Wound healing involves a complex cascade of cellular, molecular, and biochemical responses and signaling processes. It consists of successive interrelated phases, the duration of which depends on a multitude of factors. Wound treatment is a major healthcare issue that can be resolved by the development of effective and affordable wound dressings based on natural materials and biologically active substances. The proper use of modern wound dressings can significantly accelerate wound healing with minimum scar mark. Sulfated polysaccharides from seaweeds, with their unique structures and biological properties, as well as with a high potential to be used in various wound treatment methods, now undoubtedly play a major role in innovative biotechnologies of modern natural interactive dressings.
- seaweed
- sulfated polysaccharides
- alginates
- fucoidans
- carrageenans
- ulvans
- wound dressing
- wounds
1. Introduction
2. Polysaccharides from Marine Algae Used in the Development of Wound Dressings
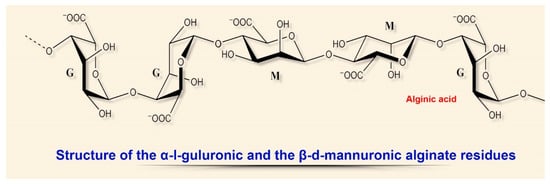
2.1. Alginates
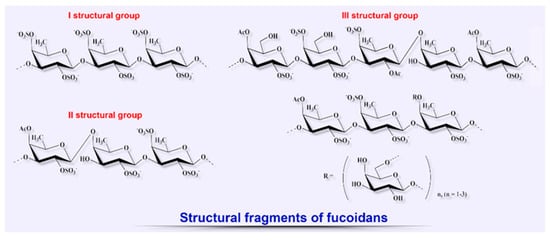
2.2. Fucoidans
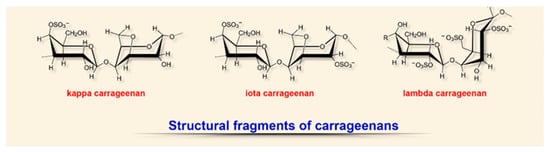
2.3. Carrageenans
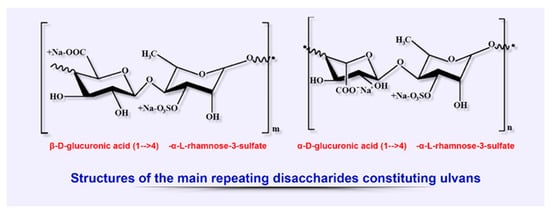
2.4. Ulvans
This entry is adapted from the peer-reviewed paper 10.3390/biomedicines8090301
References
- Basu, S.; Shukla, V. Complications of Wound Healing. In Measurements in Wound Healing; Mani, R., Romanelli, M., Shukla, V., Eds.; Springer: London, UK, 2012; Volume C, pp. 109–144.
- Bilal, M.; Iqbal, H.M.N. Marine Seaweed Polysaccharides-Based Engineered Cues for the Modern Biomedical Sector. Mar. Drugs 2019, 18, 7.
- Weller, C.D.; Team, V.; Sussman, G. First-Line Interactive Wound Dressing Update: A Comprehensive Review of the Evidence. Front. Pharmacol. 2020, 11.
- Dabiri, G.; Damstetter, E.; Phillips, T.J. Choosing a Wound Dressing Based on Common Wound Characteristics. Adv. Wound Care 2016, 5, 32–41.
- Bryant, R.; Nix, D. Acute and Chronic Wounds, 5th ed.; Mosby: Maryland Heights, MO, USA, 2016; 648p.
- Dhivya, S.; Padma, V.V.; Elango, S. Wound dressings—A review. Biomedicine (Taipei) 2015, 5, 22.
- Shah, J.B. The history of wound care. J. Am. Coll. Certif. Wound Spéc. 2011, 3, 65–66.
- Mouës, C.M.; Heule, F.; Legerstee, R.; Hovius, S.E. Five millennia of wound care products--what is new? A literature reviews. Ostomy Wound Manag. 2009, 55, 16–22.
- Broughton, G., 2nd; Janis, J.E.; Attinger, C.E. A brief history of wound care. Plast Reconstr Surg. 2006, 117 (Suppl. S7), 6S–11S.
- Aduba, D.C.; Yang, H. Polysaccharide Fabrication Platforms and Biocompatibility Assessment as Candidate Wound Dressing Materials. Bioengineering (Basel) 2017, 4, 1.
- Sweeney, I.R.; Miraftab, M.; Collyer, G. A critical review of modern and emerging absorbent dressings used to treat exuding wounds. Int. Wound J. 2012, 9, 601–612.
- Zhou, J.; Yao, D.; Qian, Z.; Hou, S.; Li, L.; Jenkins, T.; Fan, Y. Bacteria-responsive intelligent wound dressing: Simultaneous In situ detection and inhibition of bacterial infection for accelerated wound healing. Biomaterials 2018, 161, 11–23.
- Jiao, G.; Yu, G.; Zhang, J.; Ewart, H.S. Chemical Structures and Bioactivities of Sulfated Polysaccharides from Marine Algae. Mar. Drugs 2011, 9, 196–223.
- Ngo, D.-H.; Kim, S.-K. Sulfated polysaccharides as bioactive agents from marine algae. Int. J. Boil. Macromol. 2013, 62, 70–75.
- Khotimchenko, Y.S. Biologically active substances from marine aquatic organisms—A source of new pharmaceutical substances and medicines. Pac. Med. J. 2010, 2, 5–9. (In Russia)
- Kuznetsova, T.A.; Besednova, N.N.; Somova, L.M.; Makarenkova, I.D.; Plekhova, N.G.; Drobot, E.I.; Kovalev, N.N.; Usov, V.V. Experimental evaluation of the effectiveness of wound dressings based on biologically active substances from marine hydrobionts. Russ. J. Mar. Boil. 2016, 42, 427–432. (In Russia)
- Besednova, N.N.; Smolina, T.P.; Andryukov, B.G.; Kuznetsova, T.A.; Mikhailov, V.V.; Zvyagintseva, T.N. Exopolysaccharides of marine bacteria: Prospects for use in medicine. Antibiot. chemother. 2018, 63, 67–78. (In Russia)
- Michalak, I.; Chojnacka, K. Algae Biomass: Characteristics and Applications: Towards Algae-Based Products; Chojnacka, K., Wieczorek, P.P., Schroeder, G., Michalak, I., Eds.; Springer International Publishing AG: Cham, Switzerland, 2018; 143p.
- Leandro, A.; Pereira, L.; Gonçalves, A.M.M. Diverse Applications of Marine Macroalgae. Mar. Drugs 2019, 18, 17.
- Kim, S.K.; Chojnacka, K. Marine Algae Extracts. Process, Product and Application; Kim, S.K., Chojnacka, K., Eds.; Wiley: Weinheim, Germany, 2015; Volume 2, 724p.
- Bhatia, S. Natural Polymer Drug Delivery Systems: Nanoparticles, Plants, and Algae; Springer: Gurgaon, India, 2016; 225p.
- Raposo, M.F.D.J.; De Morais, A.M.M.B.; Morais, R. Marine Polysaccharides from Algae with Potential Biomedical Applications. Mar. Drugs 2015, 13, 2967–3028.
- Florez, N.; Gonzalez-Munoz, M.; Ribeiro, D.; Fernandes, E.; Dominguez, H.; Freitas, M. Algae Polysaccharides’ Chemical Characterization and their Role in the Inflammatory Process. Curr. Med. Chem. 2017, 24, 149–175.
- Hinman, C.D.; Maibach, H. Effect of Air Exposure and Occlusion on Experimental Human Skin Wounds. Nat. 1963, 200, 377–378.
- Pan, H.; Fan, D.; Duan, Z.; Zhu, C.; Fu, R.Z.; Li, X. Non-stick hemostasis hydrogels as dressings with bacterial barrier activity for cutaneous wound healing. Mater. Sci. Eng. C 2019, 105, 110118.
- Ribeiro, D.M.L.; Júnior, A.R.C.; De Macedo, G.H.R.V.; Chagas, V.L.; Silva, L.D.S.; Cutrim, B.D.S.; Santos, D.M.; Soares, B.L.L.; Zagmignan, A.; Miranda, R.; et al. Polysaccharide-Based Formulations for Healing of Skin-Related Wound Infections: Lessons from Animal Models and Clinical Trials. Biomolecules 2019, 10, 63.
- Sun, M.-L.; Zhao, F.; Chen, X.-L.; Zhang, X.-Y.; Zhang, Y.-Z.; Song, X.-Y.; Sun, C.-Y.; Yang, J. Promotion of Wound Healing and Prevention of Frostbite Injury in Rat Skin by Exopolysaccharide from the Arctic Marine Bacterium Polaribacter sp. SM1127. Mar. Drugs 2020, 18, 48.
- Sun, M.-L.; Zhao, F.; Shi, M.; Zhang, X.-Y.; Zhou, B.-C.; Zhang, Y.-Z.; Chen, X.-L. Characterization and Biotechnological Potential Analysis of a New Exopolysaccharide from the Arctic Marine Bacterium Polaribacter sp. SM1127. Sci. Rep. 2015, 5, 18435.
- Sahana, T.; Rekha, P. A bioactive exopolysaccharide from marine bacteria Alteromonas sp. PRIM-28 and its role in cell proliferation and wound healing in vitro. Int. J. Boil. Macromol. 2019, 131, 10–18.
- Delbarre-Ladrat, C.; Sinquin, C.; Lebellenger, L.; Zykwinska, A.; Colliec-Jouault, S. Exopolysaccharides produced by marine bacteria and their applications as glycosaminoglycan-like molecules. Front. Chem. 2014, 2, 85.
- Casillo, A.; Lanzetta, R.; Parrilli, M.; Corsaro, M.M. Exopolysaccharides from Marine and Marine Extremophilic Bacteria: Structures, Properties, Ecological Roles and Applications. Mar. Drugs 2018, 16, 69.
- Delattre, C.; Pierre, G.; Laroche, C.; Michaud, P. Production, extraction and characterization of microalgal and cyanobacterial exopolysaccharides. Biotechnol. Adv. 2016, 34, 1159–1179.
- Laurienzo, P. Marine Polysaccharides in Pharmaceutical Applications: An Overview. Mar. Drugs 2010, 8, 2435–2465.
- Liu, S.-B.; Qiao, L.-P.; He, H.-L.; Zhang, Q.; Chen, X.-L.; Zhou, W.-Z.; Zhou, B.-C.; Zhang, Y.-Z. Optimization of Fermentation Conditions and Rheological Properties of Exopolysaccharide Produced by Deep-Sea Bacterium Zunongwangia profunda SM-A87. PLoS ONE 2011, 6, e26825.
- Menshova, R.V.; Shevchenko, N.M.; Imbs, T.I.; Zvyagintseva, T.N.; Malyarenko, O.; Zaporoshets, T.S.; Besednova, N.N.; Ermakova, S.P. Fucoidans from Brown Alga Fucus evanescens: Structure and Biological Activity. Front. Mar. Sci. 2016, 3, 129.
- Abasalizadeh, F.; Moghaddam, S.V.; Alizadeh, E.; Akbari, E.; Kashani, E.; Fazljou, S.M.B.; Torbati, M.; Akbarzadeh, A. Alginate-based hydrogels as drug delivery vehicles in cancer treatment and their applications in wound dressing and 3D bioprinting. J. Boil. Eng. 2020, 14, 1–22.
- Kiuru, P.; D’auria, M.V.; Muller, C.D.; Tammela, P.; Vuorela, H.; Yli-Kauhaluoma, J. Exploring Marine Resources for Bioactive Compounds. Planta Med. 2014, 80, 1234–1246.
- Kordjazi, M.; Shabanpour, B.; Zabihi, E.; Faramarzi, M.A.; Feizi, F.; Gavlighi, H.A.; Feghhi, M.A.; Hosseini, S.A. Sulfated Polysaccharides Purified from Two Species of Padina Improve Collagen and Epidermis Formation in the Rat. Int. J. Mol. Cell. Med. 2013, 2, 156–163.
- Dobrinčić, A.; Balbino, S.; Zorić, Z.; Pedisić, S.; Kovačević, D.B.; Garofulić, I.E.; Dragović-Uzelac, V. Advanced Technologies for the Extraction of Marine Brown Algal Polysaccharides. Mar. Drugs 2020, 18, 168.
- Williams, C. Kaltostat. Br. J. Nurs. 1994, 3, 965–967.
- Venkatesan, J.; Anil, S.; Rao, S.; Bhatnagar, I.; Kim, S.K. Sulfated Polysaccharides from Macroalgae for Bone Tissue Regeneration. Curr. Pharm. Des. 2019, 25, 1200–1209.
- Dhaliwal, K.; Lopez, N. Hydrogel dressings and their application in burn wound care. Br. J. Community Nurs. 2018, 23, S24–S27.
- Cunha, L.; Grenha, A. Sulfated Seaweed Polysaccharides as Multifunctional Materials in Drug Delivery Applications. Mar. Drugs 2016, 14, 42.
- Aderibigbe, B.; Buyana, B. Alginate in Wound Dressings. Pharmaceutics 2018, 10, 42.
- Kaiser, D.; Hafner, J.; Mayer, D.; French, L.E.; Läuchli, S. Alginate dressing and polyurethane film versus paraffin gauze in the treatment of split-thickness skin graft donor sites: A randomized controlled pilot study. Adv. Ski. Wound Care 2013, 26, 67–73.
- Lee, K.Y.; Mooney, D.J. Alginate: Properties and biomedical applications. Prog. Polym. Sci. 2012, 37, 106–126.
- Kamoun, E.A.; Kenawy, E.-R.S.; Chen, X. A review on polymeric hydrogel membranes for wound dressing applications: PVA-based hydrogel dressings. J. Adv. Res. 2017, 8, 217–233.
- Straccia, M.C.; D’Ayala, G.G.; Romano, I.; Oliva, A.; Laurienzo, P. Alginate Hydrogels Coated with Chitosan for Wound Dressing. Mar. Drugs 2015, 13, 2890–2908.
- Ching, S.H.; Bansal, N.; Bhandari, B. Alginate gel particles—A review of production techniques and physical properties. Crit. Rev. Food Sci. Nutr. 2017, 57, 1133–1152.
- Boateng, J.S.; Matthews, K.H.; Stevens, H.N.; Eccleston, G.M. Wound Healing Dressings and Drug Delivery Systems: A Review. J. Pharm. Sci. 2008, 97, 2892–2923.
- Gurtner, G.C.; Werner, S.; Barrandon, Y.; Longaker, M.T. Wound repair and regeneration. Nature 2008, 453, 314–321.
- Boateng, J.S.; Catanzano, O. Advanced Therapeutic Dressings for Effective Wound Healing—A Review. J. Pharm. Sci. 2015, 104, 3653–3680.
- Boateng, J.S.; Burgos-Amador, R.; Okeke, O.; Pawar, H. Composite alginate and gelatin-based bio-polymeric wafers containing silver sulfadiazine for wound healing. Int. J. Boil. Macromol. 2015, 79, 63–71.
- Pawar, H.V.; Boateng, J.S.; Ayensu, I.; Tetteh, J. Multifunctional Medicated Lyophilised Wafer Dressing for Effective Chronic Wound Healing. J. Pharm. Sci. 2014, 103, 1720–1733.
- Murakami, K.; Ishihara, M.; Aoki, H.; Nakamura, S.; Nakamura, S.-I.; Yanagibayashi, S.; Takikawa, M.; Kishimoto, S.; Yokoe, H.; Kiyosawa, T.; et al. Enhanced healing of mitomycin C-treated healing-impaired wounds in rats with hydrosheets composed of chitin/chitosan, fucoidan, and alginate as wound dressings. Wound Repair Regen. 2010, 18, 478–485.
- Williams, C. Role of CarboFlex in the nursing management of wound odour. Br. J. Nurs. 2001, 10, 122–125.
- Lee, S.M.; Park, I.K.; Kim, Y.S.; Kim, H.J.; Moon, H.; Mueller, S.; Jeong, Y.-I. Physical, morphological, and wound healing properties of a polyurethane foam-film dressing. Biomater. Res. 2016, 20, 15.
- Thomas, S. Alginate dressings in surgery and wound management—Part 1. J. Wound Care 2000, 9, 56–60.
- Stoica, A.E.; Chircov, C.; Stoica, A.E. Nanomaterials for Wound Dressings: An Up-to-Date Overview. Molecules 2020, 25, 2699.
- Fertah, M.; Belfkira, A.; Dahmane, E.M.; Taourirte, M.; Brouillette, F. Extraction and characterization of sodium alginate from Moroccan Laminaria digitata brown seaweed. Arab. J. Chem. 2017, 10, S3707–S3714.
- Fan, C.; Pek, C.H.; Por, Y.C.; Lim, G.J.S. Biobrane dressing for paediatric burns in Singapore: A retrospective review. Singap. Med. J. 2018, 59, 360–365.
- McBride, C.; Kimble, R.; Stockton, K. Prospective randomised controlled trial of Algisite™ M, Cuticerin™, and Sorbact® as donor site dressings in paediatric split-thickness skin grafts. Burn. Trauma 2018, 6, 33.
- Sahana, T.G.; Rekha, P.D. Biopolymers: Applications in wound healing and skin tissue engineering. Mol. Boil. Rep. 2018, 45, 2857–2867.
- Thomas, S. Alginate dressings in surgery and wound management: Part 2. J. Wound Care 2000, 9, 115–119.
- Totty, J.P.; Bua, N.; Smith, G.; Harwood, A.; Carradice, D.; Wallace, T.; Chetter, I. Dialkylcarbamoyl chloride (DACC)-coated dressings in the management and prevention of wound infection: A systematic review. J. Wound Care 2017, 26, 107–114.
- Thomas, S. Alginate dressings in surgery and wound management: Part 3. J. Wound Care 2000, 9, 163–166.
- Hiro, M.E.; Pierpont, Y.N.; Ko, F.; Wright, T.E.; Robson, M.C.; Payne, W.G. Comparative Evaluation of Silver-Containing Antimicrobial Dressings on In Vitro and In Vivo Processes of Wound Healing. Eplasty 2012, 12, e48.
- Pires, A.L.R.; Motta, L.D.A.; Dias, A.M.; De Sousa, H.C.; Moraes, Â.M.; Braga, M.E.M. Towards wound dressings with improved properties: Effects of poly(dimethylsiloxane) on chitosan-alginate films loaded with thymol and beta-carotene. Mater. Sci. Eng. C 2018, 93, 595–605.
- Rezvanian, M.; Amin, M.C.I.M.; Ng, S.-F. Development and physicochemical characterization of alginate composite film loaded with simvastatin as a potential wound dressing. Carbohydr. Polym. 2016, 137, 295–304.
- Fallacara, A.; Baldini, E.; Manfredini, S.; Vertuani, S. Hyaluronic Acid in the Third Millennium. Polymers (Basel) 2018, 10, 701.
- Catanzano, O.; D’Esposito, V.; Acierno, S.; Ambrosio, M.; De Caro, C.; Avagliano, C.; Russo, P.; Russo, R.; Miro, A.; Ungaro, F.; et al. Alginate–hyaluronan composite hydrogels accelerate wound healing process. Carbohydr. Polym. 2015, 131, 407–414.
- Hajiali, H.; Summa, M.; Russo, D.; Armirotti, A.; Brunetti, V.; Bertorelli, R.; Athanassiou, A.; Mele, E. Alginate-lavender nanofibers with antibacterial and anti-inflammatory activity to effectively promote burn healing. J. Mater. Chem. B 2016, 4, 1686–1695.
- Hu, C.; Gong, R.H.; Zhou, F.-L. Electrospun sodium alginate/polyethylene oxide fibers and nanocoated yarns. Int. J. Polym. Sci. 2015, 2015, 1–12.
- Jayakumar, R.; Kumar, P.S.; Mohandas, A.; Lakshmanan, V.-K.; Biswas, R.; Pt, S.K.; Raja, B. Exploration of alginate hydrogel/nano zinc oxide composite bandages for infected wounds. Int. J. Nanomed. 2015, 10, 53–66.
- Xie, H.; Chen, X.; Shen, X.; He, Y.; Chen, W.; Luo, Q.; Ge, W.; Yuan, W.; Tang, X.; Hou, D.; et al. Preparation of chitosan-collagen-alginate composite dressing and its promoting effects on wound healing. Int. J. Boil. Macromol. 2018, 107, 93–104.
- Li, S.; Li, L.; Guo, C.; Qin, H.; Yu, X. A promising wound dressing material with excellent cytocompatibility and proangiogenesis action for wound healing: Strontium loaded Silk fibroin/Sodium alginate (SF/SA) blend films. Int. J. Boil. Macromol. 2017, 104, 969–978.
- Park, J.-H.; Choi, S.-H.; Park, S.-J.; Lee, Y.J.; Park, J.H.; Song, P.H.; Cho, C.-M.; Ku, S.-K.; Song, C.-H. Promoting Wound Healing Using Low Molecular Weight Fucoidan in a Full-Thickness Dermal Excision Rat Model. Mar. Drugs 2017, 15, 112.
- Li, B.; Lu, F.; Wei, X.; Zhao, R. Fucoidan: Structure and Bioactivity. Molecules 2008, 13, 1671–1695.
- Luthuli, S.; Wu, S.; Cheng, Y.; Zheng, X.; Wu, M.; Tong, H. Therapeutic Effects of Fucoidan: A Review on Recent Studies. Mar. Drugs 2019, 17, 487.
- Fitton, J.H.; Stringer, D.N.; Park, A.Y.; Karpiniec, S.S. Therapies from Fucoidan: New Developments. Mar. Drugs 2019, 17, 571.
- Zayed, A.; Ulber, R. Fucoidans: Downstream Processes and Recent Applications. Mar. Drugs 2020, 18, 170.
- Benbow, N.L.; Webber, J.L.; Karpiniec, S.; Krasowska, M.; Ferri, J.K.; Beattie, D.A. The influence of polyanion molecular weight on polyelectrolyte multilayers at surfaces: Protein adsorption and protein—polysaccharide complexation/stripping on natural polysaccharide films on solid supports. Phys. Chem. Chem. Phys. 2017, 19, 23790–23801.
- Nakamura, T.; Mizuno, S. The discovery of Hepatocyte Growth Factor (HGF) and its significance for cell biology, life sciences and clinical medicine. Proc. Jpn. Acad. Ser. B 2010, 86, 588–610.
- Lin, X.Y.; Wang, H.; Tan, Y. [Role of Hepatocyte Growth Factor in Wound Repair]. Zhongguo Yi Xue Ke Xue Yuan Xue Bao 2018, 40, 822–826.
- O’Leary, R.; Rerek, M.; Wood, E.J. Fucoidan modulates the effect of transforming growth factor (TGF)-beta1 on fibroblast proliferation and wound repopulation in in vitro models of dermal wound repair. Boil. Pharm. Bull. 2004, 27, 266–270.
- Charboneau, A.J.; Delaney, J.P.; Beilman, G. Fucoidans inhibit the formation of post-operative abdominal adhesions in a rat model [published correction appears in PLoS One. 2019; 14(1): e0211371]. PLoS ONE 2018, 13, e0207797.
- Cashman, J.D.; Kennah, E.; Shuto, A.; Winternitz, C.; Springate, C.M. Fucoidan Film Safely Inhibits Surgical Adhesions in a Rat Model. J. Surg. Res. 2011, 171, 495–503.
- Okabayashi, K.; Ashrafian, H.; Zacharakis, E.; Hasegawa, H.; Kitagawa, Y.; Athanasiou, T.; Darzi, A. Adhesions after abdominal surgery: A systematic review of the incidence, distribution and severity. Surg. Today 2014, 44, 405–420.
- Levendosky, K.; Mizenina, O.; Martinelli, E.; Jean-Pierre, N.; Kizima, L.; Rodriguez, A.; Kleinbeck, K.; Bonnaire, T.; Robbiani, M.; Zydowsky, T.M.; et al. Griffithsin and Carrageenan Combination To Target Herpes Simplex Virus 2 and Human Papillomavirus. Antimicrob. Agents Chemother. 2015, 59, 7290–7298.
- Derby, N.; Lal, M.; Aravantinou, M.; Kizima, L.; Barnable, P.; Rodriguez, A.; Lai, M.; Wesenberg, A.; Ugaonkar, S.; Levendosky, K.; et al. Griffithsin carrageenan fast dissolving inserts prevent SHIV HSV-2 and HPV infections in vivo. Nat. Commun. 2018, 9, 3881.
- Zepon, K.M.; Martins, M.M.; Marques, M.S.; Heckler, J.M.; Morisso, F.D.P.; Moreira, M.G.; Ziulkoski, A.L.; Kanis, L.A. Smart wound dressing based on κ–carrageenan/locust bean gum/cranberry extract for monitoring bacterial infections. Carbohydr. Polym. 2019, 206, 362–370.
- Yegappan, R.; Selvaprithiviraj, V.; Amirthalingam, S.; Jayakumar, R. Carrageenan based hydrogels for drug delivery, tissue engineering and wound healing. Carbohydr. Polym. 2018, 198, 385–400.
- Shankar, S.; Teng, X.; Li, G.; Rhim, J.-W. Preparation, characterization, and antimicrobial activity of gelatin/ZnO nanocomposite films. Food Hydrocoll. 2015, 45, 264–271.
- Zia, K.M.; Tabasum, S.; Nasif, M.; Sultan, N.; Aslam, N.; Noreen, A.; Zuber, M. A review on synthesis, properties and applications of natural polymer-based carrageenan blends and composites. Int. J. Boil. Macromol. 2017, 96, 282–301.
- Kuznetsova, T.A.; Andryukov, B.G.; Besednova, N.N.; Zaporozhets, T.S.; Kalinin, A.V. Marine Algae Polysaccharides as Basis for Wound Dressings, Drug Delivery, and Tissue Engineering: A Review. J. Mar. Sci. Eng. 2020, 8, 481.
- Radiation Effects in Polymeric Materials; Kumar, V.; Chaudhary, B.; Sharma, V.; Verma, K. (Eds.) Springer: Cham, Switzerland, 2019; 412p.
- Tytgat, L.; Van Damme, L.; Arevalo, M.D.P.O.; Declercq, H.; Thienpont, H.; Otteveare, H.; Blondeel, P.; Dubruel, P.; Van Vlierberghe, S. Extrusion-based 3D printing of photo-crosslinkable gelatin and κ-carrageenan hydrogel blends for adipose tissue regeneration. Int. J. Boil. Macromol. 2019, 140, 929–938.
- Li, H.; Tan, Y.J.; Li, L. A strategy for strong interface bonding by 3D bioprinting of oppositely charged κ-carrageenan and gelatin hydrogels. Carbohydr. Polym. 2018, 198, 261–269.
- Lokhande, G.; Carrow, J.K.; Thakur, T.; Xavier, J.R.; Parani, M.; Bayless, K.J.; Gaharwar, A.K. Nanoengineered injectable hydrogels from kappa-carrageenan and two-dimensional nanosilicates for wound healing application. Acta Biomater. 2018, 70, 35–47.
- Singh, D.; Singh, A.; Singh, R. Polyvinyl pyrrolidone/carrageenan blend hydrogels with nanosilver prepared by gamma radiation for use as an antimicrobial wound dressing. J. Biomater. Sci. Polym. Ed. 2015, 26, 1269–1285.
- El-Fawal, G.F.; Yassin, A.M.; El-Deeb, N.M. The Novelty in Fabrication of Poly Vinyl Alcohol/κ-Carrageenan Hydrogel with Lactobacillus bulgaricus Extract as Anti-inflammatory Wound Dressing Agent. AAPS PharmSciTech 2016, 18, 1605–1616.
- Schaude, C.; Fröhlich, E.; Meindl, C.; Attard, J.; Binder, B.; Mohr, G.J. The Development of Indicator Cotton Swabs for the Detection of pH in Wounds. Sensors 2017, 17, 1365.
- Nair, A.V.; Raman, M.; Doble, M. Cyclic β-(1→3) (1→6) glucan/carrageenan hydrogels for wound healing applications. RSC Adv. 2016, 6, 98545–98553.
- Tamayol, A.; Akbari, M.; Zilberman, Y.; Comotto, M.; Lesha, E.; Serex, L.; Bagherifard, S.; Chen, Y.; Fu, G.; Ameri, S.K.; et al. Flexible pH-Sensing Hydrogel Fibers for Epidermal Applications. Adv. Heal. Mater. 2016, 5, 711–719.
- Tziveleka, L.-A.; Ioannou, E.; Roussis, V. Ulvan, a bioactive marine sulphated polysaccharide as a key constituent of hybrid biomaterials: A review. Carbohydr. Polym. 2019, 218, 355–370.
- Manivasagan, P.; Oh, J. Marine polysaccharide-based nanomaterials as a novel source of nanobiotechnological applications. Int. J. Boil. Macromol. 2016, 82, 315–327.
- Kanno, K.; Akiyoshi, K.; Nakatsuka, T.; Watabe, Y.; Yukimura, S.; Ishihara, H.; Shin, N.; Kawasaki, Y.; Yano, D. Biocompatible Hydrogel from a Green Tide-Forming Chlorophyta. J. Sustain. Dev. 2012, 5, 38–45.
- Alves, A.; Pinho, E.D.; Neves, N.; Sousa, R.A.; Reis, R.L. Processing ulvan into 2D structures: Cross-linked ulvan membranes as new biomaterials for drug delivery applications. Int. J. Pharm. 2012, 426, 76–81.
- Toskas, G.; Hund, R.-D.; Laourine, E.; Cherif, C.; Smyrniotopoulos, V.; Roussis, V. Nanofibers based on polysaccharides from the green seaweed Ulva Rigida. Carbohydr. Polym. 2011, 84, 1093–1102.
- Kidgell, J.T.; Magnusson, M.; De Nys, R.; Glasson, C.R. Ulvan: A systematic review of extraction, composition and function. Algal Res. 2019, 39, 101422.
- Mata, L.; Magnusson, M.; Paul, N.A.; De Nys, R. The intensive land-based production of the green seaweeds Derbesia tenuissima and Ulva ohnoi: Biomass and bioproducts. J. Appl. Phycol. 2016, 28, 365–375.
- Glasson, C.R.K.; Sims, I.M.; Carnachan, S.M.; De Nys, R.; Magnusson, M. A cascading biorefinery process targeting sulfated polysaccharides (ulvan) from Ulva ohnoi. Algal Res. 2017, 27, 383–391.
- Nardelli, A.; Chiozzini, V.G.; Braga, E.S.; Chow, F. Integrated multi-trophic farming system between the green seaweed Ulva lactuca, mussel, and fish: A production and bioremediation solution. J. Appl. Phycol. 2018, 31, 847–856.
- Adrien, A.; Bonnet, A.; Dufour, D.; Baudouin, S.; Maugard, T.; Bridiau, N. Pilot production of ulvans from Ulva sp. and their effects on hyaluronan and collagen production in cultured dermal fibroblasts. Carbohydr. Polym. 2017, 157, 1306–1314.
- Hardouin, K.; Bedoux, G.; Burlot, A.-S.; Donnay-Moreno, C.; Bergé, J.-P.; Nyvall-Collén, P.; Bourgougnon, N. Enzyme-assisted extraction (EAE) for the production of antiviral and antioxidant extracts from the green seaweed Ulva armoricana (Ulvales, Ulvophyceae). Algal Res. 2016, 16, 233–239.
- Rizk, M.Z.; El-Sherbiny, M.; Borai, I.H.; Ezz, M.K.; Aly, H.F.; Matloub, A.A.; Farrag, A.E.R.; Fouad, G.I. Sulphated polysaccharides (SPS) from the green alga ulva fasciata extract modulates liver and kidney function in high fat diet-induced hypercholesterolemic rats. Int. J. Pharm. Pharm. Sci. 2016, 8, 43–55.
