This review outlines the current status and next steps for the development and use of the AOP framework in decision making regarding the safety of MNs. Opportunities and challenges toward the advancement and adoption of AOPs as part of an integrated approach to testing and assessment (IATA) of MNs are identified and specific actions proposed to advance the development, use and acceptance of the AOP framework and associated testing strategies for MN risk assessment and decision making. The intent of this review is to reflect the views of a diversity of stakeholders including experts, researchers, policymakers, regulators, risk assessors and industry representatives on the current status, needs and requirements to facilitate future use of AOPs in MN risk assessment. It incorporates the views and feedback of experts that participated in two workshops hosted as part of an Organization for Economic Cooperation and Development (OECD) Working Party on Manufactured Nanomaterials (WPMN) project titled, “Advancing AOP Development for Nanomaterial Risk Assessment and Categorization” as well as the position of several EU-funded nanosafety research consortia.
- Adverse Outcome Pathway
- Nanomaterials
- Risk Assessment
1. Overview of the AOP Framework and Current Status
1.1 Overview
AOPs clarify relationships across biological levels of organization (including molecular, subcellular, cellular, tissue, organ, organism and whole populations), using cause-and-effect relationships to connect molecular initiating events (MIE) to adverse outcomes (AO). AOs are negative biological consequences resulting from chemical exposure; they are typically measured at higher levels of biological organization and are important for regulatory decision making, e.g., concerning issues of human health (organ or organism) or environmental endpoints (organism or populations). The pathways connecting MIEs and AOs are defined by key events (KEs) which represent measurable biological changes, and key event relationships (KERs), i.e., the directed, predictive relationships among those KEs. AOPs are the units of development for the AOP framework, and represent a single, nonbranching sequence of KEs, linked by KERs, connecting a single MIE to a single AO [1].
Figure 1 gives an example of a generalized AOP and its components (Panel A), and shows how AOPs can form interlinked networks based on overlapping MIEs, KEs and AOs (Panel B) that represent the complex biology underlying disease processes [2]. Bioassays targeting the MIE and KEs in an AOP were developed, characterized and used as endpoints as part of an IATA. The AOP framework defines this entire conceptual approach that assembles and organizes mechanistic knowledge to communicate causal links between biological perturbations and adverse health outcomes which are meaningful to chemical risk assessments and regulatory decision making [3][4].
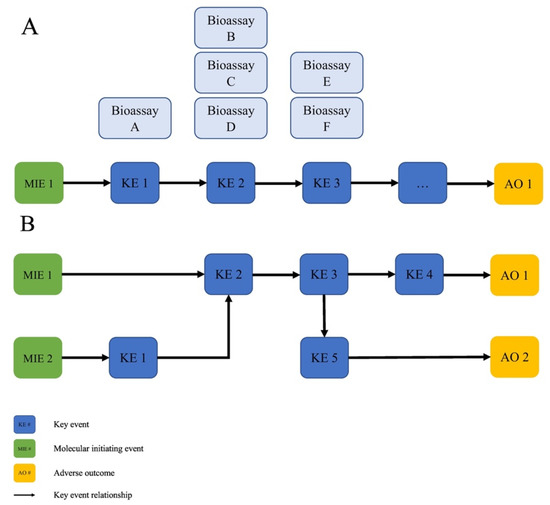
Figure 1. (A) Generalized AOP showing the relationship between MIE, KEs and AOs, and the KERs that connect them. Bioassays targeting the MIE and KEs in an AOP are characterized or developed as part of an IATA. (B) AOPs can form interlinked networks based on overlapping MIEs, KEs and AOs that better capture the complex biology of disease processes. (From [2]).
To help accelerate AOP development, strategies, principles and best practices have been established to help scientists, regulators and decision makers understand and contribute to the AOP framework. In 2014, Villeneuve et al. proposed a set of five core principles to guide AOP development [5]; these are highlighted in Box 1.
Box 1. Core Tenets of AOPs [5].
-
AOPs are not chemical-specific. Specificity limits the predictive utility of AOPs for new substances.
-
AOPs are designed with modular units. These components should be reusable to enhance flexibility, and they should be designed to accommodate differing levels of detail based on evidence.
-
AOPs are a unit of development. An individual AOP is defined as a single, nonbranching sequence of KEs, linked by KERs, connecting a single MIE to a single AO. This structure reduces the complexity, and is a practical unit for development and evaluation.
-
AOPs form networks. Multiple AOPs, sharing one or more common KE or KER, form networks that more realistically represent the complexity of biological systems needed to make accurate biological predictions of adverse toxicological outcomes.
-
AOPs should be continuously updated. New research should be used to inform and refine existing AOPs.
1.2 Current Status
Tremendous progress in AOP research and development has occurred since AOPs were first described in 2010 [4]. A survey of papers published annually on the topic from 2010–2019 indicated exponential growth, with almost 200 publications expected in 2020 based on a PubMed literature search. This growth suggests that the AOP concept is gaining widespread traction and acceptance in the academic community (Figure 2 [6]).
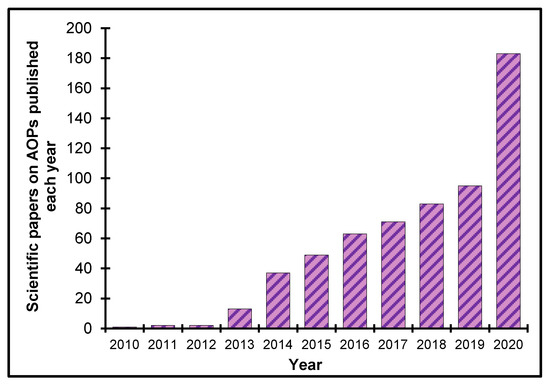
Figure 2. Scientific papers on AOPs published annually, 2010–2019. PubMed was searched for published papers containing the text words “adverse outcome pathway” on 22 May, 2020. The result for 2020 is estimated given the number of publications that have been published per month.
A number of efforts worldwide are contributing to AOP development; among these, the OECD is spearheading one of the largest. The OECD AOP Development Programme was started in 2012, and is overseen by the Extended Advisory Group on Molecular Screening and Toxicogenomics (EAGMST). Its goal is to develop, review and officially endorse AOPs. AOP development is based on voluntary contributions from member countries and stakeholders. It involves an internal review within EAGMST to ensure compliance with AOP principles, as well as an external review by subject matter experts to assess the scientific merit of the proposed pathway. There are currently nine OECD-endorsed AOPs, seven that have received approved status, fourteen under review (indicating that they are at a late stage of the endorsement process), and twenty-four proposals under active development.
The OECD, together with contributions from the U.S. EPA, the European Commission’s Joint Research Centre and the U.S. Army Engineer Research and Development Center has also led the development of the AOP Knowledge Base (AOP-KB). Launched in 2014, this web-based tool consists of five modules to enable and promote the development and application of AOPs: the e.AOP.Portal, the AOP-Wiki, the Effectopedia, the AOP Xplorer and the Intermediate Effects Database [1]. The AOP-Wiki serves as the primary repository for qualitative AOPs developed (including those endorsed by the OECD) or under development. It is intended to foster collaboration among various stakeholders contributing to AOP development following the standard OECD principles for developing and assessing AOPs [7]. The AOP Wiki currently contains more than 200 AOPs, including more than 2000 defined KEs (Figure 3).
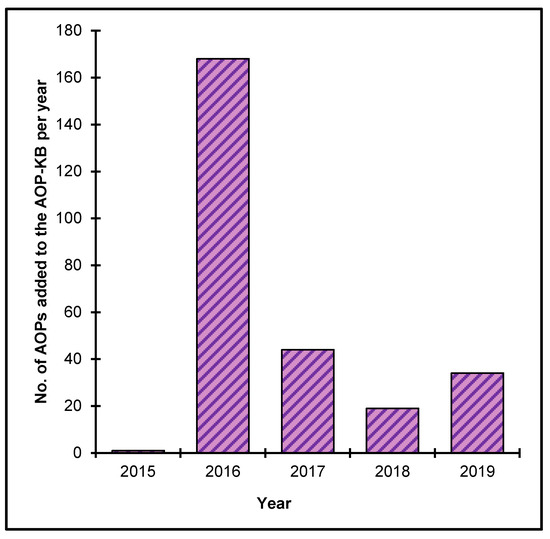
Figure 3. AOPs created in the AOP-KB from 2015–2019 (until May 22, 2020).
In addition to these efforts, various stakeholders have supported projects aimed at evaluating and promoting the development of AOPs for MNs. The EU, through its H2020 initiative, has supported several projects focused on the development and application of AOPs for MNs; these include SmartNanoTox (Smart Tools for Gauging Nano Hazards) and PATROLS (Physiologically Anchored Tools for Realistic nanOmateriaL hazard aSsessment) [8][5]. Since 2016, the OECD Working Party on Manufactured Nanomaterials (WPMN) has included in its program the project Advancing Adverse Outcome Pathway (AOP) Development for Nanomaterial Risk Assessment and Categorisation [NanoAOP Project; ENV/CHEM/NANO(2017)5].
2. Potential Applications of AOPs
Progress in AOP development has given rise to a range of potential applications. In chemical risk assessment, the AOP framework is intended to guide and develop IATA. IATA is an approach to characterizing the hazard of chemicals that integrates analyses of existing information with the generation of new information through targeted testing strategies [1]. From a risk assessment perspective, the AOP framework provides:
-
A structured framework to evaluate existing information available for a chemical of interest; potential sources include in chemico, in silico, in vitro, ex vivo, in vivo and ‘omics’ data;
-
A way to identify data gaps and efficiently generate missing information to increase confidence in decision making and assessments of risk;
-
A framework to apply an iterative approach until sufficient information is gathered for decision making.
Within this context, the AOP framework has several specific applications and benefits to improve risk assessment and decision making for chemicals; these are summarized in Table 2 [1][6][9]. Of particular interest, the AOP concept offers a systematic, mechanistic framework to develop, assess, use and interpret alternative testing strategies for chemical risk assessment and decision-making, thereby reducing reliance on new substance testing. Applications in chemical safety assessment are especially useful to regulatory toxicologists, risk assessors and risk managers, as well as to industry stakeholders responsible for product stewardship and compliance with regulatory requirements [3].
Table 2. Potential applications and benefits of the AOP framework in chemical risk assessment.
| 1. Evaluation of existing information |
|
|
|
| 2. Identification of data gaps and generation of new data |
|
|
|
|
|
| 3. Iterative decision making |
|
|
One area of proposed application of AOPs is MN risk assessment and decision-making. Because of their particulate aspects, MNs do not necessarily display classical dose-response relationships, and their toxicity is not always predictable from chemical substance models. Moreover, the biological effects of small changes in their physical and chemical makeup are not easily predicted with today’s risk assessment toolbox and models. The benefits of a framework that is better suited to assessing the impacts of such MN modifications can apply to regulators, researchers and product developers. One of the essential components of the mechanistic representation of MN-induced outcomes provided by AOPs is the MIE, that triggers the pathway, and KEs, that can be directly steered by MN interactions with biomolecules. The molecular level description of the initiating event and the underlying physical interaction make it possible to relate the physicochemical properties of the MN to the probability of a MIE via quantitative structure– or property–activity relationships (QSAR/QSPR). These relationships help to identify the MN properties of concern, and thus, enable the consequent grouping of the MNs based on their ability to induce a MIE/AOP. The main benefit of these relationships is the replacement of biological testing with in silico or in vitro screening.
While not the focus of this discussion, the AOP framework has other potential applications that extend beyond risk assessment [3]. AOPs provide utility in product discovery and development, especially in the pharmaceutical and agrochemical industries; here, they can support preclinical safety assessments to identify compounds, early in their development, which are potentially harmful to human, animal or environmental health. In medicine and health, clinicians and researchers can use AOP knowledge to understand disease pathways across multiple biological levels, informing prevention, diagnosis and treatment efforts. Other stakeholders that may benefit from AOP applications include academics (who may benefit from a unifying framework to increase the real-world impact of their work) and nongovernmental organizations (NGOs), including animal welfare and environmental NGOs. As mentioned, several focus areas in the proposed Horizon Europe Strategic Plan [10] could benefit from advancements and the adoption of AOPs in decision making.
3. Recent Progress with AOPs for Nanosafety
Projects worldwide are focusing on meeting the challenges of AOP development and promoting the use of AOPs for decision making. Below, projects contributing to the development, use and adoption of AOPs for MNs are highlighted and summarized.
3.1 Progress in AOP Development for Manufactured Nanomaterials
Inhalation exposure to powdered MNs, especially during manufacture and handling, is a highly relevant risk scenario [11]. Some studies have documented significant AOs such as fibrosis and cancer following exposure to certain forms of MNs (e.g., carbon nanotubes [12]); however, considerable uncertainty about the physical and chemical properties influencing these outcomes remains. In vivo inhalation experiments to characterize the hazard of MNs are challenging and resource-intensive. Of all standardized toxicity testing, inhalation studies are some of the most expensive to commission and complete. To simulate realistic exposure, animals have to be exposed to low dose aerosols over long periods of time (up to 2 years). Generating MN aerosols can be difficult (e.g., many are viscosity modifiers), and often, alternative exposure methods such as pharyngeal aspiration or intratracheal instillation must be used that deliver high bolus doses over short time frames [11]. The AOP framework, as part of an IATA, offers a more time- and cost-efficient alternative approach to assessing the potential risks from inhaled MNs; however, development and verification of the pathway is necessary before such an approach can be used for decision making.
The SmartNanoTox project is using results from in vivo, in vitro and in silico research to develop AOPs for adverse pulmonary effects following MN exposure. The effort is using representative sets of MNs to identify critical KEs and KERs to construct AOPs and relate them to interactions at the bio-nano interface [8]. Using the data generated from various KEs and KERs along the AOP, SmartNanoTox aims to develop quantitative structure–activity relationships (QSARs) to enable predictions of whether a MN can trigger an AOP and lead to an AO, and to enable the grouping, categorization and read-across of MNs for these endpoints [13][14]. The PATROLS project has collated and generated data to establish AOPs for both lung and liver inflammation, fibrosis and cancer AOs following MN exposure. The purpose of this activity was, based on our understanding of AOPs, to identify biomarkers which are suitable for long-term MN exposure effects that could be applied in short-term in vitro assays to develop more targeted nonanimal hazard testing strategies with potentially higher predictive value. NanoCommons has implemented a tool for predicting occurrences of MIEs using differentially expressed genes/proteins from high-throughput experiments to calculate a prioritized list of MIEs with identified biological processes, while NanoSolveIT is developing the concept of a nanomaterial fingerprint that captures the key physicochemical, biomolecular and interactional features of a MN that are predictive of its toxicity.
Other related efforts are developing data, methods and tools which are useful for AOP development [15][16][17]. For example, the EU’s NanoSolveIT project is developing: (i) innovative modelling techniques and tools for nanoinformatics; (ii) an IATA to identify the specific characteristics of MNs that are responsible for adverse effects on human health or the environment; and (iii) in silico methods, models and tools which are useful for AOP development. The grouping strategies, data and methods in the H2020 projects GRACIOUS and NanoReg 2 are being incorporated into IATA [18]. Similarly, NanoCommons aims to deliver a nanoinformatics research infrastructure including a database to facilitate the reuse of existing nanosafety data, and in silico tools for analyses and predictions of MN impacts, including tools to predict MIEs and AOPs. The Data and Knowledge on Nanomaterial (DaNa) project has compiled data on the applications of MNs and the current state of knowledge. The EU Cluster of Systems of Metadata for Official Statistics (COSMOS) is identifying common sets of metadata objects with standard definitions and methods to build better metadata repositories. NanoCommons, in collaboration with the National Cancer Institute working group on nanoinformatics, is supporting the development of metadata standards for nanosafety research, as described in another paper in this special issue [19]. All of these efforts can make significant contributions to AOP development.
3.2 Progress in the Application and Use of AOPs
One of the first AOPs officially endorsed by the OECD was the skin sensitization AOP (AOP No. 40 in the AOP-Wiki) [6]. Since its endorsement, mechanistic knowledge gained on skin sensitization has been used to develop and validate three standardized in vitro tests targeting the KEs in the AOP. These test guidelines have been published and are now accepted for regulatory use as a viable alternative to traditional animal testing for skin sensitization. The skin sensitization AOP is a success story, demonstrating how AOP development can help identify, promote development of, and validate alternative testing strategies for chemical risk assessment without the use of animals.
While significant progress has been made in AOP development, application and use over the last decade, for endpoints beyond skin sensitization, there is still a significant distance between the current reliance on, reduction of, and replacement of animal testing with nonanimal approaches for risk assessment and regulatory decision making. The OECD WPMN NanoAOP project is contributing to both the development and application of AOPs for MN risk assessment. The goal of the project was to develop a methodology to use existing nanotoxicology literature to support MN-relevant AOP development. While not developing an AOP, a case study outlines how the literature can be mined to identify and develop specific KEs to support AOP development.
As part of its outcomes, the OECD WPMN NanoAOP project convened two workshops to gain expert feedback on the current status, use and future needs for AOPs which are relevant to MNs in support of risk assessment. The consensus from experts is that currently, the primary applications of AOPs are (i) to support hazard identification; (ii) grouping, categorization and read-across; (iii) ranking and prioritizing MNs; (iv) the identification of novel biomarkers for alternative test method development; (v) for product development as part of a safer manufacturing approach; and (vi) together with ‘omics’ strategies, AOPs can be used to propose testing that could be predictive of AOs. The ultimate goal is to use AOPs as the basis for regulatory decision making for MNs; however, several challenges have been identified by experts that need to be overcome to advance the future use of AOPs for MN risk assessment. Experts suggest that to ensure the relevance of AOP frameworks for regulatory decision making, development should proceed by first choosing an AO relevant for regulators, and then developing the pathway working backwards toward an MIE.
Several projects are contributing directly to the application and use of AOPs for MN risk assessment. As described, SmartNanoTox and PATROLS are developing in vitro models, methods and computational tools for MN hazard assessment targeting the KEs in developed AOPs. This is an important step to ensure that identified KEs in an AOP can be assessed with validated methods and tests, and thus, to support their inclusion in an IATA for MN risk assessment. SmartNanoTox is constructing simplified in vitro or in silico tests for the AOPs developed in their project, targeting identified MIEs and KEs for adverse respiratory outcomes from MN inhalation. PATROLS is focused on developing mechanism-based, nonanimal methods, models and computational tools for MN hazard characterization, targeting the KEs in established AOPs. This includes in silico hazard testing systems, in vitro human tissue models, ecotoxicology models and methods for MN characterization in biological systems.
4. Challenges in the Development and Application of AOPs
Substantial progress has been made toward the development and application of AOPs, but a number of challenges remain before their full potential can be realized. A global horizon scanning exercise to identify current challenges toward the regulatory adoption of the AOP framework is one of the largest efforts to advance AOP development and application [20]. The key findings from that effort are summarized below, as are expert insights from the workshops held as part of the OECD NanoAOP project. Limitations identified through these efforts include challenges specific to MNs, as well as outstanding research and technical needs hindering AOP development more generally.
5. Summary
The adoption of AOPs for advanced materials and MNs is hindered by the lack of accepted methods for toxicity evaluation and over-reliance on resource-intensive animal testing. There is a need and an opportunity to develop smarter approaches for screening and prioritizing novel nanoscale materials for decision-making. The AOP framework is a promising and toxicologically realistic approach to help address several current challenges of chemical and nano-specific safety assessments. Significant progress has been made in AOP development, application and use over the last decade including methods, tools and assessment approaches. However, we are still a long way from reliance on—and the replacement of animal testing with—nonanimal approaches. Several challenges remain and must be addressed to realize the full potential of AOPs. Projects worldwide are focusing on meeting these challenges and advancing the use of AOPs for decision-making. International cross-disciplinary analyses and deliberation will improve the adoption and acceptance of the AOP framework, including for MNs.
This entry is adapted from the peer-reviewed paper 10.3390/nano10061229
References
- Nathalie Delrue; Magdalini Sachana; Yuki Sakuratani; Anne Gourmelon; Eeva Leinala; Robert Diderich; The Adverse Outcome Pathway Concept: A Basis for Developing Regulatory Decision-making Tools. Alternatives to Laboratory Animals 2016, 44, 417-429, 10.1177/026119291604400504.
- Sabina Halappanavar; James D. Ede; Jo Anne Shatkin; Harald F. Krug; A systematic process for identifying key events for advancing the development of nanomaterial relevant adverse outcome pathways. NanoImpact 2019, 15, 100178, 10.1016/j.impact.2019.100178.
- Annamaria Carusi; Mark R. Davies; Giovanni De Grandis; Beate I. Escher; Geoff Hodges; Kenneth M.Y. Leung; Maurice Whelan; Catherine Willett; Gerald T. Ankley; Harvesting the promise of AOPs: An assessment and recommendations. Science of The Total Environment 2018, 628–629, 1542-1556, 10.1016/j.scitotenv.2018.02.015.
- Gerald T. Ankley; Richard S. Bennett; Russell J. Erickson; Dale J. Hoff; Michael W. Hornung; Rodney D. Johnson; David R. Mount; John W. Nichols; Christine L. Russom; Patricia K. Schmieder; et al. Adverse outcome pathways: A conceptual framework to support ecotoxicology research and risk assessment. Environmental Toxicology and Chemistry 2010, 29, 730-741, 10.1002/etc.34.
- Daniel L Villeneuve; Ug Crump; Natàlia Garcia-Reyero; Markus Hecker; Thomas H. Hutchinson; Carlie A. Lalone; Brigitte Landesmann; Teresa Lettieri; Sharon Munn; Malgorzata Nepelska; et al. Adverse Outcome Pathway (AOP) Development I: Strategies and Principles. Toxicological Sciences 2014, 142, 312-320, 10.1093/toxsci/kfu199.
- Langley, G. Adverse Outcome Pathways: Will they Deliver a Superior Alternative to Animal Testing? Lush Prize: 2017. Available online: https://lushprize.org/wp/wp-content/uploads/Lush-Prize-AOP-paper-for-WC10-final.pdf (accessed on 8 September 2019).
- Organisation for Economic Cooperation and Development. Revised Guidance Document on Developing and Assessing Adverse Outcome Pathways; Series on Testing and Assessment; OECD: Paris, France, 2017.
- Sabina Halappanavar; Sybille Van Den Brule; Penny Nymark; Laurent Gaté; Carole Seidel; Sarah Valentino; Vadim Zhernovkov; Pernille Høgh Danielsen; Andrea De Vizcaya; Henrik Wolff; et al. Adverse outcome pathways as a tool for the design of testing strategies to support the safety assessment of emerging advanced materials at the nanoscale. Particle and Fibre Toxicology 2020, 17, 1-24, 10.1186/s12989-020-00344-4.
- Fadeel, B.; Farcas, L.; Hardy, B.; Vazquez-Campos, S.; Hristozov, D.; Marcomini, A.; Lynch, I.; Valsami-Jones, E.; Alenius, H.; Savolainen, K. Advanced tools for the safety assessment of nanomaterals. Nat. Nanotechnol. 2018, 13, 537–543.
- Horizon Europe. Orientations towards the First Strategic Plan Implementing the Research and Innovation Framework Programme Horizon Europe. 2019. Available online: https://clepa.eu/wp-content/uploads/2019/07/Horizon-Europe-Strategic-Planning-Summer-2019 (accessed on 8 September 2019).
- Ede, J.D.; Ong, K.J.; Goergen, M.; Rudie, A.; Pomeroy-Carter, C.A.; Shatkin, J.A. Risk analysis of cellulose nanomaterails by inhalation: Current state of science. Nanomaterials 2019, 9, 337.
- Tatsuya Kasai; Yumi Umeda; Makoto Ohnishi; Takashi Mine; Hitomi Kondo; Tetsuya Takeuchi; Michiharu Matsumoto; Shoji Fukushima; Lung carcinogenicity of inhaled multi-walled carbon nanotube in rats.. Particle and Fibre Toxicology 2016, 13, 53, 10.1186/s12989-016-0164-2.
- Hadrup, N.; Zhernovkov, V.; Jacobsen, N.R.; Voss, C.; Strunz, M.; Ansari, M.; Schiller, H.B.; Halappanavar, S.; Poulsen, S.S.; Kholodenko, B.; et al. Acute Phase Response as a Biological Mechanism-of-Action of (Nano)particle-Induced Cardiovascular Disease. Small 2020, 16, e1907476.
- Danielsen, P.H.; Knudsen, K.B.; Štrancar, J.; Umek, P.; Koklič, T.; Garvas, M.; Vanhala, E.; Savukoski, S.; Ding, Y.; Madsen, A.M.; et al. Effects of physicochemical properties of TiO2 nanomaterials for pulmonary inflammation, acute phase response and alveolar proteinosis in intratracheally exposed mice. Toxicol. Appl. Pharmacol. 2020, 386, 114830.
- Rahman, L.; Williams, A.; Gelda, K.; Nikota, J.; Wu, D.; Vogel, U.; Halappanavar, S. 21st century tools for nanotoxicology: Transcriptomic biomarker panel and precision-cut lung slice organ mimic system for the assessment of nanomaterial-induced lung fibrosis. Small 2000, 29, e2000272.
- Gaté, L.; Knudsen, K.B.; Seidel, C.; Berthing, T.; Chézeau, L.; Jacobsen, N.R.; Valentino, S.; Wallin, H.; Bau, S.; Wolff, H.; et al. Pulmonary toxicity of two different multi-walled carbon nanotubes in rat: Comparison between intratracheal instillation and inhalation exposure. Toxicol. Appl. Pharmacol. 2019, 375, 17–31.
- Yang, L.; Gradl, R.; Dierolf, M.; Möller, W.; Kutschke, D.; Feuchtinger, A.; Hehn, L.; Donnelley, M.; Günther, B.; Achterhold, K.; et al. Multimodal Precision Imaging of Pulmonary Nanoparticle Delivery in Mice: Dynamics of Application, Spatial Distribution, and Dosimetry. Small 2019, 15, e1904112.
- A. Mech; Kirsten Rasmussen; P. Jantunen; L. Aicher; M. Alessandrelli; U. Bernauer; E. A. J. Bleeker; J. Bouillard; P. Di Prospero Fanghella; R. Draisci; et al. Insights into possibilities for grouping and read-across for nanomaterials in EU chemicals legislation. Nanotoxicology 2018, 13, 119-141, 10.1080/17435390.2018.1513092.
- Papadiamantis, A.G.; Klaessig, F.C.; Exner, T.E.; Hofer, S.; Hofstaetter, N.; Himly, M.; Williams, M.A.; Doganis, P.; Hoover, M.D.; Afantis, A. Metadata stewardship in nanosafety research: Community-driven organisation of metadata schemas to support FAIR nanoscience data. Nanomaterials 2020, in press.
- Carlie Lalone; Gerald T. Ankley; Scott E. Belanger; Michelle R Embry; Geoff Hodges; Dries Knapen; Sharon Munn; Edward J. Perkins; Murray Rudd; Daniel L Villeneuve; et al. Advancing the adverse outcome pathway framework-An international horizon scanning approach. Environmental Toxicology and Chemistry 2017, 36, 1411-1421, 10.1002/etc.3805.
