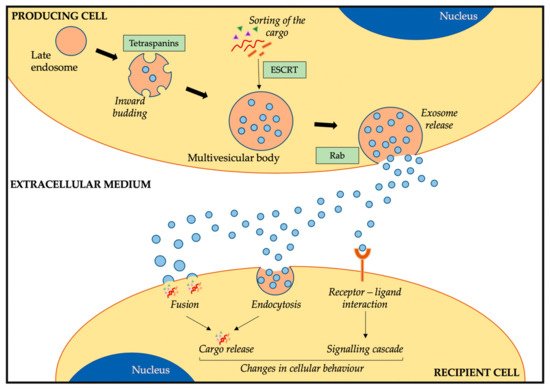
Due to the importance of metastasis in these bone sarcomas, many studies have focused on deciphering the pathways behind disease progression. Amongst the different genes identified in OS, we find overexpression of
CD155 [
77], loss of
TP53,
RB1, and
PTEN [
78], and upregulation of Notch genes [
79] to be important in metastatic OS. Moreover, different novel treatment strategies are evaluating their efficacy in OS metastatic patients through clinical trials [
75], with the aim of finding novel treatment strategies for these patients. In ES, similar studies have resulted in several genes linked to metastasis, such as
ROR1 [
80],
MSH2,
MSH6,
RPA2, and
RFC2 genes from the mismatch repair pathway [
81], PPP1R1A [
82] and TWIST1 proteins [
83], the Cad11 adhesion molecule [
84], or ERBB4 via activation of the PI3K-Akt-FAK cascade [
85]. Moreover, similar to other cancers, hypoxia has been associated with induction of metastasis in OS and ES via regulation of HIF1α through HIF1α [
86] or overexpression of CXCR4 in ES [
87], amongst others. Similar to OS, different clinical trials are evaluating the response of metastatic ES patients to different treatment approaches in order to improve survival rates [
76]. In contrast to OS and ES, CS is usually a non-metastatic disease with locally aggressive tumours [
88]. However, some evidence suggests integrins are involved in the metastatic organotropism of CS to the lungs [
89,
90].
2.1. Exosomes in OS
OS is the most common primary bone tumour, with an incidence of 0.3 per 100.000 people per year [
91]. The peak incidence follows a bimodal distribution, with most cases between 0–24 years of age and 60–85 years of age [
92]. It is an intraosseous neoplasm with origin in bone regions with active cellular growth in which the balance between osteoclasts and osteoblasts is disrupted [
93]. Contrary to other sarcomas with a clear genetic driving event, OS is characterised by high genomic instability. This results in complex karyotypes involving copy number alterations [
94] and frequency of chromotripsis (high incidence of chromosomal rearrangements in a delimited genomic region) [
95]. All these factors, adding to the heterogeneity and rarity of OS, have made difficult the successful identification of better treatment strategies for patients.
Despite improvements in diagnosis and treatment over the last decades, survival rates are still poor for an important fraction of patients. Between 10% and 20% of patients have metastasis at diagnosis, with 5-year overall survival rates (OSR) lower than 20% [
75]. For localised disease at diagnosis, survival rates are higher (5-year OSR: 60–80%), although evidence suggests 80% of patients have micrometastasis at the time of diagnosis which will be refractory to chemotherapy. This results in 30–40% of non-metastatic OS patients developing metastasis and recurrent disease [
96,
97]. Therefore, one of the focal points of OS research is to better understand the process of metastasis and how different factors modulate the TME to favour metastatic spread. This will help in identifying treatment strategies against metastatic and refractory disease, leading to improved survival rates.
Due to the role exosomes play in tumorigenesis in many cancers and the implications these small extracellular vesicles have in metastasis, it is not strange that researchers in the OS field are investigating the implications of exosomes in OS (). As exosomes are involved in cellular cross-talk via transfer of their cargo, one of the focal points has been to characterise the cargo of OS-derived exosomes. An analysis of OS-derived exosomes and conditioned media (exosome-free) revealed a different protein profile between both [
98]. Among the 250 most enriched proteins in exosomes, pathway analysis revealed an association with migration, adhesion, and angiogenesis, all important processes for cancer dissemination and modulation of the pre-metastatic niche. Following these data, the same group investigated the exosomal cargo of several OS cell lines [
99]. A difference in the miRNA content was seen between metastatic and non-metastatic OS-derived exosomes. Amongst the predicted target genes for the differential expressed miRNA, there was an enrichment of genes associated with tumour progression and metastasis. A deeper analysis of miRNAs enriched in exosomes from the most metastatic OS cell line (SAOS2) revealed that 4 miRNAs targeted 31 target genes from the same network associated with cellular adhesion and apoptosis [
99]. Therefore, data from these studies suggest that OS-derived exosomes could drive a pro-metastatic phenotype by transferring specific proteins and miRNA to other OS cells, resulting in induction of changes in migration, adhesion, and angiogenesis.
Table 1. Summary of exosome studies in OS.
| Origin Cell |
Recipient Cell |
Cargo |
Change |
Ref. |
| OS cells and conditioned media |
- |
Profiling of proteome and secretome |
Exosome proteins involved in migration, adhesion, and angiogenesis |
[98] |
| Metastatic and non-metastatic OS cell lines |
- |
Profiling of miRNAs and target genes |
miRNA of metastatic OS exosomes target metastasis-associated genes, cell adhesion, and apoptosis |
[99] |
| Metastatic and non-metastatic OS cell lines |
Osteoblasts |
miR-675
(miRNA profiling) |
Metastasis-associated exosomes induce migration and invasion in osteoblasts via miR-675/CALN1 axis |
[100] |
| Metastatic OS cell lines |
Macrophages, osteoclasts, endothelial cells |
miR-148a,
miR-21-5p
(RNA profiling) |
Induction of osteoclast-like gene expression (macrophage), increase in bone resorption (osteoclasts) and angiogenesis (endothelial cells) via miRNA transfer |
[101] |
| Metastatic and non-metastatic OS cell lines |
MSC |
TGFβ
(induction of IL6) |
Internalization of TGFβ induces IL6 production, cell growth, and lung metastasis in vivo |
[102] |
| Doxorubicin-resistant OS cell lines |
Sensitive OS cell lines |
MRP1, Pgp(multidrug resistant proteins) |
Increase in doxorubicin resistance in recipient cells; increase in MRP1 and Pgp mRNA levels. |
[103] |
| Bone marrow (conditioned media) |
Metastatic and non-metastatic OS cells |
uPA (secreted, paracrine loop) |
Increase in migration on recipient cells, induction of OS metastasis in vivo |
[104] |
| CAF |
OS cell lines |
miR-1228
(miRNA profiling) |
Increase in migration and invasion via miR-1228 transfer |
[105] |
| MSC |
OS cell lines |
miR-143 (synthetic introduction) |
Reduction of migration via exosome transfer (better than transfection) |
[106] |
| OS cell lines |
- |
Profiling of miRNA as OS biomarkers |
Better biomarker than ALP or patient stratification according to chemotherapy response |
[107,108], |
Similar to what has been described in other cancers, tumour-derived exosomes can educate the TME in order to induce pro-metastatic and tumorigenic changes [
52]. OS-derived exosomes from metastatic cells induced an increase in migration and invasion capacity of recipient osteoblasts compared to the uptake of non-metastatic OS-derived exosomes [
100]. In agreement with previous data [
99], the miRNA profile of exosomes from metastatic and non-metastatic OS cells differed, with an enrichment of target genes associated with metastasis and cancer. Amongst these, miR-675 was highly present in metastatic OS-derived exosomes. Validation studies using metastatic and non-metastatic exosome transfer and mimic treatments confirmed that the uptake of miR-675 via exosomes led to the downregulation of CALN1 expression associated with migration [
100]. A more recent study has investigated if OS-derived exosomes could induce changes to macrophages, osteoclasts, and endothelial cells, important components of the TME [
101]. Exosome internalization by macrophages induced osteoclast gene expression profile and an increase in the number of osteoclast-like cells. In addition, osteoclasts cultured with OS-derived exosomes had higher bone resorption activity in vitro [
101], suggesting that OS could induce changes in the bone microenvironment via macrophage and osteoclast modulation. Moreover, the uptake of OS-derived exosomes by endothelial cells induced an increase in pro-angiogenic factors and the formation of capillary structures in vitro, thus confirming that OS could modulate the invasiveness of cancer cells by modulating the TME [
101]. Profiling of the miRNA content of these exosomes identified miR-148a and miR-21-5p as being behind the change in the phenotype of osteoclast and endothelial activities upon exosome uptake, as overexpression of these miRNAs in recipient cells induced the expression of osteoclast markers and increased bone resorption and angiogenesis in vitro [
101]. Another example of OS-derived exosomes educating stromal cells was seen in mesenchymal stem cells (MSCs). Injection of MSCs co-cultured with metastatic OS-derived exosomes in an OS mouse model induced an increase in the tumour growth and tumour volume in vivo as well as metastatic dissemination to the lungs [
102]. Exosome profiling identified TGFβ to be present in the exosome surface, which, once in contact with MSCs, induced a signalling cascade resulting in the increase of cytokine production (IL-6 and IL-8) by MSCs [
102]. As OS cells do not produce IL-6, this release from MSCs could enhance a pro-inflammatory microenvironment favourable for metastasis.
Although not focused on the induction of a pro-metastatic phenotype in OS, Torregiani et al. [
103] identified that the transfer of exosomes from doxorubicin-resistant to chemosensitive OS cells induced an increase in the resistance to chemotherapeutics in vitro. The presence of MDR-1 and Pgp mRNA, both associated with drug resistance, was confirmed in the exosomal cargo, leading to an increase in the expression of these in recipient OS-chemosensitive cells [
103]. Therefore, exosomes from OS cells can not only educate TME cells, inducing a pro-metastatic and tumorigenic phenotype, but can educate less aggressive OS cells towards a chemo-resistant profile.
Another possibility is for exosomes released from TME-forming cells to modulate the behaviour of OS cells. In this direction, two different studies on OS have elucidated how non-OS exosomes can be taken up by OS cells inducing a pro-metastatic profile. In this line, Endo-Munoz et al. identified the urokinase plasminogen activator (uPA) and its receptor (uPAR) to be behind the metastatic potential of OS cells [
104]. Metastatic OS cells as well as exosomes derived from these contain high levels of uPA, which have been associated with a signalling cascade inducing an increase in the migration potential in vitro and metastasis in vivo [
104]. A paracrine loop in uPA signalling was identified via BM-educated OS cells. uPA was secreted by BM cells and internalised by OS cells, inducing an increase in the migration capacity and metastasis [
104]. This ability of stromal cells to induce changes in OS cells has been reported in CAF-secreted exosomes. Transfer of the exosomal cargo from CAF to OS cells increased migration and invasion in vitro compared to incubation with non-cancer fibroblast [
105]. This was induced by an enrichment of miR-1228 in CAF, which was transferred to OS cells via exosomes, resulting in a decrease in suppressor of cancer cell invasion expression and an increase in the migration capacity [
105]. This was validated by the blocking of the migration capacity when OS cells were incubated with normal fibroblasts (not enriched by miR-1228) or when expression of miR-1228 was inhibited [
105]. All these data further confirm that stromal cells can educate OS cells towards a more oncogenic phenotype, thus evidencing the importance of not only studying cancer cells but also stromal cells.
Despite not being associated with the metastatic potential of exosomes in OS, three other studies have investigated OS-derived exosomes. Shimbo et al. evaluated the ability to introduce synthetic miRNA to OS cells via exosomes in order to reduce their tumorigenic potential [
106]. Inducing the expression of miR-143 in MSCs, this miRNA was transferred to OS cells, with a subsequent reduction of the migration capacity without affecting the proliferation rate in vitro. Although the delivery efficiency was lower than using lipofection, the reduction in migration was similar and with less cytotoxicity [
106]. The two other studies investigated the ability to use OS-derived exosomes as non-invasive circulating biomarkers for disease progression [
107] and patient stratification between chemosensitive and chemoresistant profiles [
108]. This could eventually help in disease monitoring and treatment stratification in OS patients. A summary of all the data on exosomes in OS is presented in .
2.2. Exosomes in ES
ES is the second most common type of bone tumour in children and young adults, with its peak incidence between 15 and 24 years of age [
3]. The main driving event is the translocation between the
EWSR1 gene in chromosome 22 and a member of the ETS family of transcription factors, being in 85% of cases the
FLI1 gene located on chromosome 11 [
109]. However, less common ETS members have been identified as well, such as
ERG,
ETV1,
E1AF, or
FEV [
110]. This results in an aberrant transcription factor fundamental for ES tumorigenic transformation [
74]. Moreover, the cell-surface glycoprotein CD99 is expressed at high levels in 95–100% of cases [
111] and has been involved ES tumorigenesis [
112,
113]. These have become two powerful diagnostic biomarkers to distinguish ES from other small round cell sarcomas.
Although there have been improvements in the diagnosis and treatment for patients with ES, the survival is still poor [
76]. Most ES patients (75%) are diagnosed with localised disease, with a 5-year OSR of 75% [
114]. However, 25% of patients have metastatic disease at diagnosis, which is associated with dismissal survival rates (5-year OSR <30%) [
76]. Moreover, there is a proportion of patients that have refractory disease (will not respond to treatment) or will eventually recur, both being associated with poor survival rates (5-year OSR <25%) [
115]. Therefore, metastatic and recurrent disease are a main concern in ES, with researchers and clinicians studying the metastatic process to better understand its progression and to identify novel therapeutic targets. All this could ultimately lead to improved survival rates.
In parallel with the OS field, researchers in the ES field are trying to decipher the role of ES-derived exosomes in tumour progression and metastasis. Although there are fewer studies than on OS, increasing evidence is suggesting an important role of these small extracellular vesicles in ES cross-talk and TME modulation. The first evidence of exosomes secreted from ES cells was in 2013 [
116]. This first mention of ES-derived exosomes was centred on characterising the isolated vesicles focusing on the RNA content. This was used to define a panel of biomarkers to distinguish ES-exosomes from other cancer or non-cancer associated exosomes that could be identified in circulation [
116]. Although there was no validation of the role exosomes had in ES progression, different mRNAs associated with ES tumorigenesis were identified in their cargo, such as
EWSR1-FLI1 fusion or
EZH2 [
116]. Not long after this publication, another group identified
EWSR1-FLI1 mRNA to be contained in ES-derived exosomes [
117]. Moreover, xenograft ES mice released ES-derived exosomes in circulation at detectable concentrations, further evidencing the possibility of using ES-derived exosomes for diagnosis and monitoring purposes. The most important finding, however, was that ES-derived exosomes could transfer the
EWSR1-FLI1 mRNA to other ES recipient cells, but not to OS cells [
117]. This confirmed that exosomes could be implicated in the cross-talk between ES cells, modulating ES cellular behaviour towards a more tumorigenic state. A study using 3D tissue-engineered models based on scaffolds [
118] confirmed the previous findings in monolayer cultured cell lines [
116,
117]. After confirming that scaffold-derived exosomes resembled more the exosomes isolated from the patient’s plasma than those from monolayer cell cultures, they confirmed that
EZH2 was present in ES-derived exosomes [
118]. In contrast to previous data, ES-derived exosomes could transfer
EZH2 as part of their cargo to non-ES cells (osteoblasts and osteoclasts in this case), as well as ES cells [
118]. This was the first evidence that ES-derived exosomes could transfer genetic material to non-tumorigenic cells, setting the scene for further research on the cross-talk and implications of this transfer of information.
The first study that investigated the implications of the exosomal transfer in ES was in 2015 [
119]. The investigation was based in the neural differentiation triggered in ES cells by CD99 silencing via miR-34a-Notch-NFκB, antagonising the effect EWSR1-FLI1 had on ES cells [
119]. However, exosomes released from CD99-silenced ES cells, once taken up by normal ES cells, could induce a change in the phenotype similar to the stable silencing of CD99. This comprised an increase in neural differentiation and a decrease in the Notch and NFκB signalling cascade, which was associated with the transfer of high levels of miR-34a via exosomes [
119]. Further research from the same group showed that the uptake of CD99-silenced exosomes by ES cells induced a decrease in cellular growth, proliferation, and migration, as well as an increase in neural differentiation [
120]. In contrast, when ES-derived exosomes (expressing CD99) were taken up by CD99-silenced ES cells, migration capacity was regained and a decrease in neural markers took place. As CD99 expression did not change in recipient cells after exosome uptake, this modification of the cellular behaviour had to be associated with the exosomal cargo independent of CD99, as shown before [
120]. RNA and miRNA transcriptomic analysis showed that the uptake of CD99-silenced exosomes induces a change in the gene expression profile, confirming that the phenotypic alterations were driven by the transfer of genetic material [
120]. Moreover, the transfer of miR-199a-3p, the most enriched miRNA in CD99-silenced exosomes, induced a similar phenotype to the uptake of the entire exosomal cargo, thus suggesting that the change in cellular behaviour was mainly induced by the transfer of this miRNA [
120]. To put these data in a clinical context, the authors confirmed that tumours from patients with localised disease had higher levels of miR-199a-3p than those with metastatic disease, further confirming the role of this miRNA as a tumour suppressor. Data on the different studies on ES-derived exosomes are summarised in .
Table 2. Summary of exosome studies on ES.
| Origin cell |
Recipient cell |
Cargo |
Change |
Ref. |
| ES cell lines |
- |
EWSR1-FLI1, EZH2, and 10 more mRNAs
(mRNA profiling of known ES targets) |
Suitable as circulating biomarkers for ES, detectable in spike-in healthy blood samples |
[116] |
| ES cell lines |
ES cell lines |
EWSR1-FLI1 mRNA |
Labelled EWSR1-FLI1 transferred to other ES cells, not to OS |
[117] |
| ES cell lines |
Osteoblasts, osteoclasts in 3D scaffold |
EZH2 mRNA
(target of interest) |
Transfer of EZH2 mRNA to MSC (increase expression), osteoblasts (no change), and osteoclasts (reduction expression) |
[118] |
| ESCD99neg cell line model |
ES cell lines (normal CD99) |
Increased miR-34a |
Regulation of NFκB via miR-34a through reduction of Notch. Increase in neural differentiation (similar to direct CD99 silencing) |
[119] |
| ESCD99neg cell line model |
ES cell lines (normal CD99) |
miR-199a-3p
(miRNA profiling)
miR-199a-3p |
Induction of different gene expression profiles, neural differentiation, and neurogenesis; reduction of cell growth and migration (similar to miR mimic) |
[120] |
Therefore, the role that ES exosomes could play in modelling other cells and changing their cellular behaviour was confirmed. In this case, however, the change in phenotype was towards a less tumorigenic effect, which could be exploited for novel treatment strategies. Moreover, the different data on the protein and RNA content of ES-derived exosomes, including several important factors for ES tumorigenesis, suggest that this transfer of material could have important implications in TME modulation and education of neighbouring and distant cells, thus promoting the formation of pre-metastatic niches and modulation towards a more tumorigenic state.
2.3. Exosomes in CS
CS is the second most common type of bone sarcoma, with its peak incidence between 50–70 years of age [
88]. It is a heterogeneous disease characterised by slow cellular growth, the formation of a hyaline cartilaginous tissue around the cells, and low vasculature, making this bone sarcoma highly resistant to radiotherapy and chemotherapy [
88]. Moreover, 5–10% of CS patients present with advanced disease (3rd grade) associated with an elevated probability to develop metastasis [
121].
In contrast to OS and ES, there are still no data on the role exosomes could play in this malignancy. However, different studies have evaluated the role of miRNAs in angiogenesis and metastatic progression in CS. Angiogenesis is associated with worse prognosis in CS [
122]. It has been shown that CCL5 induces downregulation of miR-199a and miR-200b, not being able to regulate VEGF-A, resulting in higher VEGF-A levels in cells [
123,
124]. Similarly, miR-181a is enriched in hypoxic CS cells, which results in increased VEGF levels via the RGD16 and CXCR4 signalling cascade [
125].
Not only have miRNAs been associated with angiogenesis in CS, but several studies have linked miRNAs to tumour-suppressing mechanisms. For instance, high levels of miR-129-5p induced a decrease in SOX4, resulting in the inhibition of the proliferation, migration, and induction of apoptosis [
126]. A similar response was seen for miR-30a via SOX4, resulting in the induction of a similar phenotype transformation [
127]. Moreover, miR-141 and miR-101 have been shown to inhibit the metastatic potential of CS via regulation of c-Src [
128,
129].
Based on these data and on the evidence of the role exosomes have in OS and ES, it would not be a surprise if exosomes are shown to play a role in CS tumorigenesis. The transfer of miRNAs to modulate TME or CS cells towards a pro-angiogenic state or the education of CS cells in inhibiting tumour progression by the transfer of tumour suppressor miRNAs could be identified in the future. This might help to better understand the high radiotherapeutic and chemotherapeutic resistance of CS patients to conventional treatment. Moreover, based on data that OS-derived exosomes [
102] and ES-derived exosomes [
119,
120] can modulate cancer cells towards a less tumorigenic phenotype, reducing migration and proliferation, a similar strategy could be exploited in CS to obtain sensitivity to treatment strategies and a reduction of tumorigenesis.