The large amount of protein variants collected in public databases and the limitations in the experimental methods stimulated the development of several tools for predicting their impact on protein stability and their pathogenic effect. Accordingly, early developed tools focus on the prediction of protein stability change by estimating the variation of free energy change (ΔΔG
f) resulting from an amino acid substitution [
116,
117]. The majority of methods, which have been trained on ProTherm database [
118] or on manually collected datasets [
26], predict either the value or the sign (positive/negative) of the ΔΔG
f. More recently, once large databases collecting pathogenic variants were made available, many binary classifiers have been implemented for predicting the impact of genetic variants on human health [
119,
120,
121]. All available methods for predicting the impact of variants on protein stability or on protein pathogenicity rely on the various features extracted from protein sequence, structure, and evolutionary information. State-of-the-art methods of both types are currently used for protein engineering and for variant interpretation. In this section, we analyze the effect of the protein variants using computational approaches for predicting protein stability changes and pathogenicity, with the aim of estimating the role of protein stability on cancer mechanisms and the reliability of computational tools on this specific task.
2.1. Collection of the Protein Variant Datasets
In this work, we analyze a set of 164 missense variants from 11 proteins to understand the contribution of protein stability on the insurgence and progression of cancer (
Table S1). Among the 11 proteins, 5 of them are mainly involved in regulation activities (BRD2, BRD3, BRD4, p16, and PPARγ), 4 have catalytic activities (PIM1, PGK1, FXN, and PTPρ) while the remaining 2 (p53 and BRCA1) are involved in many biological processes. The set collects all protein variants, for which the folding ΔΔG value was experimentally determined and whose genes are reported in the COSMIC database, either as Tier 1 genes, or with putative cancer-driving evidence. The folding ΔΔG
f is calculated as the difference between the folding ΔG of the mutant and wild-type proteins (ΔΔG
f = ΔG
fmut − ΔG
fwt), i.e., it is positive for destabilizing variants. When available, the variation of the melting temperature (Δ
Tm =
Tmmut −
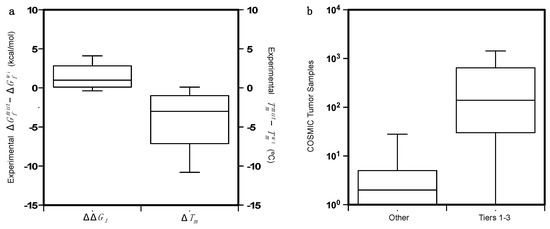
Tmwt) was also collected. The distributions of the ΔΔG
f and Δ
Tm values are plotted in a. The protein mutants are mapped on unique protein structures except in the case of p53, for which the DNA binding and oligomerization domains are considered separately. A subset of 97 variants from 9 proteins is obtained by matching our dataset with the data collected by the Cancer Mutation Census (CMC) project. This subset is composed of 24 putative cancer-driving variants annotated as “Tier 1–3” and 63 putative benign variants annotated as “
Other”.
Figure 4. (a) Distributions of the of ΔΔGf and ΔTm on the dataset of 164 protein variants. ΔTm is available only for 73 of them. (b) Comparison of the distributions of the COSMIC tumor samples for the putative cancer-driving variants (Tiers 1–3) and the benign variants (Others) annotated by the Cancer Mutation Census project.
In b we compared the distribution of the COSMIC tumor samples in which the putative cancer-driving variants (PCVs) and putative benign variants were detected. The somatic variants annotated by the CMC project are found in different tumor tissues. In particular, the hotspot mutants in the p53 DNA-binding region are detected in tumors from more than 30 tissues.
2.3. Predicting the Folding Free Energy Change of the Protein Variants
For each mutant in our dataset, we predicted the variation of the folding free energy (ΔΔG
f) using FoldX, which is one of the most accurate methods for such a task [
79]. For each mutant, we averaged the FoldX predictions on 10 models of the mutated structure. We then compared the predicted and the experimental ΔΔG
f values and calculated the performance on the set of variants, either grouped by proteins or on the whole set. In particular, we calculated three types of correlation (Pearson, Spermann, Kendall-Tau), and two error estimates, the Root Mean Square Error (RMSE) and the Mean Absolute Error (MAE). The results in
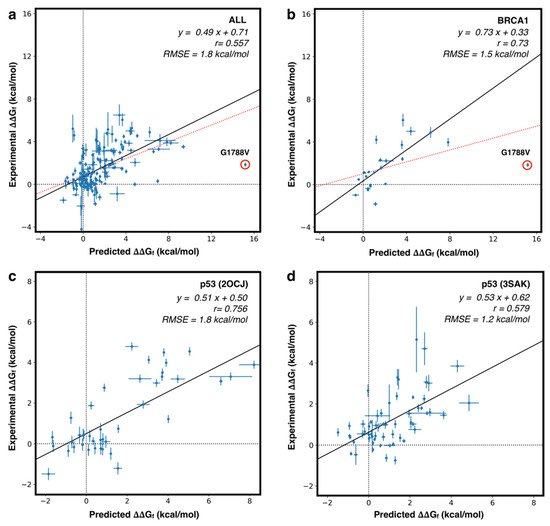
Table S2 show that, on the whole set of 164 variants, FoldX achieves a Pearson correlation coefficient (
rP) of 0.50 and a RMSE of 2.1 kcal/mol. This result can be improved by removing the variant p.G1788V of BRCA1 from the dataset. For that variant, FoldX predicts a ΔΔG
f of 15.2 kcal/mol, which is much higher than all other predicted values. Such a large predicted ΔΔG value is likely due to a limitation of FoldX, which in this case fails to identify a stable conformation of the protein mutant. After removing p.G1788V from the dataset, the
rP increased to 0.56 and the RMSE decreased to 1.8 kcal/mol. On average we observed that the predicted ΔΔG values returned by FoldX tend to be larger than the experimental values. This behavior is probably due to the implementation of the FoldX algorithm which predicts the structure of the mutant protein only considering different rotamers of the amino acid side chains. Such a process might limit the ability of the tool to identify more stable conformations that could be obtained through the rearrangement of the backbone.
Further analysis was performed after grouping the variants by proteins and calculating the performance on the resulting protein subsets. By doing so, we observed for some proteins (both domains of p53, hFXN, PGK1, and PTPρ) a rP > 0.58. For other proteins (BRDs, PIM-1, and p16) with a smaller number of mutants (≤10), we observed lower or negative correlation coefficients. The scatter plots, showing the correlation between predicted and experimental ΔΔGf values for the whole set of proteins, or for the proteins with the highest number of mutants (p53 and BRCA1), are shown in . Another interesting analysis consists in the prediction of highly destabilizing variants (ΔΔGf > 2 kcal/mol). In this case, we have used FoldX as a binary classifier, optimizing a threshold on its output.
Figure 5. Scatter plot for predicted vs experimental ΔΔGf values. (a) fitting on the whole set of 164 variants (red) and after removing the outlier BRCA1 p.G1788V (black). (b) Similar analysis performed on the whole set of variants in BRCA1 (red) and after removing BRCA1 p.G1788V variant (black). (c,d) fitting on the subset of variants in p53 core (2OCJ) and oligomeric (3SAK) domains, respectively. r: Pearson correlation coefficient. RMSE: Root Mean Square Error.
The optimization procedure, based on balancing the true positive and true negative rates, shows that FoldX can achieve an overall accuracy of 77% and a Matthews Correlation Coefficient (MCC) of 0.55 when a prediction threshold of ~1.2 kcal/mol is considered for the whole set of 164 variants. This method shows a good performance also when considering protein-specific thresholds. Indeed, for the subset of proteins with the highest number of mutants (p53 and BRCA1), the performance in the classification task reached MCC = 0.78 and AUC (Area Under the ROC curve) = 0.95 for the DNA binding domain of p53, or MCC = 0.67 and AUC = 0.90 for BRCA1. All performance measures in the classification task are summarized in
Table S3.
In general, our analysis confirms that, on average, the predicted and experimental ΔΔGf correlate well, and that the FoldX prediction can be used to estimate the impact of mutations of protein stability, in spite of the fact that the prediction error still remains ~2.0 kcal/mol. To partially address this limitation, the methods for ΔΔGf prediction can be used as binary classifiers to detect highly destabilizing protein variants.
2.4. Predicting the Pathogenicity of Protein Variants
In the last decade, several methods have been developed for predicting the pathogenicity of variants. In general, those approaches are binary classifiers, based on the analysis of evolutionary conservation. The idea behind these tools is based on the observation that mutations occurring in highly conserved regions of the protein are more likely to be pathogenic than mutations in variable regions. In the case of cancer-associated variants, the validation of the predictive methods is a difficult task due to the lack of curated sets of annotated variants. To address this issue, the COSMIC curators are annotating the somatic mutations in the Cancer Mutation Census (CMC) dataset [
19]. Currently, the CMC contains ~3 million missense variants, only ~0.1% of which were curated. Using such annotation, we analyzed the prediction of Meta-SNP, an algorithm combining the output of 4 methods, on our dataset. Initially, we analyzed the relationship between the experimental ΔΔG
f and the variant pathogenicity score returned by Meta-SNP, to test its performance in the detection of highly destabilizing variants (ΔΔG
f > 2 kcal/mol). Setting the optimized classification threshold to 0.66, we found that Meta-SNP reaches an accuracy of 73% and a Pearson correlation coefficient of 0.40 in the classification of highly destabilizing variants. We also estimated the performance of Meta-SNP in the prediction of putative cancer-driving variants (PCVs), assuming that missense variants, annotated as “Other” in the CMC database, can be classified as benign and variants in CMC annotated as classes 1–3 (Tier 1–3) can be considered putative cancer-driving variants (PCVs). For a more stringent test we calculated the performance of Meta-SNP by removing from the dataset 15 mutations used for the training of the method. Our results show that, for the subset of 82 variants annotated in CMC, by using a classification threshold of 0.71, Meta-SNP is able to predict PCVs with 77% accuracy and a Matthews correlation coefficient of 0.37.
The high fraction of false positives in the prediction of highly destabilizing variants may indicate the presence of pathogenicity mechanisms alternative to the loss of stability, while the high rate of false positives in the prediction of PCVs can be due to incorrect and/or incomplete protein variants annotation.
Although the Meta-SNP predictions result in a high fraction of false positive, the PCVs, annotated with 1 to 3 in the CMC database, are enriched in the variants with folding ΔΔGf > 2 kcal/mol with respect to the subset of CMC variant annotated as “Other”. Indeed, the relative p-value calculated by the Fisher test is <0.03.
Furthermore, the comparison of the distributions of the Meta-SNP output for Tier 1–3 and “Other” variants reveals a significant difference. The average values of the distributions of Meta-SNP outputs for Tier 1–3 and “Other” variants are 0.71 and 0.47, respectively. This difference is statistically significant, corresponding to a Kolmogorov-Smirnov p-value < 10−4.
Finally, we also tested the performance of FoldX in predicting PCVs. Our analysis revealed that, selecting a predicted ΔΔGf threshold of 2.7 kcal/mol, FoldX is able to identify Tier 1–3 variants with 73% overall accuracy and a Matthews correlation coefficient of 0.33. The comparison of the results shows that a predictor of putative pathogenic variants (Meta-SNP) is performing better than a method designed to predict folding ΔΔGf (FoldX) in the detection of PCVs.
2.5. Analysis of the Prediction on the Basis of the Amino Acid Accessibility and Conservation
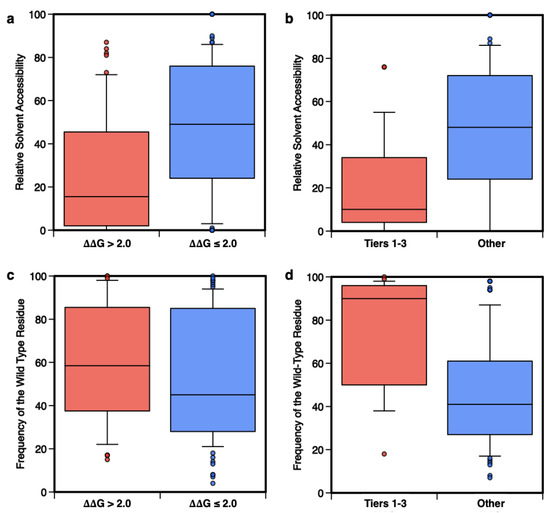
In the previous sections, we have shown that protein stability of cancer-related genes can be predicted with a good level of confidence using dedicated computational tools like FoldX. We have also observed that the pathogenicity score, calculated through a consensus method, correlates with protein stability data and with phenotypic data. Nevertheless, the prediction of PCVs, starting from protein stability predictions, is a more complex task. To this end, more experimental data on the stability of cancer proteins and their variants, and a higher level of curation of the existing databases on cancer protein variants would be needed. As an in-silico alternative for estimating the impact of protein variants on the stability and phenotypic levels, we used Meta-SNP, which is one of the state-of-the-art methods that best predict the protein variant pathogenic potential. To better analyze the results obtained by Meta-SNP, we calculated the distributions of solvent accessibility and conservation scores of the wild-type residues for the subset of highly destabilizing and PCVs.
In detail, for each mutated site we calculated the relative solvent accessibility (RSA) of the mutated residues and the frequency of the wild-type residue in the multiple sequence alignment (fWT) of possible homologs of the mutated protein.
As described in
supplementary materials, the RSA was calculated by normalizing the solvent accessibility calculated with the DSSP program [
126] and the
fWT was returned as part of the output of the Meta-SNP server (
http://snps.biofold.org/meta-snp, accessed on 1 May 2021). In the first part of our analysis, we compared the RSA values for the subset of highly destabilizing variants (ΔΔG > 2.0 kcal/mol) and the remaining ones, showing that for RSA ≤ 0.2 there is little overlap between the two distributions (a). In the same range of RSA (b), the PCVs (Tiers 1–3) can be easily discriminated from benign ones (“Other”). In both cases, using the Kolmogorov-Smirnov test to estimate the statistical difference between the two subsets in Figs. 6a and 6b, we obtained
p-values < 10
−3. In particular, b shows that the majority of PCVs (~58%) are occurring in buried regions (RSA ≤ 0.2), while ~75% of putative benign variants are in exposed regions (RSA > 0.2). The fraction of PCVs in exposed regions, which we found in our dataset, is higher than the value reported for pathogenic variants [
63], nevertheless, due to the reduced size of our dataset, such a difference is not statistically significant.
Figure 6. Distributions of the Relative Solvent Accessibility (RSA) and frequency of the wild-type residue (fwt) in the multiple sequences of homolog proteins. The distributions of RSA and fwt calculated for the subsets of 53 highly destabilizing variants (ΔΔGf > 2.0 kcal/mol) compared to the remaining 111 variants (ΔΔGf ≤ 2.0 kcal/mol) are shown in panels (a,c). In panels (b,d) the same distributions are plotted for the subsets of 24 putative cancer-driving (Tier 1–3) or 73 benign (‘Other’) variants.
In the second part of this analysis, different results are observed when the distributions of
fWT are compared. c shows that conservation is not a strong feature for the classification of highly destabilizing variants, while it is essential for the prediction of PCVs. Indeed, for
fWT > 50% the distributions of Tier 1–3 and “Other” variants have little overlap (d). The comparison between the results in c,d indicates that destabilization and conservation may indeed serve the pathogenicity prediction task as reciprocally integrating features [
25].
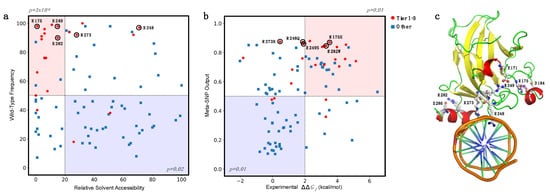
A further interesting analysis can be performed by considering the distribution in two dimensions of the RSA and fWT together for Tiers 1–3 and “Other” variants. In a we observed an enrichment for Tiers 1–3 variants in the buried (RSA < 20%) and conserved residues (fWT > 50%), with a corresponding p-value of 3 × 10−6, obtained by considering a binomial distribution with a success probability of 0.247. On the opposite side of the plot (RSA ≥ 20% and fWT ≤ 50%), we observed a depletion of PCVs (p-value = 0.02). Finally, we performed a similar analysis by combining the experimental ΔΔGf with the Meta-SNP predictions (b).
Figure 7. Enrichment and depletion of putative cancer-driving variants (Tier 1–3) in different subgroups. (a) Enrichment of Tier 1–3 variants in mutated sites with Relative Solvent Accessibility ≤20% and frequency of the wild-type residue > 50% (light red). (b) Enrichment of Tier 1–3 variants in the subset of mutants with experimental ΔΔGf > 2.0 kcal/mol and Meta-SNP output >0.5 (light red). In both cases, the opposite regions (light blue) are depleted of Tier 1–3 variants. In (a,b), the hotspot mutants R175, R248, R249, R273, and R282 are highlighted with black circles. (c) Hotspot sites in the p53 structure in interaction with DNA (PDB:1TUP). Residues R248 and R273 interact with DNA. Residues R175, R249, and R282 are likely to stabilize the protein structure by forming salt bridge interactions with D162, E171, and E286, respectively.
If we considered the subset of highly destabilizing (ΔΔGf > 2.0 kcal/mol) and predicted pathogenic (Meta-SNP output > 0.5) variants, we found an enrichment in Tier 1–3 variants with corresponding p-value of 0.01. On the opposite side of the plot, we observed a depletion of Tier 1–3 variants, again with a p-value of 0.01.
These observations confirm the hypothesis that relative solvent accessibility and amino acid conservation are important features for predicting the impact of amino acid substitution in terms of protein stability and pathogenicity. Furthermore, the combination of the experimental ΔΔGf and the predicted pathogenicity of variants allows to select a subset of variants with a significantly high probability of having a deleterious phenotypic effect.
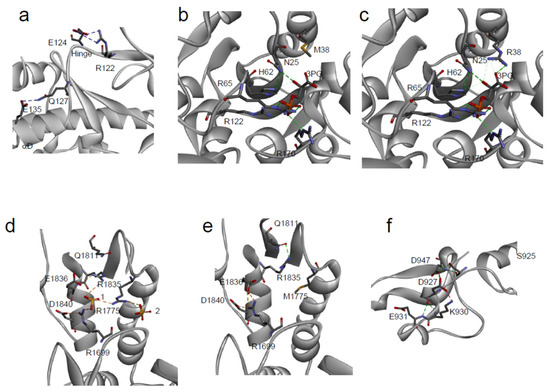
In particular, focusing on a subset of five hotspot sites of p53 [
127,
128], we observed that R248 and R273 are directly interacting with the DNA, in agreement with their high RSA, while R175, R249, and R282 (low RSA) are surrounding the DNA binding site (c). These structural aspects, combined with our predictions, support the hypothesis that the p.R248Q and p.R273H variants (with high pathogenicity score but low ΔΔG
f) have a direct impact on the protein function of DNA-interaction, while p.R175H and p.R282W (with both high pathogenicity score and high ΔΔG
f) destabilize the p53 structure. An intermediate case is p.R249S, which shows a variation of folding free energy of ~2 kcal/mol and a low RSA. Similar to R175 and R282, the presence of an oppositely charged residue (E171) in the proximity of R249 suggests that a mutation in this site can indeed reduce the stability of p53, due to a missing salt bridge interaction. Although a significant difference between predicted and experimental ΔΔG
f values (RMSE = 3.2 kcal/mol) is observed for the five hotspots, similar results are obtained when combining the Meta-SNP output with the predicted ΔΔG
f (
Figure S1). Our analysis can be compared with the experimental data on DNA-binding affinity of the p53 mutants [
82]. The data show that among the five hotspots cancer mutants shown in , the three of them with low impact on p53 stability (p.R248Q, p.R249S, and p.R273H with ΔΔG
f ≤ 2.0 kcal/mol) had no detectable binding affinity with the gadd45 promoter DNA (0% with respect to the wild type). This observation supports the hypothesis that protein–DNA interactions may play an important role in the cancer-inducing mechanism of the mutated p53. By analogy, a similar case of compromised protein–DNA or protein–protein interactions might turn out to hold for other cancer-associated mutants, which might not be in the ‘highly destabilizing’ category. The above observation is also in agreement with the possible roles hotspots cancer p53 mutants are considered to play as gain-of-function effectors [
127,
128], not only for the ‘contact’ mutants p.R248Q and p.R273H, but also for the ‘conformational’ mutants p.R175H, p.R249S, and p.R282W, since an altered p53 binding energy landscape can shift the mutated cells to different functionalities. Furthermore, the data shown in are consistent with the fact that also destabilizing variants can have gain-of-function characteristics, possibly through altered protein–protein interactions [
56].