During the past several years, an increasing number of obstacles to DNA-related transactions was found to similarly trigger DNA repair and tolerance mechanisms. Among them, RNA-containing structures have recently caught the attention of DNA biologists. These range from single ribonucleotide insertions to RNA stretches, DNA:RNA hybrids and R-loops, in which a single-stranded DNA is displaced. Such structures are observed in diverse species and represent a significant fraction of their genomes: for example, ribonucleotides are incorporated in newly synthesized DNA at an overall rate of ~1:1000 nucleotides and R-loops occupy approximately 5% of the human genome. It has become increasingly clear that these structures are relevant for genome integrity: on the one hand, genetic screens have highlighted the importance of RNA metabolism factors in the DNA damage response, and on the other hand, factors classically associated with DNA repair or genome maintenance handle RNA-containing substrates in vitro.
- DNA repair
- genetic recombination
- genetic stability
- transcription
- RNA
- ribonucleotide
- DNA:RNA hybrid
- R-loop
1. When DNA Polymerases Meet Ribonucleotides
1.1. DNA Polymerases Can Use rNTPs during DNA Synthesis
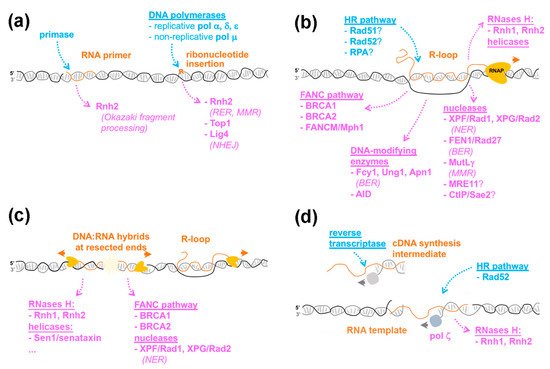
1.2. Recognition of Embedded Ribonucleotides: Genotoxicity vs. Tolerance
1.3. How Ribonucleotides Insertions Benefit Genome Homeostasis
2. R-Loop Formation: Is the Homologous Recombination Machinery Invited?
2.1. R-Loop Formation: Are There Enzymatic Activities Involved In Vivo?
2.2. Proposed Mechanisms for How HR Proteins Could Favor R-Loop Formation
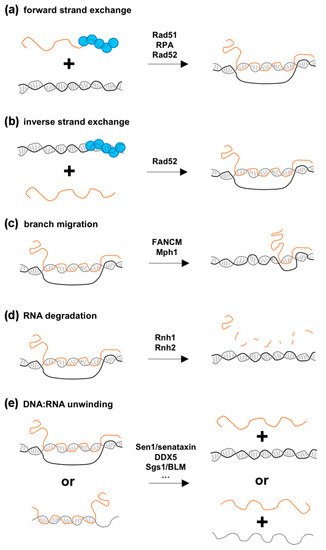
2.3. Addressing the Impact of HR-Dependent R-Loops on Genome Functions
3. R-Loop Resolution: Where DNA Repair Enzymes Come to Play
3.1. Direct and Indirect Roles of DNA Repair Factors in R-Loop Dissociation
3.2. R-Loop Processing by DNA Repair Enzymes
3.3. DNA Repair Enzymes Encountering R-Loops: A Double-Edged Sword?
4. DNA:RNA Hybrids in DNA Repair: Scaffolds or Obstacles?
4.1. DNA:RNA Hybrids Accumulation as a Consequence of DNA Damage
4.2. Do DNA:RNA Hybrids Contribute to the Outcome of DNA Repair?
4.3. Mechanisms by Which DNA:RNA Hybrids Impact the DNA Damage Response
5. When RNA Acts as a Template in DNA Repair
5.1. RNA-Templated Repair: Lessons from Yeast
5.2. Which Enzymatic Activities Are Necessary for RNA-Templated Repair?
5.3. RNA-Templated Rearrangements in Other Model Systems: Same but Different
This entry is adapted from the peer-reviewed paper 10.3390/cells10061433
