3.3. Light Aldehydes
Acetaldehyde is a key intermediate in the oxidation of hydrocarbons and alcohols, especially ethanol, which is increasingly being used as a fuel for automobiles. However, it is one of the most abundant toxic oxidative emissions from the combustion of biofuels [
140,
141], and its atmospheric reaction generates several secondary pollutants [
142,
143]. Thus, the pyrolysis mechanism study of this intermediate at various reaction conditions can help to understand the overall combustion mechanism of hydrocarbons and alcohol-based fuels [
71]. In this regard, several authors reported on the degradation kinetics and combustion chemistry of acetaldehyde. For instance, Sivaramakrishnan et al. [
144] conducted a study on the theoretical calculations of acetaldehyde (C
2H
4O) and ethoxide (C
2H
5O) potential energy surfaces (PES) and updated the kinetic model of acetaldehyde pyrolysis. The study revealed C–C bond fission with a minor contribution from the roaming mechanism to form CH
4 and CO as the main decomposition pathway of acetaldehyde during high-temperature processing. The model developed by the author incorporates a master equation for the analysis of H + CH
2CHOH as a primary reaction mechanism for the removal of CH
2CHOH. The governing H-abstraction route at the aldehydic site was found to form a carbonyl radical (R
n–CO), which quickly further decomposes to an alkyl radical (R
n) and CO. Based on that, there is a general implication that the low-temperature oxidation of the generic C
n aldehyde degraded to C
n−1 alkyl radicals [
26].
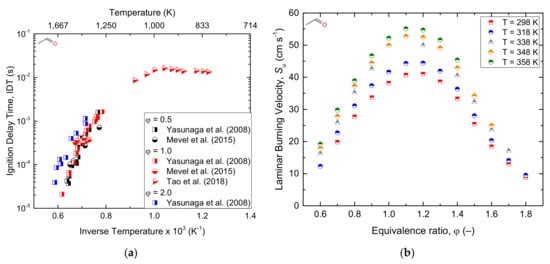
To better understand the combustion parameters, the ignition delay times of acetaldehyde behind shock tube waves under ranges of reaction conditions were reported by Mével et al. [
72]. additionally, a sensitivity analysis, energy release, and rate of production were conducted, indicating four important elementary reactions (Equations (11)–(14)) taking place during acetaldehyde pyrolysis and oxidation:
In the end, due to the huge differences observed during the research, the authors recommended the need for new experimental and detailed numerical studies. Tao et al. [
145] reported nearly 40 species in laminar and premixed flames of acetaldehyde. Christensen et al. [
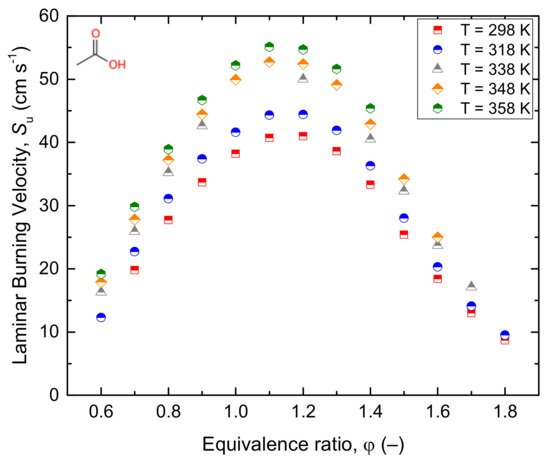
146] studied the laminar burning velocities at atmospheric pressure and different initial temperatures. Similarly, Christensen and Konnov [
147] reported the laminar burning velocity of diacetyl and the updated sub-mechanism model of acetaldehyde and CH
3CO in their model. Halstead et al. [
148] studied the kinetic development of acetaldehyde in the perspective of the cool flame feature and suggested models containing 14 steps. From the study, acetyl was found to play a significant role in the chain-branching process through CH
3CO → CH
3CO
3 → CH
3CO
3H → CH
3CO
2 + OH. The theoretical work reported by Felton et al. [
149] and the detailed kinetic model developed by Cavanagh et al. [
150] supported the result of Halstead et al. [
148].
Nevertheless, Gibson et al. [
151] came up with another cool flame phenomena of acetaldehyde to be processed by CH
3OOH (CH
3 → CH
3OO → CH
3OOH → CH
3O + OH). On the other hand, the study conducted by Kaiser et al. [
152] revealed the radical decomposition reaction (Equation (15)) and O
2 addition to acetyl (Equation (16)) as the main determining step of the chain-branching process.
Recently, researchers [
27,
153] have developed a kinetic model for the low-temperature oxidation of acetaldehyde, as well as C
3 and C
4 aldehydes. Zhang et al. [
40] studied the oxidation of acetaldehyde under a wide range of conditions and revealed CH
3OO, CH
3OOH, and HOOCOCHO as the main oxidation products. Besides, H-abstracting agents were found to be processed by H, OH, HO
2, CH
3, O
2, CH
3COOO, CH
3OO, and CH
3O. At the lean condition, OH was found to be the most important H-abstracting agent. It was concluded that CH
3COOOH and CH
3OOH are the main decomposition pathways of acetaldehyde oxidation via the chain-branching reaction, and the reactions related to methyl oxidation were reported to be very sensitive to CH
3OO and CH
3OOH under the studied conditions [
40]. Bentz et al. [
154] studied the shock tube thermal decomposition of CH
3CHO and CH
3CHO + H at a temperature within 1250–1650 K and a pressure range of 1–5 bar. Combining their results and the low-temperature data from other studies, the authors reported the acetaldehyde rate constant expression as K = 6.6 × 10
−18 exp (−800 K/T) cm
3 s
−1 for the temperature range of 300–2000 K. Moreover, Hidaka et al. [
155] studied the pyrolysis of acetaldehyde oxidation behind reflected shockwave tubes using single-pulse methods. The study considered different fuel concentrations (2.0% CH
3CHO, 4.0% CH
3CHO, and 5.0% CH
3CHO) diluted with Ar under the temperature range of 1000–1700 K and pressure of 1.2 and 3.0 atm. The (Equations (17)–(19)) reactions were mentioned to be the most important initiation reactions and (Equations (20) and (21)) as the most crucial reactions responsible for acetaldehyde pyrolysis.
Similarly, Ernst et al. [
156] conducted acetaldehyde pyrolysis behind reflected shockwaves under a temperature range of 1350–1650 K. The results revealed the decomposition as a first-order reaction with a rate constant expression of K = 1.2 × 10
16 exp (−81.74 kcal/RT) s
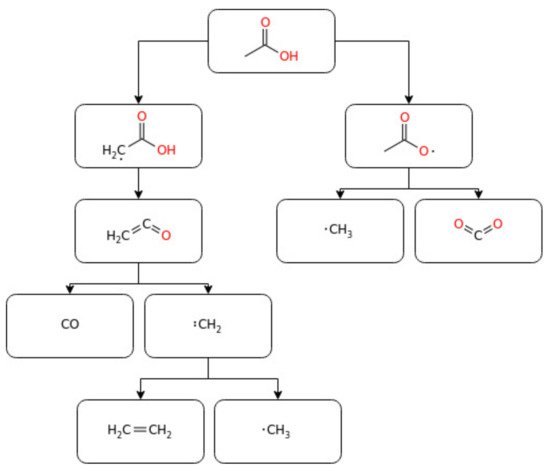
−1. The experimental ignition delay time and laminar burning velocity data of acetaldehyde oxidation reported in the current literature are shown below in . Furthermore, reports a simplified schematization of the oxidation pathway of acetaldehyde.
Figure 12. Ignition delay time (
a) and laminar burning velocity (
b) of the acetaldehyde/air mixture under different conditions. Note that laminar burning velocity measurements refer to the data reported by Christensen and Konnov [
14] exclusively.
Figure 13. A simplified reaction pathway for acetaldehyde oxidation.