Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is an old version of this entry, which may differ significantly from the current revision.
Subjects:
Andrology
The etiology and pathophysiology genesis mechanisms of benign prostatic hyperplasia and prostate cancer have not yet been fully elucidated. Men’s age is clearly the strongest risk factor. Due to its nature, prostate cancer is considered a heterogeneous disease.
- benign prostatic hyperplasia
- prostate cancer
- testosterone aging
- vascular aging
- inflamm-aging
- amyloidosis
- autophagy
- tumorigenesis
- prostate stagnation
- endothelial dysfunction
- inflammation
- nitric oxide
- oxidative stress
- testosterone replacement therapy
- aging
1. Introduction
Benign prostatic hyperplasia is a major health care expenditure in Australia, and this is trending upwards from $5.3million in 2011 to $35.2million in 2018 [1]. Data from the Australian Institute of Health and Welfare predicted that by 2020, 21 out of 100,000 Australian men will die from prostate cancer [2].
This new perspective and overview analyses and evaluates the biological aspects in the accrued interdisciplinary evidence for prostate ageing degeneration, which could provide us with answers to the etiology and pathophysiology genesis mechanisms. These include testosterone ageing, vascular ageing and inflamm-ageing along with amyloidosis and autophagy in cell regulation and homeostasis, as molecular and cellular evidence of prostate ageing in a degenerative multifactorial heterogeneous disease. These new insights into the early evolution of benign prostatic hyperplasia and prostate cancer would allow us to develop strategies for early prevention and maintaining quality of life for the ageing individual along with substantial medical cost savings.
2. Prevention
This prostate ageing degeneration hypothesis postulates that this triad of testosterone, vascular and inflamm-ageing results in conjoining nitric oxide down-regulating, vascular/endothelial dysfunction and inflammation, with the induction of amyloidosis and autophagy. These are the key etiology and pathophysiology contributors to the prostatic diseases within the evolutionary tumorigenesis microenvironment. It provides a framework for integrating new evidence into a comprehensive concept of a timeline of evolutionary cancer biology of prostate ageing as degenerative, inflammatory and neoplasm progressions of the diseases, for at least a 30 years period (Figure 1). This is a testable hypothesis where biomarkers panel sets can be used to chart the course and range of the ageing degeneration processes.
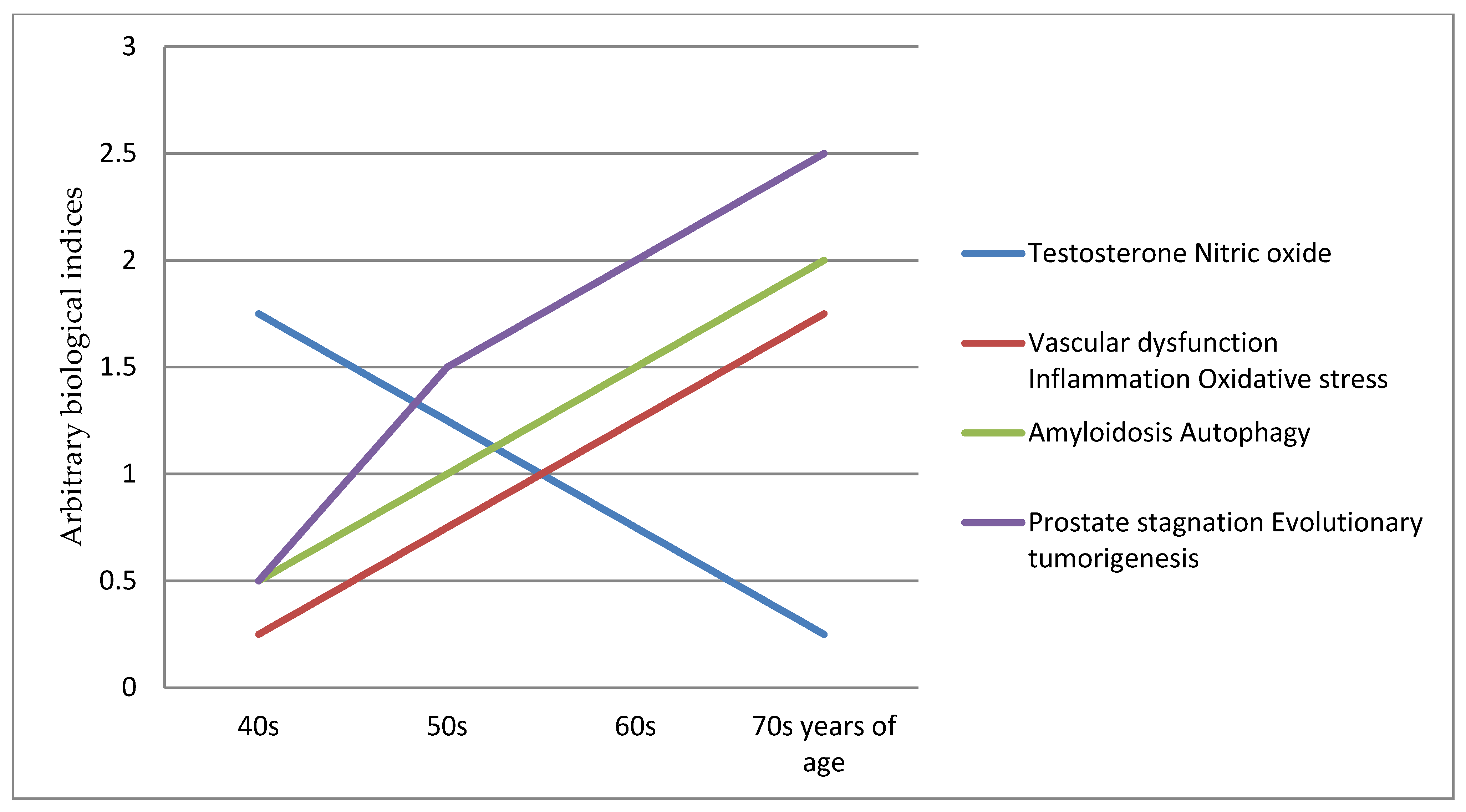 Figure 1. Prostate ageing degeneration hypothesis schematic chart.
Figure 1. Prostate ageing degeneration hypothesis schematic chart.The future paradigm shift involves an emphasis on prevention as early maintenance of healthy vascular function is necessary to preserve cell function, homeostasis and regulation [3][4], thus prolonging the function of the prostate gland, and delaying/avoiding late stage amyloidosis and autophagy dysregulation [5][6][7][8][9]. Other, potential strategies for ameliorating these biological processes of endothelial dysfunction, oxidative stress and inflammation [10][11][12] could be developed. These should also be complemented consistently with a healthy diet and lifestyle [13][14][15][16].
The key in preventive medicine is to prevent the disease from developing by catching or stopping it early; in this case between the fifth (40s) and before the seventh (60s) decade of life [17][18][19]. A potential three-pronged approach can be explored:
Testosterone replacement therapy: long term replacement therapy should be considered to maintain the vascular function; this is a topic of importance that is discussed below as it is an integral part of the prostate ageing degeneration hypothesis.
Nutraceuticals supplement: three supplement combinations [20][21][22][23][24][25][26][27][28][29] are necessary to ameliorate the biological processes of endothelial dysfunction (e.g., l-citrulline [30][31][32], l-arginine [33][34]), oxidative stress and inflammation [35]; publications on this topic are extensive and therefore it is not discussed here.
Prostate stagnation: a standard operating procedure using a patented prostate device US8182503B2 could be developed for regular periodic home use for prostate-rectal drainage [36][37][38], in order to modulate the prostate-stagnation tumorigenesis inflammatory microenvironment [39][40]; this is yet to be fully investigated.
Testosterone replacement therapy has been mired in controversy since its introduction in the 1930s up until to the present day [41][42][43][44][45]. Similarly, findings from the Women’s Health Initiative trial of continuous conjugated equine estrogens alone reported two years later, which suggested prevention of coronary heart disease in women who began hormone replacement therapy at age < 60 years and an overall reduction in breast cancer, were largely ignored [46]. This highlights the “window of opportunity and timing” hypothesis, in which the age of starting hormone replacement therapy affects its risk [47] and with “yin-yang” roles [48]. Nitric oxide is one of the most well studied and recognized female estrogen-induced vasodilators [49][50][51][52].
Important health problems in men such as type 2 diabetes, insulin resistance, erectile dysfunction, benign prostatic hyperplasia and depression have been shown to share common pathological processes, such as endothelial dysfunction and inflammation [53], and in numerous testosterone-related concomitant disease and comorbidities [54][55][56][57][58][59][60][61][62][63][64][65][66][67][68][69][70][71][72][73][74][75][76]. Men with low testosterone levels exhibit increases in cardiovascular disease risk markers [77], micro vascular dysfunction [65][78] and these are associated with higher prostate cancer aggressiveness [79]. Both aggressive and metastatic prostate cancer are influenced by metabolic alterations and cardiovascular disease [80], and the progression in hormone naïve prostate carcinomas correlates with low numbers of vascular vessels [81]. In human surgical specimens, there is evidence that local atherosclerosis of the prostatic artery is significantly associated with prostate size [82]. The use of nicorandil, a nitrate derivative to increase the blood flow, reduces the development of prostatic hyperplasia [83]. Sclerotherapy of the internal spermatic veins restores normal supply of testosterone to the prostate solely via its arterial supply, resulting in a significant decrease in prostatic volume and symptoms [84]. Findings from a study suggest that endothelial dysfunction is associated with lower urinary tract symptoms in men [65]. Experimental testosterone deprivation orchiectomy studies showed induced changes to the prostate of rats, and testosterone replacement therapy was effective in reversing such alterations [85]. In two 60-day studies, canine orchiectomy lowered prostate vascularisation [86] and blood volume [87].
Erectile dysfunction is associated with prostate cancer incidence [88] and vascular function. Sleep fragmentation, benign prostate obstruction and nocturnal frequency could decrease sleep-related erections, reflecting the patient’s relevant erectile function [89]. Long term testosterone therapy improves long term blood circulation of penile arteries, penile length and girth, erectile function, and nocturnal penile tumescence and duration [90]. Low androgen status decreased the nitric oxide production and impaired erectile function of rats [91] and electrical penile erection stimulation in mice induced angiogenesis, cell survival and proliferation, and anti-fibrosis signaling pathways [92].
Nitric oxide serves many biological functions [93][94][95]; ageing is frequently associated with l-arginine deficiency [96][97] and with the menopausal transition in women [98], as a substrate for nitric oxide synthase. Both oral l-citrulline and/or l-arginine supplementation increases nitric oxide bioavailability levels in plasma and tissue [30][31][32][33][34][99]. L-arginine restores doxorubicin-induced vascular dysfunction in cancer treatments by attenuating vascular nitric oxide release and apoptosis [20]. Emerging evidence suggests that increasing nitric oxide bioavailability or endothelial nitric oxide synthase activity activates telomerase and delays endothelial cell senescence [100].
A collaborative analysis of the worldwide data on endogenous hormones and prostate cancer risk, found no risk association [101]. In cancer, the two-concentration (biphasic) hypothesis of nitric oxide has determined that low levels of nitric oxide are cancer promoting, while high levels of nitric oxide are protective against cancer [102][103][104][105]. The acquisition of hypoxia-induced malignant phenotypes in tumor cells is impeded by nitric oxide activation of cyclic guanosine monophosphate signaling [106]. Nitric oxide promotes apoptosis and inhibits autophagy in human liver cancer cells [107]. In autophagy, tripartite motif 36 expression is increased in response to androgen and has a prostate cancer suppressive role [108][109][110]. Loss of testosterone impairs anti-tumor neutrophil function [111].
3. Conclusions
The disease criteria used by the World Health Organization were applies to human biological ageing and it has been found that aging fits the ICD-11 criteria and can be considered a disease; it is included in the extension code for “Ageing-related” (XT9T) in the “Causality” section of the ICD-11 [126].
Tissue degeneration and loss of organ function are features of ageing; conversely, cancer is a state of sustained cellular proliferation and the gain of new functions [127].
Therefore, the most advantageous and best chance strategy is early preventive intervention before tissue damage sets in, and to maintain the vascular function of the ageing prostate gland for as long as possible. Could early, long term testosterone replacement therapy be the Achilles’ heel of prostate cancer? A large preventive trial is warranted to discover the answers to this important question.
This entry is adapted from the peer-reviewed paper 10.3390/medicines8060030
References
- Morton, A.; Williams, M.; Perera, M.; Teloken, P.E.; Donato, P.; Ranasinghe, S.; Chung, E.; Bolton, D.; Yaxley, J.; Roberts, M.J. Management of benign prostatic hyperplasia in the 21st century: Temporal trends in Australian population-based data. BJU Int. 2020, 126, 18–26.
- Australian Institute of Health and Welfare Cancer Data in Australia. Available online: (accessed on 8 June 2021).
- Schaaf, M.B.; Houbaert, D.; Meçe, O.; Agostinis, P. Autophagy in endothelial cells and tumor angiogenesis. Cell Death Differ. 2019, 26, 665–679.
- Peng, Z.; Shu, B.; Zhang, Y.; Wang, M. Endothelial Response to Pathophysiological Stress. Arterioscler. Thromb. Vasc. Biol. 2019, 39, e233–e243.
- Dower, C.M.; Wills, C.A.; Frisch, S.M.; Wang, H.G. Mechanisms and context underlying the role of autophagy in cancer metastasis. Autophagy 2018, 14, 1110–1128.
- Li, X.; He, S.; Ma, B. Autophagy and autophagy-related proteins in cancer. Mol. Cancer 2020, 19, 12.
- Huang, F.; Wang, B.-R.; Wang, Y.-G. Role of autophagy in tumorigenesis, metastasis, targeted therapy and drug resistance of hepatocellular carcinoma. World J. Gastroenterol. 2018, 24, 4643–4651.
- Alvarez-Meythaler, J.G.; Garcia-Mayea, Y.; Mir, C.; Kondoh, H.; LLeonart, M.E. Autophagy Takes Center Stage as a Possible Cancer Hallmark. Front. Oncol. 2020, 10, 586069.
- Lim, J.; Murthy, A. Targeting Autophagy to Treat Cancer: Challenges and Opportunities. Front. Pharmacol. 2020, 11, 590344.
- Hu, Y.; Niu, X.; Wang, G.; Huang, J.; Liu, M.; Peng, B. Chronic prostatitis/chronic pelvic pain syndrome impairs erectile function through increased endothelial dysfunction, oxidative stress, apoptosis, and corporal fibrosis in a rat model. Andrology 2016, 4, 1209–1216.
- Nanni, S.; Grasselli, A.; Benvenuti, V.; Aiello, A.; Pantisano, V.; Re, A.; Gaetano, C.; Capogrossi, M.C.; Bacchetti, S.; Pontecorvi, A.; et al. The role of nuclear endothelial nitric oxide synthase in the endothelial and prostate microenvironments. Horm. Mol. Biol. Clin. Investig. 2011, 5, 91–96.
- Cyr, A.R.; Huckaby, L.V.; Shiva, S.S.; Zuckerbraun, B.S. Nitric Oxide and Endothelial Dysfunction. Crit. Care Clin. 2020, 36, 307–321.
- Ballon-Landa, E.; Parsons, J.K. Nutrition, physical activity, and lifestyle factors in prostate cancer prevention. Curr. Opin. Urol. 2018, 28, 55–61.
- Wilson, K.M.; Mucci, L.A. Diet and Lifestyle in Prostate Cancer. Adv. Exp. Med. Biol. 2019, 1210, 1–27.
- Rock, C.L.; Thomson, C.; Gansler, T.; Gapstur, S.M.; McCullough, M.L.; Patel, A.V.; Andrews, K.S.; Bandera, E.V.; Spees, C.K.; Robien, K.; et al. American Cancer Society guideline for diet and physical activity for cancer prevention. CA Cancer J. Clin. 2020, 70, 245–271.
- Shah, R.; Makarem, N.; Emin, M.; Liao, M.; Jelic, S.; Aggarwal, B. Mediterranean diet components are linked to greater endothelial function and lower inflammation in a pilot study of ethnically diverse women. Nutr. Res. 2020, 75, 77–84.
- Greenwald, P. A favorable view: Progress in cancer prevention and screening. Cancer Prev. 2007, 174, 3–17.
- Greenwald, P.; Dunn, B.K. Do we make optimal use of the potential of cancer prevention? Cancer Prev. II 2009, 181, 3–17.
- Hewitt, K.; Son, J.; Glencer, A.; Borowsky, A.D.; Cooperberg, M.R.; Esserman, L.J. The Evolution of Our Understanding of the Biology of Cancer Is the Key to Avoiding Overdiagnosis and Overtreatment. Cancer Epidemiol. Prev. Biomark. 2020, 29, 2463–2474.
- Li, X.; Gu, J.; Zhang, Y.; Feng, S.; Huang, X.; Jiang, Y.; Xia, Y.; Liu, Y.; Yang, X. l-arginine alleviates doxorubicin-induced endothelium-dependent dysfunction by promoting nitric oxide generation and inhibiting apoptosis. Toxicology 2019, 423, 105–111.
- Chinnapaka, S.; Zheng, G.; Chen, A.; Munirathinam, G. Nitro aspirin (NCX4040) induces apoptosis in PC3 metastatic prostate cancer cells via hydrogen peroxide (H(2)O(2))-mediated oxidative stress. Free Radic. Biol. Med. 2019, 143, 494–509.
- Mokbel, K.; Wazir, U.; Mokbel, K. Chemoprevention of Prostate Cancer by Natural Agents: Evidence from Molecular and Epidemiological Studies. Anticancer Res. 2019, 39, 5231–5259.
- Banez, M.J.; Geluz, M.I.; Chandra, A.; Hamdan, T.; Biswas, O.S.; Bryan, N.S.; Von Schwarz, E.R. A systemic review on the antioxidant and anti-inflammatory effects of resveratrol, curcumin, and dietary nitric oxide supplementation on human cardiovascular health. Nutr. Res. 2020, 78, 11–26.
- Wang, H.; Wang, L.; Xie, Z.; Zhou, S.; Li, Y.; Zhou, Y.; Sun, M. Nitric Oxide (NO) and NO Synthases (NOS)-Based Targeted Therapy for Colon Cancer. Cancers 2020, 12, 1881.
- Tan, B.L.; Norhaizan, M.E. Oxidative Stress, Diet and Prostate Cancer. World J. Mens. Health 2020.
- Termini, D.; Den Hartogh, D.J.; Jaglanian, A.; Tsiani, E. Curcumin against Prostate Cancer: Current Evidence. Biomolecules 2020, 10, 1536.
- Pascual-Geler, M.; Robles-Fernandez, I.; Monteagudo, C.; Lopez-Guarnido, O.; Rodrigo, L.; Gálvez-Ontiveros, Y.; Cozar, J.M.; Rivas, A.; Alvarez-Cubero, M.J. Impact of oxidative stress SNPs and dietary antioxidant quality score on prostate cancer. Int. J. Food Sci. Nutr. 2020, 71, 500–508.
- Jahan, N.; Chowdhury, A.; Li, T.; Xu, K.; Wei, F.; Wang, S. Neferine improves oxidative stress and apoptosis in benign prostate hyperplasia via Nrf2-ARE pathway. Redox Rep. 2021, 26, 1–9.
- Mitsunari, K.; Miyata, Y.; Matsuo, T.; Mukae, Y.; Otsubo, A.; Harada, J.; Kondo, T.; Matsuda, T.; Ohba, K.; Sakai, H. Pharmacological Effects and Potential Clinical Usefulness of Polyphenols in Benign Prostatic Hyperplasia. Molecules 2021, 26, 450.
- Hartman, W.J.; Torre, P.M.; Prior, R.L. Dietary citrulline but not ornithine counteracts dietary arginine deficiency in rats by increasing splanchnic release of citrulline. J. Nutr. 1994, 124, 1950–1960.
- Moinard, C.; Nicolis, I.; Neveux, N.; Darquy, S.; Bénazeth, S.; Cynober, L. Dose-ranging effects of citrulline administration on plasma amino acids and hormonal patterns in healthy subjects: The Citrudose pharmacokinetic study. Br. J. Nutr. 2008, 99, 855–862.
- Schwedhelm, E.; Maas, R.; Freese, R.; Jung, D.; Lukacs, Z.; Jambrecina, A.; Spickler, W.; Schulze, F.; Böger, R.H. Pharmacokinetic and pharmacodynamic properties of oral L-citrulline and L-arginine: Impact on nitric oxide metabolism. Br. J. Clin. Pharmacol. 2008, 65, 51–59.
- Morita, M.; Hayashi, T.; Ochiai, M.; Maeda, M.; Yamaguchi, T.; Ina, K.; Kuzuya, M. Oral supplementation with a combination of L-citrulline and L-arginine rapidly increases plasma L-arginine concentration and enhances NO bioavailability. Biochem. Biophys. Res. Commun. 2014, 454, 53–57.
- Suzuki, T.; Morita, M.; Hayashi, T.; Kamimura, A. The effects on plasma L-arginine levels of combined oral L-citrulline and L-arginine supplementation in healthy males. Biosci. Biotechnol. Biochem. 2017, 81, 372–375.
- Cicero, A.F.G.; Allkanjari, O.; Vitalone, A.; Busetto, G.M.; Cai, T.; Larganà, G.; Russo, G.I.; Magri, V.; Perletti, G.; della Cuna, F.S.R.; et al. Nutraceutical treatment and prevention of benign prostatic hyperplasia and prostate cancer. Arch. Ital. Urol. Androl. 2019, 91, 139–152.
- Capodice, J.L.; Stone, B.A.; Katz, A.E. Evaluation of an At-Home-Use Prostate Massage Device for Men with Lower Urinary Tract Symptoms. Open Urol. Nephrol. J. 2014, 2, 20–23.
- Takashima, J. Prostate Massage Apparatus. U.S. Patent 8,182,503, 22 May 2012.
- Feliciano, A.E. Repetitive prostate massage. In Textbook of Prostatitis; Isis Medical Media: Oxford, UK, 1999; pp. 311–318.
- Belli, C.; Trapani, D.; Viale, G.; D’Amico, P.; Duso, B.A.; Della Vigna, P.; Orsi, F.; Curigliano, G. Targeting the microenvironment in solid tumors. Cancer Treat. Rev. 2018, 65, 22–32.
- Casey, S.C.; Amedei, A.; Aquilano, K.; Azmi, A.S.; Benencia, F.; Bhakta, D.; Bilsland, A.E.; Boosani, C.S.; Chen, S.; Ciriolo, M.R.; et al. Cancer prevention and therapy through the modulation of the tumor microenvironment. Semin. Cancer Biol. 2015, 35, S199–S223.
- Krakowsky, Y.; Morgentaler, A. Risk of Testosterone Flare in the Era of the Saturation Model: One More Historical Myth. Eur. Urol. Focus 2019, 5, 81–89.
- Morgentaler, A.; Traish, A. The History of Testosterone and the Evolution of its Therapeutic Potential. Sex. Med. Rev. 2020, 8, 286–296.
- Khera, M. Controversies with testosterone therapy. Can. J. Urol. 2020, 27, 20–23.
- Yassin, A.; AlRumaihi, K.; Alzubaidi, R.; Alkadhi, S.; Al Ansari, A. Testosterone, testosterone therapy and prostate cancer. Aging Male Off. J. Int. Soc. Study Aging Male 2019, 22, 219–227.
- Kaplan, A.L.; Hu, J.C.; Morgentaler, A.; Mulhall, J.P.; Schulman, C.C.; Montorsi, F. Testosterone Therapy in Men with Prostate Cancer. Eur. Urol. 2016, 69, 894–903.
- Langer, R.D. The evidence base for HRT: What can we believe? Climacteric 2017, 20, 91–96.
- Dehaini, H.; Fardoun, M.; Abou-Saleh, H.; El-Yazbi, A.; Eid, A.A.; Eid, A.H. Estrogen in vascular smooth muscle cells: A friend or a foe? Vascul. Pharmacol. 2018, 111, 15–21.
- Fardoun, M.; Dehaini, H.; Shaito, A.; Mesmar, J.; El-Yazbi, A.; Badran, A.; Beydoun, E.; Eid, A.H. The hypertensive potential of estrogen: An untold story. Vascul. Pharmacol. 2020, 124, 106600.
- Iorga, A.; Cunningham, C.M.; Moazeni, S.; Ruffenach, G.; Umar, S.; Eghbali, M. The protective role of estrogen and estrogen receptors in cardiovascular disease and the controversial use of estrogen therapy. Biol. Sex Differ. 2017, 8, 33.
- Hildreth, K.L.; Ozemek, C.; Kohrt, W.M.; Blatchford, P.J.; Moreau, K.L. Vascular dysfunction across the stages of the menopausal transition is associated with menopausal symptoms and quality of life. Menopause 2018, 25, 1011–1019.
- Somani, Y.B.; Pawelczyk, J.A.; De Souza, M.J.; Kris-Etherton, P.M.; Proctor, D.N. Aging women and their endothelium: Probing the relative role of estrogen on vasodilator function. Am. J. Physiol. Heart Circ. Physiol. 2019, 317, H395–H404.
- Novella, S.; Dantas, A.P.; Hermenegildo, C.; Hellsten, Y. Regulatory Mechanisms of Estrogen on Vascular Ageing. Oxid. Med. Cell. Longev. 2019, 2019, 4859082.
- Hackett, G. Should All Men with Type 2 Diabetes Be Routinely Prescribed a Phosphodiesterase Type 5 Inhibitor? World J. Mens. Health 2020, 38, 271–284.
- Braun, M.; Wassmer, G.; Klotz, T.; Reifenrath, B.; Mathers, M.; Engelmann, U. Epidemiology of erectile dysfunction: Results of the “Cologne Male Survey”. Int. J. Impot. Res. 2000, 12, 305–311.
- Peters, T.J. The Relationship between LUTS and sexual function. Prostate Cancer Prostatic Dis. 2001, 4, S2–S6.
- Vlachopoulos, C.; Ioakeimidis, N.; Terentes-Printzios, D.; Stefanadis, C. The Triad: Erectile Dysfunction—Endothelial Dysfunction - Cardiovascular Disease. Curr. Pharm. Des. 2008, 14, 3700–3714.
- Traish, A.M.; Miner, M.M.; Morgentaler, A.; Zitzmann, M. Testosterone deficiency. Am. J. Med. 2011, 124, 578–587.
- Vlachopoulos, C.; Jackson, G.; Stefanadis, C.; Montorsi, P. Erectile dysfunction in the cardiovascular patient. Eur. Heart J. 2013, 34, 2034–2046.
- Iacono, F.; Prezioso, D.; Ruffo, A.; Illiano, E.; Romis, L.; Di Lauro, G.; Romeo, G.; Amato, B. Testosterone deficiency causes penile fibrosis and organic erectile dysfunction in aging men. Evaluating association among Age, TDS and ED. BMC Surg. 2012, 12, S24.
- Shimizu, S.; Tsounapi, P.; Shimizu, T.; Honda, M.; Inoue, K.; Dimitriadis, F.; Saito, M. Lower urinary tract symptoms, benign prostatic hyperplasia/benign prostatic enlargement and erectile dysfunction: Are these conditions related to vascular dysfunction? Int. J. Urol. 2014, 21, 856–864.
- Di Sante, S.; Mollaioli, D.; Gravina, G.L.; Ciocca, G.; Limoncin, E.; Carosa, E.; Lenzi, A.; Jannini, E.A. Epidemiology of delayed ejaculation. Transl. Androl. Urol. 2016, 5, 541–548.
- Andersson, K.E.; Boedtkjer, D.B.; Forman, A. The link between vascular dysfunction, bladder ischemia, and aging bladder dysfunction. Ther. Adv. Urol. 2017, 9, 11–27.
- Tsujimura, A.; Hiramatsu, I.; Aoki, Y.; Shimoyama, H.; Mizuno, T.; Nozaki, T.; Shirai, M.; Kobayashi, K.; Kumamoto, Y.; Horie, S. Atherosclerosis is associated with erectile function and lower urinary tract symptoms, especially nocturia, in middle-aged men. Prostate Int. 2017, 5, 65–69.
- de la Taille, A.; Descazeaud, A.; Robert, G. How to prevent LUTS due to BPH development and progression. Prog. Urol. 2018, 28, 821–829.
- Matsui, S.; Kajikawa, M.; Maruhashi, T.; Iwamoto, Y.; Oda, N.; Kishimoto, S.; Hashimoto, H.; Hidaka, T.; Kihara, Y.; Chayama, K.; et al. Endothelial dysfunction, abnormal vascular structure and lower urinary tract symptoms in men and women. Int. J. Cardiol. 2018, 261, 196–203.
- McMahon, C.G. Current diagnosis and management of erectile dysfunction. Med. J. Aust. 2019, 210, 469–476.
- Calogero, A.E.; Burgio, G.; Condorelli, R.A.; Cannarella, R.; La Vignera, S. Epidemiology and risk factors of lower urinary tract symptoms/benign prostatic hyperplasia and erectile dysfunction. Aging Male 2019, 22, 12–19.
- Chung, E. Sexuality in Ageing Male: Review of Pathophysiology and Treatment Strategies for Various Male Sexual Dysfunctions. Med. Sci. 2019, 7, 98.
- Liao, L.; Chuang, Y.-C.; Liu, S.-P.; Lee, K.-S.; Yoo, T.K.; Chu, R.; Sumarsono, B.; Wang, J.-Y. Effect of lower urinary tract symptoms on the quality of life and sexual function of males in China, Taiwan, and South Korea: Subgroup analysis of a cross-sectional, population-based study. Low. Urin. Tract Symptoms 2019, 11, O78–O84.
- Gur, S.; Alzweri, L.; Yilmaz-Oral, D.; Kaya-Sezginer, E.; Abdel-Mageed, A.B.; Dick, B.; Sikka, S.C.; Volkan Oztekin, C.; Hellstrom, W.J.G. Testosterone positively regulates functional responses and nitric oxide expression in the isolated human corpus cavernosum. Andrology 2020.
- Sihotang, R.C.; Alvonico, T.; Taher, A.; Birowo, P.; Rasyid, N.; Atmoko, W. Premature ejaculation in patients with lower urinary tract symptoms: A systematic review. Int. J. Impot. Res. 2020.
- Leong, D.P.; Fradet, V.; Shayegan, B.; Duceppe, E.; Siemens, R.; Niazi, T.; Klotz, L.; Brown, I.; Chin, J.; Lavallee, L.; et al. Cardiovascular Risk in Men with Prostate Cancer: Insights from the RADICAL PC Study. J. Urol. 2020, 203, 1109–1116.
- Haider, K.S.; Haider, A.; Saad, F.; Doros, G.; Hanefeld, M.; Dhindsa, S.; Dandona, P.; Traish, A. Remission of type 2 diabetes following long-term treatment with injectable testosterone undecanoate in patients with hypogonadism and type 2 diabetes: 11-year data from a real-world registry study. Diabetes. Obes. Metab. 2020, 22, 2055–2068.
- Diaconu, R.; Donoiu, I.; Mirea, O.; Bălşeanu, T.A. Testosterone, cardiomyopathies, and heart failure: A narrative review. Asian J. Androl. 2021.
- Liang, G.; Song, Y.; Liu, L.; Zhou, K.; Tian, J.; Li, J.; Shi, H.; Zhu, Q.; Wang, J.; Zheng, J.; et al. Association of hypogonadism symptoms and serum hormones in aging males. Andrologia 2021, e14013.
- Passos, G.R.; Ghezzi, A.C.; Antunes, E.; de Oliveira, M.G.; Mónica, F.Z. The Role of Periprostatic Adipose Tissue on Prostate Function in Vascular-Related Disorders. Front. Pharmacol. 2021, 12, 626155.
- Pastuszak, A.W.; Kohn, T.P.; Estis, J.; Lipshultz, L.I. Low Plasma Testosterone Is Associated with Elevated Cardiovascular Disease Biomarkers. J. Sex. Med. 2017, 14, 1095–1103.
- Corrigan, F.E., 3rd; Al Mheid, I.; Eapen, D.J.; Hayek, S.S.; Sher, S.; Martin, G.S.; Quyyumi, A.A. Low testosterone in men predicts impaired arterial elasticity and microvascular function. Int. J. Cardiol. 2015, 194, 94–99.
- Neuzillet, Y.; Raynaud, J.-P.; Dreyfus, J.-F.; Radulescu, C.; Rouanne, M.; Schneider, M.; Krish, S.; Rouprêt, M.; Drouin, S.J.; Comperat, E.; et al. Aggressiveness of Localized Prostate Cancer: The Key Value of Testosterone Deficiency Evaluated by Both Total and Bioavailable Testosterone: AndroCan Study Results. Horm. Cancer 2019, 10, 36–44.
- Di Francesco, S.; Robuffo, I.; Caruso, M.; Giambuzzi, G.; Ferri, D.; Militello, A.; Toniato, E. Metabolic Alterations, Aggressive Hormone-Naïve Prostate Cancer and Cardiovascular Disease: A Complex Relationship. Medicina 2019, 55, 62.
- Smentoch, J.; Szade, J.; Żaczek, A.J.; Eltze, E.; Semjonow, A.; Brandt, B.; Bednarz-Knoll, N. Low numbers of vascular vessels correlate to progression in Hormone-Naïve prostate carcinomas undergoing radical prostatectomy. Cancers 2019, 11, 1356.
- Haga, N.; Akaihata, H.; Hata, J.; Aikawa, K.; Yanagida, T.; Matsuoka, K.; Koguchi, T.; Hoshi, S.; Ogawa, S.; Kataoka, M.; et al. The association between local atherosclerosis of the prostatic artery and benign prostatic enlargement in humans: Putative mechanism of chronic ischemia for prostatic enlargement. Prostate 2018, 78, 1001–1012.
- Saito, M.; Tsounapi, P.; Oikawa, R.; Shimizu, S.; Honda, M.; Sejima, T.; Kinoshita, Y.; Tomita, S. Prostatic ischemia induces ventral prostatic hyperplasia in the SHR; possible mechanism of development of BPH. Sci. Rep. 2014, 4, 3822.
- Gat, Y.; Goren, M. Benign Prostatic Hyperplasia: Long-term follow-up of prostate volume reduction after sclerotherapy of the internal spermatic veins. Andrologia 2018, 50.
- Felix-Patrício, B.; Miranda, A.F.; Medeiros, J.L.J.; Gallo, C.B.M.; Gregório, B.M.; Souza, D.B.; Costa, W.S.; Sampaio, F.J.B. The prostate after castration and hormone replacement in a rat model: Structural and ultrastructural analysis. Int. Braz. J. Urol. 2017, 43, 957–965.
- Angrimani, D.S.R.; Francischini, M.C.P.; Brito, M.M.; Vannucchi, C.I. Prostatic hyperplasia: Vascularization, hemodynamic and hormonal analysis of dogs treated with finasteride or orchiectomy. PLoS ONE 2020, 15, e0234714.
- Yoon, S.; Alfajaro, M.M.; Cho, K.-O.; Choi, U.-S.; Je, H.; Jung, J.; Jang, Y.; Choi, J. Perfusion change in benign prostatic hyperplasia before and after castration in a canine model: Contrast enhanced ultrasonography and CT perfusion study. Theriogenology 2020, 156, 97–106.
- Kok, V.C.; Hsiao, Y.-H.; Horng, J.-T.; Wang, K.-L. Association Between Erectile Dysfunction and Subsequent Prostate Cancer Development: A Population-Based Cohort Study with Double Concurrent Comparison Groups. Am. J. Mens. Health 2018, 12, 1492–1502.
- Lee, D.S.; Kim, S.W.; Sohn, D.W. Association between Nocturnal Frequency and Erectile Function in Eugonadal Men with Benign Prostatic Obstruction: A Cross Sectional Study. World J. Mens. Health 2020.
- Canguven, O.; Talib, R.A.; El-Ansari, W.; Shamsoddini, A.; Salman, M.; Al-Ansari, A. RigiScan data under long-term testosterone therapy: Improving long-term blood circulation of penile arteries, penile length and girth, erectile function, and nocturnal penile tumescence and duration. Aging Male Off. J. Int. Soc. Study Aging Male 2016, 19, 215–220.
- Xiong, W.; Kong, X.; Jiang, J.; Yang, Z.; Jiang, R. Low androgen status inhibits erectile function by inducing eNOS uncoupling in rat corpus cavernosum. Andrology 2020, 8, 1875–1883.
- Kwon, M.-H.; Park, S.-H.; Song, K.-M.; Ghatak, K.; Limanjaya, A.; Ryu, D.-S.; Ock, J.; Hong, S.-S.; Ryu, J.-K.; Suh, J.-K. Penile erection induces angiogenic, survival, and antifibrotic signals: Molecular events associated with penile erection induced by cavernous nerve stimulation in mice. Int. J. Urol. Off. J. Jpn. Urol. Assoc. 2016, 23, 614–622.
- DeMartino, A.W.; Kim-Shapiro, D.B.; Patel, R.P.; Gladwin, M.T. Nitrite and nitrate chemical biology and signalling. Br. J. Pharmacol. 2019, 176, 228–245.
- Gantner, B.N.; LaFond, K.M.; Bonini, M.G. Nitric oxide in cellular adaptation and disease. Redox Biol. 2020, 34, 101550.
- Mintz, J.; Vedenko, A.; Rosete, O.; Shah, K.; Goldstein, G.; Hare, J.M.; Ramasamy, R.; Arora, H. Current Advances of Nitric Oxide in Cancer and Anticancer Therapeutics. Vaccines 2021, 9, 94.
- Reckelhoff, J.F.; Kellum, J.A.; Blanchard, E.J.; Bacon, E.E.; Wesley, A.J.; Kruckeberg, W.C. Changes in nitric oxide precursor, L-arginine, and metabolites, nitrate and nitrite, with aging. Life Sci. 1994, 55, 1895–1902.
- Moretto, J.; Guglielmetti, A.S.; Tournier-Nappey, M.; Martin, H.; Prigent-Tessier, A.; Marie, C.; Demougeot, C. Effects of a chronic L-arginine supplementation on the arginase pathway in aged rats. Exp. Gerontol. 2017, 90, 52–60.
- Klawitter, J.; Hildreth, K.L.; Christians, U.; Kohrt, W.M.; Moreau, K.L. A relative L-arginine deficiency contributes to endothelial dysfunction across the stages of the menopausal transition. Physiol. Rep. 2017, 5, e13409.
- Gambardella, J.; Khondkar, W.; Morelli, M.B.; Wang, X.; Santulli, G.; Trimarco, V. Arginine and Endothelial Function. Biomedicines 2020, 8, 277.
- Hayashi, T.; Yano, K.; Matsui-Hirai, H.; Yokoo, H.; Hattori, Y.; Iguchi, A. Nitric oxide and endothelial cellular senescence. Pharmacol. Ther. 2008, 120, 333–339.
- Roddam, A.W.; Allen, N.E.; Appleby, P.; Key, T.J. Endogenous sex hormones and prostate cancer: A collaborative analysis of 18 prospective studies. J. Natl. Cancer Inst. 2008, 100, 170–183.
- Vanini, F.; Kashfi, K.; Nath, N. The dual role of iNOS in cancer. Redox Biol. 2015, 6, 334–343.
- Melegh, Z.; Oltean, S. Targeting angiogenesis in prostate cancer. Int. J. Mol. Sci. 2019, 20, 2676.
- Soni, Y.; Softness, K.; Arora, H.; Ramasamy, R. The Yin Yang Role of Nitric Oxide in Prostate Cancer. Am. J. Mens. Health 2020, 14, 1557988320903191.
- Seabra, A.B.; Durán, N. Nitric oxide donors for prostate and bladder cancers: Current state and challenges. Eur. J. Pharmacol. 2018, 826, 158–168.
- Kim, J.; Barsoum, I.B.; Loh, H.; Paré, J.-F.; Siemens, D.R.; Graham, C.H. Inhibition of hypoxia-inducible factor 1α accumulation by glyceryl trinitrate and cyclic guanosine monophosphate. Biosci. Rep. 2020, 40.
- Zhang, X.G.; Jin, L.; Tian, Z.; Wang, J.; Yang, Y.; Liu, J.F.; Chen, Y.; Hu, C.H.; Chen, T.Y.; Zhao, Y.R.; et al. Nitric oxide inhibits autophagy and promotes apoptosis in hepatocellular carcinoma. Cancer Sci. 2019, 110, 1054–1063.
- Kimura, N.; Yamada, Y.; Takayama, K.I.; Fujimura, T.; Takahashi, S.; Kume, H.; Inoue, S. Androgen-responsive tripartite motif 36 enhances tumor-suppressive effect by regulating apoptosis-related pathway in prostate cancer. Cancer Sci. 2018, 109, 3840–3852.
- Liang, C.; Wang, S.; Qin, C.; Bao, M.; Cheng, G.; Liu, B.; Shao, P.; Lv, Q.; Song, N.; Hua, L.; et al. TRIM36, a novel androgen-responsive gene, enhances anti-androgen efficacy against prostate cancer by inhibiting MAPK/ERK signaling pathways article. Cell Death Dis. 2018, 9, 155.
- Mandell, M.A.; Saha, B.; Thompson, T.A. The Tripartite Nexus: Autophagy, Cancer, and Tripartite Motif-Containing Protein Family Members. Front. Pharmacol. 2020, 11, 308.
- Markman, J.L.; Porritt, R.A.; Wakita, D.; Lane, M.E.; Martinon, D.; Noval Rivas, M.; Luu, M.; Posadas, E.M.; Crother, T.R.; Arditi, M. Loss of testosterone impairs anti-tumor neutrophil function. Nat. Commun. 2020, 11, 1613.
- Aversa, A.; Duca, Y.; Condorelli, R.A.; Calogero, A.E.; La Vignera, S. Androgen deficiency and phosphodiesterase type 5 expression changes in aging Male: Therapeutic implications. Front. Endocrinol. 2019, 10, 225.
- Mohamad, N.V.; Wong, S.K.; Wan Hasan, W.N.; Jolly, J.J.; Nur-Farhana, M.F.; Ima-Nirwana, S.; Chin, K.Y. The relationship between circulating testosterone and inflammatory cytokines in men. Aging Male 2019, 22, 129–140.
- Malkin, C.J.; Pugh, P.J.; Morris, P.D.; Kerry, K.E.; Jones, R.D.; Jones, T.H.; Channer, K.S. Testosterone replacement in hypogonadal men with angina improves ischaemic threshold and quality of life. Heart 2004, 90, 871–876.
- Mancini, A.; Leone, E.; Festa, R.; Grande, G.; Silvestrini, A.; De Marinis, L.; Pontecorvi, A.; Maira, G.; Littarru, G.P.; Meucci, E. Effects of testosterone on antioxidant systems in male secondary hypogonadism. J. Androl. 2008, 29, 622–629.
- Popp Marin, D.; Paola Bolin, A.; De Cassia Macedo Dos Santos, R.; Curi, R.; Otton, R. Testosterone suppresses oxidative stress in human neutrophils. Cell Biochem. Funct. 2010, 28, 394–402.
- Tsikas, D.; Kinzel, M. Associations between asymmetric dimethylarginine (ADMA), nitrite-dependent renal carbonic anhydrase activity, and plasma testosterone levels in hypogonadal men. Hell. J. Cardiol. 2018, 59, 201–206.
- Aminuddin, A.; Salamt, N.; Ahmad Fuad, A.F.; Chin, K.-Y.; Ugusman, A.; Soelaiman, I.N.; Wan Ngah, W.Z. Vascular Dysfunction among Malaysian Men with Increased BMI: An Indication of Synergistic Effect of Free Testosterone and Inflammation. Medicina 2019, 55, 575.
- Bianchi, V.E. The Anti-Inflammatory Effects of Testosterone. J. Endocr. Soc. 2019, 3, 91–107.
- Moreau, K.L.; Babcock, M.C.; Hildreth, K.L. Sex differences in vascular aging in response to testosterone. Biol. Sex Differ. 2020, 11, 18.
- Tyagi, V.; Scordo, M.; Yoon, R.S.; Liporace, F.A.; Greene, L.W. Revisiting the role of testosterone: Are we missing something? Rev. Urol. 2017, 19, 16–24.
- Kohn, T.P.; Mata, D.A.; Ramasamy, R.; Lipshultz, L.I. Effects of Testosterone Replacement Therapy on Lower Urinary Tract Symptoms: A Systematic Review and Meta-analysis. Eur. Urol. 2016, 69, 1083–1090.
- Okada, K.; Miyake, H.; Ishida, T.; Sumii, K.; Enatsu, N.; Chiba, K.; Matsushita, K.; Fujisawa, M. Improved Lower Urinary Tract Symptoms Associated with Testosterone Replacement Therapy in Japanese Men with Late-Onset Hypogonadism. Am. J. Mens. Health 2018, 12, 1403–1408.
- Rastrelli, G.; Cipriani, S.; Lotti, F.; Cellai, I.; Comeglio, P.; Boddi, V.; Della, C.P.; Gacci, M.; Serni, S.; Maggi, M.; et al. Testosterone replacement therapy is able to reduce prostate inflammation in men with BPH, metabolic syndrome and hypogonadism: Preliminary results from a randomized placebo-controlled clinical trial. In Endocrine Abstracts; BioScientifica: Gifford, UK, 2019; Volume 63.
- Rastrelli, G.; Vignozzi, L.; Maggi, M. Testosterone therapy: A friend or a foe for the aging men with benign prostatic hyperplasia? Asian J. Androl. 2020, 22, 233–235.
- Khaltourina, D.; Matveyev, Y.; Alekseev, A.; Cortese, F.; Ioviţă, A. Aging Fits the Disease Criteria of the International Classification of Diseases. Mech. Ageing Dev. 2020, 189, 111230.
- Zinger, A.; Cho, W.C.; Ben-Yehuda, A. Cancer and aging - the inflammatory connection. Aging Dis. 2017, 8, 611–627.
This entry is offline, you can click here to edit this entry!
