Therefore, it is also clear that in this circumstance, the control of miR-19a levels could contribute to positive outcomes for cancer patients.
2. Roles and Mechanisms of Action of miR-19a in Clinical Cancer Features
Recently, scientific evidence has indicated the dysregulation of miRNAs in cancer initiation, progression, and aggressiveness, thus affecting the clinical features of cancer patients.
On this basis, we focused on miR-19a, probing its molecular mechanisms and analyzing its key role in different tumor types.
Several studies have shown that aberrantly expressed miRNAs contribute to the initiation and progression of brain tumors; between these, miR-19a has a central role.
Qin et al. [
40] showed that the upregulation of miR-19a-3p promoted cell proliferation, migration, and invasion by repressing the expression of PTEN, as reported in . PTEN is one of the most frequently mutated tumor suppressor genes [
41] in human cancers that plays a key role in tumor cell growth, survival, and metabolic regulation. Functionally, PTEN acts as a negative regulator of cell survival and protein synthesis via inhibition of the phosphatidylinositol 3-kinase (PI3K)/AKT [
41], one of the most important molecular pathways involved both in cell survival and in malignant neoplasms, which contributes, if altered or deregulated, to tumor pathogenesis [
42] and chemoresistance [
43].
On other hand, Chen et al. [
44] also reported that miR-19a-3p overexpression promotes cell proliferation and invasion by targeting RhoB in glioma, while its inhibition suppresses them, suggesting that miR-19a may act as an oncogene in gliomas.
The link between the Rho family and miR-19a-3p is also elucidated by Lv et al. [
45]. In their study, the authors identified the existence of a circ-EPB41L5/miR-19a/EPB41L5/RhoC/AKT regulatory axis. Circ-EPB41L5 inhibits the proliferation, migration, and invasion of glioma cells by sponging miR-19a-3p and regulating the host gene
EPB41L5 expression, which reduces the progression of glioma by inhibiting RhoC and p-AKT.
RhoC upregulation is associated with cell proliferation, contributing to the epithelial–mesenchymal transition (EMT). In addition,
RhoC improves cell motility, which consequently results in a greater ability of the tumor to become invasive [
46]. It is known that there is a connection between the increase in
RhoC expression and an advanced stage of the tumor as well as with the presence of metastases [
47]; to which certain crosstalk with angiogenic factors such as vascular endothelial growth factor (VEGF) also contributes [
48].
Downregulation of miR-19a in gliomas plays an anti-oncogenic role, which suggests its potential application as a target for gene therapy. Furthermore, because its overexpression is often associated with a poor prognosis, it could represent a new diagnostic and prognostic marker for gliomas.
Xu et al. [
49] investigated the impact of miR-19a-3p/miR-19b-3p on clinicopathologic factors and the prognosis of patients with ESCC. They observed that miR-19b-3p expression was positively correlated with tumor size, lymph node metastasis, and clinical stage, while miR-19a-3p is a prognostic indicator for progression-free survival and overall survival.
Similar results were obtained from Plum et al. [
50]. They demonstrated that upregulation of miR-19a/b is associated with tumor progression and the occurrence of lymph node metastasis in human esophageal adenocarcinoma, indicating that miR-19a/b could represent a new prognostic biomarker in this cancer form.
Many studies [
51,
52,
53] have shown that miR-19a is involved in the proliferation of human gastric cancer (GC). Yuan et al. [
51] demonstrated that miR-17-92a-1 Cluster Host Gene (MIR17HG)-derived miR-18a and miR-19a-3p coordinately mediate GC cell metastasis by directly inhibiting mothers against decapentaplegic homolog 2 (SMAD2) expression and upregulating Wingless-related integration site (Wnt)/β-catenin signaling.
Qin et al. demonstrated that the suppressor of cytokine signaling 1 (
SOCS1) is a novel target of miR-19a-3p in GC cells. They reported that miR-19a-3p expression is inversely correlated with
SOCS1 expression in GC cells, and that its overexpression markedly promotes proliferation and tumorigenicity both in vitro and in vivo [
54]. In accordance with this evidence, other studies [
55,
56] have confirmed miR-19a overexpression in GC patients, suggesting that miR-19a could represent a potential new diagnostic biomarker for GC.
Liu et al. [
57] demonstrated that miR-19a-3p is involved in colorectal cancer, promoting its proliferation and migration by targeting T cell Intracellular Antigen 1 (TIA1), thus also suggesting miR-19a as a new diagnostic and prognostic biomarker for gastrointestinal cancers.
The role of miR-19a-5p in hepatocellular carcinoma (HCC) has also been elucidated by Baik et al. [
58], who showed that the suppression of adenine nucleotide translocase 2 (ANT2) by short hairpin RNA (shRNA) downregulates miR-19a through the PI3K/Akt pathway. The knockdown of ANT2 directly downregulates miR-19a, thus resulting in the suppression of tumor growth in HCC cells and clinical samples.
Tan et al. [
59] demonstrated that high levels of miR-19a-5p correlate with poor prognosis in patients, proposing that miR-19a is a potential therapeutic target for pancreatic cancer.
Recently, it has been revealed that PLGF influences miR-19a-3p expression by modulating c-
MYC [
60]. Furthermore, a positive pairwise correlation among PLGF, c-
MYC, and miR-19a expression in gallbladder cancer (GBC) tissues has been displayed [
60]; this finding confirmed that the PLGF/c-MYC/miR-19a axis is involved in tumor progression of the gastrointestinal tract.
The upregulated expression of miR-19a-3p has also been determined in clinical tongue squamous cell carcinoma cells (TSCC) specimens [
61].
Wu et al. [
62] reported that miR-19a-3p is upregulated in laryngeal squamous cell carcinoma (LSCC) patients and is correlated with neck nodal metastasis, poor differentiation, and advanced stage, indicating that its overexpression is associated with reduced overall survival. It has also been demonstrated that miR-19a plays an influent effect in laryngeal verrucous squamous cell carcinoma (LVSCC), a highly differentiated form of LSCC. Marioni et al. [
63] reported that miR-19a expression is significantly higher in malignant glottic lesions (LSCC and LVSCC) than in benign ones. Thus, these studies indicate the oncogenic role of miR-19a in the progression of LSCC and denote it as a possible biomarker to establish an earlier diagnosis as well as a marker of differentiation in the various forms of laryngeal tumors.
miR-19a overexpression is involved in the pathophysiology of lung cancer, and is associated with the poor prognosis, metastasis, and proliferation of pulmonary cancer cells [
64]; thus, it constitutes a good biomarker and a possible target therapy for lung tumors [
65].
Gu et al. [
66] demonstrated that miR-19a-3p/miR-19b-3p promotes the proliferation and migration of lung cancer cells by targeting Microtubule-Associated Scaffold Protein 1 (MTUS1). Following these suggestions, miR-19a could represent an important diagnostic and prognostic marker for differential diagnoses of lung cancers [
67].
Some clinical studies [
68,
69] have shown that high levels of miR-19a are implicated in a more frequently large tumor size, advanced clinical stage, positive distant metastasis, and poor response to chemotherapy in osteosarcoma patients. Huang et al. [
70] and Zou et al. [
71] described that miR-19a-5p and miR-19a-3p overexpression contributes to both the risk of poor prognosis in osteosarcoma and the probability of developing metastases, respectively.
miRNAs profiling can also be useful for the characterization and classification of different thyroid carcinomas, as well as in strengthening therapeutic strategies.
Calabrese et al. [
72] showed that miR-19a-3p overexpression is correlated with a poor prognosis of thyroid cancers, highlighting its contribution to more de-differentiation and aggressiveness.
These results suggest that not only does miR-19a-3p have an important role in the malignancy of thyroid cancers [
73], but it also represents an important prognostic indicator and is a good therapeutic target [
74] for anaplastic thyroid carcinoma (ATC) patients.
The role of miR-19a has also been investigated in clear cell renal cell carcinoma (ccRCC) tissues and human cell lines, highlighting that the high expression of miR-19a-3p is correlated with poor prognosis via promoting cell proliferation and suppressing PTEN/mothers against decapentaplegic homolog 4 (SMAD4) expression [
75]. In addition, Niu et al. [
76] indicated that miR-19a-3p directly targets the 3′untranslated region (3′UTR) of RhoB, promoting tumorigenesis, cancer cell proliferation, and invasiveness, and suggesting the clinical potential of miR-19a as a molecular target in ccRCC. Ge et al. [
77] assessed the relationship of high miR-19a levels with the progression and prognosis of chromophobe renal cell carcinoma (chRCC), demonstrating that miR-19a inhibition is significantly associated with both recurrence-free survival and overall survival.
miR-19a upregulation has also been correlated with bladder cancer prognosis. The role of the PI3K/AKT pathway and its crosstalk with miR-19a-3p in bladder carcinogenesis has also been elucidated by Calderaro et al. [
78], thus opening the way to involvements between miR-19a and various molecular patterns in bladder urothelial carcinomas (UCs).
Mearini et al. [
79] reported that miR-19a-3p is overexpressed in bladder cancer carcinogenesis and its oncogenic role is dependent on targeting PTEN [
80] as well as inhibiting the expression of RhoB, in order to promote the invasion and EMT of bladder cancer cells [
81]. Furthermore, it has been shown that miR-19a overexpression is associated with the poor prognosis of bladder cancer patients [
82], representing a good starting point for future preclinical and clinical exploration.
Despite the initial controversy over the beneficial role of miR-19a in prostate cancer [
83], recent studies are consistent in indicating its crucial role in prostate tumorigenesis and progression [
84]. Its involvement includes the promotion of cell migration, invasion, and EMT in prostate cancer by directly binding to Cullin-5 (CUL5) mRNA 3′-UTR as reported by Wang et al. [
85]; the regulation of proliferation and apoptosis of prostate cancer cells by targeting the B cell translocation gene 1 (BTG1) as described by Lu et al. [
86], and the inhibition of vacuolar protein sorting-associated protein 37A (VPS37A) expression, as indicated by Fu et al. [
87].
All these data highlight the importance of miR-19a for the development of new targeted therapies and suggest its use as a prognostic biomarker in prostate cancer patients [
88].
miR-19a overexpression promotes cell proliferation, metastasis, migration, invasion, and angiogenesis in breast cancer (BC) [
89]. Sochor et al. [
90] developed and validated a composite risk score based on the expression of three miRNAs, including miR-19a, with prognostic value for BC. They reported that miR-19a overexpression is correlated specifically with bone metastasis, suggesting that it could be a good diagnostic and prognostic marker for BC. Additionally, Kawaguchi et al. [
91] showed that miR-19a overexpression is correlated with poor prognosis and the risk of multiple metastases, as well as angiogenesis and EMT.
Alunni-Fabbroni et al. [
92] indicated that miR-19a-3p shows a promising role differentiating early BC patients at different time points and from healthy controls. In addition, the regulation of miR-19a in BC is also useful in managing chemoresistance, as suggested by Liang et al. [
32]. Ouchida et al. [
93] suggested that inosine monophosphate dehydrogenase 1 (IMPDH1) and probable aminopeptidase-like 1 (NPEPL1) genes are direct targets of miR-19a in BC, while the exogenous expression of these genes is not associated with the growth suppression of MCF-7 cells. Hence, once again, the importance and usefulness of miR-19a both as a diagnostic biomarker and as a molecular target are highlighted.
Scientific findings have confirmed the involvement of miR-19a in malignant lymphoma, elucidating its crucial roles in the tumorigenesis and pathogenesis of aggressive transformed, high-grade, and refractory lymphomas, highlighting its prognostic role [
94].
Lv et al. [
95] performed bioinformatic analyses and demonstrated that 41 target genes of miR-19a are associated with the development and progression of multiple myeloma (MM), suggesting its potential role as a biomarker. Another study [
96] also confirmed that miR-19a-3p plays the role of an oncogene by regulating the PTEN/AKT/pAKT pathway in MM and promoting cell proliferation and inhibiting apoptosis.
Wang et al. [
97] demonstrated that miR-19a-3p is highly expressed in ovarian cancer tissues and cell lines and that its overexpression promotes proliferation, while its down-regulation reduces the growth of ovarian cancer cells. Furthermore, in this study, the authors suggested that the overexpression of PTEN suppresses miR-19a, promoting an effect on cancer cell growth, indicating that miR-19a expression and PTEN are inversely related in ovarian cancer tissues. The role of miR-19a has also been investigated in metastatic serous ovarian cancer (SOC). In fact, Wahab et al. [
98] described a significant differential expression of 48 miRNAs, including miR-19a, in metastatic SOC compared to healthy subjects. These studies proved a potential oncogenic role of miR-19a in ovarian cancer, suggesting that it could represent a promising marker for ovarian cancer diagnosis, prognosis, and treatment.
The role of miRNAs in cervical cancer concerns many aspects of tumor cell development and survival, including sensitivity to radiotherapy.
In their study, Wang et al. [
99] showed that the silencing of miR-19a-5p significantly improved the sensitivity of SiHa cells to radiotherapy by reducing proliferation, increasing apoptosis, upregulating BCL2-associated X (BAX), and downregulating B cell lymphoma 2 (Bcl-2) [
99].
Similar results have been shown by Xu et al. [
100], who demonstrated that both miR-19a-5p and miR-19b-5p are vastly expressed in human cervical cancer cells and are implicated in malignant HeLa and C33A cell phenotypes [
100].
In addition, miR-19a-5p and miR-19b-5p have also been shown to control CUL5 levels directly and negatively in cervical cancer cells, both emphasizing their importance and that of their target genes in tumorigenesis processes [
100].
Furthermore, it is interesting to highlight the Bcl-2 interacting mediator of cell death (BIM), an initiator of the intrinsic apoptotic pathway in both physiological and pathophysiological conditions [
101]. In fact, its reduction has often been associated with tumor promotion, while its overexpression has the ability to inhibit tumor growth and resistance to chemotherapy [
102]. Therefore,
BIM has emerged as a key mediator in the regulation of tumorigenesis, as demonstrated by several studies [
103,
104] that indicate it as a promising target in the field of anticancer therapy. In particular, the miR-17-92 cluster has been found to suppress
BIM expression in multiple myeloma cells [
105], human ovarian cancer cells [
106], and esophageal adenocarcinoma [
102]. miR-19a, as a prominent component of the miR-17-92 cluster, can facilitate tumor formation, inhibiting BIM expression and promoting the proliferation of tumor cells [
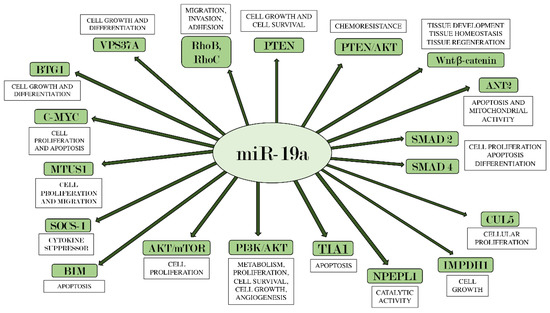
107]. The function of BIM, along with all other miR-19a targets described in this review, is shown in .
Table 1. This table summarizes the relationship between the miR-19a isoform and its target genes in several types of cancers.
Figure 1. miR-19a and its main target genes. The figure summarizes the interactions between miR-19a and the target genes on which it acts, also indicating each their main biological effects.