3. Nano/Ultrafiltration Membrane Coatings
Membranes for nanofiltration (NF) and ultrafiltration (UF) processes in the water treatment field are employed to remove the most common organic and inorganic contaminants (e.g., natural organic matter, pharmaceuticals, inorganic salts, organic dyes) with high efficiencies, through different removal mechanisms like electrostatic repulsion, size/steric exclusion, hydrophobic adsorption etc. [
198,
199,
200,
201].
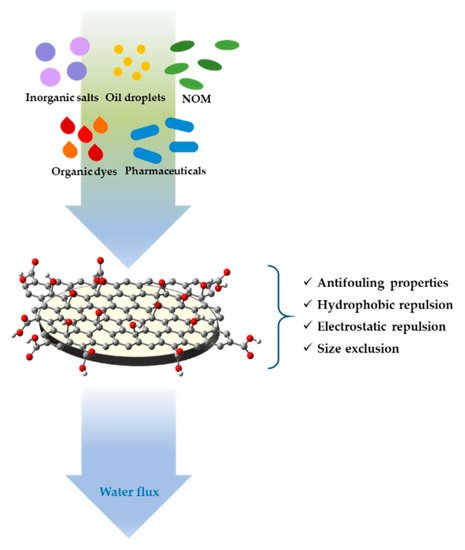
The major problem of this technology, mostly in low-pressure processes, is the membrane fouling, that negatively affects membrane performance. In recent years, a proposed solution to reduce the membrane fouling and improve their durability, selectivity, retention and permeate flux, consists in the surface modification of membranes. This can be easily done using coatings that represent the most efficient approach, because of its easy processability, which involves chemical modifications to change surface properties of membranes. In particular, membrane coatings based on nano-sized materials like graphene oxides (GOs), carbon nanotubes (MWCNT), and titanium dioxide (TiO2), thanks to their higher hydrophilicity and the capacity to reduce pore size and increase charge effects of membranes, represents a technology of growing interest in the water treatment field.
GO nanosheet coatings for membranes have good chemical stability, exceptional transport properties, and excellent mechanical stiffness and strength; in particular, the nanochannels of GO sheets 1 nm wide, by a sieving mechanism, can reject larger molecules when the water passes through the membrane filter [
202,
203,
204].
Some studies have reported the removal of several contaminants by a GO membrane in combination with polymeric membranes like polyvinylidene fluoride (PVDF), polysulfone (PS), polyamide (PA), or poly(ether sulfone) (PES), fabricated with various methods in which GO layers are bonded or unbonded together ().
Figure 2. Schematic representation of water contaminants retention of graphene oxide (GO) coated ultrafiltration (UF) membrane.
GO membranes supported on microporous substrates of PVDF exhibit high rates of rejection for organic dyes methylene blue and rhodamin WT, under a transmembrane pressure of 50 psi (0.34 MPa), in a range of 46–66% and 93–95%, respectively and NaCl and Na
2SO
4 in a range of 6–19% and 26–46% respectively, depending on the specific number of GO layers deposited [
205]. Also ceramic membrane coated with GO, at the expense of a low water permeability compared with the pristine membrane, show retention efficiencies of NOM (natural organic matter, humic acid and tannic acid), pharmaceuticals (ibuprofen and sulfamethoxazole), and inorganic salts (NaCl, Na
2SO
4, CaCl
2, and CaSO
4), in the order of 93.5%, 51.0%, and 31.4%, respectively, much higher than the pristine membrane [
206]. Moreover, the abundant oxygen-containing functional groups feature of GO (carboxyl, carbonyl, hydroxyl and epoxy, distributed at edges and structural defects of GO flakes), which causes a high negative charge on the surface of the membrane, enhancing the hydrophilicity, retention, and antifouling properties. Multiple layers of GO coated on PES ultrafiltration membrane obtained via vacuum filtration of a GO suspension, exhibit a NOM rejection in the order of 31–67%, based on the number of GO layers coated, and a water flux change of the permeate less than ±10% [
207]. GO-coated UF membranes, thanks to their underwater superoleophobicity and low oil-adhesion, are very effective in oil-in-water emulsion separation and the removing of oil droplets with sizes in the micrometer range, to obtain water with low oil/grease concentration. Porous polyamide (PA), with 200-nm 3D pores, coated with GO by vacuum filtration for antifouling oil/water separation, show an excellent antifouling performance thanks to the low oil adhesion on the membrane surface, resulting from the optimized micro-/nano-hierarchical roughness of the GO in particular with a 10 nm thickness and exhibit a 100% recovery by surface water flushing [
208]. The underwater oleophobicity of GO sheets can be tuned by oxidative etching with ultraviolet (UV) light, to create or enlarge structural defects and introducing oxygen groups around them, to improve potential applications of GO coatings in oil/water separation, oil-repellent materials, microfluidic devices, anti-bioadhesion materials, and robust antifouling materials [
209].
The most common fouling agents in water present a negative charge, so they are sensitive to a negatively charged membrane surface due to the electrostatic repulsion, as we have seen in the case of a GO-coated membrane. Several methods to enhance the surface charge of membranes have been developed, and one of them is the fabrication of electrically conducting membranes (ECM) to perform electrofiltration processes by the application of an electrical field. This type of membrane can be produced by coating UF membranes with conductive inorganic materials like carbon nanotubes (CNTs) or multiwalled carbon nanotubes (MWCNTs) that can also be used as inorganic fillers to fabricate nanocomposite membrane, to improve their performance also for the desalination of water [
210]. CNTs and MWCNTs, before their use in the fabrication of nanocomposites or coatings for their application in membranes, are treated by acid treatment, that however can lead structural damage of MWCNTs, or by coatings with some molecules like poly(vinyl alcohol), polyaniline or polydopamine, to enhance their hydrophilicity [
211]. An example is represented by a thin film made by cross-linked poly(vinyl alcohol) and carboxylated MWCNTs, deposited by pressure on a PS membrane. This system, used in an electrofiltration cell, in which a cell potential of 3–5 V and fields of 9–15 V/cm are applied, demonstrates the inhibition of a negatively charged fouling agent, represented by alginic acid, and the potential of CNT-based coatings on the reduction fouling rates [
212]. Also composite membranes based on CNT-conjugated polymers (i.e., PANI, polypyrrole), fabricated through a process of electropolymerization of aniline on a CNT substrate under acidic conditions, the latter obtained by a coating of a PS membrane with a CNT suspension performed by pressure deposition, demonstrate that the application of an anodic potential to the ECM surface, is able to degrade a model organic contaminant (methylene blue) through an electro-oxidation process, also showing an electrochemical in situ membrane cleaning capacity [
213]. To reduce membrane fouling by photo-oxidation, there is another solution that can be represented by the use of Titanium dioxide coatings. Photoactivity properties of the semiconductor titanium dioxide, exhibiting under UV irradiation, can be exploited for the photodegradation of smaller organic molecules entrapped in UF membranes. In particular, photoactive anatase membranes on asymmetric ceramic supports, prepared by slip-casting on asymmetric tubular supports in alumina, subsequently immersed in nanocrystalline anatase sols, show a high retention capacity of colloids and macromolecules, ensured by the separative top layer and the photodegradation of smaller organic molecules performed by UV irradiation of the opposite side of the membrane [
214].
Manganese oxide and iron oxide coatings for catalytic membranes are also evaluated for the retention and removal of total organic carbon (TOC) in ultrafiltration processes, which is proven to depend on the number of times the membrane was coated with the metal oxide nanoparticles. This type of membrane is produced by coating, using a layer-by-layer self-assembly technique, of commercial UF ceramic membranes, that can also be cleaned from fouling with Distilled DeIonized (DDI) water using an ozonation-filtration. Hybrid ozonation-ceramic membrane filtration performed with Mn oxide-coated membranes, have given the best results in comparison with other metal oxide coatings (titanium oxide and iron oxide), thanks to the excellent catalytic properties of manganese in the oxidation of organic material and then in the reduction of TOC in the permeate [
215].
Another application of metal coatings in filtration techniques, is their use as metal mesh coatings for application in oil/water filtration. An example is an eco-friendly iron-based, with a micro/nano-structure, metal mesh coating, produced by immersion of a stainless steel mesh in a solution in which is performed the reduction of FeCl
2 with NaBH
4, resulting in a membrane with underwater superoleophobicity, with an oil contact angle as high as 152°, that can separate oil/hot corrosive water mixed liquid efficiently, with a separation efficiency >96.2% [
216].
A further approach of water filtration in water treatment processes, consists in the use of other types of membranes like chemically-functionalized membranes (CFMs), in which are incorporated selective ligands or ion-exchangers for the extraction specifically chemical species, bulk liquid membranes (BLMs), emulsion liquid membranes (ELMs), supported liquid membranes (SLMs) in which two aqueous phases (feed and stripping) are separated by an organic liquid as interphase and polymer inclusion membranes (PIMs). PIMs, more stable than SLMs, have acquired significant importance in recent years in the field of separations and extraction of organic molecules and metal ions (Au(III), As(V), Cd(II), Co(II)) from water [
217]. This type of membrane also has important relevance in the preconcentration of antibiotics (sulfonamides and tetracyclines), which is highly influenced by sample pH, in environmental water samples [
218]. PIMs are thin, flexible and stable films made by casting a solution containing an extractant (or carrier), usually an ionic liquid for the selective extraction of the target chemical species, a plasticizer to improve the elasticity of the membrane or modify the solubility of the extracted species and a base polymer such as cellulose triacetate (CTA) or poly(vinyl chloride) (PVC) [
219]. The extractant agent in PIMs can also be represented by inorganic species that have a high capability of ion exchange and cation fixation like clays, in particular montmorillonite clay [
220]. A PIM membrane based on PVC, whit montmorillonite and ionic liquids (Aliquat 336, Alicy and thiomalic acid) like extractant and plasticising agents, for the absorption and preconcentration in particular of Sn
2+ from water samples, can be produced in two different methods: casting the clay modified with organosilanes (by a sol–gel method with dynasylan and APTES) in the solution of membrane (one pot method) or by coating the PIM membrane with the organoclay (layer by layer method).
The research of new technologies for the improvement of retention and regeneration properties of membranes in the waste water treatment, is constantly expanding, but strong evidence, as reported above, shows that the simplest and most efficient methodology for changing the surface properties of membranes, is represented by the use of membrane coatings based on various nanomaterials, which in particular allow easy cleaning, through simple washing or electrochemical methods, from fouling or oil residues for their reuse, in a circular economy perspective.