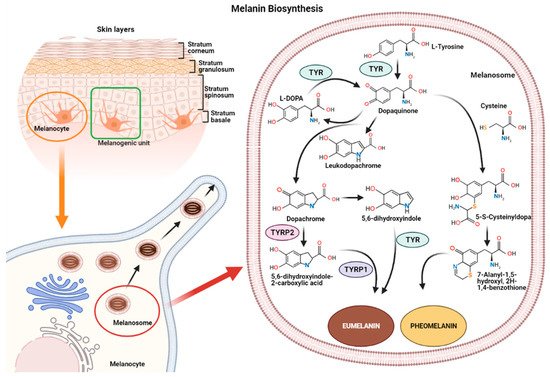
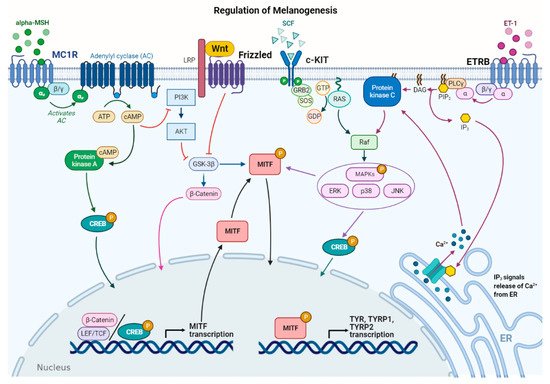
MicroRNAs (miRNAs) represent a class of non-coding RNAs of approximately 21–23 nucleotides in length. Since their discovery in 1993 [
39], miRNAs have been largely studied as essential regulators of gene expression. miRNAs are highly conserved among species and are found in different cell types and organisms, including plants, animals, and viruses [
40,
41]. It is now estimated that miRNAs target approximately 60% of genes in
humans and other mammals [
42]. Due to their role in mRNA expression regulation, miRNAs are involved in all cellular activities, including proliferation, differentiation, migration, apoptosis, and immune responses, both in normal and pathological conditions [
43,
44,
45,
46]. Several mechanisms ensure high stability in miRNAs. Since they can be easily detected in almost all bodily fluids, miRNAs are now considered important biological markers and potential therapeutic molecules [
47]. Inside cells, miRNAs derive from long precursor transcripts which give rise to mature miRNAs through a multi-step process and are then incorporated into an RNA–protein complex known as the RNA-Induced Silencing Complex (RISC) [
48]. miRNA activity mainly induces gene expression reduction by binding to sequences in the 3′ untranslated region (3′-UTR) of target genes. This can lead to mRNA target degradation, or inhibition of translation and reduction in protein levels [
45]. However, miRNA activities show higher complexity as each miRNA can target different genes and several miRNAs can regulate the same gene’s expression. Moreover, the strict interplay between long non-coding RNAs (lncRNAs) and miRNAs contributes to gene expression regulation [
40,
45,
49].
4.1. miRNA Regulating MITF
Due to the key role of
MITF as an essential regulator of the melanogenic process, several studies have investigated miRNAs with potential effects on
MITF expression and consequent regulation of mRNA levels in melanogenic enzymes [
3,
30,
31,
53] (). Some preliminary studies concerning melanogenesis regulation by miRNAs have been performed in fiber-producing animals, such as
alpaca, due to the contribution of melanin synthesis to coat color and the specific interest of
alpaca breeders in animal color coat modulation. Differences in miRNA profiles from
alpaca skins with different colored coats were identified and most differentially expressed miRNAs showed predicted targets involved in pigmentation [
54,
55]. These results led to further investigation concerning the functional role of selected miRNAs. Data obtained by Zhu Z et al. (2010) demonstrated the functional role of miR-25 in reducing
MITF mRNA and protein,
TYR, and
TYRP-1 expression in cultured melanocytes. In addition, an inverse relationship was observed between miR-25 level and coat color. Similarly, in
alpaca melanocytes, miR508-3p also directly targets
MITF, binding to the 3′-UTR of the gene. miR508-3p overexpression downregulated
MITF expression, resulting in a decrease in TYR, TYRP-2, and melanin production [
56]. Interestingly, in 2012, Dong C. et al. investigated the role of miR-137, another miR-targeting
MITF, in a transgenic mice model [
57]. Initially investigating melanoma cells lines, where its overexpression downregulated
MITF [
58], it was found that miR-137 also decreased the expression of the MITF protein and its downstream genes in transgenic mice. Notably, miR-137 had an impact on coat color, demonstrating that modulating a specific miR may significantly regulate melanogenesis, at least in animal models [
57].
Table 1. microRNAs regulating MITF in melanogenesis.
| miRNA |
Cell Model |
Target Gene |
Effect on
Melanogenesis |
Ref. |
| miR-25 |
Alpaca melanocytes |
MITF |
Negative |
[56] |
| miR508-3p |
Alpaca melanocytes |
MITF |
Negative |
[56] |
| miR-137 |
Alpaca melanocytes |
MITF |
Negative |
[58] |
| miR-675 |
Melanocytes of melasma patients, keratinocytes of melasma |
MITF |
Negative |
[5,59,60] |
| miR-218 |
Melan-a murine melanocytes, human skin OTC |
MITF |
Negative |
[61] |
| miR-183 |
B16 melanoma cells |
MITF |
Negative |
[62] |
| miR-340 |
Human epidermal melanocytes (Pig-I) |
MITF |
Negative |
[63,64] |
| miR-200a-3p |
B16-4A5 melanoma cells |
MITF |
Negative |
[52] |
| miR-148a-3p |
B16-4A5 melanoma cells |
MITF |
Negative |
[52] |
| miR-141-3p |
B16-4A5 melanoma cells |
MITF |
Negative |
[52] |
Other studies have identified miRNAs regulating skin pigmentation in
human melanocytes. Particular interest has been shown in miR-675, another miR which targets
MITF and was to be found expressed at low levels in the hyperpigmented skin of melasma patients. In melanocytes or keratinocytes derived from melasma patients, miR-675 upregulation decreased TYRP-1 and TYRP-2 expression, whereas its knockdown increased their expression. Interestingly, miR-675 was also identified in exosomes released from keratinocytes into the extracellular environment [
5,
59,
60]. Another miR involved in melanogenesis by binding the 3′-UTR in
MITF is miR-218, which downregulated TYR, TYRP-1, and DCT mRNA and protein levels, reducing melanin content in immortalized melan-a murine melanocytes. In agreement with these data, miR-218 also suppressed melanogenesis in
human pigmented skin organotypic culture (OTC) through
MITF repression. Anti-miR-218 was also found to promote melanogenesis in
human primary melanocytes [
61].
Recently, other miRNAs, including miRNA-183 cluster, miR-340, miR-141-3p, and miR-200a-3p, have been investigated for their role in regulating
MITF expression and melanogenesis [
62]. Results of this research show that the miRNA-183 cluster targets the 3′-UTR of
MITF in B16
mouse melanoma cells. miRNA-183 cluster overexpression decreased
MITF,
TYR,
TYRP-1, and
DCT expression and melanin production. Conversely, knockdown of the miRNA-183 cluster increased
MITF,
TYR,
TYRP-1, and DCT expression and, consequently, melanin levels. The miR-183 cluster was also involved in regulating the MEK/ERK signaling pathway implicated in cell proliferation and migration, by modulating mitogen-activated protein kinase 1 (MEK1), extracellular regulated protein kinases1/2 (ERK1/2), and CREB expression [
62]. miR-340, which was first identified in melanoma cell lines for its ability to bind specifically to the 3′-UTR of
MITF [
64], has been also investigated in immortalized
human epidermal melanocytes (Pig-1), where it downregulates
MITF expression and melanin synthesis [
63]. Recently, miR-141-3p and miR-200a-3p have been identified as
MITF regulators [
52]. In this study, comparing miRNA expression profiles in B16-4A5
mouse melanoma cells which were treated or untreated with α-MSH led to 13 miRNAs being identified as differentially expressed through miRNA array analysis. miR-141-3p, miR-200a-3p, and miR-148a-3p, which target
MITF, were downregulated in α-MSH-stimulated cells when compared to untreated cells. Furthermore, miR-141-3p and miR-200a-3p overexpression suppressed
MITF expression and
TYR activity in B16-4A5 cells. Notably, the inhibitory effect on melanin synthesis was confirmed in a three-dimensional tissue culture model of the
human epidermis (3D-MHE model) [
52].
4.2. miRNAs Regulating Other Genes in Melanogenesis
Besides the abovementioned studied group of miRNAs, with significant regulatory roles in MITF, other miRNAs are involved in melanogenesis by regulating the expression of other molecular targets, including melanogenic enzymes, transcription factors, or components of the signaling pathways which regulate melanogenesis ().
Several miRNAs, including miR-450b-5p, miR-1208, miR-326, miR-434-5p, miR330-5p, miR-125, miR-145, and miR-203, have been predicted as targeting
TYR and most also target other genes, including
MITF [
65,
66].
Table 2. Other microRNAs involved in melanogenesis.
| miRNA |
Cell Model |
Target Gene |
Effect on
Melanogenesis |
Ref. |
| miR-434-5p |
Mouse skin, human skin cell cultures |
TYR |
Negative |
[65] |
| miR-330-5p |
Melanoma cells, normal human melanocytes |
TYR |
Negative |
[56,67] |
| miR-203 |
Keratinocytes exposed to UV |
Kinesin Superfamily Protein 5b |
Positive |
[67] |
| miR-3196 |
Keratinocytes exposed to UV |
Unknown target gene |
Positive |
[67] |
| miR-21a-5p |
Human melanocytes |
SOX5 |
Positive |
[68] |
| miR-145 |
Murine melan-a melanocytes |
Myo5a |
Negative |
[56] |
| miR-380-3p |
Alpaca melanocytes |
SOX6 |
Negative |
[56] |
| miR-200c |
Normal human epidermal keratinocytes (NHEK) |
SOX1 |
Positive |
[69] |
| miR-27a-3p |
Alpaca and Mouse melanocytes |
Wnt3a |
Negative |
[54,70] |
| miR-379 |
Alpaca melanocytes |
IGF1R |
Negative |
[71] |
| miR-143-5p |
Human melanocytes |
Myo5a |
Negative |
[72] |
| miR-143-5p |
Alpaca melanocytes |
TAK1 |
Negative |
[73] |
| miR-125b |
WM266-4 human melanoma cells, MNT1 human melanoma cells |
SH3BP4 |
Negative |
[60] |
Wu et al. used a miR-434-5p homologue to target
TYR in cells cultured in vitro as well as in an animal model and showed efficient melanin synthesis reduction. Similarly, miR-330-5p downregulated
TYR in melanoma cells and normal melanocytes, inducing depigmentation without affecting cell proliferation [
66]. This miR has been also identified in exosomes derived from keratinocytes [
56]. In this study, exosomes carrying miR-330-5p caused a decrease in melanin production and
TYR expression in melanocytes. Similarly, miR-330-5p overexpression in melanocytes confirmed its inhibitory activity on melanogenesis. Several other miRNAs, such as miR-203, which targets
Kinesin Superfamily Protein 5b, involved in melanosome transfer, and miR-3196, with unknown target genes, have been identified in
human-derived exosomes from keratinocytes and have shown an ability to increase melanin content in melanocytes [
67]. Indeed, exosomes released by Black keratinocytes, as well as Caucasian UV-irradiated keratinocytes, were able to induce increased TYR activity and melanogenic gene expression in melanocytes, at least partly as a result of miR content.
As reported above, several miRNAs which directly target
MITF are involved in melanogenesis. Conversely,
MITF expression may be subject to more complex regulation. For example, it has been shown that miR-21a-5p overexpression downregulated the
SOX5 target, as well as β-catenin and CDK2 protein expression, in normal
human melanocytes. In agreement with
SOX5 involvement in melanogenesis, its downregulation induced an increase in
MITF and melanogenic enzymes’ expression, with consequent stimulation of melanogenesis [
68]. Several other miRNAs have been identified as regulators of SOX transcription factors [
56,
69,
74]. miR-145, which was initially identified as a miR regulated by UV treatment and forskolin in murine melan-a melanocytes, significantly modulates several genes involved in pigmentation. miR-145 overexpression or downregulation reduced or increased the expression of several genes involved in melanin biosynthesis, such as
SOX9,
MITF,
TYR, and
TYRP-1 [
74,
75], and in melanosome transfer, such as
Myosin Va (
Myo5a),
Rab27a, and
fascin1 (
Fscn1), respectively [
76]. Notably, miR-145 targeted the 3′-UTR binding site of
Myo5a, a molecular motor involved in the intracellular trafficking of vesicles and organelles. A further miR that binds an SOX family component is miR-380-3p, which targets
SOX6 by binding to the 3′-UTR region. In
alpaca melanocytes, miR-380-3p overexpression downregulated
SOX6 and increased β-catenin. Differently from studies indicating that β-catenin induces
MITF transcription, in this study, miR-380-3p overexpression caused decreased mRNA levels of melanin-related genes including
MITF,
TYR,
TYRP-1, and
DCT, suggesting other
SOX6 activities in
MITF regulation [
56].
Recently, miR-200c has been identified as a
SOX1 regulator. Specifically, miR-200c induced
SOX1 downregulation by direct binding to the
SOX1 3′-UTR, leading to intranuclear β-catenin upregulation and increased expression of
MITF-dependent gene expression as well as melanogenesis. These effects were induced in normal
human epidermal melanocytes (NHEM) treated with exosomes containing miR-200c, produced by normal
human epidermal keratinocytes (NHEK). Notably, miR-200c was found at low levels in exosomes derived from keratinocytes in vitiligo lesions compared to exosomes from NHEK, with a consequent inhibition in melanin production [
69]. Some miRNAs have also been involved in modulating the extracellular ligands which regulate melanogenic signaling pathways. An example is miR-27a-3p, which directly targets
Wnt3a, a component of the Wnt signaling pathway. miR-27a-3p was found to be expressed at higher levels in
alpaca white skin compared to brown skin [
54] and was similarly related to skin color in mice [
70]. Furthermore, its functional role in downregulating
Wnt3a, and thus inhibiting β-catenin and melanogenesis, was confirmed in
mouse melanocytes transfected with miR-27a-3p [
70]. In addition, miR-379 was shown to modulate
insulin-like growth factor receptor I (
IGFR1) in
alpaca melanocytes. Insulin-like growth factor 1 (IGF1), mainly produced in dermal cells, has been shown to improve melanogenesis through the cAMP pathway. Likewise, miR-379 overexpression reduced melanogenesis by inhibiting the cAMP response element (CRE)-binding protein (CREB)/(
MITF) pathway in
alpaca melanocytes [
71].
Finally, miRNAs involved in the regulation of melanosome processing and transfer have been also identified. Among them, miR-143-5p targets
Myo5a, which belongs to the complex
Rab27a/MLPH/Myo5a that connects melanosomes to the actin cytoskeleton in
human melanocytes [
72]. In a more recent study, Qi S. et al. inhibited miR-143-5p expression using Short Tandem Target Mimics (STTMs) in order to evaluate the functional role of miR-143-5p. As a consequence, increased melanogenic gene expression, including
MITF, TYR, and TYRP-1, MLPH, and Rab27, and melanin production were observed [
77]. This result indicates the effective possibility of modulating melanin biosynthesis by a miRNA-regulating approach. In addition, miR-143-5p overexpression induces a decrease in
TGF-β-activated kinase 1 (
TAK1) expression with consequent effects on melanocyte migration and proliferation and
MITF downregulation in
alpaca melanocytes. Therefore, miR-143-5p may regulate melanogenesis by modulating different gene targets [
73]. By means of a bioinformatic approach, followed by experimental validation, miR-125b has also been shown to directly target
SRC homology 3 domain-binding protein 4 (
SH3BP4), a gene regulated by
MITF, which may act by controlling melanogenic enzymes’ distribution to melanosomes or the mTOR signaling pathway. Indeed, when miR-125b was overexpressed in WM266-4 and MNT1
human melanoma cells, decreased levels of the melanogenic enzymes TYR and DCT were observed, although these genes are not direct miR-125b targets [
78].
4.3. miRNA Regulated by UV Rays
UV rays can modify intracellular functions in different ways: by directly or indirectly damaging DNA through reactive oxygen species (ROS), inducing apoptosis, cell cycle arrest, and carcinogenesis. The expression profiles of the miRNAs involved in these events are modified by UV irradiation [
79,
80,
81]. Among the UV-induced effects, it is well known that there is a correlation between UVB irradiation and melanogenesis stimulation [
1]. miRNA profiling in irradiated
mouse melanocytes has been helpful in identifying miRNAs involved in melanogenesis, such as miR-145 [
74]. Furthermore, several miRNAs targeting different pigmentation genes have been involved in UV ray effects, including miR-145, miR-137, miR-148, and miR-25 [
57,
82,
83].
miR-340 upregulation was identified in pigmented cells treated with UVB irradiation among miRNAs related to UV irradiation. Studies have shown that miR-340 significantly represses RhoA protein expression and stimulates melanosome transport [
81]. Recently, the functional role of miR-340 has been further investigated in immortalized
human epidermal melanocytes (Pig-1), confirming that its expression is modulated by UV and that it downregulates
MITF expression and melanin synthesis. Notably, miR-340 mimics decreased melanin content in irradiated cells [
63]. A further example of a miR potentially being involved in UV-induced pigmentation modulation is miR-21. The latter has been largely investigated in melanoma and shown to be increased by UV radiation in melanocytes, keratinocytes, and fibroblasts [
84]. In
mouse skin melanocytes, miR-21 has been shown to enhance
MITF expression by targeting
SOX5 [
85]. Inversely, miR-21a-5p has been observed with different levels of activity in UV-irradiated A375.S2
human melanoma and B16F10 melanoma
mouse cells. Indeed, in these UV-treated cells, increased melanin content was associated with an increase in α-MSH expression and reduced EGFR and Akt phosphorylation. miR-21 overexpression negatively modulated these UV-induced effects [
86]. Interestingly, this miR has been also involved in communication between melanocytes and keratinocytes. Extracellular vesicles derived from UVA-exposed melanocytes modified keratinocyte behavior by inducing miR-21 upregulation and TGF-β and IL-6/STAT3 signaling pathway activation [
87]. UVA and UVB rays also induce changes in miRNA produced by keratinocytes [
88,
89]. Accumulating evidence indicates that exosomes and exosomal miRNAs represent an effective means of communication for melanocytes and keratinocytes [
56]. Notably, in Caucasian
human keratinocytes, UV rays induce the release of miR, such as hsa-miR-3196, which may stimulate melanin synthesis in melanocytes [
67]. Recently, miR-675 production by
human keratinocytes has been shown and its role as a paracrine regulator of melanogenesis has been confirmed in vitro, indicating increased exosomal miR-675 in cells irradiated with a 585 nm light emitting diode (LED) and its ability to attenuate melanogenesis in melanocytes [
90].