Wound healing is a complex, staged process. It involves extensive communication between the different cellular constituents of various compartments of the skin and its extracellular matrix (ECM). Different signaling pathways are determined by a mutual influence on each other, resulting in a dynamic and complex crosstalk. It consists of various dynamic processes including a series of overlapping phases: hemostasis, inflammation response, new tissue formation, and tissue remodeling. Interruption or deregulation of one or more of these phases may lead to non-healing (chronic) wounds. The most important factor among local and systemic exogenous factors leading to a chronic wound is infection with a biofilm presence. In the last few years, an increasing number of reports have evaluated the effects of extremely low frequency (ELF) electromagnetic fields (EMFs) on tissue repair. Each experimental result comes from a single element of this complex process. An interaction between ELF-EMFs and healing has shown to effectively modulate inflammation, protease matrix rearrangement, neo-angiogenesis, senescence, stem-cell proliferation, and epithelialization.
- ELF-EMF
- wound
- healing
- fibroblasts
- keratinocytes
- non-healing wounds
1. Introduction
2. EMF-ELF and Wound-Repair: Mechanism of Action
2.1. ELF-EMFs and Hemostatic Phase
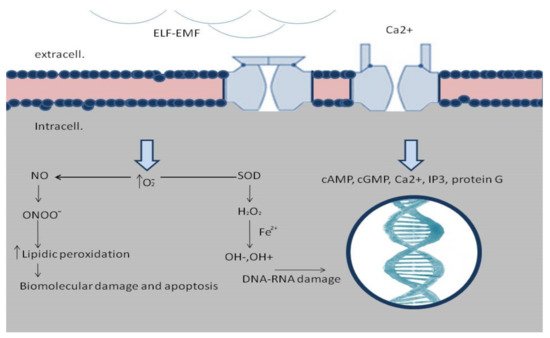
2.2. ELF-EMFs and Inflammatory Phase
2.3. ELF-EMFs and Proliferative Phase
2.4. ELF-EMFs and Remodeling Phase
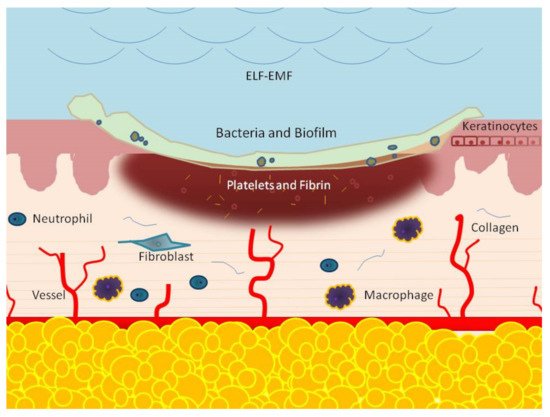
3. ELF-EMFs and Non-Healing Conditions
This entry is adapted from the peer-reviewed paper 10.3390/ijms22095037
References
- Alonso, N.; Nebreda, A.D.; Monczor, F.; Gutkind, J.S.; Davio, C.; Fernandez, N.; Shayo, C. PI3K pathway is involved in ERK signaling cascade activation by histamine H2R agonist in HEK293T cells. Biochim. Biophys. Acta 2016, 1860, 1998–2007.
- Aarabi, S.; Longaker, M.T.; Gurtner, G.C. Hypertrophic Scar Formation Following Burns and Trauma: New Approaches to Treatment. PLoS Med. 2007, 4, e234.
- Martin, P.; D’Souza, D.; Martin, J.; Grose, R.; Cooper, L.; Maki, R.; McKercher, S.R. Wound healing in the PU.1 null mouse–tissue repair is not dependent on inflammatory cells. Curr. Biol. 2013, 13, 1122–1128.
- Martin, P.; Leibovich, S.J. Inflammatory cells during wound repair: The good, the bad and the ugly. Trends Cell Biol. 2005, 15, 599–607.
- Schmidt, B.A.; Horsley, V. Intradermal adipocytes mediate fibroblast recruitment during skin wound healing. Development 2013, 140, 1517–1527.
- Xie, C.; Shi, K.; Zhang, X.; Zhao, J.; Yu, J. MiR-1908 promotes scar formation post-burn wound healing by suppressing Ski-mediated inflammation and fibroblast proliferation. Cell Tissue Res. 2016, 366, 371–380.
- Amin, Z.A.; Ali, H.M.; Alshawsh, M.A.; Darvish, P.H.; Abdulla, M.A. Application of Antrodia camphorata promotes rat’s wound healing in vivo and facilitates fibroblast cell proliferation in vitro. Evid. Based Complement. Altern. Med. 2015, 2015, 317693.
- Park, J.; Hong, Y.; Kwon, S.H.; Park, J.; Park, J. Anti-aging effects of Piper cambodianum P. Fourn. extract on normal human dermal fibroblast cells and a wound-healing model in mice. Clin. Interv. Aging 2016, 11, 1017–1026.
- Upadhyay, A.; Chattopadhyay, P.; Goyary, D.; Mazumder, P.M.; Veer, V. Ixora coccinea Enhances Cutaneous Wound Healing by Upregulating the Expression of Collagen and Basic Fibroblast Growth Factor. ISRN Pharmacol. 2014, 2014, 751824.
- Zheng, Z.; Kang, H.Y.; Lee, S.; Kang, S.W.; Goo, B.; Cho, S.B. Up-regulation of fibroblast growth factor (FGF) 9 expression and FGF-WNT/beta-catenin signaling in la-ser-induced wound healing. Wound Repair Regen. 2014, 22, 660–665.
- Nakamichi, M.; Akishima-Fukasawa, Y.; Fujisawa, C.; Mikami, T.; Onishi, K.; Akasaka, Y. Basic Fibroblast Growth Factor Induces Angiogenic Properties of Fibrocytes to Stimulate Vascular Formation during Wound Healing. Am. J. Pathol. 2016, 186, 3203–3216.
- Zigrino, P.; Brinckmann, J.; Niehoff, A.; Lu, Y.; Giebeler, N.; Eckes, B.; Kadler, K.E.; Mauch, C. Fibroblast-Derived MMP-14 Regulates Collagen Homeostasis in Adult Skin. J. Investig. Dermatol. 2016, 136, 1575–1583.
- Opalenik, S.R.; Davidson, J.M. Fibroblast differentiation of bone marrow-derived cells during wound repair. FASEB J. 2005, 19, 1561–1563.
- Werner, S.; Krieg, T.; Smola, H. Keratinocyte–Fibroblast Interactions in Wound Healing. J. Investig. Dermatol. 2007, 127, 998–1008.
- Briggs, M.; Closs, S. Patients’ perceptions of the impact of treatments and products on their experience of leg ulcer pain. J. Wound Care 2006, 15, 333–337.
- Monari, P.; Pelizzari, L.; Crotti, S.; Damiani, G.; Calzavara-Pinton, P.; Gualdi, G. The Use of PRISM (Pictorial Representation of Illness and Self Measure) in Patients Affected by Chronic Cutaneous Ulcers. Adv. Skin Wound Care 2015, 28, 489–494.
- Gurtner, G.C.; Werner, S.; Barrandon, Y.; Longaker, M.T. Wound repair and regeneration. Nat. Cell Biol. 2008, 453, 314–321.
- Eming, S.A.; Martin, P.; Tomic-Canic, M. Wound repair and regeneration: Mechanisms, signaling, and translation. Sci. Transl. Med. 2014, 6, 265sr6.
- Geng, D.Y.; Li, C.H.; Wan, X.W.; Xu, G.Z. Biochemical kinetics of cell proliferation regulated by extremely low frequency electromagnetic field. Biomed. Mater. Eng. 2014, 24, 1391–1397.
- Pirozzoli, M.C.; Marino, C.; Lovisolo, G.A.; Laconi, C.; Mosiello, L.; Negroni, A. Effects of 50 Hz electromagnetic field exposure on apoptosis and differentiation in a neuroblastoma cell line. Bioelectromagnetics 2003, 24, 510–516.
- Repacholi, M.H.; Greenebaum, B. Interaction of static and extremely low frequency electric and magneticfields with living systems: Health effects and research needs. Bioelectromagnetics 1999, 20, 133–160.
- Kaszuba-Zwoinska, J.; Chorobik, P.; Juszczak, K.; Zaraska, W.; Thor, P.J. Pulsed electromagnetic field affects intrinsic and endoplasmatic reticulum apoptosis induction pathways in MonoMac6 cell-line culture. J. Physiol. Pharmacol. 2012, 63, 537–545.
- Cheng, K.; Zou, C. Electromagnetic field effect on separation of nucleotide sequences and unwinding of a double helix during DNA replication. Med. Hypotheses 2006, 66, 148–153.
- Jasti, A.C.; Wetzel, B.J.; Aviles, H.; Vesper, D.N.; Nindl, G.; Johnson, M.T. Effect of a wound healing electromagnetic field on inflammatory cytokine gene expression in rats. Biomed. Sci. Instrum. 2001, 37, 209–214.
- Köbbert, C.; Berndt, A.; Bierbaum, T.; Sontag, W.; Breithardt, G.; Weissen-Plenz, G.; Sindermann, J.R. Low-energy electromagnetic fields promote proliferation of vascular smooth muscle cells. Electromagn. Biol. Med. 2008, 27, 41–53.
- Huo, R.; Ma, Q.; Wu, J.J.; Chin-Nuke, K.; Jing, Y.; Chen, J.; Miyar, M.E.; Davis, S.C.; Li, J. Noninvasive electromagnetic fields on keratinocyte growth and migration. J. Surg. Res. 2010, 162, 299–307.
- Yokus, B.; Akdag, M.Z.; Dasdag, S.; Cakir, D.U.; Kizil, M. Extremely low frequency magnetic fields cause oxidative DNA damage in rats. Int. J. Radiat. Biol. 2008, 84, 789–795.
- Akdag, M.Z.; Dasdag, S.; Uzunlar, A.K.; Ulukaya, E.; Oral, A.Y.; Çelik, N.; Akşen, F. Can safe and long-term exposure to extremely low frequency (50 Hz) magnetic fields affect apoptosis, re-production, and oxidative stress? Int. J. Radiat. Biol. 2013, 89, 1053–1060.
- Khaki, A.; Imani, S.A.M.; Golzar, F.S. Effects of rosmarinic acid on male sex hormones (testosterone-FSH-LH) and testis tissue apoptosis after exposure to electromagnetic field (EMF) in rats. Afr. J. Pharm. Pharmacol. 2016, 6, 248–252.
- Murbach, M.; Christopoulou, M.; Achermann, P.; Kuster, N.; Crespo-Valero, P. Exposure system to study hypotheses of ELF and RF electromagnetic field interactions of mobile phones with the central nervous system. Bioelectromagnetics 2012, 33, 527–533.
- Akdag, M.Z.; Dasdag, S.; Ketani, M.A.; Sagsoz, H. Effect of extremely low frequency magnetic fields in safety standards on structure of acidophilic and basophilic cells in anteriorpituitary gland of rats: An experimental study. J. Int. Dent. Med. Res. 2009, 2, 61–66.
- Akdag, M.Z.; Dasdag, S.; Ulukaya, E.; Uzunlar, A.K.; Kurt, M.A.; Taşkin, A. Effects of extremely low-frequency magnetic field on caspase activities and oxidative stress values in rat brain. Biol. Trace Elem. Res. 2013, 38, 238–249.
- Akdag, M.Z.; Dasdag, S.; Cakir, D.U.; Yokus, B.; Kizil, G.; Kizil, M. Do 100- and 500-μT ELF magnetic fields alter beta-amyloid protein, protein carbonyl and malondialdehyde in rat brains? Electromagn. Biol. Med. 2013, 32, 363–372.
- Lai, J.; Zhang, Y.; Zhang, J.; Liu, X.; Ruan, G.; Chaugai, S.; Tang, J.; Wang, H.; Chen, C.; Wang, D.W. Effects of 100-μT extremely low frequency electromagnetic fields exposure on hematograms and blood chemistry in rats. J. Radiat. Res. 2015, 57, 16–24.
- Liu, X.; Zhao, L.; Yu, D.; Ma, S.; Liu, X. Effects of extremely low frequency electromagnetic field on the health of workers in auto-motive industry. Electromagn. Biol. Med. 2013, 32, 551–559.
- Liboff, A. The cyclotron resonance hypothesis: Experimental evidence and theoretical constraints. In Interaction Mechanisms of Low-Level Electromagnetic Fields with Living Systems; Norden, B., Ramal, C., Eds.; Oxford University Press: London, UK, 1991; pp. 130–147.
- Piacentini, R.; Ripoli, C.; Mezzogori, D.; Azzena, G.B.; Grassi, C. Extremely low-frequency electromagnetic fields promote in vitro neurogenesis via upregulation of Ca(v)1-channel activity. J. Cell Physiol. 2008, 215, 129–139.
- Carrasco, M.A.; Hidalgo, C. Calcium microdomains and gene expression in neurons and skeletal muscle cells. Cell Calcium 2006, 40, 575–583.
- Gordon, G.A. Designed electromagnetic pulsed therapy: Clinical applications. J. Cell. Physiol. 2007, 212, 579–582.
- Han, J.; Shuvaev, V.V.; Muzykantovv, V.R. Targeted interception of signaling reactive oxygen species in the vascular endothelium. Ther. Deliv. 2012, 3, 263–276.
- Wolf, F.I.; Torsello, A.; Tedesco, B.; Fasanella, S.; Boninsegna, A.; D’Ascenzo, M.; Grassi, C.; Azzena, G.B.; Cittadini, A. 50-Hz extremely low frequency electromagnetic fields enhance cell proliferation and DNA damage: Possible involvement of a redox mechanism. Biochim. Biophys. Acta 2015, 1743, 120–129.
- Huang, C.Y.; Chang, C.W.; Chen, C.R.; Chuang, C.Y.; Chiang, C.S.; Shu, W.Y.; Fan, T.C.; Hsu, I.C. Extremely Low-Frequency Electromagnetic Fields Cause G1 Phase Arrest through the Activation of the ATM-Chk2-p21 Pathway. PLoS ONE 2014, 9, e104732.
- Eming, S.A.; Koch, M.; Krieger, A.; Brachvogel, B.; Kreft, S.; Bruckner-Tuderman, L.; Krieg, T.; Shannon, J.D.; Fox, J.W. Differential proteomic analysis distinguishes tissue repair biomarker signatures in wound exudates obtained from normal healing and chronic wounds. J. Proteome Res. 2010, 9, 4758–4766.
- Beidler, S.K.; Douillet, C.D.; Berndt, D.F.; Keagy, B.A.; Rich, P.B.; Marston, W.A. Inflammatory cytokine levels in chronic venous insufficiency ulcer tissue before and after compression therapy. J. Vasc. Surg. 2009, 49, 1013–1020.
- Sindrilaru, A.; Peters, T.; Wieschalka, S.; Baican, C.; Baican, A.; Peter, H.; Hainzl, A.; Schatz, S.; Qi, Y.; Schlecht, A.; et al. An unrestrained proinflammatory M1 macrophage population induced by iron impairs wound healing in humans and mice. J. Clin. Investig. 2011, 121, 985–997.
- Loots, M.A.; Lamme, E.N.; Zeegelaar, J.; Mekkes, J.R.; Bos, J.D.; Middelkoop, E. Differences in cellular infiltrate and extracellular matrix of chronic diabetic and venous ulcers versus acute wounds. J. Investig. Dermatol. 1998, 111, 850–857.
- Barrientos, S.; Stojadinovic, O.; Golinko, M.S.; Brem, H.; Tomic-Canic, M. Growth factors and cytokines in wound healing. Wound Repair Regen. 2008, 16, 585–601.
- Tarnuzzer, R.W.; Schultz, G.S. Biochemical analysis of acute and chronic wound environments. Wound Repair Regen. 1996, 4, 321–325.
- Vianale, G.; Reale, M.; Amerio, P.; Stefanachi, M.; Di Luzio, S.; Muraro, R. Extremely low frequency electromagnetic field enhances human keratinocyte cell growth and decreases pro-inflammatory chemokine production. Br. J. Dermatol. 2008, 158, 1189–1196.
- Pesce, M.; Patruno, A.; Speranza, L.; Reale, M. Extremely low frequency electromagnetic field and wound healing: Implication of cytokines as biological medi-ators. Eur. Cytokine Netw. 2013, 24, 1–10.
- Patruno, A.; Amerio, P.; Pesce, M.; Vianale, G.; Di Luzio, S.; Tulli, A.; Franceschelli, S.; Grilli, A.; Muraro, R.; Reale, M. Extremely low frequency electromagnetic fields modulate expression of inducible nitric oxide syn-thase, endothelial nitric oxide synthase and cyclooxygenase–2 in the human keratinocyte cell line HaCaT: Potential therapeutic effects in wound healing. Br. J. Dermatol. 2010, 162, 258–266.
- Costantini, E.; Sinjari, B.; D’Angelo, C.; Murmura, G.; Reale, M.; Caputi, S. Human Gingival Fibroblasts Exposed to Extremely Low-Frequency Electromagnetic Fields: In Vitro Model of Wound-Healing Improvement. Int. J. Mol. Sci. 2019, 20, 2108.
- Patruno, A.; Ferrone, A.; Costantini, E.; Franceschelli, S.; Pesce, M.; Speranza, L.; Amerio, P.; D’Angelo, C.; Felaco, M.; Grilli, A.; et al. Extremely low-frequency electromagnetic fields accelerate wound healing modulating MMP-9 and inflam-matory cytokines. Cell Prolif. 2018, 51, e12432.
- Gomez-Ochoa, I.; Gómez-Ochoa, P.; Gómez-Casal, F.; Cativiela, E.; Larrad-Mur, L. Pulsed electromagnetic fields decrease proinflammatory cytokine secretion (IL-1beta and TNFalpha) on human fibroblast-like cell culture. Rheumatol. Int. 2011, 31, 1283–1289.
- Mahaki, H.; Mahaki, H.; Jabarivasal, N.; Sardanian, K.; Zamani, A. Effects of various densities of 50 Hz electromagnetic field on serum IL-9, IL-10, and TNF-α levels. Int. J. Occup. Environ. Med. 2020, 11, 24–32.
- Yager, D.R.; Zhang, L.-Y.; Liang, H.-X.; Diegelmann, R.F.; Cohen, I.K. Wound Fluids from Human Pressure Ulcers Contain Elevated Matrix Metalloproteinase Levels and Activity Compared to Surgical Wound Fluids. J. Investig. Dermatol. 1996, 107, 743–748.
- Mahdavinejad, L.; Alahgholi-Hajibehzad, M.; Eftekharian, M.M.; Zaerieghane, Z.; Salehi, I.; Hajilooi, M.; Mahaki, H.; Zamani, A. Extremely Low Frequency Electromagnetic Fields Decrease Serum Levels of Interleukin-17, Transforming Growth Factor-β and Downregulate Foxp3 Expression in the Spleen. J. Interferon Cytokine Res. 2018, 38, 457–462.
- Wang, J.; Cui, J.; Zhu, H. Suppression of type I collagen in human scleral fibroblasts treated with extremely low-frequency electromagnetic fields. Mol. Vis. 2013, 19, 885–893.
- Blanpain, C.; Fuchs, E. Epidermal homeostasis: A balancing act of stem cells in the skin. Nat. Rev. Mol. Cell Biol. 2009, 10, 207–217.
- Krisp, C.; Jacobsen, F.; McKay, M.J.; Molloy, M.P.; Steinstraesser, L.; Wolters, D.A. Proteome analysis reveals antiangiogenic environments in chronic wounds of diabetes mellitus type 2 patients. Proteomics 2013, 13, 2670–2681.
- Hoffmann, D.C.; Willenborg, S.; Koch, M.; Zwolanek, D.; Müller, S.; Becker, A.K.; Metzger, S.; Ehrbar, M.; Kurschat, P.; Hellmich, M.; et al. Proteolytic processing regulates placental growth factor activities. J. Biol. Chem. 2013, 288, 17976–17989.
- Leu, A.J.; Leu, H.J.; Franzeck, U.K.; Bollinger, A. Microvascular changes in chronic venous insufficiency—A review. Cardiovasc. Surg. 1995, 3, 237–245.
- Lauer, G.; Sollberg, S.; Cole, M.; Flamme, I.; Stürzebecher, J.; Mann, K.; Krieg, T.; Eming, S.A. Expression and proteolysis of vascular endothelial growth factor is increased in chronic wounds. J. Investig. Dermatol. 2000, 15, 12–18.
- Cichon, N.; Bijak, M.; Czarny, P.; Miller, E.; Synowiec, E.; Sliwinski, T.; Saluk-Bijak, J. Increase in Blood Levels of Growth Factors Involved in the Neuroplasticity Process by Using an Extremely Low Frequency Electromagnetic Field in Post-stroke Patients. Front. Aging Neurosci. 2018, 10, 294.
- Delle Monache, S.; Alessandro, R.; Iorio, R.; Gualtieri, G.; Colonna, R. Extremely low frequency electromagnetic fields (ELF-EMFs) induce in vitro angiogenesis process in human endothelial cells. Bioelectromagnetics 2008, 29, 640–648.
- Ozgün, A.; Marote, A.; Behie, A.; Salgado, L.; Garipcan, A.; Ozgun, B. Extremely low frequency magnetic field induces human neuronal differentiation through NMDA receptor activation. J. Neural Transm. 2019, 126, 1281–1290.
- Peng, J.; Zhao, J.; Long, Y.; Xie, Y.; Nie, J.; Chen, L. Magnetic Materials in Promoting Bone Regeneration. Front. Mater. 2019, 6, 268.
- Fan, W.; Qian, F.; Ma, Q.; Zhang, P.; Chen, T.; Chen, C.; Zhang, Y.; Deng, P.; Zhou, Z.; Yu, Z. 50 Hz electromagnetic field exposure promotes proliferation and cytokine production of bone marrow mesen-chymal stem cells. Int. J. Clin. Exp. Med. 2015, 8, 7394–7404.
- Trzyna, A.; Pikula, B.; Ludwin, A.; Kocan, B.; Banas-Zabczyk, A. The influence of an electromagnetic field on adipose-derived stem/stromal cells’ growth factor secretion: Modulation of FGF-2 production by in vitro exposure. Arch. Biol. Sci. 2020, 72, 339–347.
- Rodrigues, M.; Kosaric, N.; Bonham, C.A.; Gurtner, G.C. Wound Healing: A Cellular Perspective. Physiol. Rev. 2019, 99, 665–706.
- Ahmadian, S.; Zarchi, S.R.; Bolouri, B. Effects of extremely-low-frequency pulsed electromagnetic fields on collagen synthesis in rat skin. Biotechnol. Appl. Biochem. 2006, 43, 71–75.
- Godina-Nava, J.J.; Eduardo-Ambrosio, P.; Sanchez-Dominguez, D. Comparative analyzes of 120 Hz Electromagnetic Field, respect the interferon-β and Transfer Factor effect in the recovery of chronic ulcers measuring the frequency of the lymphocytes CD4+ and CD8+ in an animal model. J. Phys. Conf. Ser. 2019, 1221, 012056.
- Percival, S.L.; Vuotto, C.; Donelli, G.; Lipsky, B.A. Biofilms and Wounds: An Identification Algorithm and Potential Treatment Options. Adv. Wound Care 2015, 4, 389–397.
- International Wound Infection Institute. Wound Infection in Clinical Practice; International Wound Infection Institute: London, UK, 2016.
- Malone, M.; Bjarnsholt, T.; McBain, A.J.; James, G.A.; Stoodley, P.; Leaper, D.; Tachi, M.; Schultz, G.; Swanson, T.; Wolcott, R.D. The prevalence of biofilms in chronic wounds: A systematic review and meta-analysis of published data. J. Wound Care 2017, 26, 20–25.
- Ganesh, K.; Sinha, M.; Mathew-Steiner, S.S.; Das, A.; Roy, S.; Sen, C.K. Chronic wound biofilm model. Adv. Wound Care 2015, 4, 382–388.
