Covid-19 has the potential to cause severe damage to many tissues, including systemic inflammatory response syndrome (SIRS), acute respiratory disease syndrome (ARDS), multiorgan involvement, and shock. One of the most feared complications are thromboembolic events which lead to severe clinical phenotypes: worsening of pulmonary conditions, oxygen desaturation, and acute respiratory distress. Management choices should be considered according to the critical or chronic setting. Main pathophysiological mechanisms underlying thrombosis from Covid-19 are discussed, including metabolic derangements and hormonal factors.
- thromboembolism
- COVID-19 infection
- pathophysiology
- obesity
- insulin
- metabolism
1. Venous Thromboembolism Diagnosed in Covid-19 Patients and Management
1.1. Clinical Diagnoses of Venous Thromboembolism
We are aware of a small number of studies that have reported the incidence of VTE in patients with Covid-19 [1][2]. A Chinese retrospective study worked in this direction by reporting a percentage of 25% (20 out of 81) of patients admitted to the ICU in whom an accident of VTE occurred. Of note, none of the patients had been managed with the use of VTE prophylaxis drugs [3]. Klok et al. [4], in a multicenter study that included 184 patients with severe Covid-19, recorded the percentage of 31% (95% confidence interval: 20% to 41%) of patients who developed an accident of VTE. All patients had been treated with VTE prophylaxis drugs, although the authors noted underdosing in 2 of the 3 recruiting centers (81). It cannot be ruled out that VTE may go undiagnosed and unrecognized in patients with severe Covid-19. This is an important aspect during the clinical evolution of Covid-19, as ARDS in those patients is potentially the cause of a vicious circle involving hypoxia, pulmonary vasoconstriction, pulmonary hypertension and right ventricular failure. The onset of pulmonary embolism is an additional clinical event that often cannot be resolved.
1.2. Medical Treatment in the Acute Setting and on Discharge
The use of drugs for systemic anticoagulation has been a crucial point for the treatment of VTE. The choice of the most suitable drug requires clinical considerations factoring in comorbidities and possible impairment of renal or hepatic function, haematological disorders such as thrombocytopenia or that of the gastrointestinal system. Multiple therapies may be considered including a change in the anticoagulant pharmacological treatment in progress during the hospitalization period, due to the critical state at hand to a more suitable regiment at the time of discharge to adapt to the convalescence period.
This pharmacological management used in critically ill hospitalized patients with VTE, in whom the administration of parenteral anticoagulants such as, for example, UFH may be preferred because it has good pharmacodynamic and pharmacokinetic requirements without known drug interactions with experimental Covid-19 therapies. It is important to underline that the effects of the therapy are obtained after a longer period with the administration of UFH (unfractionated heparin) because they depend on the achievement of a therapeutic aPTT. In addition, healthcare workers are more exposed to the risk of contamination due to frequent blood draws. Therefore, these aspects argue for the preferential choice in the administration of LMWH in patients in whom invasive procedures are not planned.
The use of oral anticoagulation with DOAC is certainly advantageous because it does not involve the need for monitoring, easing the planning of discharge, and for the outpatient management of the patient. It is possible that the worsening of the patient’s clinical condition including, for example, the deterioration of the respiratory function in patients with Covid-19, may necessitate a switch between these classes of medications. Patients who are expected to be discharged are advised to use DOAC or LMWH to limit contact with healthcare personnel because controls for INR monitoring as for vitamin K antagonists (VKA) are not required.
1.3. Pulmonary Embolism in Covid-19 Patients: Stratification and Choice of Therapy
The vast majority of patients with Covid-19 and symptoms referable to acute DVT should be treated at home with anticoagulant therapy whenever possible. However, there are a number of patients for whom hospitalization is necessary to initiate acute endovascular techniques such as local fibrinolysis or embolectomy and patients with refractory symptoms [5]. Patients at medium and high risk for VTE may need the input of a multidisciplinary team to manage their PE [6][7][8][9]. The available data are limited but report a lower mortality with the use of advanced therapies for the treatment of VTE. The caveat here is the limited data which marginally demonstrates lower mortality from routine use of advanced VTE therapies. Therefore, severe respiratory failure sustained by a PE treated with the use of catheter-guided therapies during the pandemic should be limited to the most critical patients. Likewise, the indiscriminate use of filters positioned in the inferior vena cava should be avoided [10]. The placement of a filter is recommended in restricted cases for patients experiencing recurrent PE despite optimal anticoagulant treatment or with clinically significant VTE in the context of absolute contraindications to anticoagulant therapy [11]. IVC filters do not exonerate patients from the use of anticoagulation which must be restored immediately, with the dosage of the drug gradually increased whilst considering the potential to bleed. Patients with intermediate-risk and hemodynamically stable [6][12][13][14] should be closely monitored and managed with anticoagulant therapy.
Rescue systemic fibrinolysis should be considered in patients with progressive deterioration, either systemically or by transcatheter approach. Instead, for patients with evident haemodynamic instability and high-risk PE [6][12][13][14] systemic fibrinolysis is indicated with two possible options, a percutaneous catheter approach or if this is contraindicated, systemic fibrinolysis.
1.4. Management of Antithrombotic Drugs in Patients with SARS-CoV-2 Infection and Critical Illness
Patients with severe SARSCoV-2 infection and critical illness have a higher risk of VTE. Many factors contribute to the development of VTE which is often associated with worsening clinical condition and rapid deterioration of the patient. First, they have haemostatic imbalance coupled with a systemic inflammatory state and are forced into long immobility because they are assisted with mechanical ventilation. Second, the placement of central venous catheters contributes to increasing the risk of VTE in the ICU and infections [15][16][17]. Third, nutritional shortage and liver dysfunction can interfere with the production of clotting factors [18]. A modification of the pharmacokinetics is to be considered in critically ill patients who require dosage adjustment of anticoagulant drugs [19], caused by factors related to the absorption, metabolism, and renal or hepatic elimination of these drugs to be considered with the possible presence of organ dysfunction.
Administration of anticoagulant therapy by systemic parenteral infusion is recommended in majority of patients whereby Covid-19 with acute thrombosis occurs. In these cases, the use of UFH can be used based on expected protocols, or in patients with deterioration of renal function. In the absence of an application for an urgent procedure, the use of LMWH is a reasonable alternative [20]. Anticoagulation is of particular importance in patients undergoing ECMO (extracorporeal membrane oxygenation) for whom continuous monitoring of anticoagulant drugs is required to maintain the patency of the circuit especially when hemodynamic stability is ensured by lower blood flow.
Available data are scarce to establish complication rates in SARS-CoV-2 patients, but thrombosis and haemorrhage rates can be high reaching the rate of 53% and 16% respectively as reported in other populations with respiratory failure [21]. Even more limited are the data from the surveillance of ECMO patients in critical condition for SARSCoV-2 infection. Two studies reported results with very high mortality which was five out of six patients in one series and three out of three in another [22][23]. To date concerns related to recommend target anticoagulation therapy for critically patients with overt disease from severe SARS Cov 2 infection requiring ECMO exists due to lack of insufficient data [24].
1.5. Risk Stratification Scores to Drive Pharmacological Prophylactic Treatment
Patients with Covid-19 and a lung infection who required hospitalization following clinical complications have an increased risk of VTE [6][25]. The use of prophylactic anticoagulation and the dosage are recommended by current guidelines and the position papers of professional societies, predominantly based on large randomized clinical trials that have reported a benefit concerning outcomes after hospitalization [6][26][27][28].
Several studies have reported that early initiation of appropriate prophylactic anticoagulation reduces the risk of VTE in critically ill patients [29][30][31]. The Caprini score, the International Registry of Medical Prevention on Venous Thromboembolism (IMPROVE) model and the Padua model are risk stratification tools useful for VTE risk assessment [32][33][34][35][36][37]. For example, the use of the Padua model was reported by Wang et al. highlighting that 40% of patients admitted to hospital for Covid-19 and in critical condition were at high risk of VTE. However, the study lacks data on the use either of prophylaxis for VTE or about the incidence of VTE [38]. VTE drug prophylaxis is indicated for patients admitted to the hospital for COVID-19 who have respiratory failure or comorbidities such as active cancer and heart failure [39]. This therapy should also be extended to patients with prolonged bed rest and who require a prolonged admission to the intensive care unit unless patients have specific contraindications.
In January 2020 a WHO report recommended interim guidance for daily prophylaxis with low molecular weight heparin (LMWH) or unfractionated subcutaneous heparin (UFH) to be administered twice daily [40]. In patients forced into prolonged immobilization, in whom prophylactic pharmacological treatment is contraindicated, the use of mechanical prophylaxis with intermittent pneumatic compression of the VTE should be considered [40][41]. The administration of pharmacological prophylaxis for VTE should be very rigorous to avoid episodes of failed doses which has been a common occurrence leading to worse outcomes [42]. Pregnant patients with Covid-19 deserve particular consideration and must be meticulously evaluated. This population of individuals have a higher risk of VTE that also extends to the postpartum period [43][44]. To date, there are insufficient data to draw definitive conclusions on pregnant women who have been admitted to hospital with Covid-19 even if a greater risk of VTE is conceivable. VTE risk stratification for this patient population helps consider the use of pharmacological thromboprophylaxis, especially if they have other risk factors for VTE. Further research is needed to establish the appropriate prophylactic dosage of anticoagulants based on the weight of pregnant patients [45].
It is important to point out that the problem of extended prophylaxis with LMWH [46] or direct oral anticoagulants (DOAC) involves patients once discharged from the hospital after the resolution of the acute episode of Covid-19 [47][48][49][50]. In discharged patients, although the risk of VTE is reduced, the occurrence of bleeding events is increased [51][52]. There is no detailed information that clarifies this aspect of Covid patient management. However, it would be useful to employ individualized risk stratification models for thrombotic and haemorrhagic episodes when considering prolonged, 45-day prophylactic anticoagulant therapy. The latter is indicated in discharged patients with a high risk of VTE, with reduced mobility or with active neoplastic disease, who may have a high D-dimer > 2 times the upper limit, but who are at low risk of bleeding [49][53][54].
2. Hypotheses of Thrombosis Generation and Pathophysiological Mechanisms
2.1. Covid-19 and Disseminated Intravascular Coagulation
Patients with clinically critical Covid-19 have a greater chance of developing disseminated intravascular coagulation (DIC) [55][56] which is common to many diseases that evolve with severity [57]. It has not yet been demonstrated whether Covid-19 can lead to the development of a DIC with mechanisms intrinsic to SARS Cov 2 that cause direct activation of coagulation cascades. The hypercoagulable state caused by DIC leads to an activation of the tissue factor pathway with consequent platelet consumption and a consistent bleeding diathesis. It is characterized by raised fibrin degradation products (FDPs) which are commonly elevated in Covid-19 patients suffering from thrombosis.
The ISTH DIC score calculator is commonly used to establish the diagnosis of DIC [58]. The worsening of blood coagulation in patients with a critical picture of Covid-19 is carried out by monitoring platelet counts, PT, D-dimer, and fibrinogen. The continuous monitoring of the parameters listed above is the first step for correct identification of the DIC and starting its management. The appearance of bacterial superinfections is not uncommon and must be promptly treated with aggressive antibiotic therapy. The sudden prophylaxis with LMWH can decrease the formation of thrombin and reduce its consumption, to be useful in modulating the evolution of DIC. Preliminary results are based on limited data which, however, have shown an effective benefit with the prophylactic administration of LMWH [49][52]. Regarding the discontinuation of long-acting single or dual antiplatelet therapy (DAPT), it is recommended in most patients when the diagnosis of DIC is suspected, administration of antiplatelet medications in patients who have suffered from a recent ACS or who have implanted stents are left alone. For patients with a moderate or severe clinical condition of COVID-19 in whom dual antiplatelet therapy is indicated because they have received PCI in the past 3 months or have suffered from recent MI, even in the presence of a suspected or confirmed diagnosis of DIC but without noticeable bleeding, therapy must be individually tailored. In the absence of strong evidence confirming a DIC it is reasonable to continue the DAPT, if the patient has a platelet count of at least 50,000. The administration of a single antiplatelet drug is indicated when the platelet count is between 25,000 and 50,000 while an interruption of antiplatelet therapy is recommended for a platelet count of less than 25,000. However, rigidity in the application of these indications is not necessary and therapy must be adapted according to the patient’s condition, increasing or decreasing the antiplatelet dose and paying close attention to the possible onset of thrombotic complications or bleeding. Parenteral systemic fibrinolysis with the consequent elimination of thrombi scattered in the organs favors the resolution of DIC.
Patients with Covid-19 rarely suffer from clinically evident bleeding. However, significant blood loss is common in patients who develop DIC often when septic coagulopathy is associated [59]. If bleeding occurs, treatment consists of transfusion of blood products.
First, the use of platelet concentrate is useful in patients who have DIC with active bleeding to maintain platelet counts greater than 50 × 109/L or in patients who have a high risk of bleeding because they require more invasive procedures to maintain a platelet count greater than 20 × 109/L. Second, the administration of fresh frozen plasma (15 to 25 mL/kg) in patients who experience active bleeding and whose PT or aPTT values are altered (PTT> 1.5 times normal) or who have a decrease in fibrinogen (<1.5 g/L). Third, the administration of concentrated fibrinogen or cryoprecipitate for patients in whom the values of fibrinogen are low and persistent over time (<1.5 g/L) and the administration of prothrombin complex concentrate where transfusion of fresh frozen plasma is not possible. There are currently no indications for the routine use of tranexamic acid in patients with DIC associated with COVID-19 [60].
2.2. Direct Viral Damage and Endothelitis-Driven Inflammatory Reaction
Concerns about the triggering of changes in the hemostatic process may be also related to the direct effect of SARS-CoV-2 viral particles. Varga et al. demonstrated the presence of viral elements within endothelial cells due to the expression of ACE2 receptors. Also, inflammatory cells were demonstrated, corroborating evidence of a direct viral involvement towards endothelitis development, also noted in several organs [61].
Preclinical analyses have also demonstrated the successful infection of organoids by viruses confirming their tropism for endothelial cells. Investigators used clinical-grade soluble human ACE2 to achieve the infection [62][63]. Endothelial damage further contributes to inflammatory cell infiltrate in the lungs worsening the respiratory parameters [64].
2.3. Covid-19-Associated Hyperinflammatory Syndrome (cHIS)
A further hypothesis for thrombosis generation is related to cytokine storm precipitating an initially controlled inflammation in the more complicated SIRS [57], as emerged from the observation of similar cases of individuals infected with other viral diseases such as HIV, Zika, Chikungunya, and Ebola [65][66][67].
As a consequence of dysregulated immune reactions, hypercoagulable states are established due to continued inflammation. Macrophages activation syndrome (MAS) and cytokines released play major roles by activating a pro-inflammatory cascade [68][69] (Figure 1).
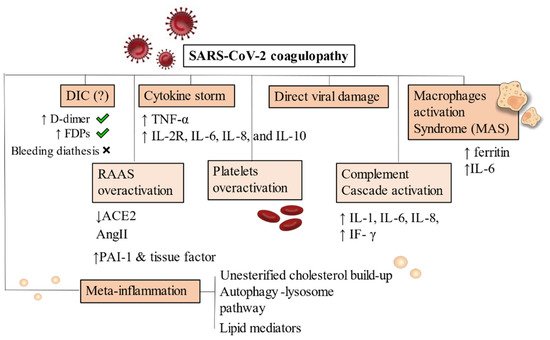
Figure 1. Pathophysiology of SARS-CoV-2 coagulopathy. DIC has been frequently noticed in Covid-19 severe patients but bleeding diathesis was a less present feature. Cytokine storm and inflammatory-driven thrombogenesis is the most known hypothesis, due to IL-2R, IL-6, IL-8, and IL-10 cascade generation. A direct viral damage has also been acknowledged to start endothelitis and endothelial damage. MAS is instead an added mechanism present on an already compromised immune condition where hyperferritinaemia and increase in IL-6 production are pathognomonic of macrophages overactivation. RAAS system, complement and platelets also drive uncontrollable responses by generating hypercytokinemia and dysregulating fibrinolysis. Increased levels of PAI-1 and tissue factor have been demonstrated. Meta-inflammation is another possible trigger for coagulopathy. Obesity, hyperinsulinemia and metabolic syndrome are strong risk factors for severity of infection in hospitalized patients. Primary conditions developing after viral infection are depicted with a darker background. RAAS overactivation, platelets and complement activation are mainly secondary mechanisms, thus they appear in a lighter background. Abbreviations: DIC: disseminated intravascular coagulation, FDPs: fibrin degradation products, ACE: angiotensin converting enzyme, AngII: angiotensin II, PAI-1: plasminogen activator inhibitor, IF-γ: interferon- γ, TNF-α: tumor necrosis factor-α, IL-6, IL-8, IL-10: interleukin 6, interleukin 8, interleukin 10. IL-2R: interleukin 2 receptor.
Recently, Webb et al. [70] proposed several criteria to diagnose the Covid-19-associated hyperinflammatory syndrome (cHIS) which has been associated with coagulopathy and hypercytokinemia leading, among the other complications, to mechanical ventilation and death in most severe cases. Criteria for the diagnosis have not been established at the time of writing. Thus, the authors proposed six of them: fever, hyperferritinaemia from macrophages activation syndrome, haematological dysfunction (N/L, neutrophil to lymphocyte ratio), hepatic injury (lactate dehydrogenase or aspartate aminotransferase), D-dimer to diagnose coagulopathy and cytokinaemia (C-reactive protein, interleukin-6, or triglycerides levels).
3. Inflammatory Cascade: The Bridge between Metabolic Derangements and Thrombogenesis
The Immunologic Dialogue of Cytokines, T Cells, and Checkpoint Proteins Accelerating Thrombosis in Atherosclerotic Lesions
Endothelial dysfunction, which is the first recognizable step of Covid-19 thrombogenesis from an inflammatory cause, is first seen in atherosclerosis development. Metabolic derangements play main roles as atherogenic stimuli such as dyslipidemias, hypertension, and obesity which stimulate a series of changes in lesion-prone vascular endothelium, towards a vasoconstrictor, prothrombotic, proliferative, inflammatory phenotype. Pro-inflammatory cytokines, oxidized lipoproteins (ox-LDL), and advanced glycation end products (AGE), as well as disturbed blood flow associated with reciprocating, low shear stress [71] lead to endothelial activation (EA). These signals transduce mainly via the pleiotropic transcription factor TF nuclear factor-κB (NF-κB), resulting in a coordinated program of genetic regulation within the endothelial cell, among which chemokines, surface expression of adhesion molecules (e.g., vascular cell adhesion molecule-1 [VCAM-1), and prothrombotic agents such as tissue factor, von Willebrand Factor and plasminogen activator inhibitor (PAI-1) [72][73].
According to the Virmani classification, the intimal xanthoma is the initial detectable small lesion constituted by foam cells with intracellular- and not extracellular- lipid accumulation.
The endothelial expression of adhesion molecules VCAM-1 and ICAM-1 drives leukocytes and platelet recruitment. Also, smooth muscle cells coming from the tunica media, migrate to the plaque attracted by the PDGF stimulus and start to produce other inflammatory cytokines, such as IL-1 and IL-6.
An interesting role is one of the activated T cells, mainly CD4+: the recognition of proteic antigens presented by macrophages on MHC type II molecules triggers a response for which Th1 cells produce IFN-ɣ, in turn activating macrophages and overly stimulating the synthesis of pro-atherogenic inflammatory cytokines. In this process, costimulatory and coinhibitory receptors on T cells direct T-cell function and determine T-cell fate. These co-signaling molecules are divided into pro-atherogenic and anti-atherogenic ones. It is known, in fact, at least in the pre-clinical setting, that CTLA-4 blocking accelerates atherosclerosis development and decreases luminal patency [74][75].
The evolution of the plaque sequentially favours the formation of pathological intimal thickening (PIT) lesions consisting of macrophage foam cells and small extracellular lipid pools. The lesions eventually progress to fibrous cap atheroma (FCA) presenting with a fibro-adipous cap and necrotic cores from extracellular lipids, cholesterol crystals, and calcifications due to the accumulation of osteocalcin, osteopontin, and Bone Morphogenetic Protein (BMP). The last stages of atherogenesis involve mainly complicated lesions with fissuration and erosion resulting in thrombus which provokes a critical reduction of blood flow, thus occluding the vessel.
During plaque progression, regulators of immune cells divide into pro-atherogenic immune checkpoint proteins CD28–CD80/86, OX40–OX40L, CD137–CD137L and CD30–CD30L and anti-atherogenic ones CTLA-4–CD80/CD86, PD-1–PD-L1/2, ICOS–ICOSL, GITR–GITRL, CD27–CD70 and TIM proteins [76]. CTLA-4 is mainly expressed on Tregs and activated CD4+ and CD8+ T-cells but also on monocytes and activated B cells. Their anti-atherogenic role has been demonstrated by preclinical and clinical studies, without a clear elucidation of the mechanism of pathogenesis.
4. Meta-Inflammation: Alterations of Metabolism Leading to Thrombus Generation
4.1. Obesity, Metabolic Syndrome and Dysregulated Lipid Metabolism
Obesity, metabolic syndrome and type 2 diabetes mellitus (T2DM) are among the best-known risk factors for both thrombosis and severe Covid-19 infection since they promote a state of chronic inflammation and impaired fibrinolysis. Meta-inflammation, i.e., chronic inflammation driven by chronically altered metabolic pathways presents with increased release of cytokines from adipocytes. Consequently, a contribution comes from macrophages (recruited in inflamed sites) and transient hypoxia which is proper of visceral fat rather than subcutaneous one.
Macrophages in particular, extend the secretion of cytokines ensuring the vascular endothelium is stimulated towards a pro-thrombotic state with upregulation of adhesion molecules and downregulation of coagulation inhibitory proteins.
Consequently, thrombin generation is increased, platelets are further activated and TNF-α and IL-6 increase the expression of tissue factor, promoting the hypercoagulable state.
Chronic inflammation is also associated with dysregulation of endogenous anticoagulant mechanisms, including tissue factor pathway inhibitor, antithrombin, and the protein C anticoagulation system.
The second important mechanism is impaired fibrinolysis. Expression of PAI-1 is markedly upregulated in visceral adipose tissue and elevated levels of PAI-1, together with tissue plasminogen activator (tPA), have been found in severe Covid-19 patients [77]. 118 hospitalized patients and 30 healthy controls were evaluated through laboratory, imaging, and clot-lysis assays. Elevated levels of those markers were associated with worse respiratory status, enhancement of ex-vivo clot lysis, and mortality. Also, strong correlations were demonstrated with neutrophil counts and circulating calprotectin, a neutrophil activation marker. Plasma levels of PAI-1 are also elevated in patients with obesity or metabolic syndrome. TNF-α is one of the key regulators of PAI-1 expression since it acts by stimulating its expression [78]. This mechanism suggests a clear link between antifibrinolytic activity and obesity-associated chronic inflammatory state.
Recent meta-analyses and case-control studies underlined the association between obesity and severe COVID-19. Also, they showed that obese patients had worse prognoses than non-obese patients (OR, 2.31; 95% CI, 1.3–4.12) [79]. In general, dyslipidemias were also associated with severe COVID-19 (relative risk (RR), 1.39; 95% CI, 1.03–1.87; p = 0.03) [80].
4.2. Hyperinsulinemia Contributes to Both Impaired Fibrinolysis and Hypercoagulable States
As a causative factor, hyperinsulinemia is related to all the metabolic conditions associated with poor outcomes from Covid-19 infection. Again, the primary mechanism is impaired fibrinolysis, due to increased PAI-1 expression [81]. It, therefore, produces a decrease in plasminogen activator activity.
The impairment of fibrinolysis by insulin is completely independent of glycemia while hyperglycemia produces effects on coagulation which is not related to insulinemia. Impaired fibrinolysis is consistently found in T2DM patients [82] (Figure 2).
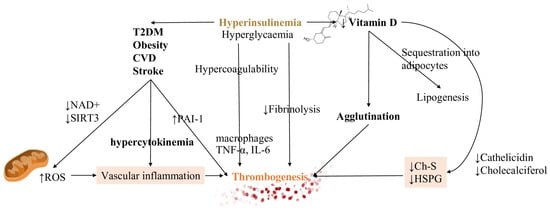
Figure 2. Hyperinsulinemia, CVD and vitamin D have a strong impact on homeostatic equilibrium. Both hyperinsulinemia (depicted in bold and colored font, in order to emphasize it) and hyperglycemia generate states of increased coagulation and decreased fibrinolysis. By driving the development of CVD, diabetes mellitus and obesity, they contribute to the inflammatory substrate of cytokines. They increase the ROS production due to the damage in decreasing both NAD+ and reduced glutathione (GSH). A reduction in vitamin D, due to sequestration into the adipocytes, leads to decreased levels of ChS and HSPG, regulators of RBCs deformation, increasing cells agglutination. These mechanisms are all responsible for thrombosis initiation. Vascular inflammation and decreasing levels of Ch-S and HSPG are represented with colored backgrounds being the main actors of thrombogenesis trigger. Also, thrombogenesis, the main effect, is outlined with a different color too. Abbreviations: Ch-S: cholesterol sulfate, HSPG: heparan sulfate proteoglycans, NAD+: nicotinamide adenine dinucleotide, PAI-1 plasminogen activator inhibitor type 1, ROS: reactive oxygen species, T2DM: type 2 diabetes mellitus, SIRT3: sirtuin 3, TNF-α: tumor necrosis factor-α, IL-6: interleukin 6.
Hyperglycemia instead stimulates clotting factors synthesis from the liver and inflammatory reaction by increasing the secretion of IL-6. Biochemical effects of hyperinsulinemia are several and entail β-oxidation and ketolysis inhibition [83].
Due to the major depletion of nicotinamide adenine nucleotides (NAD+) by glucose oxidation rather than beta-oxidation and ketolysis, NAD+ availability for mitochondrial deacetylase sirtuin 3 (SIRT3) activity decreases [77]. This process, which is NAD+ dependent, normally increases the production of NADPH reduce oxidised glutathione (GSSG) to reduced glutathione (GSH) [84]. Therefore, the final result is that insulin increases mitochondrial production of reactive oxygen species (ROS), also via generation of ceramides [85]. ROS production is then responsible for further vascular inflammation. (Figure 1).
4.3. Vitamin D Metabolism and Its Interference in the Inflammatory Pathway
Vitamin D hydroxylation requires magnesium which is notably depleted in hyperinsulinemic conditions. Other mechanisms responsible for decreased Vitamin D levels are increased renal excretion, reduced intracellular levels, and sequestration into adipocytes. The last process is related to lipogenesis which is already increased in hyperinsulinemic patients [84]. Consequences of reduced activation are decreased levels of cholesterol sulfate (Ch-S), heparan sulfate proteoglycans (HSPG), and cathelicidin synthesis. The final result is the promotion of agglutination and, subsequently, of thrombosis. An explanation for this can be found in the action of HSPGs as potent anticoagulant molecules which also buffer glycation damage. Ch-S is instead implicated in RBCs shape deformation for traveling through vascular spaces of reduced caliber [84][85][86].
4.4. Extrahepatic Vitamin K Insufficiency and Dependency of Coagulation Factors
Evidence of altered laboratory values of vitamin K have been demonstrated in severe Covid-19 cases. Other than coagulation factors, several other molecules, among which matrix Gla protein (MGP), a potent inhibitor of soft tissue calcification and elastic fibers degradation, are dependent on vitamin K.
The proposed mechanism by Janssen et al. [87], implicates that following elastic fibers degradation due to SARS-CoV-2 proteolysis, partially degraded elastic fibers show increased polarity which drives an increase in the calcium content of elastic fibers.
With an increase in MGP synthesis, vitamin K is utilized for further molecule processing (MGP carboxylation) which decreases circulating vitamin K levels and increases damage to lung tissues. As a consequence, endothelial protein S is not sufficiently carboxylated and shifts the coagulation equilibrium towards thrombogenesis [88].
An important proof that this mechanism may be deranged in Covid-19 thromboembolic events, is that diabetes, hypertension, and CVD, which are correlated with worse outcomes, are conditions related to chronic elastic fibers pathology.
4.5. Hormonal Factors Contributing to Covid-19 Thrombotic Complications
Gender differences in Covid-19 outcomes and cardiovascular diseases have raised many issues regarding the hormonal asset of patients and its interference with coagulopathy development.
Endothelial dysfunction is found in the elderly, smokers, and patients with metabolic alterations such as dyslipidemias, T2DM and arterial hypertension [89][90].
An interesting mechanism is the presence of receptors on megakaryocytes and platelet membranes for both estrogen and androgens. Also, testosterone has been observed to increase the secretion of endothelial nitric oxide eNO, a potent inhibitor of platelet activation [91][92].
The function of estrogens, in women, is also directed to regulate and enhance platelet function according to the ovarian cycle release of hormones. On the contrary, lack of testosterone i.e., hypogonadism in the elderly produces a less protective effect on hypercoagulation [93]. Consequently, this aspect should be taken into account when evaluating older patients for possible thrombosis.
This entry is adapted from the peer-reviewed paper 10.3390/metabo11060341
References
- Xie, Y.; Wang, X.; Yang, P.; Zhang, S. COVID-19 Complicated by Acute Pulmonary Embolism. Radiol. Cardiothorac. Imaging 2020, 2, e200067.
- Danzi, G.B.; Loffi, M.; Galeazzi, G.; Gherbesi, E. Acute pulmonary embolism and COVID-19 pneumonia: A random association? Eur. Hear. J. 2020, 41, 1858.
- Cui, S.; Chen, S.; Li, X.; Liu, S.; Wang, F. Prevalence of venous thromboembolism in patients with severe novel coronavirus pneumonia. J. Thromb. Haemost. 2020, 18, 1421–1424.
- Klok, F.A.; Kruip, M.J.; Van der Meer, N.J.; Arbous, M.S.; Gommers, D.A.; Kant, K.M.; Kaptein, F.H.; van Paassen, J.; Stals, M.A.; Huisman, M.V.; et al. Incidence of thrombotic complications in critically ill ICU patients with COVID-19. Thromb. Res. 2020, 191, 145–1457.
- Vedantham, S.; Goldhaber, S.Z.; Julian, J.A.; Kahn, S.R.; Jaff, M.R.; Cohen, D.J.; Magnuson, E.; Razavi, M.K.; Comerota, A.J.; Gornik, H.L.; et al. ATTRACT Trial Investigators. Pharmacomechanical catheter-directed thrombolysis for deep-vein throm-bosis. N. Engl. J. Med. 2017, 377, 2240–2252.
- Konstantinides, S.V.; Meyer, G.; Becattini, C.; Bueno, H.; Geersing, G.J.; Harjola, V.P.; Huisman, M.V.; Humbert, M.; Jennings, C.S.; Jiménez, D.; et al. ESC Scientific Document Group. 2019 ESC Guidelines for the diagnosis and management of acute pulmonary embolism developed in collaboration with the European Respiratory Society (ERS). Eur. Heart J. 2020, 41, 543–603.
- Chodakowski, J.D.; Courtney, D.M. Pulmonary embolism critical care update: Prognosis, treatment, and research gaps. Curr. Opin. Crit. Care 2018, 24, 540–546.
- Barnes, G.D.; Kabrhel, C.; Courtney, D.M.; Naydenov, S.; Wood, T.; Rosovsky, R.; Rosenfield, K.; Giri, J.; National PERT Consortium Research Committee. Diversity in the pulmonary embolism response team model: An organizational survey of the National PERT Consortium Members. Chest 2016, 150, 1414–1417.
- Rosovsky, R.; Zhao, K.; Sista, A.; Rivera-Lebron, B.; Kabrhel, C. Pulmonary embolism response teams: Purpose, evidence for efficacy, and future research directions. Res. Pr. Thromb. Haemost. 2019, 3, 315–330.
- Bikdeli, B.; Chatterjee, S.; Desai, N.R.; Kirtane, A.J.; Desai, M.M.; Bracken, M.B.; Spencer, F.A.; Monreal, M.; Goldhaber, S.Z.; Krumholz, H.M. Inferior vena cava filters to prevent pulmonary embolism: Systematic review and meta-analysis. J. Am. Coll. Cardiol. 2017, 70, 1587–1597.
- Kearon, C.; Akl, E.A.; Ornelas, J.; Blaivas, A.; Jimenez, D.; Bounameaux, H.; Huisman, M.; King, C.S.; Morris, T.A.; Sood, N.; et al. Antithrombotic therapy for VTE disease: CHEST guideline and expert panel report. Chest 2016, 149, 315–352.
- Piazza, G. Advanced Management of Intermediate- and High-Risk Pulmonary Embolism: JACC Focus Seminar. J. Am. Coll. Cardiol. 2020, 76, 2117–2127.
- Giri, J.; Sista, A.K.; Weinberg, I.; Kearon, C.; Kumbhani, D.J.; Desai, N.D.; Piazza, G.; Gladwin, M.T.; Chatterjee, S.; Kobayashi, T.; et al. Interventional Therapies for Acute Pulmonary Embolism: Current Status and Principles for the Development of Novel Evidence: A Scientific Statement From the American Heart Association. Circulation 2019, 140, e774–e801.
- Jimenez, D.; Bikdeli, B.; Marshall, P.S.; Tapson, V. Aggressive Treatment of Intermediate-Risk Patients with Acute Symptomatic Pulmonary Embolism. Clin. Chest Med. 2018, 39, 569–581.
- Pellegrini, J.A.S.; Rech, T.H.; Schwarz, P.; de Oliveira, A.C.T.; Vieceli, T.; Moraes, R.B.; Sekine, L.; Viana, M.V. Incidence of venous thromboembolism among patients with severe COVID-19 requiring mechanical ventilation compared to other causes of respiratory failure: A prospective cohort study. J. Thromb. Thrombolysis 2021, 1–11.
- Moll, M.; Zon, R.L.; Sylvester, K.W.; Chen, E.C.; Cheng, V.; Connell, N.T.; Fredenburgh, L.E.; Baron, R.M.; Cho, M.H.; Woolley, A.E.; et al. VTE in ICU Patients With COVID-19. Chest 2020, 158, 2130–2135.
- Di Minno, A.; Ambrosino, P.; Calcaterra, I.; Di Minno, M.N.D. COVID-19 and Venous Thromboembolism: A Meta-analysis of Literature Studies. Semin. Thromb. Hemost. 2020, 46, 763–771.
- Mansfield, A.; Tafur, A.; Vulih, D.; Smith, G.; Harris, P.; Ivy, S. Severe hepatic dysfunction is associated with venous thromboembolic events in phase 1 clinical trials. Thromb. Res. 2015, 136, 1169–1173.
- Smith, B.S.; Yogaratnam, D.; Levasseur-Franklin, K.E.; Forni, A.; Fong, J. Introduction to Drug Pharmacokinetics in the Critically III Patient. Chest 2012, 141, 1327–1336.
- Aryal, M.R.; Gosain, R.; Donato, A.; Pathak, R.; Bhatt, V.R.; Katel, A.; Kouides, P. Venous Thromboembolism in COVID-19: Towards an Ideal Approach to Thrombo-prophylaxis, Screening, and Treatment. Curr. Cardiol. Rep. 2020, 22, 52.
- Sklar, M.C.; Sy, E.; Lequier, L.; Fan, E.; Kanji, H.D. Anticoagulation practices during venovenous extracorporeal membrane oxy-genation for respiratory failure. A systematic review. Ann. Am. Thorac. Soc. 2016, 13, 2242–2250.
- Yang, F.; Yu, T.; Du, R.; Fan, G.; Liu, Y.; Liu, Z.; Xiang, J.; Wang, Y.; Song, B.; Gu, X.; et al. Clinical course and risk factors for mortality of adult in patients with COVID-19 in Wuhan, China: A retrospective cohort study. Lancet 2020, 395, 1054–1062.
- Yang, X.; Yu, Y.; Xu, J.; Shu, H.; Xia, J.; Liu, H.; Wu, Y.; Zhang, L.; Yu, Z.; Fang, M.; et al. Clinical course and outcomes of critically ill patients with SARS-CoV-2 pneumonia in Wuhan, China: A single-centered, retrospective, observational study. Lancet Respir. Med. 2020, 8, 475–481.
- Extracorporeal Life Support Organization. ELSO Guidelines for Cardiopulmonary Extracorporeal Life Support. v1.4. 2017. Available online: (accessed on 1 April 2020).
- Dobesh, P.P.; Trujillo, T.C. Coagulopathy, Venous Thromboembolism, and Anticoagulation in Patients with COVID-19. Pharmacother. J. Hum. Pharmacol. Drug Ther. 2020, 40, 1130–1151.
- Schunemann, H.J.; Cushman, M.; Burnett, A.E.; Kahn, S.R.; Beyer-Westendorf, J.; Spencer, F.A.; Rezende, S.M.; Zakai, N.A.; Bauer, K.A.; Dentali, F.; et al. American Society of Hematology 2018 guidelines for management of ve-nous thromboembolism: Prophylaxis for hospitalized and nonhospitalized medical patients. Blood Adv. 2018, 2, 3198–3225.
- Lopes, R.D.; de Barros ESilva, P.G.M.; Furtado, R.H.M.; Macedo, A.V.S.; Ramacciotti, E.; Damini, L.P.; Bronhara, B.; Cavalcanti, A.B.; Rosa, R.G.; Azevedo, L.C.P.; et al. Randomized Clinical Trial to Evaluate a Routine Full Anticoagulation Strategy in Patients with Coronavirus Infection (SARS-CoV2) Admitted to Hospital: Rationale and Design of the ACTION (AntiCoagulaTlon cOroNavirus)-Coalition IV Trial. Am. Heart J. 2021.
- National Institute for Health and Clinical Excellence. NICE Clinical Guideline 92: Venous Thromboembolism: Reducing the Risk. Available online: (accessed on 30 March 2020).
- Lenchus, J.D.; Biehl, M.; Cabrera, J.; De Moraes, A.G.; Dezfulian, C. In-Hospital Management and Follow-Up Treatment of Venous Thromboembolism: Focus on New and Emerging Treatments. J. Intensiv. Care Med. 2017, 32, 299–311.
- Encke, A.; Haas, S.; Kopp, I. The Prophylaxis of Venous Thromboembolism. Dtsch. Aerzteblatt Online 2016, 113, 532–538.
- Spyropoulos, A.C.; Levy, J.H.; Ageno, W.; Connors, J.M.; Hunt, B.J.; Iba, T.; Levi, M.; Samama, C.M.; Thachil, J.; Giannis, D.; et al. Scientific and Stand-ardization Committee communication: Clinical guidance on the diagnosis, prevention, and treatment of venous throm-boembolism in hospitalized patients with COVID-19. J. Thromb. Haemost. 2020, 18, 1859–1865.
- Albertsen, I.E.; Nielsen, P.B. Searching for High-Risk Venous Thromboembolism Patients Using Risk Scores: Adding to the Heap or Closing a Gap? Thromb. Haemost. 2018, 118, 1686–1687.
- Zhou, H.; Hu, Y.; Li, X.; Wang, L.; Wang, M.; Xiao, J.; Yi, Q. Assessment of the Risk of Venous Thromboembolism in Medical Inpatients using the Padua Prediction Score and Caprini Risk Assessment Model. J. Atheroscler. Thromb. 2018, 25, 1091–1104.
- Stuck, A.; Spirk, D.; Schaudt, J.; Kucher, N. Risk Assessment Models for Venous Thromboembolism in Acutely Ill Medical Patients: A Systematic Review. Thromb. Haemost. 2017, 5, 769–770.
- Liu, X.; Liu, C.; Chen, X.; Wu, W.; Lu, G. Comparison between Caprini and Padua risk assessment models for hospitalized medi-cal patients at risk for venous thromboembolism: A retrospective study. Interact. Cardiovasc. Thorac. Surg. 2016, 23, 538–543.
- Moumneh, T.; Riou, J.; Douillet, D.; Henni, S.; Mottier, D.; Tritschler, T.; Le Gal, G.; Roy, P. Validation of risk assessment models predicting venous thromboembolism in acutely ill medical inpatients: A cohort study. J. Thromb. Haemost. 2020, 18, 1398–1407.
- Mlaver, E.; Lynde, G.C.; Gallion, C.; Sweeney, J.F.; Sharma, J. Development of a Novel Preoperative Venous Thromboembolism Risk Assessment Model. Am. Surg. 2020, 86.
- Wang, T.; Chen, R.; Liu, C.; Liang, W.; Guan, W.; Tang, R.; Tang, C.; Zhang, N.; Zhong, N.; Li, S. Attention should be paid to venous thromboembolism prophylaxis in the management of COVID-19. Lancet Haematol. 2020, 7, e362–e363.
- Hunt, B.J. Hemostasis at Extremes of Body Weight. Semin. Thromb. Hemost. 2018, 44, 632–639.
- World Health Organization. Clinical Management of Severe Acute Respiratory Infection When Novel Coronavirus (2019-nCoV) Infection Is Suspected. INTERIM Guidance 28 January 2020. Available online: (accessed on 7 April 2020).
- Liew, N.C.; Alemany, G.V.; Angchaisuksiri, P.; Bang, S.M.; Choi, G.; De Silva, D.; Hong, J.M.; Lee, L.; Li, Y.J.; Rajamoney, G.N.; et al. Asian venous thromboembolism guidelines: Updated recommendations for the prevention of venous thromboembolism. Int. Angiol. 2016, 36, 1–20.
- Popoola, V.O.; Tavakoli, F.; Lau, B.D.; Lankiewicz, M.; Ross, P.; Kraus, P.; Shaffer, D.; Hobson, D.B.; Aboagye, J.K.; Farrow, N.A.; et al. Exploring the impact of route of administration on medication acceptance in hospi-talized patients: Implications for venous thromboembolism prevention. Thromb. Res. 2017, 160, 109–113.
- Bates, S.M.; Rajasekhar, A.; Middeldorp, S.; McLintock, C.; Rodger, M.A.; James, A.H.; Vazquez, S.R.; Greer, I.A.; Riva, J.J.; Bhatt, M.; et al. American Society of Hematology 2018 guidelines for management of venous thromboembolism: Venous thromboembolism in the context of pregnancy. Blood Adv. 2018, 2, 3317–3359.
- Royal College of Obstetricians and Gynaecologists. Reducing the Risk of Venous Thromboembolism during Pregnancy and the Puerperium. Green-Top Guideline No. 37a. 2015. Available online: (accessed on 1 April 2020).
- Rybstein, M.D.; DeSancho, M.T. Risk factors for and clinical management of venous thromboembolism during pregnancy. Clin. Adv. Hematol. Oncol. 2019, 17, 396–404.
- Akel, T.; Qaqa, F.; Abuarqoub, A.; Shamoon, F. Pulmonary embolism: A complication of COVID 19 infection. Thromb. Res. 2020, 193, 79–82.
- Cohen, A.T.; Harrington, R.A.; Goldhaber, S.Z.; Hull, R.D.; Wiens, B.L.; Gold, A.; Hernandez, A.F.; Gibson, C.M.; APEX Investigators. Extended thromboprophylaxis with betrixaban in acutely ill medical pa-tients. N. Engl. J. Med. 2016, 375, 534–544.
- Arigondam, A.K.; Hakeem, A.R.; Reddy, M.S.; Rela, M. An Evidence-based Protocol for Minimizing Thromboembolic Events in SARS-CoV-2 Infection. Arch. Med. Res. 2021, 52, 252–260.
- Spyropoulos, A.C.; Ageno, W.; Albers, G.W.; Elliott, C.G.; Halperin, J.L.; Hiatt, W.R.; Maynard, G.A.; Steg, P.G.; Weitz, J.I.; Suh, E.; et al. Rivaroxaban for thromboprophylaxis after hospitalization for medical ill-ness. N. Engl. J. Med. 2018, 379, 1118–1127.
- Spyropoulos, A.C.; Lipardi, C.; Xu, J.; Peluso, C.; Spiro, T.E.; De Sanctis, Y.; Barnathan, E.S.; Raskob, G.E. Modified IMPROVE VTE Risk Score and Elevated D-Dimer Identify a High Venous Thromboembolism Risk in Acutely Ill Medical Population for Extended Thromboprophylaxis. TH Open 2020, 4, e59–e65.
- Dentali, F.; Mumoli, N.; Prisco, D.; Fontanella, A.; Di Minno, M.N. Efficacy and safety of extended thromboprophylaxis for medi-cally ill patients. A meta-analysis of randomised controlled trials. Thromb. Haemost. 2017, 117, 606–617.
- Schindewolf, M.; Weitz, J.I. Broadening the Categories of Patients Eligible for Extended Venous Thromboembolism Treatment. Thromb. Haemost. 2019, 120, 14–26.
- Spyropoulos, A.C.; Lipardi, C.; Xu, J.; Lu, W.; Suh, E.; Yuan, Z.; Levitan, B.; Sugarmann, C.; De Sanctis, Y.; Spiro, T.E.; et al. Improved Benefit Risk Profile of Rivaroxaban in a Subpopulation of the MAGELLAN Study. Clin. Appl. Thromb. Hemost. 2019, 25.
- Chi, G.; Goldhaber, S.Z.; Kittelson, J.M.; Turpie, A.G.G.; Hernandez, A.F.; Hull, R.D.; Gold, A.; Curnutte, J.T.; Cohen, A.T.; Harrington, R.A.; et al. Effect of extended-duration thromboprophylaxis on venous thromboembolism and major bleeding among acutely ill hospitalized medical patients: A bivariate analysis. J. Thromb. Haemost. 2017, 15, 1913–1922.
- Zhang, A.; Leng, Y.; Zhang, Y.; Wu, K.; Ji, Y.; Lei, S.; Xia, Z. Meta-analysis of coagulation parameters associated with disease severity and poor prognosis of COVID-19. Int. J. Infect. Dis. 2020, 100, 441–448.
- Iba, T.; Levy, J.H.; Levi, M.; Thachil, J. Coagulopathy in COVID-19. J. Thromb. Haemost. 2020, 18, 2103–2109.
- Gando, S.; Shiraishi, A.; Yamakawa, K.; Ogura, H.; Saitoh, D.; Fujishima, S.; Mayumi, T.; Kushimoto, S.; Abe, T.; Shiino, Y.; et al. Japanese Association for Acute Medicine (JAAM) Focused Outcomes Research in Emergency Care in Acute Respiratory Distress Syndrome, Sepsis and Trauma (FORECAST) Study Group. Role of disseminated intravascular coagulation in severe sepsis. Thromb Res. 2019, 178, 182–188.
- Jackson Chornenki, N.L.; Dwivedi, D.J.; Kwong, A.C.; Zamir, N.; Fox-Robichaud, A.E.; Liaw, P.C.; Canadian Critical Care Translational Biology Group. Canadian Critical Care Translational Biology Group. Identification of hemostatic markers that define the pre-DIC state: A multi-center observational study. J. Thromb. Haemost. 2020, 18, 2524–2531.
- Tang, N.; Bai, H.; Chen, X.; Gong, J.; Li, D.; Sun, Z. Anticoagulant treatment is associated with decreased mortality in severe coro-navirus disease 2019 patients with coagulopathy. J. Thromb. Haemost. 2020.
- Wada, H.; Thachil, J.; Di Nisio, M.; Mathew, P.; Kurosawa, S.; Gando, S.; Kim, H.K.; Nielsen, J.D.; Dempfle, C.E.; Levi, M.; et al. Guidance for diagnosis and treatment of DIC from harmonization of the recommenda-tions from 3 guidelines. J. Thromb. Haemost. 2013.
- Varga, Z.; Flammer, A.J.; Steiger, P.; Haberecker, M.; Andermatt, R.; Zinkernagel, A.S.; Mehra, M.R.; Schuepbach, R.A.; Ruschitzka, F.; Moch, H. Endothelial cell infection and endotheliitis in COVID-19. Lancet 2020, 395, 1417–1418.
- Perico, L.; Benigni, A.; Casiraghi, F.; Ng, L.F.P.; Renia, L.; Remuzzi, G. Immunity, endothelial injury and complement-induced coagulopathy in COVID-19. Nat. Rev. Nephrol. 2021, 17, 46–64.
- Monteil, V.; Kwon, H.; Prado, P.; Hagelkrüys, A.; Wimmer, R.A.; Stahl, M.; Leopoldi, A.; Garreta, E.; Del Pozo, C.H.; Prosper, F.; et al. Inhibition of SARS-CoV-2 Infections in engineered human tissues using clinical-grade soluble human ACE2. Cell 2020, 181, 905–913.e7.
- Mehta, P.; McAuley, D.F.; Brown, M.; Sanchez, E.; Tattersall, R.S.; Manson, J.J. COVID-19: Consider cytokine storm syndromes and immunosuppression. Lancet 2020, 395, 1033–1034.
- Jackson, B.S.; Pretorius, E. Pathological Clotting and Deep Vein Thrombosis in Patients with HIV. Semin. Thromb. Hemost. 2019, 45, 132–140.
- Ramacciotti, E.; Agati, L.B.; Aguiar, V.C.R.; Wolosker, N.; Guerra, J.C.; De Almeida, R.P.; Alves, J.C.; Lopes, R.D.; Wakefield, T.W.; Comerota, A.J.; et al. Zika and Chikungunya Virus and Risk for Venous Thromboembolism. Clin. Appl. Thromb. 2019, 25.
- Smither, S.J.; O’Brien, L.M.; Eastaugh, L.; Woolley, T.; Lever, S.; Fletcher, T.; Parmar, K.; Hunt, B.J.; Watts, S.; Kirkman, E. Haemostatic Changes in Five Patients Infected with Ebola Virus. Viruses 2019, 11, 647.
- Karakike, E.; Giamarellos-Bourboulis, E.J. Macrophage Activation-Like Syndrome: A Distinct Entity Leading to Early Death in Sepsis. Front. Immunol. 2019, 10, 55.
- McGonagle, D.; Sharif, K.; O’Regan, A.; Bridgewood, C. The Role of Cytokines including Interleukin-6 in COVID-19 induced Pneumonia and Macrophage Activation Syndrome-Like Disease. Autoimmun. Rev. 2020, 19, 102537.
- Webb, B.J.; Peltan, I.D.; Jensen, P.; Hoda, D.; Hunter, B.; Silver, A.; Starr, N.; Buckel, W.; Grisel, N.; Hummel, E.; et al. Clinical criteria for COVID-19-associated hyperinflammatory syndrome: A co-hort study. Lancet Rheumatol. 2020, 2, e754–e763.
- Peng, Z.; Shu, B.; Zhang, Y.; Wang, M. Endothelial Response to Pathophysiological Stress. Arter. Thromb. Vasc. Biol. 2019, 39, e233–e243.
- Gimbrone, M.A., Jr.; Garcia-Cardena, G. Endothelial cell dysfunction and the pathobiology of atherosclerosis. Circ Res. 2016, 118, 620–636.
- Milenkovic, J.; Milojkovic, M.; Stoimenov, T.J.; Djindjic, B.; Miljkovic, E. Mechanisms of plasminogen activator inhibitor 1 action in stromal remodeling and related diseases. Biomed. Pap. Med. Fac. Palacky Univ. Olomouc 2017, 161, 339–347.
- Poels, K.; van Leent, M.M.; Reiche, M.E.; Kusters, P.J.; Huveneers, S.; de Winther, M.P.; Mulder, W.J.; Lutgens, E.; Seijkens, T.T. Antibody-Mediated Inhibition of CTLA4 Aggravates Atherosclerotic Plaque In-flammation and Progression in Hyperlipidemic Mice. Cells 2020, 9, 1987.
- Matsumoto, T.; Sasaki, N.; Yamashita, T.; Emoto, T.; Kasahara, K.; Mizoguchi, T.; Hayashi, T.; Yodoi, K.; Kitano, N.; Saito, T.; et al. Overexpression of Cytotoxic T-Lymphocyte-Associated Antigen-4 Prevents Ath-erosclerosis in Mice. Arter. Thromb. Vasc. Biol. 2016, 36, 1141–1151.
- Foks, A.; Kuiper, J. Immune checkpoint proteins: Exploring their therapeutic potential to regulate atherosclerosis. Br. J. Pharmacol. 2017, 174, 3940–3955.
- Zuo, Y.; Warnock, M.; Harbaugh, A.; Yalavarthi, S.; Gockman, K.; Zuo, M.; Madison, J.A.; Knight, J.S.; Kanthi, Y.; Lawrence, D.A. Plasma tissue plasminogen activator and plasminogen activator inhibitor-1 in hospitalized COVID-19 patients. Sci. Rep. 2021, 11, 1–9.
- Engin, A. Endothelial Dysfunction in Obesity. Adv. Exp. Med. Biol. 2017, 960, 345–379.
- Yang, J.; Hu, J.; Zhu, C. Obesity aggravates COVID-19: A systematic review and meta-analysis. J. Med. Virol. 2020, 93, 257–261.
- Hariyanto, T.I.; Kurniawan, A. Dyslipidemia is associated with severe coronavirus disease 2019 (COVID-19) infection. Diabetes Metab. Syndr. Clin. Res. Rev. 2020, 14, 1463–1465.
- Picard, F.; Adjedj, J.; Varenne, O. Diabetes Mellitus, a prothrombotic disease. Ann. Cardiol. D’angeiologie 2017, 66, 385–392.
- Bryk, A.H.; Satała, D.; Natorska, J.; Rąpała-Kozik, M.; Undas, A. Interaction of glycated and acetylated human α2-antiplasmin with fibrin clots. Blood Coagul. Fibrinolysis 2020, 31, 393–396.
- Yazıcı, D.; Sezer, H. Insulin Resistance, Obesity and Lipotoxicity. Adv. Exp. Med. Biol. 2017, 960, 277–304.
- Cooper, I.D.; Crofts, C.A.P.; DiNicolantonio, J.J.; Malhotra, A.; Elliott, B.; Kyriakidou, Y.; Brookler, K.H. Relationships between hyperinsulinaemia, magnesium, vitamin D, thrombosis and COVID-19: Rationale for clinical management. Open Hear. 2020, 7, e001356.
- Hansen, M.E.; Tippetts, T.S.; Anderson, M.C.; Holub, Z.E.; Moulton, E.R.; Swensen, A.C.; Prince, J.T.; Bikman, B.T. Insulin Increases Ceramide Synthesis in Skeletal Muscle. J. Diabetes Res. 2014, 2014, 1–9.
- Dias, I.H.; Ferreira, R.; Gruber, F.; Vitorino, R.; Rivas-Urbina, A.; Sanchez-Quesada, J.L.; Silva, J.V.; Fardilha, M.; de Freitas, V.; Reis, A. Sulfate-based lipids: Analysis of healthy human fluids and cell extracts. Chem. Phys. Lipids 2019, 221, 53–64.
- Janssen, R.; Visser, M.P.J.; Dofferhoff, A.S.M.; Vermeer, C.; Janssens, W.; Walk, J. Vitamin K metabolism as the potential missing link between lung damage and thromboembolism in Coronavirus disease 2019. Br. J. Nutr. 2020, 1–8.
- Iba, T.; Levy, J.H. Sepsis-induced Coagulopathy and Disseminated Intravascular Coagulation. Anesthesiology 2020, 132, 1238–1245.
- Hayden, M.R. Endothelial activation and dysfunction in metabolic syndrome, type 2 diabetes and coronavirus disease 2019. J. Int. Med. Res. 2020, 48.
- Hotta, Y.; Kataoka, T.; Kimura, K. Testosterone Deficiency and Endothelial Dysfunction: Nitric Oxide, Asymmetric Dimethylarginine, and Endothelial Progenitor Cells. Sex. Med. Rev. 2019, 7, 661–668.
- Giagulli, V.A.; Guastamacchia, E.; Magrone, T.; Jirillo, E.; Lisco, G.; De Pergola, G.; Triggiani, V. Worse progression of COVID-19 in men: Is testosterone a key factor? Andrology 2021, 9, 53–64.
- Zhang, Y.; Xiao, M.; Zhang, S.; Xia, P.; Cao, W.; Jiang, W.; Chen, H.; Ding, X.; Zhao, H.; Zhang, H.; et al. Coagulopathy and Antiphospholipid Antibodies in Patients with Covid-19. N. Engl. J. Med. 2020, 382, e38.
- Greinacher, A.; Thiele, T.; Warkentin, T.E.; Weisser, K.; Kyrle, P.A.; Eichinger, S. Thrombotic Thrombocytopenia after ChAdOx1 nCov-19 Vaccination. N. Engl. J. Med. 2021.
