotide-gated ion channel 6—is involved in heat shock responses. Plant J. 2012, 70, 1056–1069, doi:10.1111/j.1365-313X.2012.04969.x.
- Maksyutova, N.N.; Viktorova, L.V. A comparative study of the effect of abscisic acid and cAMP on protein synthesis in wheat caryopses under drought conditions. Biochemistry (Mosc) 2003, 68, 424–428, doi:10.1023/a:1023651913998.
- Swieiawska, B.; Jaworski, K.; Pawelek, A.; Grzegorzewska, W.; Szewczuk, P.; Szmidt-Jaworska, A. Molecular cloning and characterization of a novel adenylyl cyclase gene, HpAC1, involved in stress signaling in Hippeastrum x hybridum. Plant Physiol. Bioch. 2014, 80, 41–52, doi:10.1016/j.plaphy.2014.03.010.
- Ahn, S.J.; Shin, R.; Schachtman, D.P. Expression of KT/KUP genes in Arabidopsis and the role of root hairs in K+ uptake. Plant Physiol. 2004, 134, 1135–1145, doi:10.1104/pp.103.034660.
- Han, M.; Wu, W.; Wu, W.H.; Wang, Y. Potassium transporter KUP7 is involved in K(+) acquisition and translocation in Arabidopsis root under K(+)-limited conditions. Mol. Plant 2016, 9, 437–446, doi:10.1016/j.molp.2016.01.012.
- Kohler, C.; Merkle, T.; Neuhaus, G. Characterisation of a novel gene family of putative cyclic nucleotide- and calmodulin-regulated ion channels in Arabidopsis thaliana. Plant J. 1999, 18, 97–104, doi:10.1046/j.1365-313x.1999.00422.x.
- Arazi, T.; Kaplan, B.; Fromm, H. A high-affinity calmodulin-binding site in a tobacco plasma-membrane channel protein coincides with a characteristic element of cyclic nucleotide-binding domains. Plant Mol. Biol. 2000, 42, 591–601, doi:10.1023/a:1006345302589.
- Hua, B.G.; Mercier, R.W.; Zielinski, R.E.; Berkowitz, G.A. Functional interaction of calmodulin with a plant cyclic nucleotide gated cation channel. Plant Physiol. Bioch. 2003, 41, 945–954, doi:10.1016/j.plaphy.2003.07.006.
- Leng, Q.; Mercier, R.W.; Yao, W.; Berkowitz, G.A. Cloning and first functional characterization of a plant cyclic nucleotide-gated cation channel. Plant Physiol. 1999, 121, 753–761, doi:10.1104/pp.121.3.753.
- Kugler, A.; Kohler, B.; Palme, K.; Wolff, P.; Dietrich, P. Salt-dependent regulation of a CNG channel subfamily in Arabidopsis. BMC Plant Biol. 2009, 9, 140, doi:10.1186/1471-2229-9-140.
- Guo, K.M.; Babourina, O.; Christopher, D.A.; Borsics, T.; Rengel, Z. The cyclic nucleotide-gated channel, AtCNGC10, influences salt tolerance in Arabidopsis. Physiol. Plant 2008, 134, 499–507, doi:10.1111/j.1399-3054.2008.01157.x.
- Tunc-Ozdemir, M.; Tang, C.; Ishka, M.R.; Brown, E.; Groves, N.R.; Myers, C.T.; Rato, C.; Poulsen, L.R.; McDowell, S.; Miller, G.; et al., A cyclic nucleotide-gated channel (CNGC16) in pollen is critical for stress tolerance in pollen reproductive development. Plant Physiol. 2013, 161, 1010–1020, doi:10.1104/pp.112.206888.
- Finka, A.; Cuendet, A.F.; Maathuis, F.J.; Saidi, Y.; Goloubinoff, P. Plasma membrane cyclic nucleotide gated calcium channels control land plant thermal sensing and acquired thermotolerance. Plant Cell 2012, 24, 3333–3348, doi:10.1105/tpc.112.095844.
- Witters, E.; Valcke, R.; van Onckelen, H. Cytoenzymological analysis of adenylyl cyclase activity and 3 ‘: 5 ‘-cAMP immunolocalization in chloroplasts of Nicotiana tabacum. New Phytol. 2005, 168, 99–107, doi:10.1111/j.1469-8137.2005.01476.x.
- Molchan, O.V.; Sokolovsky, S.G.; Volotovsky, I.D. The phytochrome control of the cAMP endogenous level in oat seedlings. Russ. J. Plant Physl. 2000, 47, 463–467.
- Pietrowska-Borek, M.; Nuc, K. Both cyclic-AMP and cyclic-GMP can act as regulators of the phenylpropanoid pathway in Arabidopsis thaliana seedlings. Plant Physiol. Bioch. 2013, 70, 142–149, doi:10.1016/j.plaphy.2013.05.029.
- Miller, G.; Suzuki, N.; Rizhsky, L.; Hegie, A.; Koussevitzky, S.; Mittler, R. Double mutants deficient in cytosolic and thylakoid ascorbate peroxidase reveal a complex mode of interaction between reactive oxygen species, plant development, and response to abiotic stresses. Plant Physiol. 2007, 144, 1777–1785, doi:10.1104/pp.107.101436.
- de Pinto, M.C.; Locato, V.; Paradiso, A.; De Gara, L. Role of redox homeostasis in thermo-tolerance under a climate change scenario. Ann. Bot. 2015, 116, 487–496, doi:10.1093/aob/mcv071.
- Miller, G.; Schlauch, K.; Tam, R.; Cortes, D.; Torres, M.A.; Shulaev, V.; Dangl, J.L.; Mittler, R. The plant NADPH oxidase RBOHD mediates rapid systemic signaling in response to diverse stimuli. Sci. Signal. 2009, 2, ra45, doi:10.1126/scisignal.2000448.
- Gilroy, S.; Suzuki, N.; Miller, G.; Choi, W.G.; Toyota, M.; Devireddy, A.R.; Mittler, R. A tidal wave of signals: Calcium and ROS at the forefront of rapid systemic signaling. Trends Plant Sci. 2014, 19, 623–360, doi:10.1016/j.tplants.2014.06.013.
- Steinhorst, L.; Kudla, J. Calcium and reactive oxygen species rule the waves of signaling. Plant Physiol. 2013, 163, 471–485, doi:10.1104/pp.113.222950.
- Bose, J.; Rodrigo-Moreno, A.; Shabala, S. ROS homeostasis in halophytes in the context of salinity stress tolerance. J. Exp. Bot. 2014, 65, 1241–1257, doi:10.1093/jxb/ert430.
- Pottosin, I.; Velarde-Buendia, A.M.; Bose, J.; Zepeda-Jazo, I.; Shabala, S.; Dobrovinskaya, O. Cross-talk between reactive oxygen species and polyamines in regulation of ion transport across the plasma membrane: Implications for plant adaptive responses. J. Exp. Bot. 2014, 65, 1271–1283, doi:10.1093/jxb/ert423.
- Zhu, J.K. Regulation of ion homeostasis under salt stress. Curr. Opin. Plant Biol. 2003, 6, 441–445, doi:10.1016/s1369-5266(03)00085-2.
- Shabala, S.; Cuin, T.A. Potassium transport and plant salt tolerance. Physiol. Plant 2008, 133, 651–669, doi:10.1111/j.1399-3054.2007.01008.x.
- Jones, J.D.G.; Dangl, J.L. The plant immune system. Nature 2006, 444, 323–329, doi:10.1038/nature05286.
- Wu, Y.; Zhou, J.M. Receptor-like kinases in plant innate immunity. J. Integr. Plant Biol. 2013, 55, 1271–1286, doi:10.1111/jipb.12123.
- Tang, D.Z.; Wang, G.X.; Zhou, J.M. Receptor kinases in plant-pathogen interactions: More than pattern recognition. Plant Cell 2017, 29, 618–637, doi:10.1105/tpc.16.00891.
- Couto, D.; Zipfel, C. Regulation of pattern recognition receptor signalling in plants. Nat. Rev. Immunol. 2016, 16, 537–552, doi:10.1038/nri.2016.77.
- Cui, H.T.; Tsuda, K.; Parker, J.E. Effector-Triggered Immunity: From pathogen perception to robust defense. Annu. Rev. Plant Biol. 2015, 66, 487–511, doi:10.1146/annurev-arplant-050213-040012.
- Wang, W.; Feng, B.M.; Zhou, J.M.; Tang, D.Z. Plant immune signaling: Advancing on two frontiers. J. Integr. Plant Biol. 2020, 62, 2–24, doi:10.1111/jipb.12898.
- Kurosaki, F.; Nishi, A. Stimulation of calcium influx and calcium cascade by cyclic AMP in cultured carrot cells. Arch. Biochem. Biophys. 1993, 302, 144–151, doi:10.1006/abbi.1993.1192.
- Cooke, C.J.; Smith, C.J.; Walton, T.J.; Newton, R.P. Evidence that cyclic-AMP is involved in the hypersensitive response of Medicago-sativa to a fungal elicitor. Phytochemistry 1994, 35, 889–895, doi:10.1016/S0031-9422(00)90633-2.
- Zhao, J.; Guo, Y.Q.; Fujita, K.; Sakai, K. Involvement of cAMP signaling in elicitor-induced phytoalexin accumulation in Cupressus lusitanica cell cultures. New Phytol. 2004, 161, 723–733, doi:10.1111/j.1469-8137.2004.00976.x.
- Jiang, J.; Fan, L.W.; Wu, W.H. Evidences for involvement of endogenous cAMP in Arabidopsis defense responses to Verticillium toxins. Cell Res. 2005, 15, 585–592, doi:10.1038/sj.cr.7290328.
- Ma, W.; Qi, Z.; Smigel, A.; Walker, R.K.; Verma, R.; Berkowitz, G.A. Ca2+, cAMP, and transduction of non-self perception during plant immune responses. Proc. Natl. Acad. Sci. USA 2009, 106, 20995–21000, doi:10.1073/pnas.0905831106.
- Lomovatskaya, L.A.; Kuzakova, O.V.; Romanenko, A.S.; Goncharova, A.M. Activities of adenylate cyclase and changes in cAMP concentration in root cells of pea seedlings infected with mutualists and phytopathogens. Russ. J. Plant Physiol. 2018, 65, 588–597, doi:10.1134/S1021443718030056.
- Mauch-Mani, B.; Slusarenko, A.J. Production of salicylic acid precursors is a major function of phenylalanine ammonia-lyase in the resistance of Arabidopsis to Peronospora parasitica. Plant Cell 1996, 8, 203–212, doi:10.1105/tpc.8.2.203.
- Huang, J.; Gu, M.; Lai, Z.; Fan, B.; Shi, K.; Zhou, Y.H.; Yu, J.Q.; Chen, Z. Functional analysis of the Arabidopsis PAL gene family in plant growth, development, and response to environmental stress. Plant Physiol. 2010, 153, 1526–1538, doi:10.1104/pp.110.157370.
- Shine, M.B.; Yang, J.W.; El-Habbak, M.; Nagyabhyru, P.; Fu, D.Q.; Navarre, D.; Ghabrial, S.; Kachroo, P.; Kachroo, A. Cooperative functioning between phenylalanine ammonia lyase and isochorismate synthase activities contributes to salicylic acid biosynthesis in soybean. New Phytol. 2016, 212, 627–636, doi:10.1111/nph.14078.
- Torres, M.A.; Jones, J.D.G.; Dangl, J.L. Reactive oxygen species signaling in response to pathogens. Plant Physiol. 2006, 141, 373–378, doi:10.1104/pp.106.079467.
- Torres, M.A, ROS in biotic interactions. Physiol. Plant 2010, 138, 414–429, doi:10.1111/j.1399-3054.2009.01326.x.
- Qi, J.S.; Wang, J.L.; Gong, Z.Z.; Zhou, J.M., Apoplastic ROS signaling in plant immunity. Curr. Opin. Plant Biol.2017, 38, 92–100, doi:10.1016/j.pbi.2017.04.022.
- Bolwell, G.P. Role of active oxygen species and NO in plant defence responses. Curr. Opin. Plant Biol. 1999, 2, 287–294, doi:10.1016/S1369-5266(99)80051-X.
- Bindschedler, L.V.; Minibayeva, F.; Gardner, S.L.; Gerrish, C.; Davies, D.R.; Bolwell, G.P. Early signalling events in the apoplastic oxidative burst in suspension cultured French bean cells involve cAMP and Ca2+. New Phytol. 2001, 151, 185–194, doi:10.1046/j.1469-8137.2001.00170.x.
- Davies, D.R.; Bindschedler, L.V.; Strickland, T.S.; Bolwell, GP. Production of reactive oxygen species in Arabidopsis thaliana cell suspension cultures in response to an elicitor from Fusarium oxysporum: Implications for basal resistance. J. Exp. Bot. 2006, 57, 1817–1827, doi:10.1093/jxb/erj216.
- Clough, S.J.; Fengler, K.A.; Yu, I.C.; Lippok, B.; Smith, R.K.; Bent, A.F. The Arabidopsis dnd1 “defense, no death” gene encodes a mutated cyclic nucleotide-gated ion channel. Proc. Natl. Acad. Sci. USA 2000, 97, 9323–9328, doi:10.1073/pnas.150005697.
- Balague, C.; Lin, B.Q.; Alcon, C.; Flottes, G.; Malmstrom, S.; Kohler, C.; Neuhaus, G.; Pelletier, G.; Gaymard, F.; Roby, D. HLM1, an essential signaling component in the hypersensitive response, is a member of the cyclic nucleotide-gated channel ion channel family. Plant Cell 2003, 15, 365–379, doi:10.1105/tpc.006999.
- Yoshioka, K.; Moeder, W.; Kang, H.G.; Kachroo, P.; Masmoudi, K.; Berkowitz, G.; Klessig, D.F. The chimeric Arabidopsis CYCLIC NUCLEOTIDE-GATED ION CHANNEL11/12 activates multiple pathogen resistance responses. Plant Cell 2006, 18, 747–763, doi:10.1105/tpc.105.038786.
- Dangl, J.L.; Dietrich, R.A.; Richberg, M.H. Death don’t have no mercy: Cell death programs in plant-microbe interactions. Plant Cell 1996, 8, 1793–1807.doi:10.1105/tpc.8.10.1793.
- Grant, M.; Brown, I.; Adams, S.; Knight, M.; Ainslie, A.; Mansfield, J. The RPM1 plant disease resistance gene facilitates a rapid and sustained increase in cytosolic calcium that is necessary for the oxidative burst and hypersensitive cell death. Plant J. 2000, 23, 441–450, doi:10.1046/j.1365-313x.2000.00804.x.
- Lecourieux, D.; Raneva, R.; Pugin, A. Calcium in plant defence-signalling pathways. New Phyto.l 2006, 171, 249–269, doi:10.1111/j.1469-8137.2006.01777.x.
- Lamotte, O.; Courtois, C.; Dobrowolska, G.; Besson, A.; Pugin, A.; Wendehenne, D. Mechanisms of nitric-oxide-induced increase of free cytosolic Ca2+ concentration in Nicotiana plumbaginifolia cells. Free Radic. Bio. Med. 2006, 40, 1369–1376, doi:10.1016/j.freeradbiomed.2005.12.006.
- Delledonne, M.; Xia, Y.J.; Dixon, R.A.; Lamb, C. Nitric oxide functions as a signal in plant disease resistance. Nature 1998, 394, 585–588, doi:10.1038/29087.
- Durner, J.; Wendehenne, D.; Klessig, D.F. Defense gene induction in tobacco by nitric oxide, cyclic GMP, and cyclic ADP-ribose. Proc. Natl. Acad. Sci. USA 1998, 95, 10328–10333, doi:10.1073/pnas.95.17.10328.
- de Jong, C.F.; Laxalt, A.M.; Bargmann, B.O.R.; de Wit, P.J.G.M.; Joosten, M.H.A.J.; Munnik, T. Phosphatidic acid accumulation is an early response in the Cf-4/Avr4 interaction. Plant J. 2004, 39, 1–12, doi:10.1111/j.1365-313X.2004.02110.x.
- Andersson, M.X.; Kourtchenko, O.; Dangl, J.L.; Mackey, D.; Ellerstrom, M. Phospholipase-dependent signalling during the AvrRpm1- and AvrRpt2-induced disease resistance responses in Arabidopsis thaliana. Plant J. 2006, 47, 947–959, doi:10.1111/j.1365-313X.2006.02844.x.
- D’Ambrosio, J.M.; Couto, D.; Fabro, G.; Scuffi, D.; Lamattina, L.; Munnik, T.; Andersson, M.X.; Alvarez, M.E.; Zipfel, C.; Laxalt, A.M. Phospholipase C2 affects MAMP-triggered immunity by modulating ROS production. Plant Physiol. 2017, 175, 970–981, doi:10.1104/pp.17.00173.
- Yang, T.B.; Poovaiah, B.W. A calmodulin-binding/CGCG box DNA-binding protein family involved in multiple signaling pathways in plants. J. Biol. Chem. 2002, 277, 45049–45058, doi:10.1074/jbc.M207941200.
- Du, L.Q.; Ali, G.S.; Simons, K.A.; Hou, J.G.; Yang, T.B.; Reddy, A.S.N.; Poovaiah, B.W. Ca2+/calmodulin regulates salicylic-acid-mediated plant immunity. Nature 2009, 457, 1154–1158, doi:10.1038/nature07612.
- Zhou, W.B.; Freed, C.R. DJ-1 up-regulates glutathione synthesis during oxidative stress and inhibits A53T alpha-synuclein toxicity. J. Biol. Chem. 2005, 280, 43150–43158, doi:10.1074/jbc.M507124200.
- Wei, H.R.; Persson, S.; Mehta, T.; Srinivasasainagendra, V.; Chen, L.; Page, G.P.; Somerville, C.; Loraine, A. Transcriptional coordination of the metabolic network in Arabidopsis. Plant Physiol. 2006, 142, 762–774, doi:10.1104/pp.106.080358.
- Shirasu, K. The HSP90-SGT1 Chaperone complex for NLR immune sensors. Annu. Rev. Plant Biol. 2009, 60, 139–164, doi:10.1146/annurev.arPlant59.032607.092906.
- Yadeta, K.A.; Elmore, J.M.; Creer, A.Y.; Feng, B.M.; Franco, J.Y.; Rufian, J.S.; He, P.; Phinney, B.; Coaker, G. A Cysteine-rich protein kinase associates with a membrane immune complex and the cysteine residues are required for cell death. Plant Physiol. 2017, 173, 771–787, doi:10.1104/pp.16.01404.
- Ito, M.; Takahashi, H.; Sawasaki, T.; Ohnishi, K.; Hikichi, Y.; Kiba, A. Novel type of adenylyl cyclase participates in tabtoxinine-beta-lactam-induced cell death and occurrence of wildfire disease in Nicotiana benthamiana. Plant Signal. Behav. 2014, 9, 27420, doi:10.4161/psb.27420.
- Rich, T.C.; Webb, K.J.; Leavesley, S.J. Can we decipher the information content contained within cyclic nucleotide signals? J. Gen. Physiol. 2014, 143, 17–27, doi:10.1085/jgp.201311095.
- Wheeler, J.I.; Freihat, L.; Irving, H.R. A cyclic nucleotide sensitive promoter reporter system suitable for bacteria and plant cells. BMC Biotechnol. 2013, 13, 97, doi:10.1186/1472-6750-13-97.
- Jiang, J.Y.; Falcone, J.L.; Curci, S.; Hofer, A.M. Interrogating cyclic AMP signaling using optical approaches. Cell Calcium 2017, 64, 47–56, doi:10.1016/j.ceca.2017.02.010.
- Klausen, C.; Kaiser, F.; Stuven, B.; Hansen, J.N.; Wachten, D. Elucidating cyclic AMP signaling in subcellular domains with optogenetic tools and fluorescent biosensors. Biochem. Soc. Trans. 2019, 47, 1733–1747, doi:10.1042/BST20190246.
- Rahman, N.; Buck, J.; Levin, L.R. pH sensing via bicarbonate-regulated “soluble” adenylyl cyclase (sAC). Front. Physiol. 2013, 4, 343, doi:10.3389/fphys.2013.00343.
- Paramonov, V.M.; Mamaeva, V.; Sahlgren, C.; Rivero-Muller, A. Genetically-encoded tools for cAMP probing and modulation in living systems. Front. Pharmacol. 2015, 6, 196, doi:10.3389/fphar.2015.00196.
