The application of nanotechnology, molecular biotechnologies, and nano-sciences for medical purposes has been termed nanomedicine, a promising growing area of medical research. The aim of this paper is to provide an overview of and discuss nanotechnology applications in the early epochs of life, from transplacental transfer to neonatal/pediatric conditions. Diagnostic and therapeutic applications, mainly related to the respiratory tract, the neurosensory system, and infections, are explored and discussed. Preclinical studies show promising results for a variety of conditions, including for the treatment of pregnancy complications and fetal, neonatal, and pediatric diseases. However, given the complexity of the functions and interactions between the placenta and the fetus, and the complex and incompletely understood determinants of tissue growth and differentiation during early life, there is a need for much more data to confirm the safety and efficacy of nanotechnology in this field.
- nanotechnology
- nanomedicine
- child health
- nanodrug delivery
- fetus
- newborn
Introduction
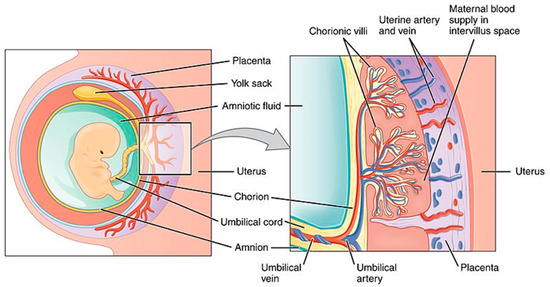
Transplacental Drug Delivery System
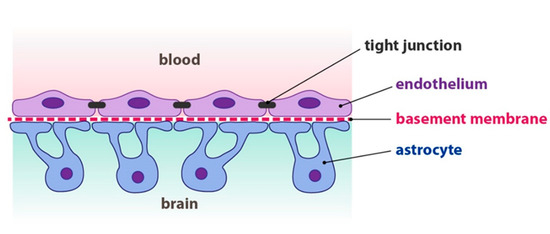
Nervous System
Respiratory System
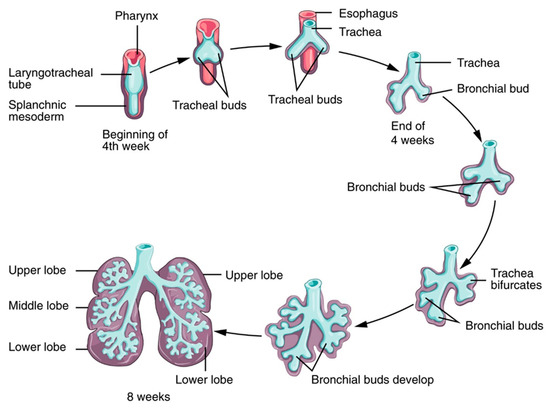
The phases are characterized by morphogenesis of the conducting airways, formation of alveoli (the terminal regions of the lungs where gas exchange occurs), differentiation of cells with region-specific functions of the lung (such as protection from foreign substances), efficient movement of air into and out of the lung, and efficient gas exchange at the alveolar–capillary interface. These processes are finely regulated by a complex network of cell signals, cell-derived molecules, hormones, and genetic expression.
One of the key components of the respiratory system is pulmonary surfactant. Pulmonary surfactant covers and stabilizes the alveoli and has peculiar biophysical properties, including the reduction of alveolar superficial tension and defense against microorganisms and toxic substances.
Surface tension is the force that develops at the interface between two or more fluids or between fluids and a solid wall. The forces of cohesion that act between the molecules of a fluid mean that those on the outer surface are subjected to a resulting force directed inwards; there is therefore a tendency of molecular stray from the limit surface of the liquid, which tends to contract and assume the minimum possible extent.
In the lungs, there is an interface between the liquid that covers the alveolar walls and the gas at their internal. Again, the surface liquid layer tends to contract and expel the air outwards, resulting in the collapse of the alveoli. The pressure generated within the alveoli due to surface tension is called collapse pressure and, according to Laplace’s law, is directly proportional to twice the surface tension and inversely proportional to the radius of the alveoli. Thus, the increase in surface tension determines an increment in the collapse pressure of the alveoli. In the physiological lung, surface tension is greatly reduced by the presence of surfactant, a mixture of different phospholipids, proteins, and ions produced by type II alveolar epithelial cells that cover about 10% of the area of the alveolar walls. Surfactant production begins at around the 20th week of gestation and reaches significant amounts in the amniotic fluid only after the 28th week [11].
The main components of surfactant are phospholipid dipalmitoylphosphatidylcholine, apoproteins, and calcium ions (see below). The tensioactive capacity lies in the amphibious dipalmitoylphosphatidylcholine at the liquid–gas interface. The absence of surfactant can lead to atelectasis (collapse of the alveoli) and respiratory insufficiency. Quantitatively, the surface tension of normal liquids covering the alveoli without surfactants is 50 dine/cm; this decreases to 5–30 dine/cm with surfactant. Consequently, the collapse pressure for medium-sized alveoli (r = 100 µm) falls from about 18 to 4 cm H2O.
To summarize, lung surfactant has important functions:
- increases lung compliance and therefore reduces the work required to expand the lung;
- contributes to the stability of the alveoli by preventing their collapse;
- limits the risk of pulmonary edema by decreasing the recall force of fluids inside the alveoli;
- allows the expansion of the lung at birth during the first respiratory efforts.
Moreover, surfactant can be used as a shuttle for the delivery of drugs and nanocarriers. Pulmonary surfactant is composed of approximately 90% lipids and 10% proteins. Four proteins—SP-A, -B, -C, and -D—are important for surfactant functions. SP-A and SP-D are large hydrophilic glycoproteins involved in pulmonary host defense through binding to inhaled particles and pathogens. SP-B and SP-C are smaller hydrophobic proteins that regulate the very low surface tension observed in the lung. The expression of surfactant proteins is regulated by genetic and environmental factors, including inflammation [12][13].
Interactions between surfactant and nanoparticles depend on the physico-chemical properties of the nanoparticles and also on the molecular composition, dynamic surface phase behavior, and monolayer biomechanics of the pulmonary surfactant [14][15][16]. The interaction between nanoparticles and surfactant has been studied with different methods. Hu et al. [14] showed that many factors, including surface charge, degree of hydrophobicity, and acquisition of a lipoprotein corona from endogenous surfactant, influence nanoprotein adsorption and blood stream translocation. Hydrophobic nanoparticles, such as carbon-based nanomaterials, can be retained at the surfactant lining layer and can lead to increased inflammation. Hydrophilic nanoparticles can be used for systemic drug delivery due to their rapid absorption and translocation to other organs and tissues. Anionic nanoparticles may inhibit the biophysical function of pulmonary surfactant by binding to surfactant proteins and interfering with their functions. Cationic nanoparticles may be taken up by cells and may cause acute toxicity. Therefore, neutral nanoparticles might be the safest option for pulmonary drug delivery.
In experimental models, some authors reported that hydrophilic nanomaterials (halloysite and bentonite) induced concentration-dependent damage on surfactant phospholipid function [17]. The shape and porous structure of nanoparticles are important factors to consider.
Other authors found that nanoparticles have different effects on lung inflammation and function depending on their metal properties: compared to non-metal nanoparticles (which elicit a dose-dependent inflammatory effect), silver nanoparticles do not induce significant inflammation, but only change in lung elastance [18][19].
On the other hand, the interaction between nanoparticles and surfactant may offer important therapeutic advantages: delivering drugs or nanoparticles in combination with surfactant can improve drug stability and prevent degradation and clearance in vivo, thereby allowing the delivery of macromolecular drugs. For example, nanovesicles carrying antibiotics have been successfully tested in animal models and clinical trials are ongoing [20][21]. Other potential applications include the diagnosis and therapy of different pulmonary diseases, vaccines, and cancer therapy.
Surfactant itself (animal-derived or synthetic) is an established therapy for respiratory distress syndrome, particularly among newborns: the administration of this drug is invasive, as it requires endotracheal instillation through a tube or a catheter. However, this maneuver can be associated with lung function deterioration and long-term complications among infants, and studies aimed at finding the most convenient method of administration are ongoing [22][23]. Other potential applications include the diagnosis and therapy of different pulmonary diseases, vaccines, and cancer therapy. Some authors designed inhalable nanoparticles mimicking some of the lipid components of surfactant [24]. Hopefully, technological advancement in this field will eventually lead to the production of inhalable surfactant preparations, and invasive surfactant administration will eventually be overcome.
Respiratory tract infections are among the most frequent worldwide and represent one of the most important causes of death in childhood. Antimicrobial resistance is a major challenge, as it results in increased morbidity and mortality worldwide. Since the development of new antimicrobials is expensive and has scarce success, one strategy to overcome this issue could be the application of synthetic products with antimicrobial properties. Moreover, newborns and young children have still immature metabolic pathways that can result in increased drug toxicity and poorly known drug interactions, potentially resulting in transient or persistent adverse events. Polymers with intrinsic antimicrobial effects or that can be conjugated with antimicrobials might replace antimicrobials. A comprehensive review of these compounds has been recently published [25]. Furthermore, nanomodified endotracheal tubes have been shown to reduce bacterial adhesion on the inner surface of the tube itself and potentially reduce the likelihood of ventilator-acquired infections [26].
Lower airway inflammation can be the result of different diseases and is often characterized by neutrophil leukocytes recruitment. Current therapies locally administered have low efficacy as multiple systems reduce drug availability, i.e., hydrophobicity and clearance by local defense systems. Some authors developed a delivery platform that takes advantage of the extracellular proteolysis of a microgel to deliver nanoparticle-embedded hydrophobic drugs to neutrophils and then to the lower airways [27]. In another study, Vij et al. successfully tested, in animal models, the efficacy of a PEGylated immuno-conjugated PLGA-nanoparticle to selectively deliver a drug to neutrophil cells [28].
Furthermore, the delivery of siRNAs to endothelial cells has been investigated in animal models for the treatment of pulmonary inflammatory conditions and other diseases [29].
Asthma is a common chronic inflammatory disease of the lungs starting in childhood and is characterized by intermittent airway obstruction, bronchial hyper-reactivity, and chronic airway inflammation. Diagnostic tests include the measurement of lung exhaled air flow and characteristics by means of specific instruments, i.e., spirometers. The electronic nose (E-nose) is a novel device based on nanosensors capable of detecting specific volatile organic compounds in exhaled gas, thus confirming an asthma diagnosis and allowing stratification and subtype characterization [30]. Asthma therapy relies mainly on the inhalation of certain drugs; however, poor deposition of the inhaled drug in the lung presents a challenge for the effectiveness of therapy. Drug nanoformulations are currently being evaluated following promising preclinical studies [31][32][33].
Concerning respiratory function monitoring and diagnostics, Bhattacharjee et al. [34] developed a point-of-care testing device consisting of a mouthpiece, paper-sensor, micro-heater assemblage, and monitoring unit which could facilitate the diagnosis of chronic obstructive lung diseases. The sensor was developed by depositing gold and cadmium sulfide nanoparticles on a paper surface in which the former enhanced the electrical and thermal conductivities while the latter allowed high precision humidity sensing.
Clinical studies are needed in order to assess the safety and efficacy of nanoparticles as drug carriers for respiratory and systemic conditions.
Ocular System
Future Directions
This entry is adapted from the peer-reviewed paper 10.3390/app10124323
References
- Nicola Luigi Luigi; Nanomedicine: Insights from a Bibliometrics-Based Analysis of Emerging Publishing and Research Trends. Medicina 2019, 55, 785, 10.3390/medicina55120785.
- Stefano Nobile; Lucio Nobile; Nanotechnology for biomedical applications: Recent advances in neurosciences and bone tissue engineering. Polymer Engineering & Science 2017, 57, 644-650, 10.1002/pen.24595.
- Thádia Evelyn De Araújo; Iliana Claudia Balga Milián; Guilherme De Souza; Rafaela José Da Silva; Alessandra Monteiro Rosini; Pâmela Mendonça Guirelli; Priscila Silva Franco; Bellisa Freitas Barbosa; Eloisa Amália Vieira Ferro; Idessania Nazareth Costa; et al. Experimental models of maternal–fetal interface and their potential use for nanotechnology applications. Cell Biology International 2019, 44, 36-50, 10.1002/cbin.11222.
- Baozhen Zhang; Ruijing Liang; Mingbin Zheng; Lintao Cai; Xiujun Fan; Surface-Functionalized Nanoparticles as Efficient Tools in Targeted Therapy of Pregnancy Complications.. International Journal of Molecular Sciences 2019, 20, 3642, 10.3390/ijms20153642.
- Lucie Valero; Khair Alhareth; Jenifer Espinoza Romero; Warren Viricel; Jeanne Leblond; Audrey Chissey; Hélène Dhotel; Caroline Roques; Danielle Campiol Arruda; Virginie Escriou; et al. Liposomes as Gene Delivery Vectors for Human Placental Cells. Molecules 2018, 23, 1085, 10.3390/molecules23051085.
- Mary G. Prieve; Pierrot Harvie; Sean D. Monahan; Debashish Roy; Allen G. Li; Teri L. Blevins; Amber E. Paschal; Matt Waldheim; Eric C. Bell; Anna Galperin; et al. Targeted mRNA Therapy for Ornithine Transcarbamylase Deficiency. Molecular Therapy 2018, 26, 801-813, 10.1016/j.ymthe.2017.12.024.
- Irina Burd; Fan Zhang; Tahani Dada; Manoj K. Mishra; Talaibek Borbiev; Wojciech G. Lesniak; Haitham Baghlaf; Sujatha Kannan; Rangaramanujam M. Kannan; Fetal uptake of intra-amniotically delivered dendrimers in a mouse model of intrauterine inflammation and preterm birth. Nanomedicine: Nanotechnology, Biology and Medicine 2014, 10, 1343-1351, 10.1016/j.nano.2014.03.008.
- Limin Wei; Longquan Shao; Xiaoli Feng; Aijie Chen; Yanli Zhang; Jianfeng Wang; Central nervous system toxicity of metallic nanoparticles. International Journal of Nanomedicine 2015, 10, 4321-4340, 10.2147/IJN.S78308.
- Kevin Liaw; Ozgul Gok; Louis DeRidder; Sujatha Kannan; Rangaramanujam M. Kannan; Quantitative assessment of surface functionality effects on microglial uptake and retention of PAMAM dendrimers. Journal of Nanoparticle Research 2018, 20, 111, 10.1007/s11051-018-4219-1.
- Meng Zheng; Wei Tao; Yan Zou; Omid C. Farokhzad; Bingyang Shi; Nanotechnology-Based Strategies for siRNA Brain Delivery for Disease Therapy. Trends in Biotechnology 2018, 36, 562-575, 10.1016/j.tibtech.2018.01.006.
- Moore, K.L.; Persaud, T.V.N.; Torchia, M.G. . The Developing Human: Clinically Oriented Embryology; Elsevier—Health Sciences Division: Amsterdam, the Netherlands, 2019; pp. 181-192.
- Sara D’Aronco; Manuela Simonato; Luca Vedovelli; Aldo Baritussio; Giuseppe Verlato; Stefano Nobile; Chiara Giorgetti; Matteo Nespeca; Virgilio P. Carnielli; Paola Cogo; et al. Surfactant protein B and A concentrations are increased in neonatal pneumonia. Pediatric Research 2015, 78, 401-406, 10.1038/pr.2015.123.
- 13. Nespeca, M.; Giorgetti, C.; Nobile, S.; Ferrini, I.; Simonato, M.; Verlato, G.; Cogo, P.; Carnielli, V.P.; Does Whole-Body Hypothermia in Neonates with Hypoxic–Ischemic Encephalopathy Affect Surfactant Disaturated-Phosphatidylcholine Kinetics?. PLoS ONE 2016, 11, e0153328, 10.1371/journal.
- Guoqing Hu; Bao Jiao; Xinghua Shi; Russell P. Valle; Qihui Fan; Yi Y. Zuo; Physicochemical Properties of Nanoparticles Regulate Translocation across Pulmonary Surfactant Monolayer and Formation of Lipoprotein Corona. ACS Nano 2013, 7, 10525-10533, 10.1021/nn4054683.
- Mridula V. Dwivedi; Rakesh Kumar Harishchandra; Olga Koshkina; Michael Maskos; Hans-Joachim Galla; Size Influences the Effect of Hydrophobic Nanoparticles on Lung Surfactant Model Systems. Biophysical Journal 2014, 106, 289-298, 10.1016/j.bpj.2013.10.036.
- Moritz Beck-Broichsitter; Clemens Ruppert; Thomas Schmehl; Andreas Günther; Werner Seeger; Biophysical inhibition of synthetic vs. naturally-derived pulmonary surfactant preparations by polymeric nanoparticles. Biochimica et Biophysica Acta (BBA) - Biomembranes 2014, 1838, 474-481, 10.1016/j.bbamem.2013.10.016.
- Dorota Kondej; Tomasz R. Sosnowski; Effect of clay nanoparticles on model lung surfactant: a potential marker of hazard from nanoaerosol inhalation.. Environmental Science and Pollution Research 2015, 23, 4660-9, 10.1007/s11356-015-5610-4.
- Danielle Botelho; Bey F. Leo; Christopher Massa; Srijata Sarkar; Terry Tetley; Kian F. Chung; Shu Chen; Mary P. Ryan; Alexandra Porter; Elena N. Atochina-Vasserman; et al. Exposure to Silver Nanospheres Leads to Altered Respiratory Mechanics and Delayed Immune Response in an in Vivo Murine Model. Frontiers in Pharmacology 2018, 9, 213, 10.3389/fphar.2018.00213.
- Sinbad Sweeney; Bey Fen Leo; Shu Chen; Nisha Abraham-Thomas; Andrew J. Thorley; Andrew Gow; Stephan Schwander; Junfeng Jim Zhang; M.S.P. Shaffer; Kian Fan Chung; et al. Pulmonary surfactant mitigates silver nanoparticle toxicity in human alveolar type-I-like epithelial cells. Colloids and Surfaces B: Biointerfaces 2016, 145, 167-175, 10.1016/j.colsurfb.2016.04.040.
- Ching-Yun Hsu; Calvin T. Sung; Ibrahim A Aljuffali; Chun-Han Chen; Kai-Yin Hu; Jia-You Fang; Intravenous anti-MRSA phosphatiosomes mediate enhanced affinity to pulmonary surfactants for effective treatment of infectious pneumonia. Nanomedicine: Nanotechnology, Biology and Medicine 2018, 14, 215-225, 10.1016/j.nano.2017.10.006.
- Mohammad Doroudian; Ronan MacLoughlin; Fergus Poynton; Adriele Prina-Mello; Seamas Donnelly; Nanotechnology based therapeutics for lung disease. Thorax 2019, 74, 965-976, 10.1136/thoraxjnl-2019-213037.
- Efficacy of a new technique - INtubate-RECruit-SURfactant-Extubate - . , , , .
- Stefano Nobile; Paolo Marchionni; Giovanni Vento; Valentina Vendettuoli; Claudio Marabini; Alessandra Lio; Cinzia Ricci; Domenica Mercadante; MariaRosa Colnaghi; Fabio Mosca; et al. New Insights on Early Patterns of Respiratory Disease among Extremely Low Gestational Age Newborns. Neonatology 2017, 112, 53-59, 10.1159/000456706.
- Heidi M Mansour; Meenach; Vogt; Ronald C McGarry; Kimberly W Anderson; J. Zach Hilt; Samantha A Meenach; Frederick G Vogt; Design, physicochemical characterization, and optimization of organic solution advanced spray-dried inhalable dipalmitoylphosphatidylcholine (DPPC) and dipalmitoylphosphatidylethanolamine poly(ethylene glycol) (DPPE-PEG) microparticles and nanoparticles for targeted respiratory nanomedicine delivery as dry powder inhalation aerosols. International Journal of Nanomedicine 2013, 8, 275-293, 10.2147/IJN.S30724.
- Nor Fadhilah Kamaruzzaman; Li Peng Tan; Ruhil Hayati Hamdan; Siew Shean Choong; Weng Kin Wong; Amanda Gibson; Alexandru Chivu; Maria De Fatima Pina; Antimicrobial Polymers: The Potential Replacement of Existing Antibiotics?. International Journal of Molecular Sciences 2019, 20, 2747, 10.3390/ijms20112747.
- Mary C Machado; Daniel Cheng; Keiko M Tarquinio; Thomas J Thomas Webster; Nanotechnology: Pediatric Applications. Pediatric Research 2010, 67, 500-504, 10.1203/pdr.0b013e3181d68e78.
- Joscelyn C. Mejías; Osric A. Forrest; Camilla Margaroli; David A. Frey Rubio; Liliana Viera; Jindong Li; Xin Xu; Amit Gaggar; Rabindra Tirouvanziam; Krishnendu Roy; et al. Neutrophil-targeted, protease-activated pulmonary drug delivery blocks airway and systemic inflammation. JCI Insight 2019, 4, 131468, 10.1172/jci.insight.131468.
- Neeraj Vij; Taehong Min; Manish Bodas; Aakruti Gorde; Indrajit Roy; Neutrophil targeted nano-drug delivery system for chronic obstructive lung diseases. Nanomedicine: Nanotechnology, Biology and Medicine 2016, 12, 2415-2427, 10.1016/j.nano.2016.06.008.
- James E. Dahlman; Carmen Barnes; Omar F. Khan; Aude Thiriot; Siddharth Jhunjhunwala; Taylor E. Shaw; Yiping Xing; Hendrik B. Sager; Gaurav Sahay; Lauren Speciner; et al. In vivo endothelial siRNA delivery using polymeric nanoparticles with low molecular weight. Nature Nanotechnology 2014, 9, 648-655, 10.1038/nnano.2014.84.
- Silvano Dragonieri; Giorgio Pennazza; Pierluigi Carratu; Onofrio Resta; Electronic Nose Technology in Respiratory Diseases. Lung 2017, 195, 157-165, 10.1007/s00408-017-9987-3.
- Bhavna; Farhan J. Ahmad; Gaurav Mittal; Gaurav K. Jain; Geena Malhotra; Roop K. Khar; Aseem Bhatnagar; Nano-salbutamol dry powder inhalation: A new approach for treating broncho-constrictive conditions. European Journal of Pharmaceutics and Biopharmaceutics 2009, 71, 282-291, 10.1016/j.ejpb.2008.09.018.
- Nashwa El-Gendy; Eric M. Gorman; Eric J. Munson; Cory J. Berkland; Budesonide nanoparticle agglomerates as dry powder aerosols with rapid dissolution.. Journal of Pharmaceutical Sciences 2009, 98, 2731-46, 10.1002/jps.21630.
- Joscelyn C. Mejías; Krishnendu Roy; In-vitro and in-vivo characterization of a multi-stage enzyme-responsive nanoparticle-in-microgel pulmonary drug delivery system.. Journal of Controlled Release 2019, 316, 393-403, 10.1016/j.jconrel.2019.09.012.
- Samarjit Dey; Anirban Bhattacharjee; Debasis Pradhan; Prithwis Bhattacharyya; Daniala Chhunthang; Akash Handique; Angkita Barman; Mohd Yunus; How useful is extravascular lung water measurement in managing lung injury in intensive care unit?. Indian Journal of Critical Care Medicine 2017, 21, 494-499, 10.4103/ijccm.ijccm_40_17.
- Nobile, S.; Gnocchini, F.; Pantanetti, M.; Battistini, P.; Carnielli, V.P.; The importance of oxygen control reaffirmed: Experience of ROP reduction at a single tertiary-care NICU. . J. Pediatr. Ophtalmol. Strab. 2014, 51, 112-115, .
- Farkhondeh Chaharband; Narsis Daftarian; Mozhgan Rezaei Kanavi; Reyhaneh Varshochian; Maliheh Hajiramezanali; Parisa Norouzi; Ehsan Arefian; Fatemeh Atyabi; Rassoul Dinarvand; Trimethyl chitosan-hyaluronic acid nano-polyplexes for intravitreal VEGFR-2 siRNA delivery: Formulation and in vivo efficacy evaluation. Nanomedicine: Nanotechnology, Biology and Medicine 2020, 26, 102181, 10.1016/j.nano.2020.102181.
- J. M. Lakawicz; W. J. Bottega; J. L. Prenner; H. F. Fine; An analysis of the mechanical behaviour of a detaching retina. Mathematical Medicine and Biology: A Journal of the IMA 2014, 32, 137-161, 10.1093/imammb/dqt023.
- Joseph M. Lakawicz; William J. Bottega; Howard F. Fine; Jonathan L. Prenner; On the mechanics of myopia and its influence on retinal detachment. Biomechanics and Modeling in Mechanobiology 2019, 19, 603-620, 10.1007/s10237-019-01234-1.
- Fernando J. Cabrera; Daniel C. Wang; Kartik Reddy; Ghanashyam Acharya; Crystal S. Shin; Challenges and opportunities for drug delivery to the posterior of the eye.. Drug Discovery Today 2019, 24, 1679-1684, 10.1016/j.drudis.2019.05.035.
- Asaf Shoval; Amos Markus; Zhixin Zhou; Xia Liu; Rémi Cazelles; Itamar Willner; Yossi Mandel; Anti-VEGF-Aptamer Modified C-Dots-A Hybrid Nanocomposite for Topical Treatment of Ocular Vascular Disorders.. Small 2019, 15, e1902776, 10.1002/smll.201902776.
- Fitsum Feleke Sahle; Sangyoon Kim; Kumar Kulldeep Niloy; Faiza Tahia; Cameron V. Fili; Emily Cooper; David J. Hamilton; Tao L. Lowe; Nanotechnology in regenerative ophthalmology. Advanced Drug Delivery Reviews 2019, 148, 290-307, 10.1016/j.addr.2019.10.006.
- Nicola Di Trani; Priya Jain; Corrine Ying Xuan Chua; Jeremy S. Ho; Giacomo Bruno; Antonia Susnjar; Fernanda P. Pons-Faudoa; Antons Sizovs; R. Lyle Hood; Zachary W. Smith; et al. Nanofluidic microsystem for sustained intraocular delivery of therapeutics. Nanomedicine: Nanotechnology, Biology and Medicine 2019, 16, 1-9, 10.1016/j.nano.2018.11.002.
