Trichuris sp. infection has appeared as a pathological burden in the population, but the immunomodulation features could result in an opportunity to discover novel treatments for diseases with prominent inflammatory responses. Regarding the immunological aspects, the innate immune responses against Trichuris sp. are also responsible for determining subsequent immune responses, including the activation of innate lymphoid cell type 2 (ILC2s), and encouraging the immune cell polarization of the resistant host phenotype. Nevertheless, this parasite can establish a supportive niche for worm survival and finally avoid host immune interference. Trichuris sp. could skew antigen recognition and immune cell activation and proliferation through the generation of specific substances, called excretory/secretory (ESPs) and soluble products (SPs), which mainly mediate its immunomodulation properties.
- antigen
- innate lymphoid cells 2 (ILC2s)
- Th1
- Th2
- interleukin-13
- interleukin-10
1. Basic Immunology Concept against Trichuris trichiura
1.1. Innate Immune System Also Determines the Fate of Infection
1.2. Antibody-Dependent Cell-mediated Cytotoxicity (ADCC): Is It Reliable for Trichuriasis?
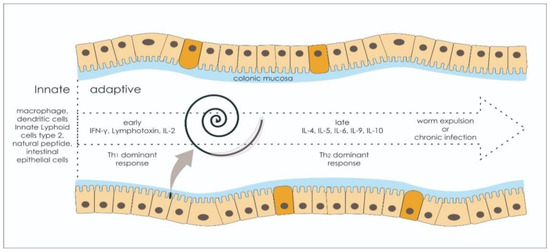
1.3. Adaptive Immune Response: Worm Expulsion versus Chronic Infection
2. Immunomodulation Properties of Trichuris sp. and Clinical Implications: Focus on the Role of Excretory/Secretory (ESPs) and Soluble Products (SPs)
2.1. Secreted Products Modulate Pattern Recognition Receptor (PRR)
2.2. Secreted Product Skewed Innate Immune System
2.3. Secreted Product Produce Deviant Cytokine Response
This entry is adapted from the peer-reviewed paper 10.3390/life11030188
References
- King, C.; Low, C.; Nutman, T. IgE production in human helminth infection. Reciprocal interrelationship between IL-4 and IFN-gamma. J. Immunol. 1993, 150, 1873–1880.
- Belhassen-García, M.; Pardo-Lledías, J.; del Villar, L.P.; Muro, A.; Velasco-Tirado, V.; de Castro, A.B.; Vicente, B.; García, M.I.G.; Bellido, J.L.M.; Cordero-Sánchez, M. Relevance of eosinophilia and hyper-IgE in immigrant children. Medicine 2014, 93, e46.
- Lillywhite, J.; Bundy, D.; Didier, J.; Cooper, E.; Bianco, A. Humoral immune responses in human infection with the whipworm Trichuris trichiura. Parasite Immunol. 1991, 13, 491–507.
- Schachter, J.; de Oliveira, D.A.; da Silva, C.M.; de Barros Alencar, A.C.M.; Duarte, M.; da Silva, M.M.P.; Ignácio, A.C.d.P.R.; Lopes-Torres, E.J. Whipworm Infection Promotes Bacterial Invasion, Intestinal Microbiota Imbalance, and Cellular Immunomodulation. Infect. Immun. 2020, 88.
- Else, K.; Entwistle, G.; Grencis, R. Correlations between worm burden and markers of Th1 and Th2 cell subset induction in an inbred strain of mouse infected with Trichuris muris. Parasite Immunol. 1993, 15, 595–600.
- Blackwell, N.M.; Else, K.J. B cells and antibodies are required for resistance to the parasitic gastrointestinal nematode Trichuris muris. Infect. Immun. 2001, 69, 3860–3868.
- Houlden, A.; Hayes, K.S.; Bancroft, A.J.; Worthington, J.J.; Wang, P.; Grencis, R.K.; Roberts, I.S. Chronic Trichuris muris infection in C57BL/6 mice causes significant changes in host microbiota and metabolome: Effects reversed by pathogen clearance. PLoS ONE 2015, 10, e0125945.
- Holm, J.B.; Sorobetea, D.; Kiilerich, P.; Ramayo-Caldas, Y.; Estellé, J.; Ma, T.; Madsen, L.; Kristiansen, K.; Svensson-Frej, M. Chronic Trichuris muris infection decreases diversity of the intestinal microbiota and concomitantly increases the abundance of lactobacilli. PLoS ONE 2015, 10, e0125495.
- Taylor, M.D.; Betts, C.J.; Else, K.J. Peripheral cytokine responses to Trichuris muris reflect those occurring locally at the site of infection. Infect. Immun. 2000, 68, 1815–1819.
- Bancroft, A.J.; Else, K.J.; Humphreys, N.E.; Grencis, R.K. The effect of challenge and trickle Trichuris muris infections on the polarisation of the immune response. Int. J. Parasitol. 2001, 31, 1627–1637.
- Glover, M.; Colombo, S.A.; Thornton, D.J.; Grencis, R.K. Trickle infection and immunity to Trichuris muris. PLoS Pathog. 2019, 15, e1007926.
- Kringel, H.; Iburg, T.; Dawson, H.; Aasted, B.; Roepstorff, A. A time course study of immunological responses in Trichuris suis infected pigs demonstrates induction of a local type 2 response associated with worm burden. Int. J. Parasitol. 2006, 36, 915–924.
- Coackley, G.; Harris, N.L. The Intestinal Epithelium at the Forefront of Host–Helminth Interactions. Trends Parasitol. 2020, 36, 761–772.
- Ogra, P.L.; Mestecky, J.; Lamm, M.E.; Strober, W.; McGhee, J.R.; Bienenstock, J. Handbook of Mucosal Immunology; Academic Press: Cambridge, MA, USA, 2012.
- Klementowicz, J.E.; Travis, M.A.; Grencis, R.K. Trichuris muris: A model of gastrointestinal parasite infection. In Seminars in Immunopathology; Springer: Berlin/Heidelberg, Germany, 2012; pp. 815–828.
- Cortés, A.; Muñoz-Antoli, C.; Toledo, R.; Esteban, J.G. Th2 and Th1 Responses: Clear and Hidden Sides of Immunity Against Intestinal Helminths. Trends Parasitol. 2017, 33, 1–16.
- Deschoolmeester, M.L.; Manku, H.; Else, K.J. The innate immune responses of colonic epithelial cells to Trichuris muris are similar in mouse strains that develop a type 1 or type 2 adaptive immune response. Infect. Immun. 2006, 74, 6280–6286.
- Bellaby, T.; Robinson, K.; Wakelin, D. Induction of differential T-helper-cell responses in mice infected with variants of the parasitic nematode Trichuris muris. Infect. Immun. 1996, 64, 791–795.
- Stadnyk, A.W.; Kearsey, J.A. Pattern of proinflammatory cytokine mRNA expression during Trichinella spiralis infection of the rat. Infect. Immun. 1996, 64, 5138–5143.
- Ludwig-Portugall, I.; Layland, L.E. TLRs, Treg, and B celss, an interplay of regulation during helminth infection. Front. Immunol. 2012, 3, 1–7.
- Sahputra, R.; Else, K.J.; Rückerl, D.; Couper, K.; Müller, W. The essential role played by B cells in supporting protective immunity against Trichuris muris infection is by controlling the Th1/Th2 balance in the mesenteric lymph nodes and depends on host genetic background. Front. Immunol. 2019, 10, 2842.
- Sanders, T.J.; McCarthy, N.E.; Giles, E.M.; Davidson, K.L.; Haltalli, M.L.; Hazell, S.; Lindsay, J.O.; Stagg, A.J. Increased production of retinoic acid by intestinal macrophages contributes to their inflammatory phenotype in patients with Crohn’s disease. Gastroenterology 2014, 146, 1278–1288.e1272.
- Fort, M.M.; Cheung, J.; Yen, D.; Li, J.; Zurawski, S.M.; Lo, S.; Menon, S.; Clifford, T.; Hunte, B.; Lesley, R. IL-25 induces IL-4, IL-5, and IL-13 and Th2-associated pathologies in vivo. Immunity 2001, 15, 985–995.
- Jang, S.; Morris, S.; Lukacs, N.W. TSLP promotes induction of Th2 differentiation but is not necessary during established allergen-induced pulmonary disease. PLoS ONE 2013, 8, e56433.
- Kitajima, M.; Lee, H.C.; Nakayama, T.; Ziegler, S.F. TSLP enhances the function of helper type 2 cells. Eur. J. Immunol. 2011, 41, 1862–1871.
- Murakami-Satsutani, N.; Ito, T.; Nakanishi, T.; Inagaki, N.; Tanaka, A.; Vien, P.T.X.; Kibata, K.; Inaba, M.; Nomura, S. IL-33 promotes the induction and maintenance of Th2 immune responses by enhancing the function of OX40 ligand. Allergol. Int. 2014, 63, 443–455.
- Marillier, R.G.; Michels, C.; Smith, E.M.; Fick, L.C.; Leeto, M.; Dewals, B.; Horsnell, W.G.; Brombacher, F. IL-4/IL-13 independent goblet cell hyperplasia in experimental helminth infections. BMC Immunol. 2008, 9, 1–9.
- Sharba, S.; Navabi, N.; Padra, M.; Persson, J.; Quintana-Hayashi, M.; Gustafsson, J.; Szeponik, L.; Venkatakrishnan, V.; Sjöling, Å.; Nilsson, S. Interleukin 4 induces rapid mucin transport, increases mucus thickness and quality and decreases colitis and Citrobacter rodentium in contact with epithelial cells. Virulence 2019, 10, 97–117.
- Motran, C.C.; Silvane, L.; Chiapello, L.S.; Theumer, M.G.; Ambrosio, L.F.; Volpini, X.; Celias, D.P.; Cervi, L. Helminth infections: Recognition and Modulation of the immune Response by innate immune Cells. Front. Immunol. 2018, 9, 664.
- Capron, A.; Dessaint, J.-P.; Haque, A.; Capron, M. Antibody-Dependent Cell-Mediated Cytotoxicity against Parasites. Immun. Concomitant Immun. Infect. Dis. 1982, 31, 234–267.
- Tang, H.; Ming, Z.; Liu, R.; Xiong, T.; Grevelding, C.G.; Dong, H.; Jiang, M. Development of adult worms and granulomatous pathology are collectively regulated by T-and B-cells in mice infected with Schistosoma japonicum. PLoS ONE 2013, 8, e54432.
- Matsuda, S.; Tani, Y.; Yamada, M.; Yoshimura, K.; Arizono, N. Type 2-biased expression of cytokine genes in lung granulomatous lesions induced by Nippostrongylus brasiliensis infection. Parasite Immunol. 2001, 23, 219–226.
- Behm, C.A.; Ovington, K.S. The Role of Eosinophils in Parasitic Helminth Infections: Insights from Genetically Modified Mice. Parasitol. Today 2000, 16, 202–209.
- Klion, A.D.; Nutman, T.B. The role of eosinophils in host defense against helminth parasites. J. Allergy Clin. Immunol. 2004, 113, 30–37.
- BETTS, C.J.; ELSE, K.J. Mast cells, eosinophils and antibody-mediated cellular cytotoxicity are not critical in resistance to Trichuris muris. Parasite Immunol. 1999, 21, 45–52.
- Yasuda, K.; Kuroda, E. Role of eosinophils in protective immunity against secondary nematode infections. Immunol. Med. 2019, 42, 148–155.
- Hurst, R.J.; Else, K.J. Trichuris muris research revisited: A journey through time. Parasitology 2013, 140, 1325–1339.
- Wakelin, D. Acquired immunity to Trichuris muris in the albino laboratory mouse. Parasitology 1967, 57, 515–524.
- Mirchandani, A.S.; Besnard, A.G.; Yip, E.; Scott, C.; Bain, C.C.; Cerovic, V.; Salmond, R.J.; Liew, F.Y. Type 2 innate lymphoid cells drive CD4+ Th2 cell responses. J. Immunol. 2014, 192, 2442–2448.
- Curry, A.; Else, K.; Jones, F.; Bancroft, A.; Grencis, R.; Dunne, D. Evidence that cytokine-mediated immune interactions induced by Schistosoma mansoni alter disease outcome in mice concurrently infected with Trichuris muris. J. Exp. Med. 1995, 181, 769–774.
- Tritten, L.; Tam, M.; Vargas, M.; Jardim, A.; Stevenson, M.M.; Keiser, J.; Geary, T.G. Excretory/secretory products from the gastrointestinal nematode Trichuris muris. Exp. Parasitol. 2017, 178, 30–36.
- Lightowlers, M.W.; Rickard, M.D. Excretory-secretory products helminth parasite: Effects on host immune responses. Parasitology 1988, 96, S123–S166.
- Hewitson, J.P.; Grainger, J.R.; Maizels, R.M. Helminth immunoregulation: The role of parasite secreted proteins in modulating host immunity. Mol. Biochem. Parasitol. 2009, 167, 1–11.
- Maizels, R.M.; Smits, H.H.; McSorley, H.J. Modulation of Host Immunity by Helminths: The Expanding Repertoire of Parasite Effector Molecules. Immunity 2018, 49, 801–818.
- Briggs, N.; Wei, J.; Versteeg, L.; Zhan, B.; Keegan, B.; Damania, A.; Pollet, J.; Hayes, K.S.; Beaumier, C.; Seid, C.A. Trichuris muris whey acidic protein induces type 2 protective immunity against whipworm. PLoS Pathog. 2018, 14, e1007273.
- Summan, A.; Nejsum, P.; Williams, A.R. Modulation of human dendritic cell activity by Giardia and helminth antigens. Parasite Immunol. 2018, 40, e12525.
- Ottow, M.; Klaver, E.; van der Pouw Kraan, T.; Heijnen, P.; Laan, L.; Kringel, H.; Vogel, D.; Dijkstra, C.; Kooij, G.; Van Die, I. The helminth Trichuris suis suppresses TLR4-induced inflammatory responses in human macrophages. Genes Immun. 2014, 15, 477–486.
- Klaver, E.; van der Pouw Kraan, T.; Laan, L.; Kringel, H.; Cummings, R.; Bouma, G.; Kraal, G.; Van Die, I. Trichuris suis soluble products induce Rab7b expression and limit TLR4 responses in human dendritic cells. Genes Immun. 2015, 16, 378–387.
- Laan, L.C.; Williams, A.R.; Stavenhagen, K.; Giera, M.; Kooij, G.; Vlasakov, I.; Kalay, H.; Kringel, H.; Nejsum, P.; Thamsborg, S.M. The whipworm (Trichuris suis) secretes prostaglandin E2 to suppress proinflammatory properties in human dendritic cells. FASEB J. 2017, 31, 719–731.
- Harnett, W. Secretory products of helminth parasites as immunomodulators. Mol. Biochem. Parasitol. 2014, 195, 130–136.
- Rehwinkel, J.; Gack, M.U. RIG-I-like receptors: Their regulation and roles in RNA sensing. Nat. Rev. Immunol. 2020, 13, 1–15.
- Duque-Correa, M.A.; Schreiber, F.; Rodgers, F.H.; Goulding, D.; Forrest, S.; White, R.; Buck, A.; Grencis, R.K.; Berriman, M. Development of caecaloids to study host-pathogen interactions: New insights into immunoregulatory functions of Trichuris muris extracellular vesicles in the caecum. Int. J. Parasitol. 2020, 50, 707–718.
- Krivan, S.; Kapelouzou, A.; Vagio, S.; Tsilimigras, D.I.; Katsimpoulas, M.; Moris, D.; Aravanis, C.V.; Demesticha, T.D.; Schizas, D.; Mavroidis, M.; et al. Increased expression of Toll-like receptors 2, 3, 4 and 7 mRNA in the kidney and intestine of a septic mouse model. Sci. Rep. 2019, 9, 1–9.
- Wiria, A.E.; Hamid, F.; Wammes, L.J.; Prasetyani, M.A.; Dekkers, O.M.; May, L.; Kaisar, M.M.; Verweij, J.J.; Guigas, B.; Partono, F. Infection with soil-transmitted helminths is associated with increased insulin sensitivity. PLoS ONE 2015, 10, e0127746.
- Rocha, D.; Caldas, A.; Oliveira, L.; Bressan, J.; Hermsdorff, H. Saturated fatty acids trigger TLR4-mediated inflammatory response. Atherosclerosis 2016, 244, 211–215.
- Qi, X.; Qin, L.; Du, R.; Chen, Y.; Lei, M.; Deng, M.; Wang, J. Lipopolysaccharide upregulated intestinal epithelial cell expression of Fn14 and activation of Fn14 signaling amplify intestinal TLR4-mediated inflammation. Front. Cell. Infect. Microbiol. 2017, 7, 315.
- Kooij, G.; Braster, R.; Koning, J.J.; Laan, L.C.; van Vliet, S.J.; Los, T.; Eveleens, A.M.; van der Pol, S.M.; Förster-Waldl, E.; Boztug, K. Trichuris suis induces human non-classical patrolling monocytes via the mannose receptor and PKC: Implications for multiple sclerosis. Acta Neuropathol. Commun. 2015, 3, 1–14.
- Klaver, E.J.; Kuijk, L.M.; Laan, L.C.; Kringel, H.; van Vliet, S.J.; Bouma, G.; Cummings, R.D.; Kraal, G.; van Die, I. Trichuris suis-induced modulation of human dendritic cell function is glycan-mediated. Int. J. Parasitol. 2013, 43, 191–200.
- Bono, M.a.R.; Elgueta, R.l.; Sauma, D.; Pino, K.; Osorio, F.; Michea, P.; Fierro, A.; Rosemblatt, M. The essential role of chemokines in the selective regulation of lymphocyte homing. Cytokine Growth Factor Rev. 2007, 18, 33–43.
- Laufer, J.M.; Legler, D.F. Beyond migration—Chemokines in lymphocyte priming, differentiation, and modulating effector functions. J. Leucoc. Biol. 2018, 104, 301–312.
- Yang, L.; Yan, Y. Protein kinases are potential targets to treat inflammatory bowel disease. World J. Gastrointest. Pharmacol. Ther. 2014, 5, 209–217.
- Brown, J.F.; Chang, Q.; Soper, B.D.; Tepperman, B.L. Protein kinase C mediates experimental colitis in the rat. Am. J. Physiol. 1999, 276, G583–G590.
- Alhallaf, R.; Agha, Z.; Miller, C.M.; Robertson, A.A.; Sotillo, J.; Croese, J.; Cooper, M.A.; Masters, S.L.; Kupz, A.; Smith, N.C. The NLRP3 inflammasome suppresses protective immunity to gastrointestinal helminth infection. Cell Rep. 2018, 23, 1085–1098.
- Zaiss, M.M.; Maslowski, K.M.; Mosconi, I.; Guenat, N.; Marsland, B.J.; Harris, N.L. IL-1β suppresses innate IL-25 and IL-33 production and maintains helminth chronicity. PLoS Pathog 2013, 9, e1003531.
- Chenery, A.L.; Alhallaf, R.; Agha, Z.; Ajendra, J.; Parkinson, J.E.; Cooper, M.M.; Chan, B.H.; Eichenberger, R.M.; Dent, L.A.; Robertson, A.A. Inflammasome-independent role for NLRP3 in controlling innate antihelminth immunity and tissue repair in the lung. J. Immunol. 2019, 203, 2724–2734.
- Duque-Correa, M.A.; Karp, N.A.; McCarthy, C.; Forman, S.; Goulding, D.; Sankaranarayanan, G.; Jenkins, T.P.; Reid, A.J.; Cambridge, E.L.; Reviriego, C.B. Exclusive dependence of IL-10Rα signalling on intestinal microbiota homeostasis and control of whipworm infection. PLoS Pathog. 2019, 15, e1007265.
- Mansfield, L.; Urban Jr, J. The pathogenesis of necrotic proliferative colitis in swine is linked to whipworm induced suppression of mucosal immunity to resident bacteria. Vet. Immunol. Immunopathol. 1996, 50, 1–17.
- Montacute, R.; Foley, K.; Forman, R.; Else, K.J.; Cruickshank, S.M.; Allan, S.M. Enhanced susceptibility of triple transgenic Alzheimer’s disease (3xTg-AD) mice to acute infection. J. Neuroinflammation 2017, 14, 50.
- Kühn, R.; Löhler, J.; Rennick, D.; Rajewsky, K.; Müller, W. Interleukin-10-deficient mice develop chronic enterocolitis. Cell 1993, 75, 263–274.
- Ebner, F.; Hepworth, M.; Rausch, S.; Janek, K.; Niewienda, A.; Kühl, A.; Henklein, P.; Lucius, R.; Hamelmann, E.; Hartmann, S. Therapeutic potential of larval excretory/secretory proteins of the pig whipworm Trichuris suis in allergic disease. Allergy 2014, 69, 1489–1497.
- Bancroft, A.J.; McKenzie, A.N.; Grencis, R.K. A critical role for IL-13 in resistance to intestinal nematode infection. J. Immunol. 1998, 160, 3453–3461.
- Bancroft, A.J.; Levy, C.W.; Jowitt, T.A.; Hayes, K.S.; Thompson, S.; Mckenzie, E.A.; Ball, M.D.; Dubaissi, E.; France, A.P.; Bellina, B. The major secreted protein of the whipworm parasite tethers to matrix and inhibits interleukin-13 function. Nat. Commun. 2019, 10, 1–11.
