Partial breast irradiation (PBI) presents an opportunity to improve quality of life during treatment with a significantly shorter treatment duration. By targeting less non-target breast tissue, excess radiation exposure and resulting toxicities are also reduced. Proton therapy represents a precision radiotherapy technology that builds on these advantages by further limiting the normal tissue exposure to unnecessary radiation dose not only to uninvolved breast tissue but also the underlying thoracic organs including the heart and lungs.
- breast cancer
- proton therapy
- precision medicine
- partial breast irradiation
- radiation therapy
1. Introduction
2. Invasive Partial Breast Irradiation Techniques
3. External Beam Photon Partial Breast Irradiation
4. Proton Partial Breast Irradiation
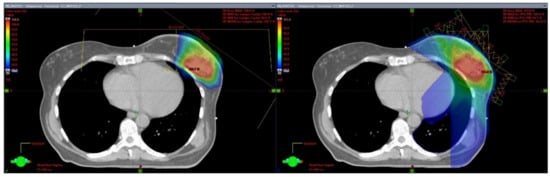
|
Study |
PT Type |
N |
Dose (Gy (RBE)) |
PT Details |
MD-Reported Cosmesis |
Patient-Reported Cosmesis |
PT Toxicity |
Disease Control |
|---|---|---|---|---|---|---|---|---|
|
Passive Scattering |
19 |
32 Gy in 8 fx BID |
1–3 fields (1 field treated per fx) |
7 yr Good-excellent: 62% |
7 yr Good-excellent 92% |
69% Telangiectasias 54% Pigmentation changes 62% Other late skin tox |
7 yr LF: 11% PP |
|
|
NCC, Korea [29] |
Passive Scattering |
30 |
30 Gy in 5 fx QD |
1–2 fields |
3 yr Good-excellent: 69% (89% with 2-field plan) |
-- |
Increased toxicity with single field plan |
3 yr LF 0% 3 yr DF 0% |
|
Passive Scattering |
90 |
40 Gy in 4 Gy × 10 fx QD |
2–4 fields |
5 yr Good-excellent: 90% |
5 yr Good-excellent 90% |
Acute: 62% Gr1–2RT dermatitis Late: 7% Gr 1 telangiectasia1% Fat necrosis |
5 yr IBTR-FS 97% 5 yr OS 95% |
|
|
PCG BRE007 [33] |
Uniform Scanning Passive Scattering |
37 1 |
40 Gy in 10 fx QD |
≥3 fields (≥2 treated daily) |
3 yr Good-excellent: 100% |
BCTOS 4: 13.2% (nipple appearance, breast shape, scar tissue) |
Gr 2–7 events ≥Gr 3–none |
3 yr LF and DF: 0% |
|
MDACC [34] |
Passive Scattering |
100 |
34 Gy in 10 fx BID |
≥ 2 fields |
2 yr Good-excellent: 84% |
2 yr Good-excellent 96% |
Acute Gr 2: 1% Breast pain 2% Hyperpigmentation 1% Pruritus 12% Dermatitis Late Gr 2: 5% Fatigue 1% Hyperpigmentation 2% Dermatitis |
2 yr LF 0% 2 yr OS 100% |
|
Mayo [35] |
Pencil Beam Scanning |
76 |
21.9 Gy in 3 fx QD |
2 fields |
NR |
1 yr Good-excellent 98% |
≥ Gr 2 acute or late tox-none |
NR |
5. Cost Effectiveness
While patients with left-sided tumors being treated to the internal mammary nodes are more susceptible to developing late cardiac toxicities from radiotherapy and thus may have significant clinical and cost effective benefit from PT [37], the cost effectiveness of PT for PBI has been called into question as a potential adverse factor in patient QOL and treatment accessibility. In the series from MD Anderson, the median out of pocket cost for patients was $700 (IQE $100-$1600), and the mean time away from work was 5 days (IQR 2-5 days) for the 5-day treatment course. A cost effectiveness analysis of 8 schedules of PBI and WBRT revealed a lower total Medicare reimbursement for PT PBI ($13,833) compared with IMRT-WBI ($19,599) or multi-lumen brachytherapy PBI ($14,859), in contrast with the costs of the other 4 regimens evaluated ($6,771-$13,149) [38]. These findings suggest minimal disruption in patient’s life from a financial standpoint, including considerations of cost and impact on livelihood.
6. Conclusion
With ongoing advancements in precision delivery of PT allowing for continued refinement of treatment approaches and further widening of the margin of benefit of proton PBI, this technique will likely continue to expand its role in the treatment of EBC. Further investigations will elucidate if a subset of patients may benefit most from the use of PT for PBI, such as those with large tumors, limited breast volume, and medial tumor location. In addition, patients with other special circumstances, such as unfavorable anatomy, connective tissue disorders, and prior irradiation, may also be considered more strongly for the use of PT for PBI.
This entry is adapted from the peer-reviewed paper 10.3390/jpm11040289
References
- Rebecca L. Siegel; Kimberly D. Miller; Hannah E. Fuchs; Ahmedin Jemal; Cancer Statistics, 2021. CA: A Cancer Journal for Clinicians 2021, 71, 7-33, 10.3322/caac.21654.
- Carol E. DeSantis; Jiemin Ma; Mia M. Gaudet; Lisa A. Newman; Kimberly D. Miller Mph; Ann Goding Sauer; Ahmedin Jemal; Rebecca L. Siegel; Breast cancer statistics, 2019. CA: A Cancer Journal for Clinicians 2019, 69, 438-451, 10.3322/caac.21583.
- American Cancer Society. Cancer Facts & Figures 2021. Atlanta, Ga: American Cancer Society; 2021.
- Early Breast Cancer Trialists' Collaborative Group. Lancet 2011, 378(9804): 1707-1716.
- Umberto Veronesi; Natale Cascinelli; Luigi Mariani; Marco Greco; Roberto Saccozzi; Alberto Luini; Marisel Aguilar; Ettore Marubini; Twenty-Year Follow-up of a Randomized Study Comparing Breast-Conserving Surgery with Radical Mastectomy for Early Breast Cancer. New England Journal of Medicine 2002, 347, 1227-1232, 10.1056/nejmoa020989.
- Bernard Fisher; Stewart Anderson; John Bryant; Richard G. Margolese; Melvin Deutsch; Edwin R. Fisher; Jong Jeong; Norman Wolmark; Twenty-Year Follow-up of a Randomized Trial Comparing Total Mastectomy, Lumpectomy, and Lumpectomy plus Irradiation for the Treatment of Invasive Breast Cancer. New England Journal of Medicine 2002, 347, 1233-1241, 10.1056/nejmoa022152.
- Candace Correa; Eleanor E. Harris; Maria Cristina Leonardi; Benjamin D. Smith; Alphonse G. Taghian; Alastair M. Thompson; Julia White; Jay R. Harris; Accelerated Partial Breast Irradiation: Executive summary for the update of an ASTRO Evidence-Based Consensus Statement. Practical Radiation Oncology 2017, 7, 73-79, 10.1016/j.prro.2016.09.007.
- https://clinicaltrials.gov/ct2/show/NCT04084730
- Robert R. Kuske; Kathryn Winter; Douglas W. Arthur; John Bolton; Rachel Rabinovitch; Julia White; William Hanson; R.M. Wilenzick; Phase II trial of brachytherapy alone after lumpectomy for select breast cancer: Toxicity analysis of RTOG 95-17. International Journal of Radiation Oncology*Biology*Physics 2006, 65, 45-51, 10.1016/j.ijrobp.2005.11.027.
- Umberto Veronesi; Roberto Orecchia; Patrick Maisonneuve; Giuseppe Viale; Nicole Rotmensz; Claudia Sangalli; Alberto Luini; Paolo Veronesi; Viviana Galimberti; Stefano Zurrida; et al. Intraoperative radiotherapy versus external radiotherapy for early breast cancer (ELIOT): a randomised controlled equivalence trial. The Lancet Oncology 2013, 14, 1269-1277, 10.1016/s1470-2045(13)70497-2.
- Chirag Shah; Alvaro Martinez; Matthew Kolar; Frank Vicini; Modern Approaches for Breast Brachytherapy. Seminars in Radiation Oncology 2019, 30, 61-67, 10.1016/j.semradonc.2019.08.004.
- Benjamin D Smith; Thomas A Buchholz; Henry M Kuerer; Intraoperative radiotherapy for early breast cancer.. The Lancet 2010, 376, 1141-4, 10.1016/S0140-6736(10)61530-9.
- Jayant S Vaidya; David J Joseph; Jeffrey S Tobias; Max Bulsara; Frederik Wenz; Christobel Saunders; Michael Alvarado; Henrik L Flyger; Samuele Massarut; Wolfgang Eiermann; et al. Targeted intraoperative radiotherapy versus whole breast radiotherapy for breast cancer (TARGIT-A trial): an international, prospective, randomised, non-inferiority phase 3 trial. The Lancet 2010, 376, 91-102, 10.1016/s0140-6736(10)60837-9.
- Jayant S Vaidya; Max Bulsara; Michael Baum; Frederik Wenz; Samuele Massarut; Steffi Pigorsch; Michael Alvarado; Michael Douek; Christobel Saunders; Henrik L Flyger; et al. Long term survival and local control outcomes from single dose targeted intraoperative radiotherapy during lumpectomy (TARGIT-IORT) for early breast cancer: TARGIT-A randomised clinical trial. BMJ 2020, 370, m2836, 10.1136/bmj.m2836.
- Strnad V, Ott OJ, Hildebrandt G, et al.; 5-year results of accelerated partial breast irradiation using sole interstitial multicatheter brachytherapy versus whole-breast irradiation with boost after breast-conserving surgery for low-risk invasive and in-situ carcinoma of the female breast: a randomised, phase 3, non-inferiority trial. Breast Diseases: A Year Book Quarterly 2016, 387, 229-238, 10.1016/j.breastdis.2016.04.028.
- Lorenzo Livi; Icro Meattini; Livia Marrazzo; Gabriele Simontacchi; Stefania Pallotta; Calogero Saieva; Fabiola Paiar; Vieri Scotti; Carla De Luca Cardillo; Paolo Bastiani; et al. Accelerated partial breast irradiation using intensity-modulated radiotherapy versus whole breast irradiation: 5-year survival analysis of a phase 3 randomised controlled trial. European Journal of Cancer 2015, 51, 451-463, 10.1016/j.ejca.2014.12.013.
- Frank A Vicini; Reena S Cecchini; Julia R White; Douglas W Arthur; Thomas B Julian; Rachel A Rabinovitch; Robert R Kuske; Patricia A Ganz; David S Parda; Michael F Scheier; et al. Long-term primary results of accelerated partial breast irradiation after breast-conserving surgery for early-stage breast cancer: a randomised, phase 3, equivalence trial. The Lancet 2019, 394, 2155-2164, 10.1016/s0140-6736(19)32514-0.
- Adam L. Liss; Merav A. Ben-David; Reshma Jagsi; James A. Hayman; Kent A. Griffith; Jean M. Moran; Robin B. Marsh; Lori J. Pierce; Decline of cosmetic outcomes following accelerated partial breast irradiation using intensity modulated radiation therapy: results of a single-institution prospective clinical trial.. International Journal of Radiation Oncology*Biology*Physics 2014, 89, 96-102, 10.1016/j.ijrobp.2014.01.005.
- Sarah C. Darby; Marianne Ewertz; Paul McGale; Anna M. Bennet; Ulla Blom-Goldman; Dorthe Scavenius Brønnum; Candace Correa; David Cutter; Giovanna Gagliardi; Bruna Gigante; et al. Risk of Ischemic Heart Disease in Women after Radiotherapy for Breast Cancer. New England Journal of Medicine 2013, 368, 987-998, 10.1056/nejmoa1209825.
- K. Shin; S. Moon; N. Jang; D. Lee; M. Yoon; S. Park; J. Kim; H. Yoo; Dosimetric Comparison of Four Different External Beam Partial Breast Irradiations: Proton, Tomotherapy, Intensity Modulated and 3D Conformal Radiation Therapy. International Journal of Radiation Oncology*Biology*Physics 2007, 69, S221-S222, 10.1016/j.ijrobp.2007.07.1197.
- F Vicini; V Remouchamps; M Wallace; M Sharpe; J Fayad; L Tyburski; N Letts; L Kestin; G Edmundson; J Wong; et al. Ongoing clinical experience utilizing 3D conformal external beam radiotherapy to deliver partial breast irradiation in patients with early stage breast cancer treated with breast conserving therapy. International Journal of Radiation Oncology*Biology*Physics 2003, 57, S364-S365, 10.1016/s0360-3016(03)01271-9.
- Alphonse G. Taghian; Kevin R. Kozak; Angela Katz; Judith Adams; Hsiao-Ming Lu; Simon N. Powell; Thomas F. DeLaney; Accelerated partial breast irradiation using proton beams: Initial dosimetric experience. International Journal of Radiation Oncology*Biology*Physics 2006, 65, 1404-1410, 10.1016/j.ijrobp.2006.03.017.
- Kevin R. Kozak; Angela Katz; Judith Adams; Elizabeth M. Crowley; Jacqueline A. Nyamwanda; Jennifer K. Feng; Karen P. Doppke; Thomas F. DeLaney; Alphonse G. Taghian; Dosimetric comparison of proton and photon three-dimensional, conformal, external beam accelerated partial breast irradiation techniques. International Journal of Radiation Oncology*Biology*Physics 2006, 65, 1572-1578, 10.1016/j.ijrobp.2006.04.025.
- Sung Ho Moon; Kyung Hwan Shin; Tae Hyun Kim; Myonggeun Yoon; Soah Park; Doo-Hyun Lee; Jong Won Kim; Dae Woong Kim; Sung Yong Park; Kwan Ho Cho; et al. Dosimetric comparison of four different external beam partial breast irradiation techniques: Three-dimensional conformal radiotherapy, intensity-modulated radiotherapy, helical tomotherapy, and proton beam therapy. Radiotherapy and Oncology 2008, 90, 66-73, 10.1016/j.radonc.2008.09.027.
- T.M. Hansen; G. Bartlett; E.M. Mannina; S.P. Srivastava; J.A. Cox; I.J. Das; Dosimetric Comparison of Treatment Techniques: Brachytherapy, IMRT, and Proton Beam in Partial-Breast Irradiation. International Journal of Radiation Oncology*Biology*Physics 2015, 93, E8, 10.1016/j.ijrobp.2015.07.564.
- Kevin R. Kozak; Barbara L. Smith; Judith Adams; Ellen Kornmehl; Angela Katz; Michele Gadd; Michelle Specht; Kevin Hughes; Valeria Gioioso; Hsiao-Ming Lu; et al. Accelerated partial-breast irradiation using proton beams: Initial clinical experience. International Journal of Radiation Oncology*Biology*Physics 2006, 66, 691-698, 10.1016/j.ijrobp.2006.06.041.
- Sigolène Galland-Girodet; Itai Pashtan; Shannon M. MacDonald; Marek Ancukiewicz; Ariel E. Hirsch; Lisa A. Kachnic; Michelle Specht; Michele Gadd; Barbara L. Smith; Simon N. Powell; et al. Long-term Cosmetic Outcomes and Toxicities of Proton Beam Therapy Compared With Photon-Based 3-Dimensional Conformal Accelerated Partial-Breast Irradiation: A Phase 1 Trial. International Journal of Radiation Oncology*Biology*Physics 2014, 90, 493-500, 10.1016/j.ijrobp.2014.04.008.
- Vivek Verma; Charles B Simone; Mark V Mishra; Quality of Life and Patient-Reported Outcomes Following Proton Radiation Therapy: A Systematic Review. JNCI Journal of the National Cancer Institute 2017, 110, 341-353, 10.1093/jnci/djx208.
- Ji Hyun Chang; Nam Kwon Lee; Ja Young Kim; Yeon-Joo Kim; Sung Ho Moon; Tae Hyun Kim; Joo-Young Kim; Dae Yong Kim; Kwan Ho Cho; Kyung Hwan Shin; et al. Phase II trial of proton beam accelerated partial breast irradiation in breast cancer. Radiotherapy and Oncology 2013, 108, 209-214, 10.1016/j.radonc.2013.06.008.
- David A. Bush; Jerry D. Slater; Carlos Garberoglio; Sharon Do; Sharon Lum; James M. Slater; Partial Breast Irradiation Delivered With Proton Beam: Results of a Phase II Trial. Clinical Breast Cancer 2011, 11, 241-245, 10.1016/j.clbc.2011.03.023.
- David A. Bush; Sharon Do; Sharon Lum; Carlos Garberoglio; Hamid Mirshahidi; Baldev Patyal; Roger Grove; Jerry D. Slater; Partial Breast Radiation Therapy With Proton Beam: 5-Year Results With Cosmetic Outcomes. International Journal of Radiation Oncology*Biology*Physics 2014, 90, 501-505, 10.1016/j.ijrobp.2014.05.1308.
- Sandra L. Teichman; Sharon Do; Sharon Lum; Theodore S. Teichman; William Preston; Shelly E. Cochran; Carlos A. Garberoglio; Roger Grove; Carol A. Davis; Jerry D. Slater; et al. Improved long‐term patient‐reported health and well‐being outcomes of early‐stage breast cancer treated with partial breast proton therapy. Cancer Medicine 2018, 7, 6064-6076, 10.1002/cam4.1881.
- I.J. Choi; K. Prabhu; W.F. Hartsell; C.C. Sinesi; C.E. Vargas; R.K. Benda; O. Cahlon; A.L. Chang; Clinical Outcomes after Proton Partial-Breast Radiotherapy for Early-Stage, Hormone Receptor-Positive Breast Cancer: 3-Year Outcomes of a Phase II Trial. International Journal of Radiation Oncology*Biology*Physics 2019, 105, E19, 10.1016/j.ijrobp.2019.06.659.
- Dario Pasalic; Eric A. Strom; Pamela K. Allen; Tyler D. Williamson; Falk Poenisch; Richard A. Amos; Wendy A. Woodward; Michael C. Stauder; Simona F. Shaitelman; Benjamin D. Smith; et al. Proton Accelerated Partial Breast Irradiation: Clinical Outcomes at a Planned Interim Analysis of a Prospective Phase 2 Trial. International Journal of Radiation Oncology*Biology*Physics 2021, 109, 441-448, 10.1016/j.ijrobp.2020.09.009.
- Robert W. Mutter; Krishan R. Jethwa; Karthik Gonuguntla; Nicholas B. Remmes; Thomas J. Whitaker; Tina J. Hieken; Kathryn J. Ruddy; Lisa A. McGee; Kimberly S. Corbin; Sean S. Park; et al. 3 fraction pencil-beam scanning proton accelerated partial breast irradiation: early provider and patient reported outcomes of a novel regimen. Radiation Oncology 2019, 14, 1-9, 10.1186/s13014-019-1417-7.
- Søren M. Bentzen; John R. Yarnold; Reports of Unexpected Late Side Effects of Accelerated Partial Breast Irradiation—Radiobiological Considerations. International Journal of Radiation Oncology*Biology*Physics 2010, 77, 969-973, 10.1016/j.ijrobp.2010.01.059.
- Vivek Verma; Chirag Shah; Jean-Claude M. Rwigema; Timothy Solberg; Xiaofeng Zhu; Charles B. Simone Ii; Cost-comparativeness of proton versus photon therapy. Chinese Clinical Oncology 2016, 5, 56-56, 10.21037/cco.2016.06.03.
- Valentina Ovalle; Eric A. Strom; Joy Godby; Simona F. Shaitelman; Michael C. Stauder; Richard A. Amos; Henry M. Kuerer; Wendy A. Woodward; Karen E. Hoffman; Proton Partial-Breast Irradiation for Early-Stage Cancer: Is It Really So Costly?. International Journal of Radiation Oncology*Biology*Physics 2016, 95, 49-51, 10.1016/j.ijrobp.2015.07.2285.
