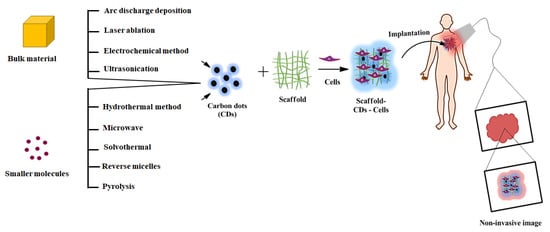
Regeneration of damaged tissues or organs is one of the significant challenges in tissue engineering and regenerative medicine. Many researchers have fabricated various scaffolds to accelerate the tissue regeneration process. However, most of the scaffolds are limited in clinical trials due to scaffold inconsistency, non-biodegradability, and lack of non-invasive techniques to monitor tissue regeneration after implantation. Recently, carbon dots (CDs) mediated fluorescent scaffolds are widely explored for the application of image-guided tissue engineering due to their controlled architecture, light-emitting ability, higher chemical and photostability, excellent biocompatibility, and biodegradability.
- carbon dots
- fluorescent scaffold
- image-guided tissue engineering
1. Background
2. Synthesis of Carbon Dots
2.1. Top-Down Approach
2.1.1. Arc Discharge Deposition Method
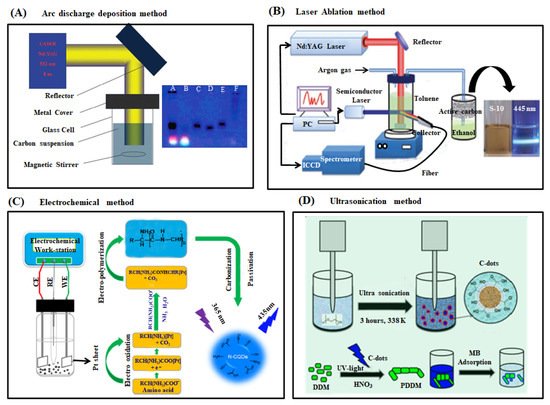
2.1.2. Laser Ablation Method
2.1.3. Electrochemical Synthesis
2.1.4. Ultrasonication
2.2. Bottom-Up Approaches
2.2.1. Hydrothermal Method
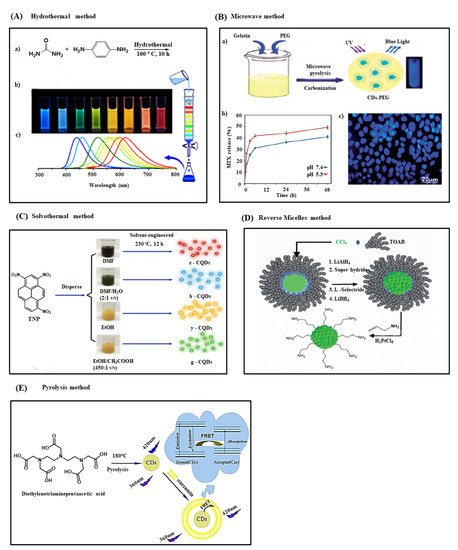
2.2.2. Microwave Method
2.2.3. Solvothermal Method
2.2.4. Reverse Micelles Method
2.2.5. Pyrolysis Method
| Synthetic Methods | Carbon Source | Reaction Condition | Size of CDs (nm) | Quantum Yield (%) | Application | Study Authors | |
|---|---|---|---|---|---|---|---|
| Top-down approaches | Arc discharge deposition | Arc soot | Electrodes, agarose gel | 1 | 1.6 | - | Xu et al. (2004) [64] |
| Arc discharge deposition | oxCNT | Electrodes | - | - | - | Bottini et al. (2006) [65] | |
| Arc discharge deposition | Graphite | Electrodes, current 100–150 A | 4–6 | 2.3–8.7 | - | Dey et al. (2014) [66] | |
| Laser ablation | Graphite powder and cement | Nd:YAG laser (1064 nm, 10 Hz) | 5 | 4–10 | - | Sun et al. (2006) [72] | |
| Laser ablation | Graphite powder | Nd:YAG laser (wavelength 1.64 µm, intensity 6.0 × 106 W cm−2), 2 h | 3.2–3.3 | 3–8 | - | Hu et al. (2009) [73] | |
| Laser ablation | Nano-carbon material | Nd:YAG laser wavelength 532 nm |
- | - | - | Li et al. (2011) [67] | |
| Electrochemical carbonization | Alcohols | Current density 15–100 mA cm−2, 4 h | 2.1–4.3 | 15.9 | Bioimaging | Deng et al. (2014) [74] | |
| Electrochemical carbonization | Amino acids | 1–10 V, 2 h | 2.95 ± 0.12 | 46.2 | Cellular imaging, Fe3+ ions detection, and fiber staining | Niu et al. (2017) [69] | |
| Electrochemical carbonization | Carbon fibers | 0.5–2.5 V, 2 h | 2.2 ± 0.6–3.3 ± 0.6 | 1.47 ± 0.20 | - | Bao et al. (2011) [75] | |
| Ultrasonication | Food waste | 20 kHz, 45 min | 4 | 2.85 | Bioimaging | Part et al. (2014) [77] | |
| Ultrasonication | Activated carbon | 40 kHz, 2 h | 5–10 | 5 | - | Li et al. (2011) [78] | |
| Ultrasonication | PEG-400 | 20 kHz, 230 V AC, 3 h | 6 | - | Catalyst | Maruthapandi et al. (2018) [70] | |
| Bottom-up approaches | Hydrothermal | Urea, p-phenylenediamine | 160 °C, 10 h | 2.6 | 35 | Bioimaging | Ding et al. (2016) [80] |
| Hydrothermal | Orange juice | 120 °C, 2.5 h | 2.5 | 26 | Cellular imaging | Sahu et al. (2012) [81] | |
| Hydrothermal | Chitosan | 180 °C, 10 h | 2–10 | 6.6 | Metal ion detection | Zhan et al. (2019) [82] | |
| Hydrothermal | Citric acid, ethylenediamine | 150–300 °C, 5 h |
2–6 | 80 | Printing ink, biosensor, and composites | Zhu et al. (2013) [83] | |
| Microwave | PEG-200 and saccharides | 500 W, 2–10 min | 2.75 ± 0.45–3.65 ± 0.6 | 3.1–6.3 | - | Zhu et al. (2009) [90] | |
| Microwave | Glycerol and TTDDA | 700 W | 3.5 | 12.02 | Cellular imaging | Liu et al. (2011) [91] | |
| Microwave | Lemon and onion biomass | 1450 W, 6 min | 6.15 | 23.6 | Detection of riboflavin | Monte-Filho et al. (2019) [92] | |
| Microwave | Gelatin and PEG | 600 W, 10 min | 6 | 34 | Bioimaging and anticancer | Arsalani et al. (2019) [84] | |
| Microwave | Aspirin and hydrazine | 500 W, 8 min | 2–5.5 | 23 | Cellular imaging and bioimaging | Xu et al. (2016) [93] | |
| Solvothermal | Spinach | 150 °C, 6 h | 3–11 | 15.34 | Bioimaging | Li et al. (2017) [95] | |
| Solvothermal | 1,3,6-trinitropyrene | 230 °C, 12 h | 2.25–3.98 | 6.4–59 | Bioimaging | Zhan et al. (2018) [85] | |
| Reverse micelles | CCl4, TOAB, and allylamine | Stir for 30 min | 2–6 | 27 | - | Linehan et al. (2014) [86] | |
| Solvothermal assisted reverse micelles | DS and Brij L4 | Stir for 30 min, 180 °C, 10 h | 5 ± 0.7 | 2.6 | - | Prikhozhdenko et al. (2018) [97] | |
| Reverse micelles | Glucose and AOT | 160 °C | ~2–4 | 35 | - | Kwon et al. (2012) [98] | |
| Pyrolysis | Glycerol and PEG | 230 °C, 30 min | 5.5 ± 1.1 | 20 | Cellular imaging and drug delivery | Lai et al. (2012) [99] | |
| Pyrolysis | DTPA | 180 °C | 5 | 17 | Detection of curcumin | Shi et al. (2015) [87] | |
| Pyrolysis | CA, EDA | 150 °C, 20 min | 5 | 88 | - | Yin et al. (2019) [101] | |
3. Properties of Carbon Dots
3.1. Mechanical Strength Concept
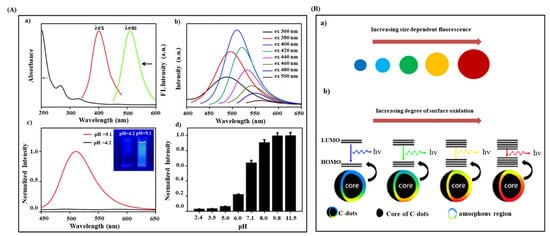
3.2. Optical Properties of CDs
4. CDs Mediated Scaffold for Tissue Engineering Application
| Composite of CQDs | Tissue Regenerative Scaffold and Methodology | Physicochemical Properties | Invasive/Non-Invasive Imaging Devices and Performances | Cellular Type | Salient Observations | Biomedical Applications | Study Authors |
|---|---|---|---|---|---|---|---|
| SF-CQD-PLA | Electrospun nanofiber | Higher porosity, better swelling ability, and Young’s modulus of 1610 kPa | CLSM/Cell proliferation | H9c2 rat myoblast cells | Increased cell viability and cardiac-maker gene expression | Tissue engineering and nursing care | Yan et al. (2019) [49] |
| CP-CQD-PCL | Electrospun nanofiber | Decreased fiber diameter, increased surface hydrophilicity, and Young’s modulus of 2.83 ± 0.23 MPa | SEM/Cell morphology and adhesion | MG-63 human osteoblast cells | Increased cell proliferation and ALP activity; more drug release at lower concentration | Bone tissue engineering | Ghorghi et al. (2020) [108] |
| CMCS-CQDAG-ODex | Injectable hydrogel/Chemical cross-linking method | Self-healing, stretchable, compressive property, pH dependent release of CQDs, and compressive modulus of 11.441 kPa | Fluorescence and optical microscopy/Cell adhesion survival and RBC imaging |
NIH-3T3 mouse embryonic fibroblast cells | Inhibition of biofilm formation; low drug resistance; better antibacterial and anti-inflammation; higher collagen deposition | Skin tissue regeneration | Li et al. (2021) [112] |
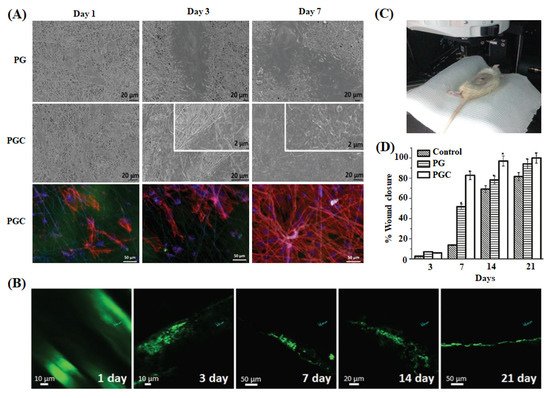
| BC-CQDs-Si NPs-SF& PVA-CQDs-Si NPs-SF | Nanofiber/Spray printing and electrospinning method | Moderate swelling, biodegradability, and Young’s modulus of 320 MPa & 70 MPa | Optical microscopy/Cell migration and tissue regeneration | NIH-3T3 fibroblasts | Better antibacterial activity; enhanced cell viability, proliferation, and re-epithelialization | Skin tissue and hair regeneration | Abolghasemzade et al. (2021) [110] |
| PCL-CNDs-gelatin | Electrospun nanofiber | Fiber diameter of 698 ± 420 nm, pore diameter of 2.93 ± 1.13 µm, photo stability, and Young’s modulus of 0.866 ± 0.06 MPa | SEM and two-photon microscopy/Cell attachment, growth, cytotoxicity, and skin regeneration kinetics | Human fibroblasts and keratinocyte cells | Increased antioxidant activity; accelerated cell adhesion, migration, and proliferation | Skin tissue regeneration | Pal et al. (2017) [146] |
| CQDs-chitosan | Film/Solvent casting method | Decrease in water absorption and moisture permeation and 1.3-fold flexibility | - | - | Enhanced antioxidant activity | Skin tissue regeneration and drug delivery | Kandra et al. (2020) [147] |
| PCL-PVA-TCP-CD | Electrospun nanofiber | Fiber diameter range of 300–414.8 nm and contact angle of 33–45° | SEM and CLSM/Cell attachment, morphology viability, and proliferation | Human buccal fat pad-derived stem cells (hBFPSCs) | Enhanced ALP activity, osteogenic differentiation, and cell proliferation | Bone tissue engineering | Shafiei et al. (2019) [145] |
| CDP-f-PU | Injectable hydrogel/Chemical cross-linking method | Moderate biodegradation | Phase contrast and CLSM/Cell viability, adhesion, migration, morphology, and vascularization pattern | MG-63 human osteoblasts | Better biocompatibility, osteoblast adhesion, proliferation, and differentiation | Bone tissue engineering | Gogoi et al. (2017) [148] |
| CD-HAp-PU | Film/Chemical cross-linking method | Dose dependent thermal stability and Young’s modulus of 28.42 ± 0.35 MPa | Fluorescence microscopy/Cell morphology, viability, and proliferation | MG-63 human osteoblasts | Higher cytocompatibility, cell proliferation, and ALP activity | Bone tissue engineering | Gogoi et al. (2016) [116] |
| Chitosan-CDs | Hydrogel/Solvent casting method | Higher pH sensitivity and Young’s modulus of 2.90 ± 0.15 GPa | Fluorescence microscopy/Cell viability and proliferation | L929 mouse fibroblasts | More antibacterial activity; better biocompatibility | Skin tissue regeneration | Omidi et al. (2017) [109] |
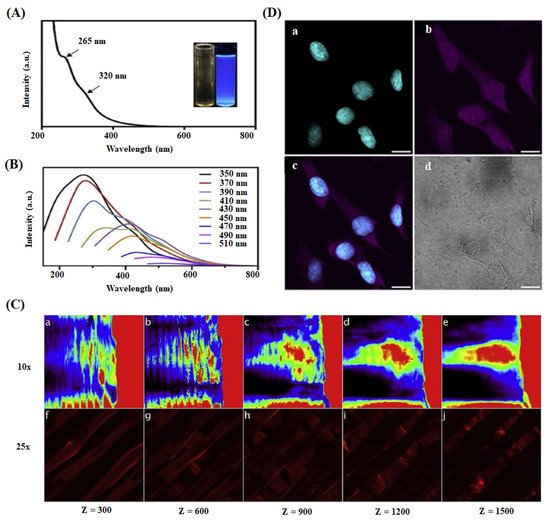
| PLA-CQDs | Scaffold/3D Printing method | Long term photostability and pH flexible fluorescence | Two-photon & multi-photon microscopy/Cell-scaffold imaging | RL-14 human fetal ventricular cardiomyocyte cell | Excellent biocompatibility; deep cellular and scaffold imaging | Tissue engineering | Dehghani et al. (2018) [26] |
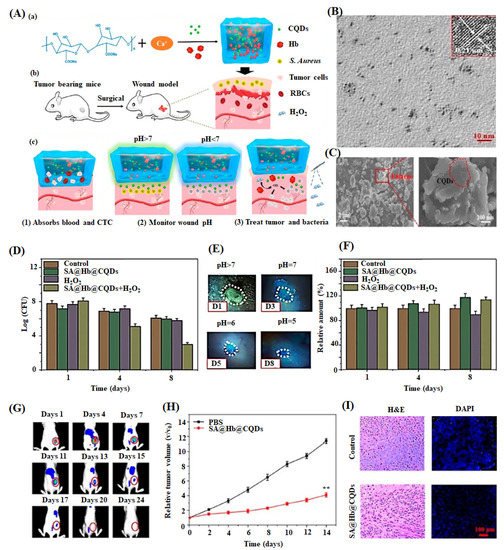
| SA-Hb-CQDs | Injectable hydrogel/Phase separation | Contact angle of 42.5°, higher pH sensitivity, and excellent biodegradability | CLSM/Cell viability | NIH-3T3 fibroblasts | Enhanced biocompatibility and antibacterial activity | Skin tissue regeneration | Zhang et al. (2020) [149] |
This entry is adapted from the peer-reviewed paper 10.3390/ijms22105378
