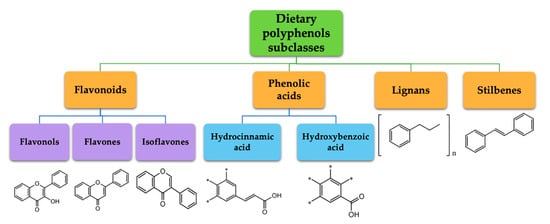
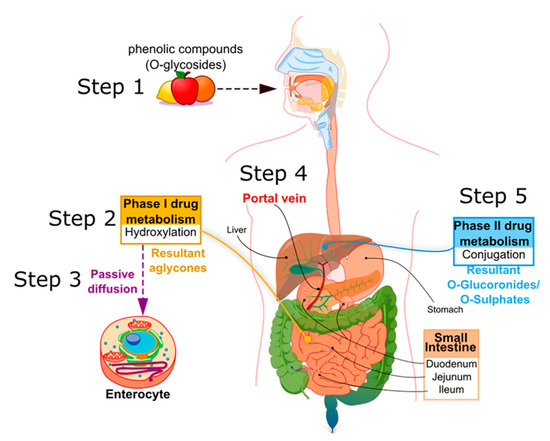
Biotransformation, during the metabolic processes in humans, is determined by the structural characteristics of the specific phenolic compound [
9]. This is because the chemical structure of the compound is specific in promoting the action of only selected intestinal enzymes and gut microbiota species. It has been shown that gut microbiota are involved in the release of phenolic aglycones and hepatic O-glucuronides [
28,
34]. According to the different structural subfamilies, PCs undergo intestinal bio-transformations by specific microbiota families [
31,
35,
36,
37,
38,
39,
40]. Furthermore, it was observed that each single phenolic compound metabolized generates numerous metabolite byproducts, usually two or three, but also many more, i.e., glycosylated quercetin produces up to 20 metabolites. According to Del Rio [
41], all these modifications during absorption have a profound influence on the biological activity of the resultant phenolic metabolites, as these may play an active role within different pathways in the human body. An example is the activation of the transcription factor nuclear factor (erythroid-derived 2)-like 2 (Nrf-2) [
42]. It was shown that protocatechuic acid, a metabolite from anthocyanins, is a known Nrf2 activator [
43,
44]. Likewise, caffeic acid metabolites have Nrf2 activating properties [
45,
46,
47]. In addition, another protective function pathway was found to be influenced by phenolic metabolites; it was shown that methylated scutellare presents an inhibitory effect on H
2O
2-induced cytotoxicity in PC12 cells, thus indicating protective activity [
48]. In order to be able to perform all the metabolic reactions, it is necessary that PC, as well as all xenobiotics, administered orally, enter the intestinal epithelium to reach the blood and lymphatic circulation [
49]. The transcellular mechanisms required to permit the entry of these compounds into the intestinal mucosa are as follows: passive diffusion, carrier-mediated active facilitated transport, and paracellular transit in tight junctions [
50,
51]. Nevertheless, generally, the majority of drugs enter the cells by passive diffusion [
52]. PCs, characterized by low molecular weights, and that are sufficiently hydrophobic and non-charged, are permitted to be transported by passive diffusion [
24]. This involves the production of biliary salts and the formation of micelles, which permeate through the translocation of the apical membrane of the enterocytes [
53]. Some PCs, such as hydroxytyrosol, tyrosol, p-cumaric acid, apigenin, and luteolin, are selectively combined in micelles and absorbed differently [
54]. In addition, in an in vitro study using Caco-2 cells to test the permeability of six dietary polyphenols (caffeic acid, chrysine, gallic acid, quercetin, resveratrol, and rutin), it was shown that several chemical-physical features are related to the passive diffusion transport capability of molecules through cells. These factors include the lipophilicity (expressed as partition coefficient logarithm, LogP), molecular weight, ionization state the number of rotatable bonds (RB), and number of hydrogen-bonding acceptor/donor (HBA/HBD), respectively [
55]. Moreover, it was observed that the scarcely lipophilic ferulic acid (FA) passes through transcellular transport, by tight junction [
56]. Regarding structurally complex PCs, such as gallotannins and ellagitannins, hydrolysis processes permit conversion into smaller molecules, thereby facilitating assimilation in simpler forms by enterocytes. However, this conversion reaction cannot occur in the small intestine, so they pass directly into the large intestine where they are fermented by the microbiota and then can be absorbed by passive diffusion at the level of the colon [
9,
57,
58]. All these transport mechanisms influence the bioavailability of these compounds in humans [
49].