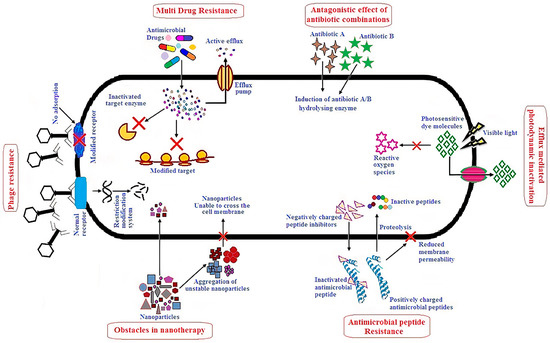
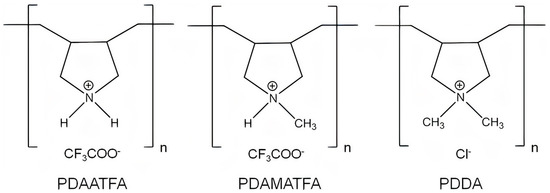
2.1. Structure and Antimicrobial Activity of ASA with AMPs
AMPs have been considered as amphipathic, cationic polymers with less than 50 amino acid residues, often displaying secondary structures such as α−helix [
7,
8,
9,
48]. Their main role in the innate immune system as an indispensable line of defense against pathogens in different body parts of mammals, plants, and other animals has been well documented [
9]. In humans, AMPs present in oral and nasal mucosae could activate anti−inflammatory cells to sites of damaged tissue [
49]. In fish, constant exposure to various types of pathogens has led to an immune system based on AMPs [
50]. The cationic character determined AMPs’ interactions with the oppositely charged bacteria cell wall and penetration in the cell membrane. Destabilization of the membrane electrochemical potential allowed AMP insertion in the plasmatic membrane of the bacteria, its rupture and bacterial cell death [
7,
9,
51,
52]. Major issues against AMPs applications have been related to AMPs’ toxicity to eukaryotic cells, poor stability in vivo with eventual degradation during transportation to their target cells and organs [
32,
51,
53,
54].
AMPs have been classified according to their origin [
8]. When they were extracted from bacteria or fungi, they belong to the nonribosomal synthetized peptides (NRAMP) class. When extracted from eukaryotic cells, they belong to the ribosomal synthetized peptides (RAMP) class. Gramicidin, vancomycin and polymyxin B are examples of NRAMPs, while nisin and melittin are RAMPs [
8,
55]. AMPs usually have one or more secondary structures such as α−helix, β−sheet, αβ, and non−αβ [
56]. A huge structural diversity of AMPs have the common feature of positive charge and amphipathic nature [
57].
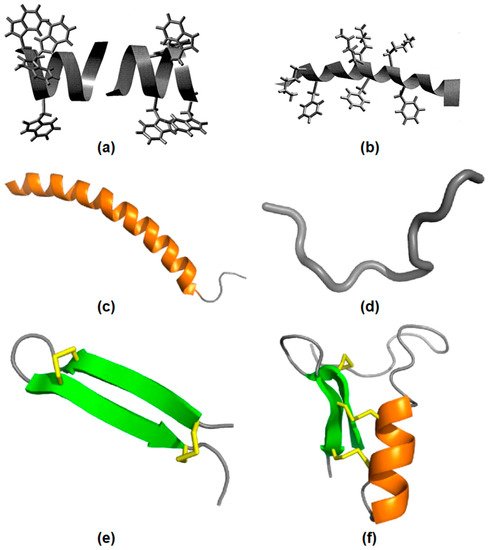
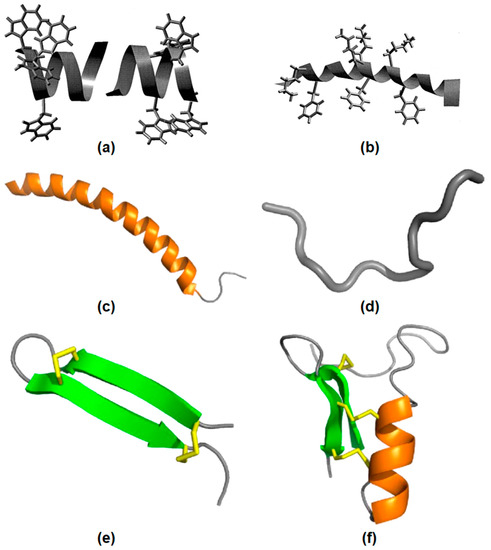
Figure 2 illustrates AMPs structures. In
Figure 2a, gramicidin A incorporated in bilayer membranes can be seen as a peptide dimer traversing the bilayer with four tryptophan side−chains as anchors at the membrane interface.
Figure 2b shows the structure of the antimicrobial frog skin peptide magainin as determined by nuclear magnetic resonance (NMR) spectroscopy in the presence of sodium dodecyl sulfate (SDS) micelles with the side−chains of lysine and phenylalanine residues [
57].
Figure 2c shows LL−37 peptide adopting a typical α−helical (orange) conformation in the presence of micelles.
Figure 2d shows indolicidin in an extended conformation.
Figure 2e shows the spider−derived β−hairpin peptide gomesin with β−sheets (green) typically stabilized by disulfide bonds (yellow).
Figure 2f shows phormicin with both α−helix and β−sheet secondary structures [
58].
Figure 2. AMPs structural features. (
a) The gramicidin A structure in membranes. (
b) The magainin structure in micelles, adapted from [
57], Copyright 1999, with permission from Elsevier. (
c) The LL−37 peptide structure in micelles. (
d) The indolicidin structure. (
e) The gomesin structure stabilized by disulfide bonds. (
f) The insect CSαβ−defensin phormicin, Adapted with permission from [
58]. Copyright 2019 Elsevier.
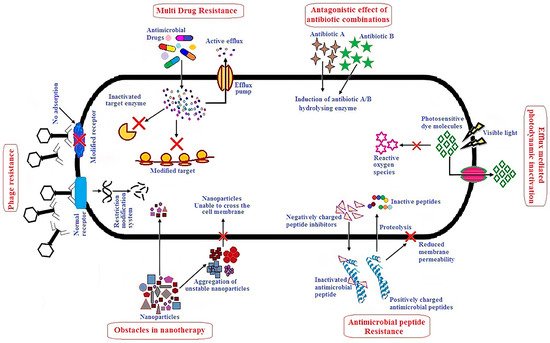
AMPs affect bacteria by inhibiting enzymatic activity, DNA or protein synthesis, or by piercing bacterial cell walls and membranes [
7,
9,
51,
52,
59].
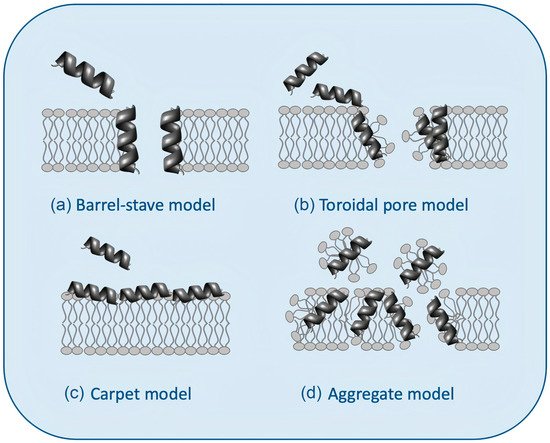
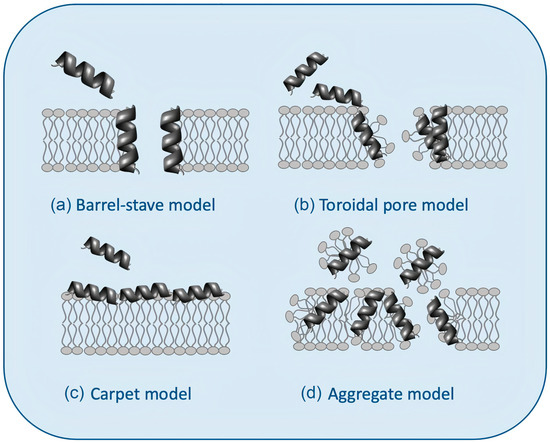
Figure 3 illustrates possible mechanisms of action for AMPs [
60].
Figure 3a shows the barrel−stave model: the AMPs approach the lipid bilayer in parallel orientation but eventually penetrate it perpendicularly, keeping intermolecular peptide interactions.
Figure 3b shows the “toroidal pore” model with two stages: at low concentrations (inactive state), peptides remain parallel to the plane of the bilayer; from a critical concentration, peptide molecules reorient perpendicularly penetrating the hydrophobic region of the bilayer (active state) and, together with some lipid molecules, adopt a multi−pore configuration with irreversible rupture of the plasma membrane.
Figure 3c shows the carpet model: peptides remain parallel to the lipid bilayer until reaching a threshold concentration above which the membrane becomes unstable and disintegrates, forming micelles, in the so−called aggregate or “detergent−like” model (
Figure 3d) [
9,
52,
60,
61,
62].
Figure 3. Models for the interaction between antimicrobial peptides (AMPs) and bilayer membranes: (
a) the barrel−stave model, (
b) the toroidal pore model, (
c) the carpet model, and (
d) the aggregate or “detergent−like” model was adapted from [
60].
The establishment of structure–function relationships for AMPs has been deemed a difficult task [
63]. More than 2000 natural or synthetic AMPs with different lengths, sequences, 3−dimensional (3−D) structures and intermolecular interactions have been described. Moreover, AMPs high sensitivity to their environment has been reported from their medium−dependent−conformations. A good example is the behavior of gramicidin D (Gr) in different media [
64].
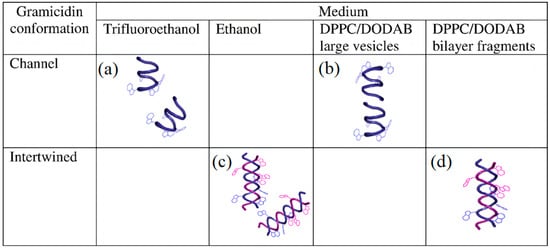
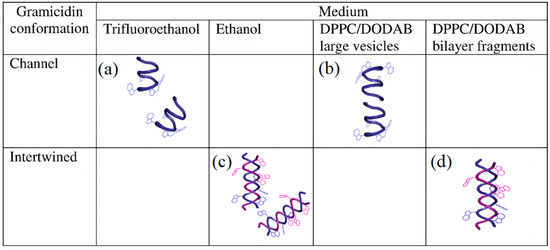
Figure 4 illustrates Gr conformations in different media depicted from Gr circular dichroism (CD) and intrinsic fluorescence spectra [
64].
Figure 4a,b shows Gr beta−helix in trifluoroethanol and large lipid vesicles (LV), respectively.
Figure 4c,d illustrates the intertwined Gr conformation in ethanol and nanosized lipid bilayer fragments (BF), respectively. The dimeric Gr functional channel has been described as a pore spanning lipid bilayers. This pore has been associated with an ionic imbalance and bacterial cell death. Curiously, Gr channels only have been observed in LV. On the other hand, Gr intertwined dimers in the non−channel conformation only occur at the borders of cationic bilayer fragments, as shown in
Figure 4d. Both LVs and BFs shield Gr tryptophans against quenching by acrylamide. However, the Stern–Volmer quenching constant is slightly higher for Gr in BFs than in LVs, confirming that the peptide was more exposed to the water medium in BFs than in LVs [
64].
Figure 4. Medium−dependent gramicidin (Gr) conformation: (
a) Gr beta−helix in trifluoroethanol, (
b) Gr beta−helix and dimeric channel in large bilayer vesicles (LVs), (
c) Gr intertwined beta−helices in ethanol, and (
d) Gr intertwined beta−helices in lipid bilayer fragments (BFs). Gr molecules sense a nonpolar medium in the LV bilayer and acquire its functional channel conformation. Gr molecules sense a polar medium in the BF bilayer and become intertwined. The lipids in LVs or BFs are dipalmitoylphosphatidyl choline (DPPC) and dioctadecyl dimethyl ammonium bromide (DODAB) at a 1:1 molar ratio. Reprinted with permission from [
64]. Copyright 2012 Elsevier.
2.2. ASA with AMPs for Preserving Activity and Reducing Toxicity
An important issue regarding AMPs performance against bacteria has been the formulation [
8]. The Gr behavior in different media can be used to exemplify the importance of the formulation. Furthermore, Gr formulation plays a central role not only on activity but also on toxicity, as discussed below.
Gr extracted from
Bacillus brevis contains a group of peptides composed of 80%, 6% and 14% of gramicidin A, B, and C, respectively [
64,
65]. Due to Gr toxicity against eukaryotic cells, its use over a range of low concentrations has been limited to topical applications avoiding systemic administration [
66,
67].
In assemblies with dioctadecyldimethylammonium bromide (DODAB) bilayers, both DODAB and Gr interacted with
Escherichia coli and
Staphylococcus aureus. Thereby DODAB antimicrobial activity against Gram−negative bacteria [
42,
43] has been combined with Gr activity against Gram−positive bacteria [
67]. This combination broadens the spectrum of antimicrobial activity. In addition, the toxicity against yeast eukaryotic cells of the DPDAB/Gr formulation has been tested and yielded improved yeast viability in comparison to the one of Gr alone [
67].
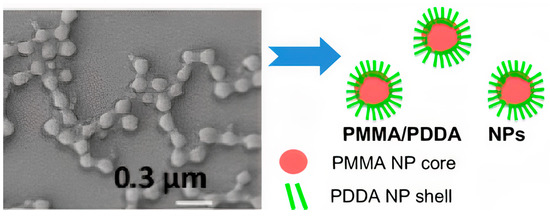
Gr has also been formulated in lipid polymer NPs [
68]. The insertion of Gr functional channels on DODAB supported bilayers has been achieved thanks to the optimization of the construction onto negatively charged polystyrene sulfate (PSS) NPs. Firstly, PSS NPs have been covered with a positively charged DODAB bilayer, which increased the zeta−average diameter by 8–10 nm, changed the zeta−potential of the NPs from negative to positive, and yielded a narrow size distribution for the PSS/DODAB/Gr NPs [
68]. This formulation has been displaying broad and high antimicrobial activity at very small concentrations of the antimicrobials, namely, 0.057 and 0.0057 mM for DODAB and Gr concentrations, respectively. The results emphasized the advantages of highly organized, nanostructured, and lipid polymer cationic NPs to achieve hybrid combinations of antimicrobials with broad−spectrum activity at tiny DODAB and Gr concentrations [
68]. Further applications for these Gr formulations using NPs have been envisaged in the biomedical field for treating burns, wounds, ulcers, caries, and pulp infections in dentistry, as antifouling, antimicrobial, and antibiofilm coatings on surfaces or embedded in hydrogels [
8,
18,
35,
63,
66,
69,
70].
Since the 1960s, the AMP nisin, a RAMP lantibiotic, has been widely employed as a food preservative to extend the shelf life of dairy products (lantibiotics refers to AMPs produced by bacteria) [
52,
66]. In contrast to Gr, nisin has been considered nontoxic to eukaryotic cells and effective against food spoiling bacteria, showing stability over a pH and temperature range plus low susceptibility to enzymatic proteolysis [
52].
Nisin has also been formulated as films released from polymer/nisin multilayers; whereas nisin/polyacrylic acid (PAA) layers disintegrated in 24 h in water solution, nisin/dextran sulfate (DX) films were stable for 14 days without releasing nisin; both films hampered the spread of
Staphylococcus epidermidis biofilms in disk diffusion tests; therapeutic utility proposed for nisin/PAA films was treating burns and wounds due to the quick nisin release, whereas nisin/DX coatings would impart steady sterilization of surfaces over long periods of time [
71].
The AMP melittin, the main component of bee venom, has been formulated on a variety of lipid or polymer based−assemblies [
55,
72]. In model membrane and cell culture studies, certain melittin analogues have been proposed as anticancer, antimicrobial, and low hemolytic activity [
73]. The interest for this AMP has been increasing due to possible uses in a variety of cancer treatments [
74,
75,
76,
77,
78,
79] despite the high in vivo cytotoxicity and hemolytic activity in intravenous applications [
80]. Apoptosis of cancer cells has been often reported in association with melittin; for example, cancer cell growth was inhibited via the increase of death receptor 3 expression and inactivation of NF−kappa beta in lung cancer cells [
76]. A graphene formulation facilitated melittin piercing of the cell wall, causing cell lysis in Gram−negative and −positive bacteria [
81].
The acronym ESKAPE pathogens have been employed to encompass E:
Enterococcus faecium, S:
Staphylococcus aureus or
Stenotrophomonas maltophilia, K:
Klebsiella pneumoniae or C:
Clostridioides difficile, A:
Acinetobacter baumannii, P:
Pseudomonas aeruginosa, E:
Enterobacter spp., or
Enterobacteriaceae. These MDR bacteria have been concerning physicians due to very few options left for treating infected patients; AMPs have been considered important for reversing this situation [
7,
82]. Recently, the synergy between antibiotics and certain AMPs has been described in a murine, sub−cutaneous abscess model caused by ESKAPE pathogens [
82]. The bacteria organization on surfaces as single and multispecies biofilms has required several techniques for proper evaluation of unconventional agents, including AMPs in the treatment of biofilm infections [
83,
84]. Designing and optimizing AMPs will have to consider that the targets reached may not be the same; peptides could be active against several kinds of cells with activity and selectivity resulting from interaction with multiple target cell components; the cellular composition has been affecting the AMP–target cell interaction and also the design of novel AMPs [
85].
Various pathogens, such as polymyxin−sensitive
Salmonella species, have been able to penetrate macrophages, where they persisted and multiplied; modifications of NPs, liposomes, and mesoporous silica with specific cell ligands have been enabling them with penetration into macrophages and killing of intracellular pathogens [
86]. Metal−based NPs, including gold NPs, have been proposed as particularly promising platforms for the intracellular delivery of AMPs, such as polymyxins eliminating intracellular
Salmonella Enterica Serovar Typhimurium [
87].
Polymyxin has been deemed of critical medical importance against severe nosocomial multidrug−resistant Gram−negative bacteria causing nosocomial pneumonia. Several polymyxin formulations have been developed for parenteral use (for treatment of cystic fibrosis, pneumonia, bacteremia, and urinary tract infections), inhalation (cystic fibrosis, pneumonia), and topical use (optic and ophthalmic solutions). The most common polymyxin side effects have been dose−dependent nephrotoxicity and neurotoxicity; since polymyxins were essentially not absorbed by the gastrointestinal tract, their encapsulation into suitable carriers improved intestinal permeability, thereby allowing novel formulations administered by the oral route [
88].
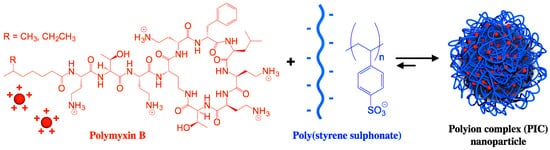
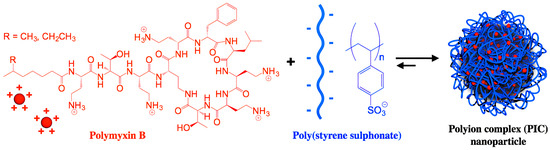
Figure 5 shows a polymer−based formulation for polymyxin B based on the electrostatic attraction between the cationic peptide and the anionic poly (styrene sulphonate) polymer as reproduced from [
89,
90]; the antimicrobial activity of the polymyxin AMP was influenced by the degree of polymerization of the poly−ion (DP): a low DP improved antimicrobial activity, while a high DP improved the NPs stability [
89].
Figure 5. Nanoparticles of polymyxin B and poly (styrene sulphonate) active against
Pseudomonas aeruginosa. Reprinted with permission from [
90]. Copyright 2017 Elsevier.
The relative high burden of methicillin−resistant
S. aureus (MRSA) has been a major concern in healthcare; vancomycin, a glycopeptide antibiotic inhibiting cell wall biosynthesis, has remained the drug of choice for treatment of severe MRSA infections for many years. Unfortunately, vancomycin−resistant
S. aureus strains have been disclosed in the 1990s; their polygenic molecular basis of resistance was due to stepwise mutations in genes encoding molecules predominantly involved in cell envelope biosynthesis [
91]. Resistance has been associated with persistent infections, vancomycin treatment failure, and poor clinical outcomes.
S. aureus strains isolated from humans, pigs, and cattle have created intermediate resistance to vancomycin [
92]. Vancomycin formulated as nanoplexes of the antibiotic with dextran sulfate sodium salt has recently addressed MRSA infections; the size, polydispersity, and zeta potential of the optimized nanoplexes were 84.6 ± 4.3 nm, 0.449 ± 0.024, and −33.0 ± 4.9 mV, respectively, with 90.4 ± 0.8% complexation efficiency and 62.3 ± 0.2% drug loading; in vivo studies using a BALB/c mouse skin infection model revealed that nanoplexes reduced MRSA burden by 2.3−fold compared to bare vancomycin [
93]. Liposomal vancomycin topical formulations have also produced similar results against MRSA, reconfirming the importance of the formulation for fighting drug−resistant microbia [
94].
AMPs have been fighting not only bacteria but also other pathogens, such as fungi [
95], viruses, and protozoa [
15]. Besides, their versatility allowed extensions for treating from skin wounds to cancer. Therefore, future studies with AMPs are necessary, for instance, with the improvement of its stability and its scaling−up projection in the industry [
51], to go beyond the combat against antimicrobial resistance. Recent advances in antimicrobial polymers in general [
96] or natural and synthetic AMPs, in particular, have been reviewed [
50,
95,
97].
Major applications for AMPs have also been the subject of important review articles such as the use of AMPs for drug design and therapeutics [
98,
99], natural additives for food preservation [
52,
100,
101], prevention of caries, and pulpal infections due to dental plaques and similar others [
102,
103].
The most important types of carriers for AMPs were liposomes [
64,
67,
69,
70,
101], nanostructured lipid carriers [
104], lyotropic lipid phases (cubic and hexagonal) [
105], lipid nanodisks, and bilayer fragments [
10,
12], NPs of several types such as biomimetic [
20,
28,
32,
68], polymeric [
61,
89,
106], magnetic [
107,
108], metal−AMPs designed as metallodrugs with nuclease, and protease activity [
109] or silver co−spinned with nisin in polymeric nanofibers [
110], hydrogels [
36,
37,
111], silver in alginate hydrogels [
112], or fabrics [
113,
114,
115].
Lipid−based liquid crystals as carriers for antimicrobial peptides emphasized the importance of more fluid lipid phases for the antimicrobial effect [
105].
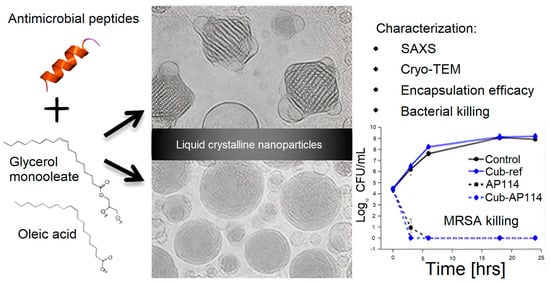
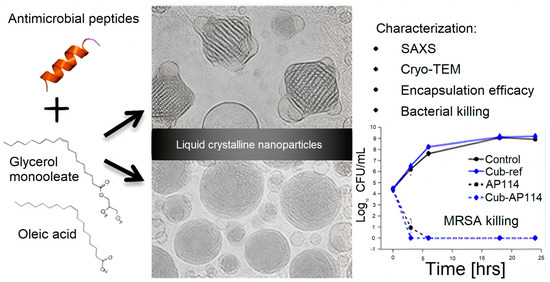
Figure 6 shows lipid−based liquid crystals from cubic and hexagonal phases; these lyotropic liquid crystalline (LC) structures consisting of cubic glycerol monooleate/water and hexagonal glycerol monooleate/oleic acid/water assemblies have been examined as carriers for AMPs. Certain AMPs had their antimicrobial activity preserved, whereas others had their activity reduced by the carriers; LC−structured gels or NPs had the capability of solubilizing both hydrophilic and hydrophobic substances, as well as being biocompatible and biodegradable; depending on AMP nature, LC showed no effect on AMPs antimicrobial activity or a diminished effect on this property.
Figure 6. Lyotropic liquid−crystalline (LC) nanoparticles (NPs) tested as carriers for AMPs. Reproduced with permission from [
105]. Copyright 2016 American Chemical Society.
Importantly, several AMPs formulations have been proposed against multidrug resistance. For example, liposomal AMPs combined with vancomycin exhibited improved activity against intracellular MRSA; after selecting AMPs with high antimicrobial activity, the selected peptides were lipidated, combined with model membranes (liposomes), and tested for intracellular activity against MRSA infecting human embryonic kidney epithelial cells in culture (HEK−293). They possessed good cell penetration to act against the intracellular MRSA; in addition, there was sustained release for the AMPs with a consequent improvement in the bioavailability [
116].
Mycobacterium tuberculosis is intrinsically resistant to many antibiotics due to mutations that lead to novel strains. AMPs with metal complexes have been proposed as advantageous combinations since metal complexes associated with known AMPs often present different mechanisms of action with respect to single peptides: the destruction of bacterial plasma membranes as well as hydrolytic or oxidative cleavage of nucleic acids promoted by metal−based compounds followed from their role in the generation of reactive oxygen species able to degrade biomolecules [
117]; the formulations complexing metal with AMPs could fight drug resistance against tuberculosis [
118]. Adding antimicrobial and antibiofilm activities to AMPs via covalently bound metal−binding motifs improved their activities in certain cases; when combined with meropenem, streptomycin, or chloramphenicol, certain variants showed synergistic effects against
E. coli (KpC+ 1812446) biofilms; the addition of motif also improved the survival rate of mice in a systemic infection model and reduced the hemolytic activity of the wild−type AMP [
119].
Besides the AMPs, the cationic antimicrobial polymers (APs) represent another extremely promising class of antimicrobial molecules. APs are briefly presented and discussed regarding their outstanding properties in the next section.