Rheumatoid arthritis (RA) is a systemic inflammatory joint disease affecting about 1% of the population worldwide. Current treatment approaches do not ensure a cure for every patient. Moreover, classical regimens are based on nontargeted systemic immune suppression and have significant side effects. Biological treatment has advanced considerably but efficacy and specificity issues remain. Gene therapy is one of the potential future directions for RA therapy, which is rapidly developing. Several gene therapy trials done so far have been of moderate success, but experimental and genetics studies have yielded novel targets. As a result, the arsenal of gene therapy tools keeps growing. Currently, both viral and nonviral delivery systems are used for RA therapy.
- rheumatoid arthritis
- gene therapy
- AAV
- miRNA
- siRNA
- lncRNA
- circRNA
- monoclonal antibodies
- gene delivery
- CIA
- clinical trials
1. Introduction
2. Targets for RA Gene Therapy
2.1. Anti- and Proinflammatory Cytokines
2.2. Matrix Degradation Enzymes
3. RA Gene Therapy Delivery
3.1. Local or Systemic Delivery
3.2. Ex Vivo or In Vivo
3.3. RNA Therapeutics
3.3.1. RNA Interference
3.3.2. Circular RNA
3.3.3. Nonviral RNA Delivery Vehicles
3.4. (Epi)Genome Editing
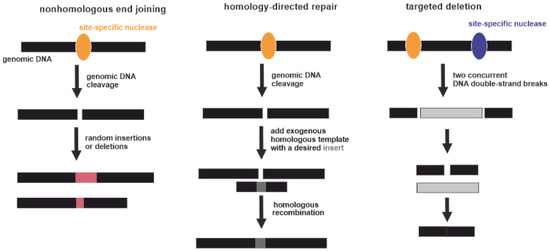
3.5. Viral Vectors
they are some of the most commonly used gene therapy vehicles. The first clinical trials of gene therapeutic drugs based on retroviruses were carried out 29 years ago [130]. This was an attempt to insert a “correct” copy of the adenosine deaminase gene into the T-lymphocyte genome of patients with severe combined immunodeficiency mediated by a lack of this enzyme. At that time, retroviruses were the only feasible way to modify the host DNA, because their life cycle includes integration into the host genome. Initially, constructs based on the murine leukemia virus (MuLV) were used as vectors. MuLV inserts the target fragment predominantly at the beginning of random transcription units of the host genome, which can lead to dysregulation of the expression of the oncogene/oncosupressor. This has led to the development of malignancies during retroviral gene therapy [131,132]. Such consequences can be avoided using a specific type of retrovirus: self-inactivating lentivirus [133]. However, the risk of malignancies cannot be fully excluded. Thus, the introduction of lentivirus-based constructs into clinical practice is limited and must include a very careful risk/benefit analysis, which is unlikely to be favorable in the case of RA.
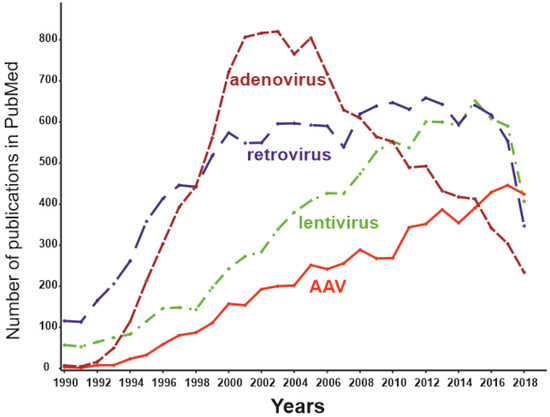
therapy vectors [134]. Adeno-associated viruses (AAVs) are among such persisting benign viruses. Currently, vectors created on the basis AAVs are recognized as safe and effective, being one of the most promising methods of gene therapy [135]. The number of publications dedicated to AAVs peaked in 2018 (Figure 4), while the use of other virus vectors has been declining. AAVs are small non-enveloped viruses with a dsDNA genome of about 4800 nucleotides. AAVs are not able to replicate independently and require several proteins complemented by adenoviruses, hence the name [124]. Viral ssDNA genome penetrates the nucleus after host cell infection. Most of the viral genomes do not integrate into the genome of the host cell, remaining in the form of episomes. At the same time, approximately 0.1% of the naturally occurring AAV genomes can be integrated into the genome, with integration taking place at a specific location in the 19th human chromosome. Recombinant AAV episomal DNA lacks the ability to integrate into host genomic DNA. Thus, the genotoxic effect inevitable for retroviral constructs has not been observed using AAV vectors [136]. As the AAV genome exists as an epichromosome, it can be removed by the cell, as well as diluted by successive cell division. Thus, the number of viruses in dividing cells is gradually reduced. This limits the use of AAVs in gene therapy to short-term gene delivery and transfection of slowly dividing cells. In the mid-1990s, it was shown that transgenes delivered to rat neural tissue by an AAV vector was stably expressed for at least several months [137]. A decade later Mochizuki et al. demonstrated correction of hyperphenylalaninemia by AAV-mediated gene transfer in mice with phenylketonuria. Interestingly, within 2 weeks of treatment, all animals had no symptoms of the disease, and the therapeutic effect persisted for 40 weeks only in male mice [138]. The main cellular components of the synovium and the most promising targets for RA gene therapy are FLSs and macrophage-like synoviocytes (MLSs) [139]. Due to their epichromosomal nature, AAVs are “diluted” during cell division. The lifespan of MLSs is estimated to be 3–6 months [140], and they can be maintained for several months in vitro [141]. Thus, the key cells involved in the pathogenesis of RA are not fast-dividing cells; therefore, AAV-based technology is generally appropriate for RA gene therapy.
DNA fragments over 4000 nucleotides in length cannot be efficiently transduced [142]. This can be circumvented by using a dual vector system [143]. Dysferlin-deficient mice were treated systemically by two AAV vectors to provide reconstitution of the full 6.5 kb dysferlin cDNA. The treatment provided long-term gene expression and functional improvement for dysferlinopathy [144]. However, the clinical usability of such systems remains questionable. Currently, 13 natural AAV serotypes are known and are termed AAV1–AAV13 [145]. Noteworthy,
by definition, a serotype (serological type) is a serologically distinguishable strain of a virus. Strikingly, AAV6, AAV10, and AAV11 in these terms represent one serotype [146]. The serological features of the more than 100 other AAV strains are not currently known; hence, these strains are termed AAV variants. These hundreds of uncharacterized variants potentially can represent novel serotypes. Known AAV serotypes have tropism to different human tissues. For example, in mice, the primary targets for AAV serotypes 1, 2, 5, 6, 7, and 9 are the liver and hindlimbs. AAV8 and 9 transduce tissues more ubiquitously in comparison with other serotypes. A transgene delivered byAAV9 was detected, besides other targets, in tissues protected by additional barriers, such as the testes and brain [147]. According to other authors, AAV1 is targeted to the nervous system and skeletal muscle; AAV2 has broad tropism; AAV5 is targeted to retinal pigmented epithelia and photoreceptors; AAV6 to skeletal muscle and lung; AAV8 to the liver, skeletal muscle, heart, and pancreas; and AAV9 to the liver, skeletal muscle, and lung [124]. An AAV directed evolution technique allows to select from a variety of synthesized variants the one most suitable for a particular application. Moreover, the wild type AAV encoding capsid can be modified to create gene libraries and then select the most appropriate novel variants [135]. For example, a novel recombinant AAV-PHP.B transfers genes after systemic administration throughout the CNS with an efficiency that is at least 40-fold greater than that of the natural viruses [148]. Hemphill et al. tested the ability of AAV1–AAV6 to transduce equine joint tissue explants (cartilage or synovium) and cultured chondrocytes or synoviocyte monolayers [149]. Tissue explants were considered as a model more closely related to in vivo transduction. AAV2 transduced around 80% of cells in cartilage explants, whereas AAV1 and AAV4 demonstrated near-zero transduction efficiency. Thus, AAV is a promising vector for RA gene therapy.
The prevalence of total IgG against different AAV serotypes among healthy humans is in the range of 37–72% [150]. Recently 34%, 83 out of 243 healthy blood donors, were found to be AAV-positive according to the PCR [151]. In most people, the immune response emerges 2 years after infection by wild-type AAV [143]. Interestingly, the longest reported transgene expression caused by a parenterally administered AAV is 10 years [152]. Thus, it is likely that the intended therapeutic effect may be exerted, even after the immune response to the vector. However, when using AAVs as vectors, the possible immune response should be considered, especially in the case of repeated administrations. Nonetheless, this circumstance may be resolved by repeated use of vectors constructed from different AAV serotypes [153]. Other methods are also possible. For example, synthetic variants obtained by structure-guided antigenic epitope evolution allows evading neutralizing antibodies (NAbs) [154]. Using an AAV capsid from other host species may also hide the virus from the pre-existing NAbs [155]. Empty capsids can act as bait for antibodies, promoting the distribution of the target vector [156]. Recently, it was demonstrated that synthetic vaccine particles encapsulating rapamycin coadministered with AAV vectors prevent the induction of anti-capsid humoral and cell-mediated responses [157]. Thus, immunogenicity does not seem to be an unpassable barrier to the use of AAV-based gene therapy. Many other attempts to create safe and efficient viral vectors have been made. Alphaviruses,
flaviviruses, picornaviruses, rhabdoviruses, and paramyxoviruses are among the viruses that have been tried as delivery vehicles in viral-based gene therapy [158]. However, these approaches have not yet been applied to RA therapy, and most of them do not appear as the best choice in the case of RA. Viral vectors described in this manuscript are summarized in the Table 2.
4. Future Directions
apparatus of human somatic cells. This is achieved by transferring nucleic acids in the form of DNA or RNA into the patient’s body. It is noteworthy that these molecules do not necessarily transfer genetic information directly. Apart from well-described transient expression and genome editing tools, it may be, for example, a new class of “smart therapeutics” represented by nucleic acids with enzymatic activity. RNA-cleaving DNAzymes can cut mRNA, directly causing target gene silencing [161]. Additionally, potential drugs may regulate microRNA processing, thereby initiating target gene mRNA interference [162]. Mammalian articular cartilage is considered to have poor regenerative properties [163]. At the
same time, other vertebrates can regenerate not only joints but al so entire limbs [164]. Recently, it was demonstrated that human cartilage has a hitherto unappreciated innate regenerative capacity associated with microRNA expression [165]. MiRNA-21, miRNA-31, and miRNA-181c are upregulated during limb regeneration in zebrafish, bichir, and axolotl [164]. A significant gradient of miRNA expression (proximal to distal) was observed for miRNA-21 and miRNA-31 [165]. Moreover, miRNA-181c was enriched in osteoarthritis-affected cartilage as compared with healthy cartilage. Currently, it is unclear if these regeneration mechanisms can be used in RA treatment.
universe may be much more complex than it was supposed to be. For example, modified ribonucleotides in siRNA evade immune response without loss of RNAi activity [166]. Recently, increased concentrations of adenosine deaminases acting on RNA (ADARs) were identified in the synovium of patients with RA [167]. Adenosine deamination results in the adenosine-to-inosine (A-to-I) editing event in mRNA and can modulate the level of protein expression. Cathepsin S is an extracellular matrix degradation enzyme involved in antigen presentation. The noncoding part of its mRNA demonstrated an increased number of A-to-I events in samples from patients with RA. Moreover, cathepsin S expression in the synovium was significantly increased in RA samples [167]. Interestingly, both ADAR expression and the individual adenosine RNA editing rate of cathepsin S decreased after treatment only in patients with a good clinical response to TNFis. Thus, the measurement of ADAR concentration may be a good marker for the prognosis of TNFi efficiency. As ADAR expression and activity are increased after stimulation with TNF-α, ADAR inhibitors may be considered a potential RA treatment.
5. Conclusions
disease. It is likely that the existing therapy is aimed at correcting the consequences of an unknown cause. At the same time, emerging data allows looking at the problem from a different perspective. Currently, there are dozens of potential RA gene therapy targets and comparing their significance is difficult. Risk/benefit and, potentially, cost/benefit ratios are not favorable for many classical gene therapy approaches, and the issue of complete cure at one shot or repeated administration of a transgene is important. Therefore, despite many trials and advancements, RA gene therapy remains in its infancy as compared to monogenic diseases.
This entry is adapted from the peer-reviewed paper 10.3390/biomedicines8010009
