Teak (Tectona grandis L.f.) is one of the most valuable timber species, and is cultivated in agroforestry systems in many countries across the tropical and subtropical regions of the world.
- agroforestry
- allelopathy
- decomposition
- homegarden
1. Introduction
Teak (Tectona grandis L.f.), belonging to the family of Lamiaceae, is a large deciduous tree up to 40 m in height. The leaves are ovate (40 cm long, 20 cm wide) and hairy on the lower surface. It has small, fragrant white flowers attached in panicles at the end of its branches. Teak is one of the most valuable timber species because of its beautiful appearance and durable property [1][2]. Although native to South and Southeast Asia, the significant economic potential of teak wood led the species to be introduced into the agroforestry systems of many countries across tropical Asia, Africa and Central and South America [3][4][5][6][7].
Home gardens in tropical and subtropical countries surround residences of local inhabitants, and create small forest-like structures. They are considered to be the traditional agroforestry systems characterized by complexity of the structure and multiple functions. Home gardens consist of various kinds of tree species, with crops, livestock, poultry and fishes—those species have been selected by local inhabitants for their preference, productivity and sociocultural aspects. Home gardens provide various food and goods, including commodities such as animal products, fruits, vegetables, folk medicine, ornamentals, fodder, timber and fuel [8][9][10][11][12][13]. Teak is one of the most essential trees in home gardens in South Asia, because it is a very precious wood species and important in folk remedies [8][14][15]. Hot water extracts of teak barks are applied for the treatment of bronchitis, biliousness, hyperacidity, diabetes, dysentery, and leprosy. Water extracts of teak leaves are used in pruritus, stomatitis, ulcers and wounds. Hot water extracts of teak roots are applied for anuria treatments. Oil extracts of the flowers are useful for scabies and hair growth. It has also been used as an important plant in Ayurvedic treatments [2][16].
Evidence of the pharmacological properties of teak plants has been accumulated over the past decades. Ethanol extracts of teak leaves have shown significant wound healing activity [17]. Ethanol extracts of teak roots have hyperglycemic activity [18]. Ethanol extracts of teak barks show anti-inflammatory and analgesic potentials [19]. Petroleum ether extracts of teak seeds have hair growth activity [20]. Many compounds with pharmacological activities were also isolated from various parts of teak plants [2][16][21].
Some plants have shown excellent weed control abilities as soil additives and/or in intercropping, due to their characteristics of allelopathy [22][23]. Plants produce hundreds of secondary metabolites. Some of those compounds are released into the surrounding environments through root exudation, volatilization, leaching and decomposition of the plants. Those compounds with allelopathic activity are able to inhibit the growth and germination of neighboring plant species [24][25][26]. Therefore, allelopathy of plants is potentially useful for weed management options in several agriculture settings, including agroforestry systems, for the reduction of commercial herbicide dependency [27][28]. Many phytotoxic substances with allelopathic activity in teak have already been isolated and characterized. However, there has been no review article about the phytotoxic substances involved in teak allelopathy.
2. Ecological Impact of Teak
3. Allelopathic Property of Teak
3.1. Leachate
3.2. Effects of Teak Leaves
3.3. Extract of Teak Soil
3.4. Extracts of Teak Leaves
| Source | Inhibition | Target Plant Species | Reference |
|---|---|---|---|
| Leachate from leaves | Germination, plant growth | Vigna unguiculata, Momordica charantia, Solanum melongena | [37] |
| Cicer arietinum | [38] | ||
| Leaf mulch | Rhizome growth | Turmeric | [15] |
| Leaf powder | Weed emergence | Cyndon dactylon, Echinochloa colona, Cyperus rotundus, Cyperus difformis, Amaranthus viridis, Chenopodium album, Melilotus alba | [39] |
| Extracts | |||
| Soil under teak trees | Germination, plant growth | Tomato | [40][41] |
| Fallen leaf | Plant growth, protein content | Vigna mungo, Vigna radiata | [42] |
| Germination, | Echinochloa colona, Cyperus difformis | [43] | |
| Weed emergence | Cyndon dactylon, Echinochloa colona, Cyperus rotundus, Cyperus difformis, Amaranthus viridis, Chenopodium album, Melilotus alba | [39] | |
| Fresh leaf | Germination, plant growth | Vigna mungo | [44] |
| Germination, plant growth | Plumbago zeylanica | [45] | |
| Rice, maize, Vigna radiate, Vigna umbellate, Arachis hypogeae | [46] | ||
| Germination | Luffa cylindrical, Abelmoschus esculentus, Brassica juncea | [47] | |
| Plant growth, contents of chlorophyll and carotenoid | Chilli, Vigna radiata | [48] | |
| Plant growth, contents of chlorophyll and carotenoid | Pennisetum glaucum, Eleusine coracana | [49] | |
| Plant growth | Amaranthus spinosus | [50] | |
| Root | Germination, seedling growth | Hibiscus esculentus | [51] |
| Leaf, root, bark | Plant growth | Maize | [52] |
4. Phytotoxic Substances with Allelopathic Activity in Teak
Phytotoxic substances with allelopathic activity identified in teak are discussed in this section. All phytotoxic substances listed in Table 2 and Figure 1 were isolated from fresh teak leaves with water. Naphthotectone (3) inhibited the germination and seedling growth of wheat (Triticum aestivum L.), onion (Allium cepa L.), tomato (Lycopersicon esculentum L.), and lettuce (Lactuca sativa L.) [53]. Rhinocerotinoic acid (8) suppressed the germination and seedling growth of wheat and lettuce [54]. 2-Oxokovalenic acid (9) and 19-hydroxyferruginol (11) inhibited the germination and seedling growth of wheat, onion, lettuce; 3β-hydroxy-7,8-dihydro-β-ionol (15) inhibited the seedling growth of wheat, onion and tomato; and 3β-hydroxy-7,8-dihydro-β-ionone (16) inhibited the seedling growth of wheat, onion, tomato and lettuce [55]. Other compounds listed in Table 2 inhibited the seedling growth of wheat [54][55][56]. Although those compounds were isolated and identified from teak leaves for potential use as a source of natural herbicide model and/or bioactive compounds, the allelopathic effects of those compounds were determined only by crop plants. It may be necessary to determine the activity of those compounds on weed species.
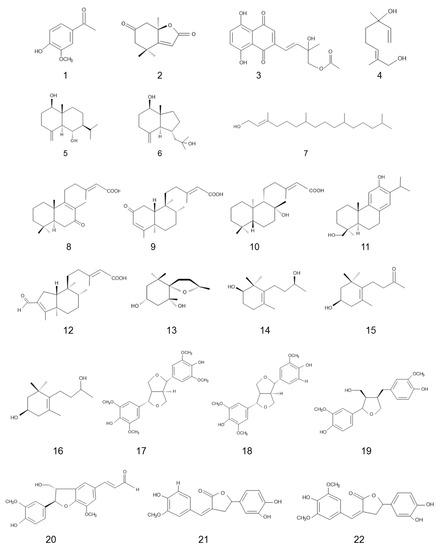
| Phytochemical Class | Compound | Terget Plant Species | Inhibition | Reference |
|---|---|---|---|---|
| Phenolic | 1: Acetovanillone | Wheat | Plant growth | [56] |
| Benzofuran | 2: Dehydrololiolide | Wheat | Plant growth | [55] |
| Anthra quinone | 3: Naphthotectone | Wheat, onion, tomato, lettuce | Plant growth, germination | [53] |
| Monoterpene | 4: (6RS)-(E)-2,6-Dimethyl-2,7-octadiene-1,6-diol | Wheat | Plant growth | |
| Sesquterprne | 5: lβ-6α-Dihydroxy-4(15)-eudesmene | Wheat | Plant growth | [54] |
| 6: (1S,3aR,4R,7aS)-1-(2-hydroxypropan-2-yl)-3a-methyl-7-methyleneoctahydro-1H-inden-4-ol | Wheat | Plant growth | [54] | |
| Diterpene | 7: Phytol | Wheat | Plant growth | [54] |
| 8: Rhinocerotinoic acid | Wheat, lettuce | Plant growth, germination | [54] | |
| 9: 2-Oxokovalenic acid | Wheat, onion, lettuce | Plant growth, germination | [54] | |
| 10: Lab-13-en-8β-ol-15-oic acid | Wheat, onion, lettuce | Plant growth | [54] | |
| 11: 19-Hydroxyferruginol | Wheat, onion, lettuce | Plant growth, germination | [54] | |
| 12: Solidagonal acid | Wheat | Plant growth | [54] | |
| Apocarotenoid | 13: Tectoionol A | Wheat | Plant growth | [55] |
| 14: Tectoionol B | Wheat | Plant growth | [55] | |
| 15: 3β-Hydroxy-7,8-dihydro-β-ionol | Wheat, onion, tomato | Plant growth | [55] | |
| 16: 3β-Hydroxy-7,8-dihydro-β-ionone | Wheat, onion, tomato, lettuce | Plant growth | [55] | |
| Phenylpropanoid | 17: Syringaresinol | Wheat | Plant growth | [56] |
| 18: Medioresinol | Wheat | Plant growth | [56] | |
| 19: Lariciresinol | Wheat | Plant growth | [56] | |
| 20: Balaphonin | Wheat | Plant growth | [56] | |
| 21: Tectonoelin A | Wheat | Plant growth | [56] | |
| 22: Tectonoelin B | Wheat | Plant growth | [56] |
Several phenolics were also identified in teak barks and leaves [39][50][57]. Phenolic compounds have been found in a wide range of plants and soils, and often mentioned as putative allelopathic substances [58][59]. The importance and contribution of those phenolics found in teak are not clear because no information regarding the phytotoxic activity of those compounds for teak allelopathy is available in the literature. However, gallic and ellagic acids were identified in teak leaf extracts [60], and the allelopathic activity of those compounds isolated from other plant sources were reported [61][62]. Therefore, some phenolics in teak plants may contribute to the allelopathy of teak. Phenolic compounds inhibit some enzyme activities and physiological processes, such as plant hormone functions, water balance and mineral uptake, as well as stomatal functions, respiration, and photosynthesis [58][63].
A number of secondary metabolites in many classes have been isolated and identified from various parts of teak plants, such as barks, flowers, fruits, leaves and roots. Those compounds were quinones, terpenes, apocarotenoids, phenolics, flavonoids, saponins, lignans and norlignans [16][64]. Teak wood shows resistance to termite and fungal damages, and napthoquinones and anthraquinones contribute a resistance property [65][66][67][68]. Some other compounds were also related to the pharmacological activities of teak [2][16][21][35][57]. Although those compounds have been associated with the pharmacological effects and property of its wood characteristics, some of those compounds may possess phytotoxic activity.
This entry is adapted from the peer-reviewed paper 10.3390/app11083314
References
- Tangmitcharoen, S.; Owens, J.N. Floral biology, pollination, pistil receptivity, and pollen tube growth of teak (Tectona grandis Linn f.). Ann. Bot. 1997, 79, 227–241.
- Nidavani, R.B.; Mahalakshmi, A.M. Teak (Tectona grandis Linn.): A renowned timber plant with potential medicinal values. Int. J. Pharm. Sci. 2014, 6, 48–54.
- Pandey, D.; Brown, C. Teak: A global overview. Unasylva 2000, 51, 3–13.
- Healey, S.P.; Gara, R.I. The effect of a teak (Tectona grandis) plantation on the establishment of native species in an abandoned pasture in Costa Rica. Ecol. Manag. 2003, 176, 497–507.
- Kenny, A.L.; Pickens, J.B.; Orr, B. Land allocation with the introduction of teak: A case study of smallholder farms in Southern Togo. J. Sustain. For. 2014, 33, 776–795.
- Newby, J.C.; Cramb, R.A.; Sakanphet, S. Forest transitions and rural livelihoods: Multiple pathways of smallholder teak expansion in Northern Laos. Land 2014, 3, 482–503.
- Udayana, C.; Andreassen, H.P.; Skarpe, C. Understory diversity and composition after planting of teak and mahogany in Yogyakarta, Indonesia. J. Sustain. For. 2020, 39, 494–510.
- Kumar, B.M.; George, S.J.; Chinnamani, S. Diversity, structure and standing stock of wood in the homegardens of Kerala in peninsular. Indian Agrofor. Syst. 1994, 25, 243–262.
- Michon, G.; Mary, F. Conversion of traditional village gardens and new economic strategies of rural households in the area of Bogor, Indonesia. Agrofor. Syst. 1994, 25, 31–58.
- Roshetko, J.M.; Delaney, M.; Hairiah, K.; Purnomosidhi, P. Carbon stocks in Indonesian homegarden systems: Can smallholder systems be targeted for increased carbon storage? Am. J. Altern. Agric. 2002, 17, 138–148.
- Pandey, C.B.; Rai, R.B.; Singh, L.; Singh, A.K. Homegardens of Andaman and Nicobar, India. Agric. Syst. 2007, 92, 1–22.
- Mohri, H.; Lahoti, S.; Saito, O.; Mahalingam, A.; Gunatilleke, N.; Irhamc; Hoang, V.T.; Hitinayake, G.; Takeuchi, K.; Herath, S. Assessment of ecosystem services in homegarden systems in Indonesia, Sri Lanka, and Vietnam. Ecosyst. Serv. 2013, 5, e125–e136.
- Bhat, S.; Bhandary, M.J.; Rajanna, L. Plant diversity in the homegardens of Karwar, Karnataka, India. Biodiversitas 2014, 15, 229–235.
- Peyre, A.; Guidal, A.; Wiersum, K.F.; Bongers, F. Dynamics of homegarden structure and function in Kerala, Indian. Agrofor. Syst. 2006, 66, 101–115.
- Lakshmi, P.G.S.; John, J. Allelopathic effect of leaf loppings of homestead trees on turmeric (Curcuma longa Linn.). J. Trop. Agric. 2015, 53, 227–232.
- Vyas, P.; Yadav, D.K.; Khandelwal, P. Tectona grandis (teak)—A review on its phytochemical and therapeutic potential. Nat. Prod. Res. 2019, 33, 2338–2354.
- Varma, S.B.; Giri, S.P. Study of wound healing activity of Tectona grandis Linn. leaf extract on rats. Anc. Sci. Life 2013, 32, 241–244.
- Varma, S.B.; Jaybhaye, D.L. Antihyperglycemic activity of Tectona grandis Linn. bark extract on alloxan induced diabetes in rats. Nat. Prod. Res. 2010, 24, 1059–1068.
- Asif, M. In vivo analgesic and antiinflammatory effects of Tectona grandis Linn. stem bark extracts. MJPS 2011, 9, 1–11.
- Jaybhaye, D.; Varma, S.; Gagne, N.; Bonde, V.; Gite, A.; Bhosle, D. Effect of Tectona grandis Linn. Seeds on hair growth activity of albino mice. Int. J. Ayurveda Res. 2010, 1, 211–215.
- Dégbé, M.; Debierre-Grockiego, F.; Tété-Bénissan, A.; Débare, H.; Aklikokou, K.; Dimier-Poisson, I.; Gbeassor, M. Extracts of Tectona grandis and Vernonia amygdalina have anti-Toxoplasma and pro-inflammatory properties in vitro. Parasite 2018, 25, 11.
- Semidey, N. Allelopathic crops for weed management in cropping systems. In Allelopathy Update. Basic and Applied Aspects; Narwal, S.S., Ed.; Science Publishers Inc.: Enfield, NH, USA, 1999; Volume 2, pp. 271–281.
- Caamal-Maldonado, J.A.; Jiménez-Osornio, J.J.; Torres-Barragán, A.; Anaya, A.L. The use of allelopathic legume cover and mulch species for weed control in cropping systems. Agron. J. 2001, 93, 27–36.
- Field, B.; Jordán, F.; Osboourn, A. First encounters-deployment of defense-related natural products by plant. New Phytol. 2006, 172, 193–207.
- Bais, H.P.; Weir, T.L.; Perry, L.G.; Gilroy, S.; Vivanco, J.M. The role of root exudates in rhizosphere interactions with plants and other organisms. Annu. Rev. Plant Biol. 2006, 57, 233–266.
- Belz, R.G. Allelopathy in crop/weed interactions—An update. Pest Manag. Sci. 2007, 63, 308–326.
- Narwal, S.S. Allelopathy in weed management. In Allelopathy Update. Basic and Applied Aspects; Narwal, S.S., Ed.; Science Publishers Inc.: Enfield, NH, USA, 1999; Volume 2, pp. 203–254.
- Duke, S.O.; Dayan, F.E.; Romagni, J.G.; Rimando, A.M. Natural products as sources of herbicide, current status and future trends. Weed Res. 2000, 40, 99–111.
- Saha, S. Vegetation composition and structure of Tectona grandis (teak, Family Verbanaceae) plantations and dry deciduous forests in central India. For. Ecol. Manag. 2001, 48, 159–167.
- Falcão, J.C.F.; Dáttilo, W.; Izzo, T.J. Efficiency of different planted forests in recovering biodiversity and ecological interactions in Brazilian Amazon. For. Ecol. Manag. 2015, 339, 105–111.
- Imron, M.A.; Tantaryzard, M.; Satria, R.A.; Maulana, I.; Pudyatmoko, K. Understory avian community in a teak forest of Cepu, Central Java. J. Trop. For. Sci. 2018, 30, 509–518.
- Oliveira, A.T.M.; Bernardo, C.S.S.; Melo, F.R.D.; dos Santos-Filho, M.; Peres, C.A.; Canale, G.R. Primate and ungulate responses to teak agroforestry in a southern Amazonian landscape. Mamm. Biol. 2019, 96, 45–52.
- Sahoo, U.K. Allelopathic studies of understorey weeds by agroforestry trees in home gardens of Mizoram. J. Exp. Biol. Agric. Sci. 2013, 1, 248–257.
- Ikhajiagbe, B.; Ogwu, M.C.; Lawrence, A.E. Single-tree influence of Tectona grandis Linn. f. on plant distribution and soil characteristics in a planted forest. Bull. Natl. Res. Cent. 2020, 44, 29.
- Suryanti, V.; Kusumaningsih, T.; Marliyana, S.D.; Setyono, H.A.; Trisnawati, E.W. Identification of active compounds and antioxidant activity of teak (Tectona grandis) leaves. Biodiversitas 2020, 21, 946–952.
- Jha, K.K. Litter production and leaf litter decomposition dynamics in an age series Tectona grandis Linn.f. plantations of moist Tarai Sal forest. Indian For. 2010, 136, 433–450.
- John, J.; Sreekumar, K.M.; Rekha, P. Allelopathic effects of leaf leachates of multipurpose trees on vegetables. Allelopathy J. 2007, 19, 507–516.
- Das, C.R.; Mondal, N.K.; Aditya, P.; Datta, J.K.; Banerjee, A.; Das, K. Allelopathic potentialities of leachates of leaf litter of some selected tree species on gram seeds under laboratory conditions. Asian J. Exp. Biol. Sci. 2012, 3, 59–65.
- Kole, R.K.; Paul, P.; Saha, S.; Das, S. Chemistry and bio-efficacy of teak leaf for weed control in wheat. Allelopathy J. 2016, 39, 191–204.
- Mensah, E.E.; Owusu-Mensah, I.; Oppong, E.; Saka, M.O. Allelopathic effect of topsoil extract from Tectona grandis L. plantation on the germination of Lycopersicum esculentum. J. Biol. Agric. Health 2015, 5, 117–122.
- Owusu-Mensah, I.; Mensah, E.E. Polynomial modelling of allelopathic effect of topsoil extract. Br. J. Res. 2015, 2, 132–141.
- Manimegalai, A. Allelopathic effect of Tectona grandis leaves on protein content changes of black gram and green gram. Int. J. Curr. Sci. 2012, 4, 30–34.
- Kole, R.K.; Karmakar, P.R.; Poi, R.; Mazumdar, D. Allelopathic inhibition of teak leaf extract: A potential pre-emergent herbicide. J. Crop. Weed 2011, 7, 101–109.
- Evangeline, V.R.; Prakash, E.J.J.; Samuel, A.S.; Jayakumar, M. Allelopathic potential of Tectona grandis L. on the germination and seedling growth of Vigna mungo (L.) Hepper. Pak. J. Weed Sci. Res. 2012, 18, 65–70.
- Biswas, K.; Das, A.P. Allelopathic effects of teak (Tectona grandis L.f.) on germination and seedling growth of Plumbago zeylanica L. Pleione 2016, 10, 262–268.
- Bhatt, B.P.; Singh, J.K.; Barooah, L.; Imtimongla. Phytotoxic influence of agroforestry tree species on food crops in Eastern Himalaya, India. Allelopathy J. 2010, 25, 485–496.
- Kumari, N.; Srivastava, P.; Mehta, S.; Lemtur, M.; Das, B. Allelopathic effects of some promising agro forestry tree species on different annual crops. Econ. Environ. Conserv. 2016, 22, 225–236.
- Leela, P.; Arumugam, K. Allelopathic influence of teak (Tectona grandis L.) leaves on growth responses of green gram (Vigna radiata (L.) Wilczek) and chilli (Capsicum frutescens L.). Int. J. Curr. Biotechnol. 2014, 2, 55–58.
- Leela, P. Phytotoxic effect of Tectona grandis (L.f.) leaf extracts on growth and developmental changes of Pennisetum glaucum (L.) R.BR. and Eleusine coracana (Gaertn). Int. Educ. Appl. Sci. Res. 2017, 6, 7–10.
- Erida, G.; Saidi, N.; Hasanuddin; Syafruddin. Herbicidal potential of methanolic extracts of Pinus merkusii Jungh. et de Vriese, Acacia mangium Willd., Jatropha curcas L., Tectona grandis L.f. and Terminalia catappa L. on Amaranthus spinosus L. Allelopathy J. 2020, 49, 201–216.
- Abugre, S.; Apetorgbor, A.K.; Antwiwaa, A.; Apetorgbor, M.M. Allelopathic effects of ten tree species on germination and growth of four traditional food crops in Ghana. J. Agric. Technnol. 2011, 7, 825–834.
- Sahoo, U.K.; Upadhyaya, K.; Meitei, C.B. Allelopathic effects of Leucaena leucocephala and Tectona grandis on germination and growth of maize. Allelopathy J. 2007, 20, 135–144.
- Lacret, R.; Varela, R.M.; Molinillo, J.M.G.; Nogueiras, C.; Macías, F.A. Anthratectone and naphthotectone, two quinones from bioactive extracts of Tectona grandis. J. Chem. Ecol. 2011, 37, 1341–1348.
- Macías, F.A.; Lacret, R.; Varela, R.M.; Nogueiras, C.; Molinillo, J.M.G. Isolation and phytotoxicity of terpenes from Tectona grandis. J. Chem. Ecol. 2010, 36, 396–404.
- Macías, F.A.; Lacret, R.; Varela, R.M.; Nogueiras, C.; Molinillo, J.M.G. Bioactive apocarotenoids from Tectona grandis. Phytochemistry 2008, 69, 2708–2715.
- Lacret, R.; Varela, R.M.; Molinillo, J.M.G.; Nogueiras, C.; Macías, F.A. Tectonoelins, new norlignans from a bioactive extract of Tectona grandis. Phytochem. Lett. 2012, 5, 382–386.
- Balogun, A.O.; Lasode, O.A.; McDonald, A.G. Devolatilisation kinetics and pyrolytic analyses of Tectona grandis (teak). Bioresour. Technol. 2014, 156, 57–62.
- Inderjit. Plant phenolics in allelopathy. Bot. Rev. 1996, 62, 186–202.
- Dalton, B.R. The occurrence and behavior of plant phenolic acids in soil environments and their potential involvement in allelochemical interference interactions: Methodological limitations in establishing conclusive proof of allelopathy. In Principals and Practices in Plant. Ecology: Allelochemical Interactions, Inderjit; Dakshini, K.M.M., Foy, C.L., Eds.; CRC Press: Boca Raton, FL, USA, 1999; pp. 57–74.
- Nayeem, N.; Karvekar, M.D. Isolation of phenolic compounds from the methanolic extract of Tectona grandis. Res. J. Pharm. Biol. Chem. Sci. 2010, 1, 221–225.
- Rudrappa, T.; Bonsall, J.; Gallagher, J.L.; Seliskar, D.M.; Bais, H.P. Root-secreted allelochemical in the noxious weed Phragmites australis deploys a reactive oxygen species response and microtubule assembly disruption to execute rhizotoxicity. J. Chem. Ecol. 2007, 33, 1898–1918.
- Liu, J.; Li, D.; Wang, D.; Liu, Y.; Song, H. Allelopathic effects, physiological responses and phenolic compounds in litter extracts of Juniperus rigida Sieb. et Zucc. Chem. Biodivers. 2017, 14, e1700088.
- Einhellig, F.A. Mode of action of allelochemical action of phenolic compounds. In Chemistry and Mode of Action of Allelochemicals; Macías, F.A., Galindo, J.C.G., Molino, J.M.G., Cutler, H.G., Eds.; CRC Press: Boca Raton, FL, USA, 2004; pp. 217–238.
- Kumara, R.; Tsvetkovc, D.E.; Varshneyb, V.K.; Nifantievc, N.E. Chemical constituents from temperate and subtropical trees with reference to knotwood. Ind. Crops Prod. 2020, 145, 112077.
- Gupta, K.P.; Singh, P. A naphthoquinone derivative from Tectona grandis Linn. J. Asian Nat. Prod. Res. 2004, 6, 237–240.
- Lukmandaru, G.; Takahashi, K. Radial distribution of quinones in plantation teak (Tectona grandis L.f.). Ann. For. Sci. 2009, 66, 605.
- López, L.I.L.; Flores, S.D.N.; Belmares, S.Y.S.; Galindo, A.S. Naphthoquinones: Biological properties and synthesis of lawsone and derivatives—A structured review. Vitae 2014, 21, 248–258.
- Rosamah, E.; Ferliyanti, F.; Kuspradini, H.; Dungani, R.; Aditiawati, P. Chemical content in two teak woods (Tectona grandis Linn.F.) that has been used for 2 and 60 years. J. Biol. Sci. Technol. Manag. 2020, 2, 15–19.
