Mycotoxins are secondary metabolites synthesized by an array of fungal genera, usually Fusarium, Penicillium and Aspergillus.
- ergot alkaloid mycotoxins
- cereals
- cereal-based product safety
- quality
- characteristics
- control strategies
- occurrence
- toxicity
- health impacts
1. Introduction
Cereals and related foodstuff are rich in nutrients and energy, and for this reason they play an important role in the diet. The abundance of nutrients contained in cereals such as fat, protein and minerals are also favorite nutrients for the growth of toxin-producing fungal species [1][2][3][4][5]. Crops and especially cereals can be infected in both pre-harvest and post-harvest stage by phytopathogenic fungi [2], with many factors being responsible for their contamination such as temperature, water activity, and substrate composition [6]. Mycotoxins are toxic chemicals substances and arise as products of secondary fungal metabolism, posing a very serious risk to food safety worldwide [7]. Mycotoxins have low molecular weight and cannot be detected with the naked eye, smell, or taste. In addition, their presence in food does not cause changes in organoleptic characteristics [8][9]. The term “mycotoxicosis” was used for the first time about 60 years ago to describe all the toxic effects of mycotoxins on human and animal health [10]. The effects of mycotoxins on human and animal health are carcinogenic, teratogenic, neurotoxic, hepatotoxic, and nephrotoxic [11].
Ergot alkaloids (EAs) is one class of mycotoxins, mainly produced by fungal Claviceps species, whose effects have been known since the fifth century AD. Ergotism, the oldest known mycotoxicosis, is the human and animal disease historically known as St. Anthony’s Fire, or holy fire, which occurs after consuming EAs contaminated food or feed [12][13][14]. The first documented ergotism epidemic occurred in 944–945 AD in France, and led to the deaths of about 10,000 people [15]. From the 9th to 14th centuries, the outbreak of ergotism was widespread in the eastern regions of France, Germany, and Russia [16]. Ergot is derived from the old French word “argot”, meaning the cock’s spur [17]. Cereal contamination and damage may also occur with other mycotoxins, such as aflatoxins (AFs); ochratoxin A (OTA); zearalenone (ZEN); fumonisins (FBs); enniatins (ENs); and trichothecenes (TCs) such as deoxynivalenol (DON), T-2, and HT-2 toxins (T-2, HT-2) [7][8], with AFs being extremely harmful to the health of humans and animals [12][18][19].
According to their structure, EAs are classified into three groups, namely, clavines, lysergic acid amides, and peptides (ergopeptines) [20]. EAs are classified as a tryptophan-derived alkaloid, and their analytical classification has been reviewed by Arroyo-Manzanares et al. [21]. Although there are more than known 80 EAs, all having a tetracyclic ergoline ring in their structure [6], the most commonly produced EAs for Claviceps species are the following 12: the ergometrine (Em), ergotamine (Et), ergocristine (Ecr), ergokryptine (Ekr), ergosine (Es), and ergocornine (Eco), and their inine-epimeric forms ergotaminine (Etn), egometrinine (Emn), egocristinine (Ecrn), ergokryptinine (Ekrn), ergocroninine (Econ), and ergosinine (Esn) [2][3][22][23]. Ergotamine and ergosine are very stable alkaloids and their concentrations are not significantly affected by heating. In contrast, for ergocristine, ergokryptine, ergocornine, and ergometrine, heating can decrease the concentrations of these alkaloids [24]. Each alkaloid coexists under two forms, the ergopeptines and ergopeptinines, which are different in terms of toxicity and biological and physicochemical properties [24][25], with ergopeptins being the left-hand rotation isomers of EAs (suffix: -in) and ergopeptinines being the right-hand rotation isomers of EAs (suffix: -inin). Among them, ergopeptines are biologically active and ergopeptinines are less biologically active [26]. Aqueous acidic or alkaline solutions accelerate the conversion of ergopeptines to ergopeptinines. On the other hand, ergopeptinines can also be converted in ergopeptines in methanol, aqueous organic solvent, and acids [27]. Therefore, it is important that assays performed for EAs must include both epimers in order to avoid underestimation of the total quantity of EAs as rapid epimerization can occur [28]. Solvents, temperature, and pH are the main factors on which the isomerization depends [29][30]. Individual fungal strain, host plant, growth conditions, and geographical area are the main factors which significantly affect the amount and pattern of EAs [31]. Convolvulaceae, Poaceae, and Polygalaceae are some families of higher plants in which EAs have also been detected [32]. Claviceps purpurea, Claviceps fusiformis, Claviceps africana, Epichloë spp., and Neotyphodium spp. are some EA producers [12][33][34]. Moreover Blansia, Penicillium, and Aspergillus also produce some quantity of EAs [20]. Serious damage and losses can be caused to the production of cereals by the aforementioned species. Recent damage to the cultivation of Sorghum bicolor caused by C. africana has led to financial losses due to reduced seed quality and yield, creating difficulties in harvesting and threshing and problems in the international trade in contaminated seeds [35]. Moreover, the poisoning of people through the consumption of food contaminated with sclerotia (the over-wintering bodies of the fungus) in developing countries shows that the problem of ergotism has not been definitively solved and remains a significant threat. A total of 47 deaths were reported as having been due to ergotism during the spring of 1978 in Ethiopia after consumption of barley contaminated with C. purpurea sclerotia [36].
On the other hand, the proven pharmaceutical and therapeutic nature of EAs has focused research on their laboratory production for commercial purposes [37]. In the last 20 years, EAs biosynthesis has achieved important progress [32]. Various fermentation techniques have been used to commercially produce EAs for the manufacture of medicines. The addition of various organic and inorganic ingredients to a fermentation medium has improved the industrial production of EAs [38]. For the past two decades, EAs have been utilized in the medical field for treatments related to migraines, brain diseases, hypertension, central nervous system diseases, Parkinson’s disease, and in gynecology as prolactin inhibitors [15][35][39]. EAs show structural similarities to the neurotransmitters noradrenaline, dopamine, adrenaline, and serotonin, enabling binding to the biogenic amine receptor and the interruption of neurotransmission and linking their biological activity to a number of drugs in the pharmatech industry [15][40].
Claviceps purpurea from the Clavicipitaceae family is the most widespread Claviceps species, with greatest concern, all over the Europe. During C. purpurea infection, healthy kernels are replaced by purple-black-colored sclerotia (also known as ergots, or ergot bodies) that contain high concentrations of various EAs. Moreover, due to the color of sclerotia, the term purpurea has been adopted for the parasitic fungus C. purpurea [41][42][43].
Diseases caused by EAs producing fungi on cereals are also characterized by the term ergot [17]. Pure ergots are soluble in organic solvents while others such as those of the lysergic acid group show partial solubility in water [11]. The young and usually unfertilized ovary (the female parts of the flower) of the living host plant is parasitized by fungal hyphae at the time of flowering, and after about a month, alkaloid-containing sclerotia appear in the healthy kernels [44]. The size of sclerotia compared to the size of healthy grains can be up to 10 times larger [45]. The presence of sclerotia is linearly related to the detection and identification of EAs, reducing the quality of cereals, while their absence does not imply the absence of EAs since there are cases where EAs were detected without the presence of sclerotia [46][47]. Sclerotia are the final step of the disease, and they contain 0.01–0.50% ergot alkaloids [25]. The number of ergots left in perennial cereal fields after harvest is a significant source of contamination [48] as each ergot has the potential to produce thousands of ascospores [49]. In the case of perennial grass seed crops, it is very difficult to implement ergot control agronomic practices related to deep tillage or crop rotation [50]. The chemical structure of the most common EAs and the structure of the tetracyclic ergoline ring system are presented in Figure 1.
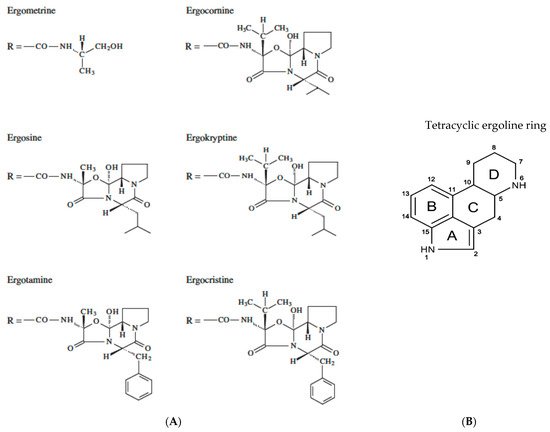
Figure 1. (A) Chemical structure of the most common ergot alkaloids (EAs). (B) The structure of the tetracyclic ergoline ring system.
The period of rainfall and mainly the period of flowering are the two critical periods in which the fungus parasitizes the seed heads of living plants [51]. EAs could be detected in grains even when ergot bodies were removed by hand-cleaning procedure [52]. The sclerotia are harvested together with the cereals or grass and can thus lead to contamination of cereal-based food and feed products with EAs [53]. Moreover, during the milling of the cereals, sclerotia are not easily separated from the healthy grains, and instead they are fragmented and can be transferred to finished product, the flour [54]. Cleaning protocols become even more unreliable when fungal sclerotia are produced in dry climates, having a similar size to the grains, making them difficult to remove [17]. Cleaning and processing of cereals can remove up to 82% of the sclerotia [55] without this meaning that they completely eliminate the problem of contamination of cereals with EAs, which can be detected at high levels [56], such as, for example, 7255 mg/kg that was reported in German rye flours [17].
At least 400 species of the Poaceae family have been found to be parasitized by C. purpurea, among them the weedy grasses, with cereals such as rye, wheat, triticale, barley, millet, and oats being the most infected [57][58][59]. Some cereals such as rye become more frequently infected with Claviceps, while others such as maize become less infected [28].
Commission Regulation (EC) No. 1881/2006 sets maximum levels for certain contaminants in foodstuffs among them and main mycotoxins [60]. Commission Regulation (EC) No. 2015/2940 sets a limit of 0.5 g/kg for the ergot sclerotia content in unprocessed cereals, with the exception of corn and rice, for human consumption [61], and the EU Council Directive 2002/32/EC has established a maximum limit of 0.1% by body weight (b.w.) for all feed containing unground cereals [62]. According to the scientific opinion of the European Agency for Food Safety Authority (EFSA) for EAs in food and feed, which was established in July 2012, the setting of a group acute reference dose (ARfD) of 1 µg/kg b.w. and a group tolerable daily intake (TDI) of 0.6 µg/kg b.w. per day for the sum of the EAs were reported [63][64]. Canada and the United States have set 300 mg ergot per kilogram grain as maximum permissible level, and 0.01% of the total ergot alkaloid content in grain was established by China [51]. A 0.02% w/w limit on sclerotia content in cereals will be expected to be established by the EU, and its implementation is expected to begin in July 2021 [65].
Among all available techniques, liquid chromatography-tandem mass spectrometry (LC–MS/MS), is the fundamental tool for quantitative mycotoxin analysis, especially in the case of multiple mycotoxin analysis. Given that many food categories are contaminated with more than one mycotoxin, this technique is constantly gaining the interest of researchers [66]. For EA analysis, in addition to LC–MS/MS technique [67], liquid chromatography coupled with fluorescence detectors (LC–FLD), is also used as a reliable alternative [68] as well as QuEChERS techniques (Quick, Easy, Cheap, Effective, Rugged, and Safe) [69].
2. Occurrence of Major Ergot Alkaloid Mycotoxins
According to the annual report of 2019 of the Rapid Alert System for Food and Feed (RARFF), mycotoxins are the most frequently reported hazard [70]. Although ergot alkaloids mycotoxins were not included in the last annual report of RASFF (in 2019), this does not mean that there is no risk of this class of mycotoxins for human and animal health. In actuality, for 2019, according to the last annual report of RASFF, only 1 of the 32 notifications for mycotoxins concerned cereals, specifically Belgian wheat flour with toxin T-2 and toxin HT-2. Moreover, very recently it was emphasized that the detection of mycotoxins worldwide has reached a very high level of 60–80%, and even the lowest presence and detection of mycotoxins should not be overlooked because of their combined action and their synergistic negative health effects on human and animal health [71].
EFSA has recommended that detailed data on ergot alkaloids in food and feedstuffs should be continued due to their high toxic effects. Available analytical results from food samples collected between 2011 and 2016 in 15 different European countries allowed EFSA to express its scientific opinion about human and animal dietary exposure to ergot alkaloids [46]. The products found with the highest contamination were “rye milling products”, “mixed wheat and rye bread and rolls”, “rye bread and rolls”, and “rye flakes”, proving that rye is more contaminated among cereals. In fact, rye is a cross-pollinator with large open florets, and this explains its great sensitivity to contamination by ergot alkaloids [72]. The German Federal Institute for Risk Assessment (BfR) in 2013, taking into account the report of EFSA, reported that pregnant women and children are the two risk groups with the most serious adverse health effects. In addition, the value of 64 μg/kg of EAs is not safe for children aged two to five years, due to their low body weight in relation to bread consumption [73].
As well as the fact that Claviceps species can contaminate the most important cereal grains, the presence of these mycotoxins is expected in some cereal-based foods such as bread, pasta, pizza, breakfast cereals, noodles, cakes and sweet snacks, biscuits, savory fine bakery products, cookies, and baby foods. All these foods are part of the daily human diet that is consumed by infants, children, adolescents, and the elderly, and therefore must be safe. It has recently been shown that EA is more common in foods containing rye and wheat. Malysheva et al. reported high rates of contamination from EAs in rye- and wheat-containing foods in northern Europe. Rye-containing foods were found to have 1121 μg/kg and wheat-containing foods were found to have 591 μg/kg, respectively [74]. In a recent Swiss study, 16 Claviceps metabolites (ergine, ergocornine, ergocorninin, ergocristine, ergocristinine, ergocryptine, ergocryptinine, ergometrine, ergometrinine, ergosine, ergosinin, ergotamine, ergotaminine, secalonic acid B, secalonic acid D, and secalonic acid F) were detected in 253 barley samples, originating from fields across Switzerland, recording a small percentage of positive samples (3–17%) depending on the metabolite [75]. Ιn western Canada, six ergot alkaloids (ergocornine, ergocristine, ergocryptine, ergometrine, ergosine, and ergotamine) were detected in 49 out of 67 naturally contaminated cool-season barley grains, with the mean concentrations ranging from 121 to 555 μg kg−1 [52].
EAs have been reported both in European countries such as Italy [57], France [76], Slovenia [41], the United Kingdom [25], Albania [77], Poland [78][79], the Netherlands [80], and Germany [43], as well as in other countries around the world such as Canada [72][81][82], China [51], Brazil [83], and Australia [84]. The last 10 years have shown an increased occurrence both in Canada [85][86] and the United States [87].
Recent representative studies with presence of EAs in different food products and in different countries are presented in Table 1.
Table 1. Presence of ergot alkaloids in different food products in different countries.
| Product | Country | No. of Samples | Positive Samples (%) | Range (µg/Kg) | Method | Reference |
|---|---|---|---|---|---|---|
| Rye-based products Wheat-based products |
Italy | 16 55 |
85 | 2.6–188.6 2.5–1142.6 |
LC–MS/MS | [57] |
| Cereals | Canada | 228 | n.a. 1 | 65–1140 | LC–MS/MS | [72] |
| Barley | Czech Republic | 15 | n.d. 2 | n.d. 2 | UHPLC–MS | [88] |
| Cereal products | China | 123 | n.d. 2 | n.d. 2 | LC–MS/MS | [51] |
| Cereal grains | Slovenia | 517 | 17 | 14–4217 | LC–MS/MS | [41] |
| Winter rye | German Austria Poland |
372 | 40 | 0.52–3673.9 | HPLC | [23] |
| Cereals and cereal products | European countries 3 | 1065 | 59 | 1–12340 | LC–MS/MS | [74] |
| Organic rye flour Conventional rye flour |
Denmark | 17 17 |
88 88 |
n.a-100 n.a-230 |
HPLC–FLD | [53] |
| Rye grain | Poland | 18 | 83 | 0.8–25 | LC–IT–MS/MS | [78] |
| Rye flour | 34 | 94 | 0.9–1215.5 | |||
| Rye bran | 12 | 100 | 1.5–46 | |||
| Rye flakes | 1 | 100 | 114–136.5 | |||
| Cereal-based food | The Netherlands | 113 | 54 | n.a.–115.4 | LC–MS/MS | [80] |
| Cereals and cereal products | Belgium | 122 | 85 | 1–1145 | LC–MS/MS | [13] |
| Cereal samples | Algeria | 60 | 20 | 3.7–76.0 | QuEChERS–UHPLC–MS/MS | [89] |
| Rye product samples | Germany | 39 | 92 | n.d.–739.7 | HPLC–FLD | [90] |
| Wheat and maize samples |
European countries 4 | 13 wheat 15 maize |
77 n.d. 2 |
n.a. 1 | LC–MS/MS | [91] |
1 n.a.: not available in the publication. 2 n.d.: not detected. 3 European countries: Belgium, Czech Republic, Denmark, Estonia, Finland, France, Germany, Italy, Poland, Sweden, Switzerland, the Netherlands, the UK. 4 European countries: Belgium, Spain, Italy, Switzerland, Lithuania, Hungary.
3. Toxicity and Health Impacts of Major Ergot Alkaloid Mycotoxins
EAs cause severe health problems both in humans and animals, leading mainly to ergotism disease that occurs in two forms: a convulsive form and a gangrenous form [92]. Ergotism is one of the oldest known human diseases caused by mycotoxins. Hallucinations, convulsions, agalactia, burning sensation, vasoconstriction, and gangrenous loss of limbs are some of symptoms in humans after consuming foods contaminated with EAs [72][81][93]. Moreover, other symptoms such as nausea, alterations in endocrine function, vomiting, weakness, numbness, adverse effects to the cardiovascular system, and even death have also been reported [21][94][95][96]. In Ethiopia in 2001, a gangrenous ergotism outbreak was caused after consumption of contaminated barley [97].
Pigs, cattle, sheep, rabbits, and poultry have shown symptoms after being infected with EAs causing financial problems to both breeders and the meat industry [14][98]. Animals can become infected with EAs after eating EA-contaminated grains, and various diseases can affect any part of their body. In poultry in particular, both the comb of their legs and the beak can turn black. Some other general clinical signs whose expression may differ significantly between animal species concern disorders of the nervous system, dyspnea, and agalactia [45]. Necrosis of the extremities is another symptom that may not be seen in high-temperature environments, although animals suffer from overheating [99]. Alterations in the liver and intestine of pigs have also been reported when the diets they received contained ergotia in quantities close to the established European limits (1 g sclerotia/kg), indicating the need to update these limits and the possible adoption of lower limits [96].
Despite the recognized toxic risk that is reported according to literature, EAs have been used for their biomedical applications, as they exhibit antibacterial, antiproliferative, and antioxidant effects. EAs are efficiently used as therapeutic agents in ocular pharmacology, particularly against glaucoma [94].
4. Control Strategies of Major Ergot Alkaloid Mycotoxins
Generally, fungi are ubiquitous, and mycotoxin contamination is difficult to predict as many factors are responsible for annual fluctuations. Every year, climatic conditions, growing, harvesting, fungal growth, weather, and crop management are different, and the result in the appearance of mycotoxins in important agricultural products is also different [99]. Each strategy has its own effectiveness, as many parameters, such as cost, safety issues, and the initial contamination levels, determine its success [100]. Mycotoxins can infect crops at any stage of the growing process, including harvesting, transportation, and storage [101]. Control of EA mycotoxins in cereals should focus on two main stages, namely, pre-harvest and post-harvest.
4.1. Pre-Harvest Ergot Alkaloid Prevention Strategies
The adoption of Good Agricultural Practice (GAP), Good Manufacturing Practice (GMP), and Hazard Analysis and Critical Control Points (HACCP) practices play a significant role in the management of mycotoxins [11].
4.1.1. Biological Control
Consumer concerns about the application of pesticides on the one hand and the development of resistance of many phytopathogenic fungi to the use of fungicides on the other hand have led to the search for more environmentally friendly solutions to the problem of contamination of agricultural production with mycotoxins. Fighting many phytopathogenic fungi using biological means can work collaboratively with chemicals, although biological agents such as microorganisms have a limited range of targets compared to chemical fungicides [102].
Yeast, bacteria, fungi, and enzymes have been used as biocontrol agents in order to cause degradation or absorption of many mycotoxins [103][104][105][106]. Transformation, adsorption, degradation, or adsorption are some of the mechanisms by which agricultural products of plant or animal origin as well as animal feed can be detoxified from mycotoxins [103].
In fact, the number of published works on the biological control of EAs is limited. Bacteria Pseudomonas aureofaciens and fungus Trichoderma lignorum have been tested for biological control against EAs, but no significant effect on sclerotia germination was reported [107]. Fungus Fusarium roseum has also been tested effectively against sclerotia in wheat [31].
Rhodococcus erythropolis bacteria have been used to degrade EAs, although the mechanism of reaction is not completely clear [108].
4.1.2. Good Agricultural Practices
The role of sclerotia in the survival of the fungus Claviceps is extremely important as they are the overwintering structures of the pathogen. Moreover, a period of cold temperatures is required in order to germinate in the next spring [109]. The use of calcium cyanamide as fertilizer in the spring proved to be very effective as it caused a reduction of germination of ergots that were in the soil by 40–50%. Agricultural practices related to crop rotation, weed control, and herbicide application, fertilizers for nutrient enrichment, could contribute to controlling EA mycotoxins by helping to maintain a robust and therefore non-vulnerable crop [31]. Given that sclerotia can survive from one to three years, crop rotation can reduce the amount of sclerotia produced. Weed control is extremely helpful as sclerotia can also be produced both from the grass weeds of the main crop and headlands of field [82].
The use of uninfected seeds should be the main preventive action against the cereal contamination by EAs. In case the sclerotia mature and fall to the soil, it is recommended that fungicides be applied to the soil in order to reduce or even prevent their germination [110]. Sclerotia can be also found in the soil during sowing as the seed is not completely clean but also contains sclerotia. In this case, the sclerotia should be treated with a fungicide to minimize the possibility of producing large numbers of ascospores [31]. In both treatments, a reduction in the amount of ascospores as well as the primary inoculum available in the spring is achieved. A good cultivation practice that could also help to reduce the amount of sclerotia is to bury them through a deep plowing. The use of resistant or less susceptible varieties are some useful cultivation practices in order to reduce the content of sclerotia in cereal crops [31].
The treatment of many fungal diseases is achieved by many means, with the use of fungicides undoubtedly being the most widely used practice. The active ingredients found in fungicides vary and all primarily aim to reduce the growth of the pathogen as well as to prevent seeding [111]. The use of chemical fungicides could possibly contribute for the control of EAs.
4.2. Harvest and Post-Harvest Ergot Alkaloids Prevention Strategies
As the grains ripen and are ready for harvest, sclerotia control can be partially effective as long as they are visible. A harvesting practice that can lead to crops with low rates of sclerotia is late harvest because possible winds will favor the removal of sclerotia from the mother plants. In the case of parcels where visible contamination by sclerotia is important, it is preferable to harvest these pieces separately and not to mix the heavily contaminated grain seed with the uninfected seed, nor to use it as animal feed. Burial of significantly contaminated grain is more appropriate in this case. Additionally, a post-harvest handling for the removal of sclerotia in loads with moderate contamination is the use of gravity sorters and the use of color sorters [112].
4.2.1. Decontamination
The applied decontamination techniques are mainly aimed in two directions. The first aims to degrade mycotoxins to non- or less toxic compounds, and the second to prevent the reduction of nutrients contained in agricultural products. Every degradation creates products that are often unknown and have unknown toxicity, and therefore the term decontamination is often not completely synonymous with the term detoxification [113][114].
Heating is a physical way to convert the toxic forms of EAs (ergotamine) to less toxic forms of EAs (ergotaminine). Among the basic six different alkaloids, ergotamine and ergosine are very stable ergot alkaloids in heat treatments, while on the contrary, ergocristine, ergokryptine, ergocornine, and ergometrine are affected more from heating and their concentration may be reduced. Even in this case, however, the problem of contamination of feed with EAs is not solved since conversion of contaminated feed into non-contaminated feed cannot be achieved. UV light (302 nm) has also been used for epimerization of EAs, but for ergotamine and ergosine there were no signs of any epimerization, whereas for ergocristine, ergometrine, ergokryptine, and ergocornine, epimerization was reported at 74–90%, 2–17%, 9–25%, and 39–70%, respectively [24]. In addition, the high cost of irradiation units often makes the use of this technology prohibitive [115].
For cases of minor infection by mycotoxins, some physical decontamination techniques, such as sorting, washing, milling, and floating, could be a good choice, especially for the removal of heavy weight contaminated particles [116].
4.2.2. Fate of Ergot Alkaloids during Food Processing
Grain processing is an approach by which EAs could be possible eliminate [117], although common processing techniques such as boiling, cooking, baking, frying, and pasteurization fail to reduce most mycotoxins that are considered stable chemical compounds [10][118][119]. From the limited published data, relative with EAs decontamination, most of them are focused on rye and rye products, with differences in results. The different rate of degradation that occurs is attributed to the different nature of ergot contamination [120].
Cookies baked with rye flour were studied, and from the results the researchers proved a degradation of EAs (2–30%) during baking and a shift from ergopeptins to ergopeptinines for all EAs [29]. In a very recent study, Tittlemier et al. studied the fate of EAs during laboratory-scale durum processing and production and cooking of pasta. The authors reported that the loss of EAs during the production or cooking of pasta was not fixed but changes were observed during the grinding and cooking of pasta between R-enantiomers and S-enantiomers [120]. In addition, the results of the study showed that a large percentage (84%) of the EAs were found in the outer regions of the seeds (bran, shorts, feeds), although S-enantiomers were identified in a larger proportion. The effect of bread baking on the stability of ergot alkaloids in wheat and rye flour was also investigated in two recent studies. From the results of the studies, baking of wheat flour reduced the alkaloid concentrations by 59% [121], whereas only 22% elimination of ergot alkaloids was reported by Bryła et al. after baking of rye flour [117].
This entry is adapted from the peer-reviewed paper 10.3390/agronomy11050931
References
- Smaoui, S.; Bra, O.B.; Hlima, H.B. Mycotoxins analysis in cereals and related foodstuffs by liquid chromatography-tandem mass spectrometry techniques. J. Food Qual. 2020.
- Lee, H.J.; Ryu, D. Worldwide occurrence of mycotoxins in cereals and cereal-derived food products: Public health perspectives of their co-occurrence. J. Agric. Food Chem. 2017, 65, 7034–7051.
- Khaneghah, A.M.; Farhadi, A.; Nematollahi, A.; Vasseghian, Y.; Fakhri, Y. A systematic review and meta-analysis to investigate the concentration and prevalence of trichothecenes in the cereal-based food. Trends Food Sci. Technol. 2020, 102, 193–202.
- Kyei, N.N.A.; Boakye, D.; Gabrysch, S. Maternal mycotoxin exposure and adverse pregnancy outcomes: A systematic review. Mycotoxin Res. 2020, 28, 1–13.
- Varzakas, T. Quality and safety aspects of cereals (wheat) and their products. Crit. Rev. Food Sci. Nutr. 2016, 56, 2495–2510.
- Kifer, D.; Sulyok, M.; Jakšić, D.; Krska, R.; Klarić, Š.M. Fungi and their metabolites in grain from individual households in Croatia. Food Addit. Contam. Part B 2021, 16, 1–12.
- Bessaire, T.; Mujahid, C.; Mottier, P.; Desmarchelier, A. Multiple mycotoxins determination in food by LC-MS/MS: An international collaborative study. Toxins 2019, 11, 658.
- Winter, G.; Pereg, L. A review on the relation between soil and mycotoxins: Effect of aflatoxin on field, food and finance. Eur. J. Soil Sci. 2019, 70, 882–897.
- Agriopoulou, S. Physicochemical Study Using Liquid Chromatography Technique on the Effect of Ozone on Aflatoxins’ Degradation and Elimination, in Pure Substrates and Foods. Ph.D. Thesis, University of Patras, Patras, Greece, 2015.
- Kaushik, G. Effect of processing on mycotoxin content in grains. Crit. Rev. Food Sci. Nutr. 2015, 55, 1672–1683.
- Pleadin, J.; Frece, J.; Markov, K. Mycotoxins in food and feed. Adv. Food Nutr. Res. 2019, 89, 297–345.
- Agriopoulou, S.; Stamatelopoulou, E.; Varzakas, T. Advances in occurrence, importance, and mycotoxin control strategies: Prevention and detoxification in foods. Foods 2020, 9, 137.
- Di Mavungu, J.D.; Malysheva, S.V.; Sanders, M.; Larionova, D.; Robbens, J.; Dubruel, P.; van Peteghem, C.; de Saeger, S. Development and validation of a new LC-MS/MS method for the simultaneous determination of six major ergot alkaloids and their corresponding epimers. Application to some food and feed commodities. Food Chem. 2012, 135, 292–303.
- Korn, A.K.; Gross, M.; Usleber, E.; Thom, N.; Köhler, K.; Erhardt, G. Dietary ergot alkaloids as a possible cause of tail necrosis in rabbits. Mycotoxin Res. 2014, 30, 241–250.
- Hulvova, H.; Galuszka, P.; Frebortova, J.; Frebort, I. Parasitic fungus Claviceps as a source for biotechnological production of ergot alkaloids. Biotechnol. Adv. 2013, 31, 79–89.
- Janik, E.; Niemcewicz, M.; Ceremuga, M.; Stela, M.; Saluk-Bijak, J.; Siadkowski, A.; Bijak, M. Molecular aspects of mycotoxins—A serious problem for human health. Int. J. Mol. Sci. 2020, 21, 8187.
- Krska, R.; Crews, C. Significance, chemistry and determination of ergot alkaloids: A review. Food Addit. Contam. Part A 2008, 25, 722–731.
- Agriopoulou, S.; Koliadima, A.; Karaiskakis, G.; Kapolos, J. Kinetic study of aflatoxins’ degradation in the presence of ozone. Food Control. 2016, 61, 221–226.
- Agriopoulou, S. Enniatins: An emerging food safety issue. EC Nutr. 2016, 3, 1142–1146.
- Shahid, M.G.; Nadeem, M.; Gulzar, A.; Saleem, M.; Rehman, H.U.; Ghafoor, G.Z.; Hayyat, M.U.; Shahzad, L.; Arif, R.; Nelofer, R. Novel ergot alkaloids production from Penicillium citrinum employing response surface methodology technique. Toxins 2020, 12, 427.
- Arroyo-Manzanares, N.; Gámiz-Gracia, L.; García-Campaña, A.M.; di Mavungu, J.D.; de Saeger, S. Ergot alkaloids: Chemistry, biosynthesis, bioactivity, and methods of analysis. In Fungal Metabolites, Reference Series in Phytochemistry; Mérillon, J.M., Ramawat, K., Eds.; Springer: Berlin/Heidelberg, Germany, 2017; pp. 887–929.
- Holderied, I.; Rychlik, M.; Elsinghorst, P.W. Optimized analysis of ergot alkaloids in rye products by liquid chromatography-fluorescence detection applying lysergic acid diethylamide as an internal standard. Toxins 2019, 11, 184.
- Kodisch, A.; Oberforster, M.; Raditschnig, A.; Rodemann, B.; Tratwal, A.; Danielewicz, J.; Korbas, M.; Schmiedchen, B.; Eifler, J.; Gordillo, A.; et al. Covariation of ergot severity and alkaloid content measured by HPLC and one Elisa method in inoculated winter rye across three isolates and three European countries. Toxins 2020, 12, 676.
- Schummer, C.; Zandonella, I.; van Nieuwenhuyse, A.; Moris, G. Epimerization of ergot alkaloids in feed. Heliyon 2020, 6, e04336.
- Crews, C.; Anderson, W.A.C.; Rees, G.; Krska, R. Ergot alkaloids in some rye-based UK cereal products. Food Addit. Contam. Part B 2009, 2, 79–85.
- Pierri, L.; Pitman, I.H.; Rae, I.D.; Winkler, D.A.; Andrews, P.R. Conformational analysis of the ergot alkaloids ergotamine and ergotaminine. J. Med. Chem. 1982, 137, 937–942.
- Komerova, E.L.; Tolkachev, O.N. The chemistry of peptide alkaloids. Part 1. Classification and chemistry of ergot peptides. Pharm. Chem. J. 2001, 35, 504–513.
- Arroyo-Manzanares, N.; Rodríguez-Estévez, V.; García-Campaña, A.M.; Castellón-Rendón, E.; Gámiz-Gracia, L. Determination of principal ergot alkaloids in swine feeding. J. Sci. Food Agric. 2021.
- Merkel, S.; Dib, B.; Maul, R.; Koppen, R.; Koch, M.; Nehls, I. Degradation and epimerization of ergot alkaloids after baking and in vitro digestion. Anal. Bioanal. Chem. 2012, 404, 2489–2497.
- Hafner, M.; Sulyok, M.; Schuhmacher, R.; Crews, C.; Krska, R. Stability and epimerisation behaviour of ergot alkaloids in various solvents. World Mycotoxin J. 2008, 1, 67–78.
- Battilani, P.; Costa, L.G.; Dossena, A.; Gullino, M.L.; Marchelli, R.; Galaverna, G.; Pietri, A.; Dall’Asta, C.; Giorni, P.; Spadaro, D.; et al. Scientific information on mycotoxins and natural plant toxicants. EFSA Supporting Publ. 2009, 6, 24E.
- Wallwey, C.; Li, S.-M. Ergot alkaloids: Structure diversity, biosynthetic gene clusters and functional proof of biosynthetic genes. Nat. Prod. Rep. 2011, 28, 496–510.
- Eady, C. The impact of alkaloid-producing epichloë endophyte on forage ryegrass breeding: A New Zealand perspective. Toxins 2021, 13, 158.
- Shimshoni, J.A.; Cuneah, O.; Sulyok, M.; Krska, R.; Sionov, E.; Barel, S.; Harel, Y.M. Newly discovered ergot alkaloids in Sorghum ergot Claviceps africana occurring for the first time in Israel. Food Chem. 2017, 219, 459–467.
- Haarmann, T.; Rolke, Y.; Giesbert, S.; Tudzynski, P. Plant diseases that changed the world. Ergot: From witchcraft to biotechnology. Mol. Plant Pathol. 2009, 10, 563–577.
- WHO. Selected Mycotoxins: Ochratoxins, Trichothecenes, Ergot; World Health Organization: Geneva, Switzerland, 1990.
- Shahid, M.G.; Baig, S.; Saleem, M.; Arif, R.; Ghafoor, G.; Liaqat, A. Qualitative and quantitative analysis of ergot alkaloids produced by Aspergillus niger through surface culture fermentation process. Pak. J. Bot. 2018, 50, 2423–2428.
- Shahid, M.G.; Nadeem, M.; Baig, S.; Cheema, T.A.; Atta, S.; Ghafoor, G. Screening and optimization of some inorganic salts for the production of ergot alkaloids from Penicillium species using surface culture fermentation process. Pak. J. Pharm. Sci. 2016, 29, 407–414.
- Young, C.A.; Schardl, C.L.; Panaccione, D.G.; Florea, S.; Takach, J.E.; Charlton, N.D.; Moore, N.; Webb, J.S.; Jaromczyk, J. Genetics, genomics and evolution of ergot alkaloid diversity. Toxins 2015, 7, 1273–1302.
- Klotz, J.L. Activities and effects of ergot alkaloids on livestock physiology and production. Toxins 2015, 7, 2801–2821.
- Babič, J.; Tavčar-Kalcher, G.; Celar, F.A.; Kos, K.; Červek, M.; Jakovac-Strajn, B. Ergot and ergot alkaloids in cereal grains intended for animal feeding collected in Slovenia: Occurrence, pattern and correlations. Toxins 2020, 12, 730.
- Miedaner, T.; Geiger, H.H. Biology, genetics, and management of ergot (Claviceps spp.) in rye, sorghum, and pearl millet. Toxins 2015, 7, 659–678.
- Appelt, M.; Ellner, F.M. Investigations into the occurrence of alkaloids in ergot and single sclerotia from the 2007 and 2008 harvests. Mycotoxin Res. 2009, 25, 95–101.
- Tudzynski, P.; Neubauer, L. Ergot alkaloids. In Biosynthesis and Molecular Genetics of Fungal Secondary Metabolites; Martín, J.F., García-Estrada, C., Zeilinger, S., Eds.; Springer: Berlin/Heidelberg, Germany, 2014; pp. 303–316.
- Tkachenko, A.; Benson, K.; Mostrom, M.; Guag, J.; Reimschuessel, R.; Webb, B. Extensive evaluation via blinded testing of an UHPLC-MS/MS method for quantitation of ten ergot alkaloids in rye and wheat grains. J. AOAC Int. 2021, 20, 1–9.
- Arcella, D.; Ruiz, J.Á.G.; Innocenti, M.L.; Roldán, R. Human and animal dietary exposure to ergot alkaloids. EFSA J. 2017, 15.
- Stroka, J.; Gonçalves, C. Mycotoxins in food and feed: An overview. In Encyclopedia of Food Chemistry; Laurence, M., Shahidi, F., Varelis, P., Eds.; Academic Press: Cambridge, MA, USA, 2018; pp. 401–419.
- Dung, J.K.S.; Alderman, S.C.; Walenta, D.L.; Hamm, P.B. Spatial patterns of ergot and quantification of sclerotia in perennial ryegrass seed fields in eastern Oregon. Plant Dis. 2016, 100, 1110–1117.
- Alderman, S.C.; Walenta, D.L.; Hamm, P.B.; Martin, R.C.; Dung, J.; Kosman, E. Afternoon ascospore release in Claviceps purpurea optimizes perennial ryegrass infection. Plant Dis. 2015, 99, 1410–1415.
- Pscheidt, J.W.; Ocamb, C.M. Pacific Northwest Plant Disease Management Handbook; Oregon State University: Corvallis, OR, USA, 2016; Available online: (accessed on 7 April 2021).
- Guo, Q.; Shao, B.; Du, Z.; Zhang, J. Simultaneous determination of 25 ergot alkaloids in cereal samples by ultraperformance liquid chromatography-tandem mass spectrometry. J. Agric. Food Chem. 2016, 64, 7033–7039.
- Shi, H.; Schwab, W.; Liu, N.; Yu, P. Major ergot alkaloids in naturally contaminated cool-season barley grain grown under a cold climate condition in western Canada, explored with near-infrared (NIR) and fourier transform mid-infrared (ATR-FT/MIR) spectroscopy. Food Control 2019, 102, 221–230.
- Storm, I.D.; Rasmussen, P.H.; Strobel, B.W.; Hansen, H.C.B. Ergot alkaloids in rye flour determined by solid-phase cation-exchange and high-pressure liquid chromatography with fluorescence detection. Food Addit. Contam. 2008, 25, 338–346.
- Pitt, J.I.; Miller, J.D. A concise history of mycotoxin research. J. Agric. Food Chem. 2017, 65, 7021–7033.
- Krska, R.; Crews, C. HPLC/MS/MS Method for the Determination of Ergot Alkaloids in Cereals. Available online: (accessed on 20 March 2021).
- EFSA. Opinion of the scientific panel on contaminants in food chain on a request from the commission related to ergot as undesirable substance in animal feed. EFSA J. 2005, 225, 1–27.
- Debegnach, F.; Patriarca, S.; Brera, C.; Gregori, E.; Sonego, E.; Moracci, G.; de Santis, B. Ergot alkaloids in wheat and rye derived products in Italy. Foods 2019, 8, 150.
- Kralova, F.I.; Frebortova, J.; Pencık, A.; Frébort, I. Overexpression of Trp-related genes in Claviceps purpurea leading to increased ergot alkaloid production. New Biotechnol. 2021, 61, 69–79.
- Liu, M.; Shoukouhi, P.; Bisson, K.R.; Wyka, S.A.; Broders, K.D.; Menzies, J.G. Sympatric divergence of the ergot fungus, Claviceps purpurea, populations infecting agricultural and nonagricultural grasses in North America. Ecol. Evol. 2021, 11, 273–293.
- Commission Regulation (EC). No 1881/2006 setting maximum levels for certain contaminants in foodstuffs. Off. J. Eur. Union 2006, 364, 5–24.
- European Union. Commission regulation (EU) 2015/1940 of 28 October 2015 amending regulation (EC) No 1881/2006 as regards maximum levels of ergot sclerotia in certain unprocessed cereals and the provisions on monitoring and reporting. Off. J. Eur. Union 2015, 283, 3–8.
- European Communities. Directive 2002/32/EC of the European parliament and of the council of 7 May 2002 on undesirable substances in animal feed. Off. J. Eur. Comm. 2002, 140, 10–22.
- Alexander, J.; Benford, D.; Boobis, A.; Ceccatelli, S.; Cottrill, B.; Cravedi, J.P.; di Domenico, A.; Doerge, D.; Dogliotti, E.; Edler, L.; et al. Scientific opinion on ergot alkaloids in food and feed. EFSA J. 2012, 10, 1–58.
- Ünüsan, N. Systematic review of mycotoxins in food and feeds in Turkey. Food Control 2019, 97, 1–14.
- Brennan, J. Ergot alkaloids—Avoiding a bad trip on new regulations. In Milling Wheat Conference; Agriculture and Horticulture Development Board: Kenilworth, UK, 2020.
- Agriopoulou, S.; Stamatelopoulou, E.; Varzakas, T. Advances in analysis and detection of major mycotoxins in foods. Foods 2020, 9, 518.
- Chung, S.W.C. A critical review of analytical methods for ergot alkaloids in cereals and feed and in particular suitability of method performance for regulatory monitoring and epimer-specific quantification. Food Addit. Contam. Part A 2021, 30, 1–16.
- Kniel, B.; Meißner, M.; Koehler, P.; Schwake-Anduschus, C. Studies on the applicability of HPLC-FLD and HPLC-MS/MS for the determination of ergot alkaloids in rye-containing breads. J. Consum. Prot. Food Saf. 2018, 13, 69–78.
- Walker, K.; Duringer, J.; Craig, A.M. Determination of the ergot alkaloid ergovaline in tall fescue seed and straw using a QuEChERS extraction method with high-performance liquid chromatography-fluorescence detection. J. Agric. Food Chem. 2015, 63, 4236–4242.
- RASFF (Rapid Alert System for Food and Feed). Available online: (accessed on 20 March 2021).
- Eskola, M.; Kos, G.; Elliott, C.T.; Hajšlová, J.; Mayar, S.; Krska, R. Worldwide contamination of food-crops with mycotoxins: Validity of the widely cited ‘FAO estimate’ of 25%. Crit. Rev. Food Sci. Nutr. 2020, 60, 2773–2789.
- Tittlemier, S.A.; Drul, D.; Roscoe, M.; McKendry, T. Occurrence of ergot and ergot alkaloids in Western Canadian wheat and other cereals. J. Agric. Food Chem. 2015, 63, 6644–6650.
- Schnaufer, R.; Baitinger, A.; Vollmer, G. Ergot Alkaloids in Rye Flours and Rye Breads. 2013. Available online: (accessed on 20 March 2021).
- Malysheva, S.V.; Larionova, D.A.; di Mavungu, J.D.; de Saeger, S. Pattern and distribution of ergot alkaloids in cereals and cereal products from European countries. World Mycotoxin J. 2014, 7, 217–230.
- Drakopoulos, D.; Sulyok, M.; Krska, R.; Logrieco, A.F.; Vogelgsang, S. Raised concerns about the safety of barley grains and straw: A Swiss survey reveals a high diversity of mycotoxins and other fungal metabolites. Food Control 2021, 125, 107919.
- Orlando, B.; Maumené, C.; Piraux, F. Ergot and ergot alkaloids in French cereals: Occurrence, pattern and agronomic practices for managing the risk. World Mycotoxin J. 2017, 10, 327–337.
- Topi, D.; Jakovac-Strajn, B.; Pavšič-Vrtač, K.; Tavčar-Kalcher, G. Occurrence of ergot alkaloids in wheat from Albania. Food Addit. Contam. Part A 2017, 34, 1333–1343.
- Bryła, M.; Szymczyk, K.; Jędrzejczak, R.; Roszko, M. Application of liquid chromatography/ion trap mass spectrometry technique to determine ergot alkaloids in grain products. Food Technol. Biotechnol. 2015, 53, 18–28.
- Bryła, M.; Ksieniewicz-Woźniak, E.; Podolska, G.; Waśkiewicz, A.; Szymczyk, K.; Jędrzejczak, R. Occurrence of ergot and its alkaloids in winter rye harvested in Poland. World Mycotoxin J. 2018, 11, 635–646.
- Mulder, P.P.; Pereboom-de Fauw, D.P.; Hoogenboom, R.L.; de Stoppelaar, J.; de Nijs, M. Tropane and ergot alkaloids in grain-based products for infants and young children in the Netherlands in 2011–2014. Food Addit. Contam. Part B 2015, 8, 284–290.
- Grusie, T.; Cowan, V.; Singh, J.; McKinnon, J.; Blakley, B. Proportions of predominant Ergot alkaloids (Claviceps purpurea) detected in Western Canadian grains from 2014 to 2016. World Mycotoxin J. 2018, 11, 259–264.
- Menzies, J.G.; Turkington, T.K. An overview of the ergot (Claviceps purpurea) issue in western Canada: Challenges and solutions. Can. J. Plant Pathol. 2015, 37, 40–51.
- Ilha, M.R.; Loretti, A.P.; Barro, C.S. Hyperthermic syndrome in dairy cattle associated with consumption of ergots of Claviceps purpurea in Southern Brazil. Vet. Hum. Toxicol. 2003, 45, 140–145.
- Blaney, B.J.; Molloy, J.B.; Brock, I.J. Alkaloids in Australian rye ergot (Claviceps purpurea) sclerotia: Implications for food and stockfeed regulations. Animal Product. Sci. 2009, 49, 975–982.
- Menzies, J.G.; Klein-Gebbinck, H.W.; Gordon, A.; O’Sullivan, D.M. Evaluation of Claviceps purpurea isolates on wheat reveals complex virulence and host susceptibility relationships. Can. J. Plant Pathol. 2017, 39, 307–317.
- Xue, A.G.; Chen, Y.; Ai-Rewashdy, Y. Diseases of spring wheat in central and eastern Ontario in 2017. Can. Plant Dis. Surv. 2017, 98, 148–149.
- Wyka, S.A.; Mondo, S.J.; Liu, M.; Nalam, V.; Broders, K.D. A large accessory genome, high recombination rates, and selection of secondary metabolite genes help maintain global distribution and broad host range of the fungal plant pathogen Claviceps purpurea. BioRxiv 2020.
- Rubert, J.; Dzuman, Z.; Vaclavikova, M.; Zachariasova, M.; Soler, C.; Hajslova, J. Analysis of mycotoxins in barley using ultra high liquid chromatography high resolution mass spectrometry: Comparison of efficiency and efficacy of different extraction procedures. Talanta 2012, 99, 712–719.
- Carbonell-Rozas, L.; Mahdjoubi, C.K.; Arroyo-Manzanares, N.; Gámiz-Gracia, L.; García-Campaña, A.M. Determination and occurrence of ergot alkaloids in cereal samples from Algeria. Toxins 2021.
- Müller, C.; Kemmlein, S.; Klaffke, H.; Krauthause, W.; Preiss-Weigert, A.; Wittkowski, R. A basic tool for risk assessment: A new method for the analysis of ergot alkaloids in rye and selected rye products. Mol. Nutr. Food Res. 2009, 53, 500–507.
- Arroyo-Manzanares, N.; de Ruyck, K.; Uka, V.; Gámiz-Gracia, L.; García-Campaña, A.M.; de Saeger, S.; di Mavungu, J.D. In-house validation of a rapid and efficient procedure for simultaneous determination of ergot alkaloids and other mycotoxins in wheat and maize. Anal. Bioanal. Chem. 2018, 410, 5567–5581.
- Usleber, E.; Bauer, J.I.; Gross, M. Two forms of historic ergotism—Two groups of toxins? In Proceedings of the 40th Mycotoxin Workshop, Munich, Germany, 11–13 June 2018; p. 32. Available online: (accessed on 13 December 2020).
- Waret-Szkuta, A.; Larraillet, L.; Oswald, I.P.; Legrand, X.; Guerre, P.; Martineau, G.-P. Unusual acute neonatal mortality and sow agalactia linked with ergot alkaloid contamination of feed. Porc. Health Manag. 2019, 5, 24.
- Baldim, I.; Oliveira, W.P.; Kadian, V.; Rao, R.; Yadav, N.; Mahant, S.; Lucarini, M.; Durazzo, A.; da Ana, R.; Capasso, R.; et al. Natural ergot alkaloids in ocular pharmacotherapy: Known molecules for novel nanoparticle-based delivery systems. Biomolecules 2020, 10, 980.
- Coufal-Majewski, S.; Stanford, K.; McAllister, T.; Blakley, B.; McKinnon, J.; Chaves, A.V.; Wang, Y. Impacts of cereal ergot in food animal production. Front. Vet. Sci. 2016, 3, 15.
- Maruo, V.M.; Bracarense, A.P.; Metayer, J.-P.; Vilarino, M.; Oswald, I.P.; Pinton, P. Ergot alkaloids at doses close to EU regulatory limits induce alterations of the liver and intestine. Toxins 2018, 10, 183.
- Urga, K.; Debella, A.; Medihn, Y.W.; Agata, N.; Bayu, A.; Zewdie, W. Laboratory studies on the outbreak of gangrenous ergotism associated with consumption of contaminated barley in Arsi, Ethiopia. Ethiop. J. Health Dev. 2002, 16, 317–323.
- Craig, A.M.; Klotz, J.L.; Duringer, J.M. Cases of ergotism in livestock and associated ergot alkaloid concentrations in feed. Front. Chem. 2015, 3, 1–6.
- Bryden, W.L. Mycotoxin contamination of the feed supply chain: Implication for animal productivity and feed security. Anim. Feed Sci. Technol. 2012, 173, 134–158.
- Mavrommatis, A.; Giamouri, E.; Tavrizelou, S.; Zacharioudaki, M.; Danezis, G.; Simitzis, P.E.; Zoidis, E.; Tsiplakou, E.; Pappas, A.C.; Georgiou, C.A.; et al. Impact of mycotoxins on animals’ oxidative status. Antioxidants 2021, 10, 214.
- Mahato, D.K.; Devi, S.; Pandhi, S.; Sharma, B.; Maurya, K.K.; Mishra, S.; Dhawan, K.; Selvakumar, R.; Kamle, M.; Mishra, A.K.; et al. Occurrence, impact on agriculture, human health, and management strategies of zearalenone in food and feed: A review. Toxins 2021, 13, 92.
- Chulze, S.N.; Palazzini, J.M.; Torres, A.M.; Barros, G.; Ponsone, M.L.; Geisen, R. Biological control as a strategy to reduce the impact of mycotoxins in peanuts, grapes and cereals in Argentina. Food Addit. Contam. Part A 2014, 32, 37–41.
- Nešić, K.; Habschied, K.; Mastanjević, K. Possibilities for the biological control of mycotoxins in food and feed. Toxins 2021, 13, 198.
- Agriopoulou, S.; Stamatelopoulou, E.; Sachadyn-Król, M.; Varzakas, T. Lactic acid bacteria as antibacterial agents to extend the shelf life of fresh and minimally processed fruits and vegetables: Quality and safety aspects. Microorganisms 2020, 8, 952.
- Hahn, I.; Thamhesl, M.; Apfelthaler, E.; Klingenbrunner, V.; Hametner, C.; Krska, R.; Schatzmayr, G.; Moll, W.-D.; Berthiller, F.; Schwartz-ZimmermannH, H.E. Characterisation and determination of metabolites formed by microbial and enzymatic degradation of ergot alkaloids. World Mycotoxin J. 2015, 8, 393–404.
- Li, C.; Kong, Q.; Mou, H.; Jiang, Y.; Du, Y.; Zhang, F. Biotransformation of alkylamides and alkaloids by lactic acid bacteria strains isolated from Zanthoxylum bungeanum meal. Bioresour. Technol. 2021, 330, 124944.
- Dabkevicius, Z.; Semaskiene, R. Control of ergot (Claviceps purpurea (FR.) Tul.) ascocarpus formation under the ipact of chemical and biological seed dressing. Plant protection science. In Proceedings of the 6th Conference of EFPP, Prague, Czech Republic, 8–14 September 2002; Taborsky, V., Polak, J., Lebede, A., Kudela, V., Eds.; Lithuanian Institute of Agriculture: Kėdainiai, Lithuania, 2002.
- Lyagin, I.; Efremenko, E. Enzymes for detoxification of various mycotoxins: Origins and mechanisms of catalytic action. Molecules 2019, 24, 2362.
- Uppala, S.S.; Wu, B.M.; Alderman, S.C. Effects of temperature and duration of preconditioning cold treatment on sclerotial germination of Claviceps purpurea. Plant Dis. 2016, 100, 2080–2086.
- Dung, J.K.S.; Kaur, N.; Walenta, D.L.; Alderman, S.C.; Frost, K.E.; Hamm, P.B. Reducing Claviceps purpurea sclerotia germination with soil-applied fungicides. Crop Prot. 2018, 106, 146–149.
- Thabit, T.M.A.; Abdelkareem, E.M.; Bouqellah, N.A.; Shokr, S.A. Triazole fungicide residues and their inhibitory effect on some trichothecenes mycotoxin excretion in wheat grains. Molecules 2021, 26, 1784.
- Bailey, K.L.; Gossen, B.D.; Gugel, R.K.; Morrall, R.A.A. Diseases of Field Crops in Canada, 3rd ed.; The Canadian Phytopathological Society and University Extension Press; University of Saskatchewan: Saskatoon, SK, Canada, 2003; pp. 89–93.
- Suman, M. Last decade studies on mycotoxins’ fate during food processing: An overview. Curr. Opin. Food Sci. 2021, 41, 70–80.
- Ji, C.; Fan, Y.; Zhao, L. Review on biological degradation of mycotoxins. Anim. Nutr. 2016, 2, 127–133.
- Calado, T.; Venâncio, A.; Abrunhosa, L. Irradiation for mold and mycotoxin control: A review. Compr. Rev. Food Sci. Food Saf. 2014, 13, 1049–1061.
- Neme, K.; Mohammed, A. Mycotoxin occurrence in grains and the role of postharvest management as a mitigation strategies. A review. Food Control 2017, 78, 412–425.
- Bryła, M.; Ksieniewicz-Woźniak, E.; Waśkiewicz, A.; Podolska, G.; Szymczyk, K. Stability of ergot alkaloids during the process of baking rye bread. LWT 2019, 110, 269–274.
- Milani, J.; Maleki, G. Effects of processing on mycotoxin stability in cereals. J. Food Sci. Agric. 2014, 94, 2372–2375.
- Karlovsky, P.; Suman, M.; Berthiller, F.; de Meester, J.; Eisenbrand, G.; Perrin, I.; Oswald, I.P.; Speijers, G.; Chiodini, A.; Recker, T.; et al. Impact of food processing and detoxification treatments on mycotoxin contamination. Mycotoxin Res. 2016, 32, 179–205.
- Tittlemier, S.A.; Drul, D.; Roscoe, M.; Turnock, D.; Taylor, D.; Fu, B.X. Fate of ergot alkaloids during laboratory scale durum processing and pasta production. Toxins 2019, 11, 195.
- Meleard, B. Degradation and Epimerization of Wheat Ergot Alkaloids during French Baking Test. 2016. Available online: (accessed on 7 April 2021).
