Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is an old version of this entry, which may differ significantly from the current revision.
Subjects:
Plant Sciences
Jasmonic acid (JA), a lipid-derived molecule, plays an essential function in controlling many different plant developmental and stress responses.
- phytohormone
- jasmonic acid
- JA signaling
1. Introduction
During growth and development, plants are constantly battling against a challenging environment. These adverse or unfavorable environmental conditions are often categorized as: (i) abiotic stresses, such as ultraviolet (UV) radiation, flood, drought, heat, cold, heavy metal toxicity and nutrient deficiency, and (ii) biotic stresses, such as pathogen infection and animal herbivory [1]. Within plant cells, various signal transduction pathways work collaboratively to convey and integrate stress stimuli, and ultimately orchestrate processes of plant growth, development and defense responses [2,3,4]. Phytohormones are among the most important signaling molecules that are involved in the signaling network that regulates these processes [5,6,7,8,9,10,11,12].
Jasmonic acid (JA) and its metabolic derivatives, such as jasmonic acid isoleucine (JA-Ile) and methyl jasmonate (MeJA), collectively known as jasmonates (JAs), are a class of lipid-derived, natural and widely distributed phytohormones in higher plants. JAs have been studied for decades as key signaling compounds involved in many aspects of plant development and stress responses [9,13,14,15,16,17,18]. Upon stress stimuli, such as wounding, herbivory or necrotrophic pathogen infection, plant cells trigger a rapid increase of JAs, which lead to the activation of defense responses and reproduction, as well as the inhibition of growth for plant fitness [19,20,21,22,23]. Moreover, through the crosstalk network, JAs often work in concert with other phytohormones, such as abscisic acid (ABA), auxin, cytokinin (CK), ethylene (ET), gibberellic acid (GA) and salicylic acid (SA), to balance between growth- and defense-related processes, thereby conferring plants acclimation to the changing environments [10,11,24].
2. JA Signaling
2.1. JA Perception and Signal Transduction
The generally accepted “relief of repression” model for JA perception is built upon decades of research beginning with the identification of the core co-receptor complex for JA-Ile, that is composed of the F-box protein CORONATINE INSENSITIVE 1 (COI1) containing SKP1-CULLIN1-F-box-type (SCF) E3 ubiquitin ligase complex SCFCOI1, JASMONATE ZIM DOMAIN (JAZ) proteins and inositol pentakisphosphate (InsP5) [34,35,36,57,58,59,60].
Under normal conditions, where little or no nuclear JA-Ile is present, certain TFs, such as MYC2 (a basic helix-loop-helix (bHLH) family TF and key activator of JA responses), are repressed by a series of JASMONATE ZIM DOMAIN (JAZ) proteins through direct interaction. MYC2 binds to the G-box motif at the promoter regions of the JA-responsive genes and activates their expression [61] (Figure 2). Most JAZ family members have been shown to interact with MYC2. When binding to MYC2, the JAZ protein recruits the TOPLESS (TPL) and TPL-related (TPR) co-repressors directly or through the adaptor protein NOVEL INTERACTOR OF JAZ (NINJA) to repress the transcriptional activity of MYC2 (Figure 2). The transcriptional repression function of the TPL co-repressors involves the further recruitment of the chromatin modifying HISTONE DEACETYLASE (HDA) complex that “switches off” the targeted region by chromatin condensation [62,63,64]. Members of HDAs such as HDA6 and HDA19 have been shown to participate in JA responses [65,66].
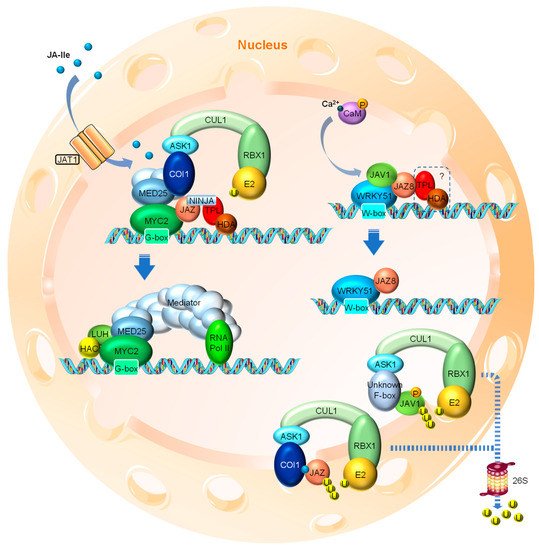
Figure 2. Simplified model of JA signaling in Arabidopsis. When nuclear bioactive JA levels are low, the master transcription factors, such as MYC2, are repressed through the interaction with JAZ proteins that recruit other co-repressors, such as NINJA, TPL and HDA, to form a repressor complex at the promoter regions of JA-responsive genes. In addition, MED25 also physically interacts with MYC2 while bound with COI1, the F-box subunit of the SCFCOI1 E3 ubiquitin ligase complex. In the case of the JA biosynthesis gene AOS, its transcription is repressed by the JJW complex composed of JAV1, JAZ8 and WRKY51. In both cases, the expression of JA-responsive genes is restrained. It is unclear whether the JJW complex also recruits co-repressors, such as TPL and HDA. When a certain developmental or environmental cue triggers the import of bioactive JA (e.g., JA-Ile) into the cell nucleus presumably through the action of JAT1, elevated levels of JA-Ile cause the formation of COI1-JA-JAZ co-receptor complex. The interaction between COI1 and JAZ leads to the dissociation of JAZ and MYC2, as well as the dissociation of COI1 and MED25. As a result, JAZ is degraded via the 26S proteasome and the enhanced interaction between MED25 and MYC2 ultimately leads to MED25-mediated transcriptional activation of the target genes. In the case of JJW-regulated AOS, stress-induced fast Ca2+ influx leads to the CaM-mediated phosphorylation of JAV1. JAV1 phosphorylation causes the disintegration of the JJW complex and AOS transcriptional activation. Phosphorylated JAV1 is subjected to E3 ubiquitin ligase-mediated ubiquitination and 26S proteasomal degradation, although the F-box protein responsible for the specific recognition of JAV1 remains to be identified. JAZ, JASMONATE ZIM DOMAIN; NINJA, NOVEL INTERACTOR OF JAZ; TPL, TOPLESS; HDA, HISTONE DEACETYLASE; MED25, MEDIATOR25; COI1, CORONATINE INSENSITIVE1; ASK1, ARABIDOPSIS SKP1-RELATED1; CUL1, CULLIN1; SCF, SKP1-CULLIN1-F-box; HAC1, HISTONE ACETYLTRANSFERASE1; LUH, LEUNIG_HOMOLOG; JAV1, JASMONATE ASSOCIATED VQ DOMAIN PROTEIN1; AOS, ALLENE OXIDE SYNTHASE; CaM, Calmodulin. RBX1 is a RING finger protein that recruits the E2 ubiquitin-conjugating enzyme to the C-terminus of CUL1.
When certain developmental or environmental cues cause a cellular burst of JA, the active derivative JA-Ile is transferred into the nucleus by JAT1 and promotes the formation of the SCFCOI1-JAZ co-receptor complex, resulting in the ubiquitination of JAZ and its subsequent degradation via the 26S proteasome [13,33,34,35,36,37]. The degradation of JAZ protein thus releases the inhibitory effect on the TFs such as MYC2 (Figure 2), which initiates JA signaling cascades by transcriptional activation of numerous downstream TF genes, such as ETHYLENE RESPONSE FACTOR1 (ERF1) and OCTADECANOID-RESPONSIVE ARABIDOPSIS59 (ORA59), and defense-related responsive genes, such as VEGETATIVE STORAGE PROTEIN2 (VSP2).
Among the 13 Arabidopsis JAZ proteins identified, five (i.e., JAZ5, JAZ6, JAZ7, JAZ8 and JAZ13) contain the ETHYLENE RESPONSIVE FACTOR-associated amphiphilic repression (EAR) motif responsible for the direct interaction of the protein with TPL co-repressors. Therefore, these JAZs can directly recruit TPLs in the absence of NINJA [67,68]. In addition, a few non-canonical JAZs (such as JAZ8 and JAZ13), which harbor a divergent JA-associated (Jas) domain exhibiting little interaction with COI1, can interact with MYC2 while directly recruiting TPLs through their EAR motifs. Therefore, they are considered as adapters linking TPL to other non-EAR-containing JAZ proteins through heterodimerization [16,67,68,69]. Recently, JAZ8 was shown to form a co-repressor complex with JASMONATE-ASSOCIATED VQ DOMAIN PROTEIN1 (JAV1) and WRKY 51 to bind and repress JA biosynthesis genes, highlighting the role of these non-canonical, EAR motif-containing JAZs [70]. Additionally, a protein named EAR-motif-Containing Adaptor Protein (ECAP) has been reported as a novel adaptor protein that directly interacts with JAZ6 and JAZ8 for the recruitment of the TOPLESS-RELATED2 (TPR2) co-repressor to repress JA responses. Genetic evidence shows that ECAP plays an important part in many JA-regulated processes, including anthocyanin accumulation, JA biosynthesis and defense-related gene activation [71]. Intriguingly, both JAZ6 and JAZ8 harbor an EAR motif which is thought to be adequate for TPL and TPR recruitment. The emerging function of ECAP suggests a novel counterpart of NINJA and a more sophisticated mechanism of gene repression.
Over the last decade, another key player in JA signaling, MEDIATOR25 (MED25), has been added to the picture [72,73,74]. MED25 is a subunit of the Mediator transcriptional coactivator complex, an evolutionarily conserved multi-subunit complex that plays an essential role in the RNA Polymerase II (Pol II)-dependent transcription throughout eukaryotes [75,76,77]. MED25 physically interacts with COI1 and MYC2, bringing COI1 in close proximity to JAZ, which binds to and represses MYC2 when no or little nuclear JA-Ile is present. Nuclear JA-Ile acts as “molecular glue” to promote the formation of the SCFCOI1-JAZ co-receptor complex, leading to the weakened interaction between COI1 and MED25, as well as subsequent JAZ degradation. This conformational change also strengthens the interaction between MYC2 and MED25 due to the release of the competitive/interfering effect by JAZ. MED25 then recruits the rest of the Mediator complex and RNA Pol II, as well as other coactivators, such as HISTONE ACETYLTRANSFERASE1 (HAC1) and LEUNIG_HOMOLOG (LUH), to the promoter region of MYC2 target genes for transcriptional activation [74,78,79,80] (Figure 2).
Recently, a WRKY TF and VQ domain protein involved mechanism of herbivory- and wound-induced JA biosynthesis has been reported [70]. The WRKY TFs, a large TF family in plants, have been shown to be crucial in a broad range of developmental and physiological processes, as well as various stress responses [81,82]. The WRKY51 TF forms a heterotrimer complex with JAZ8 and JAV1, a VQ domain protein family member previously identified to specifically modulate JA-regulated plant defense [83]. At resting stage, the JAV1-JAZ8-WRKY51 (JJW) complex represses the expression of JA biosynthesis genes, such as AOS, through binding to the W-box motifs in their promoter regions (Figure 2). The herbivory-caused injury rapidly induces cytosolic Ca2+ influx, leading to the calmodulin (CaM)-dependent phosphorylation of JAV1 and its subsequent degradation. The disintegration of the JJW complex results in the activation of JA biosynthesis gene expression [70] (Figure 2). Intriguingly, the phosphorylation and degradation of JAV1 triggered by Ca2+/CaM is independent of JA-Ile elicitation [70], which seems in contrast to earlier data suggesting that JA triggers the degradation of JAV1 in a COI1-dependent manner [83]. Resolving these conflicting findings clearly needs further exploration. It also remains to be determined whether the JJW-directed mechanism is acting in parallel to the canonical JAZ-regulated mechanisms (e.g., JAZ-MYC) and whether inputs from different upstream signals (Ca2+ vs. JA-Ile) can be simultaneously perceived by the promoters of JA biosynthesis genes (e.g., AOS) that are also directly regulated by MYC2 [84]. Finally, whether MED25 is involved in the JJW regulation needs additional confirmation. Regardless, this finding is undoubtedly a milestone that adds another layer of transcriptional regulation to wound-induced JA biosynthesis, highlighting both the regulatory mechanism of JA biosynthesis and the function of the non-canonical JAZ proteins like JAZ8. As discussed before, JAZ8 is believed to be resistant to COI1-mediated degradation due to its divergent Jas domain. Therefore, no JAZ8 degradation has been reported by Yan et al. [70]. JAZ8 now has exhibited interaction with not only MYC2 but also WRKY51, it is highly likely that JAZ8 also recruits TPL and HDA co-repressors to the JJW complex since the repression of the JJW complex largely depends on the EAR motif of JAZ8.
Other evidence also suggests that WRKY33 and WRKY57 interact with two VQ proteins SIGMA FACTOR BINDING PROTEIN1 (SIB1) and SIB2 while potentially being able to interact with JAZ4 and JAZ8, although further study is needed to elucidate the details of the molecular context [85,86].
2.2. JA-Regulated Transcription Factors
In addition to MYC2 serving as the main transcriptional regulator of JA-induced gene activation, other members of the MYC TF family as well as members of other TF families have also been shown to be directly involved in controlling JA-regulated gene expression (Figure 3). MYC3 and MYC4 are also targets of JAZ repressors (e.g., JAZ3 and JAZ5) and act additively with MYC2 to activate JA response in the vegetative tissue, especially the JA-dependent defense response against wounding and herbivory [87,88]. MYC2, MYC3, MYC4 and MYC5 interact with at least two R2R3-MYB TFs, MYB21 and MYB24, to form a MYC-MYB transcription complex (Figure 3). Both MYC and MYB are repressed by JAZ suppressor and are activated by JA to cooperatively regulate stamen development in Arabidopsis [89,90,91,92]. In rice, data have also shown that the JA-responsive R2R3-type MYB TFs, JAMYB and its homolog, are transcription activators directly regulated by JA [93]. JAMYB binds to the AG-motif-like motif in the promoter region of Argonaute18 (AGO18) gene, which encodes a core RNA silencing component that promotes AGO1-mediated antiviral RNAi [94]. The transactivation activity of JAMYB is normally repressed by JAZ6. The JA accumulation elicited by rice stripe virus coat protein triggers the ubiquitination and proteasomal degradation of JAZ6, relieving the repression of JAMYB to activate the expression of AGO18. Elevated accumulation of AGO18 ultimately leads to enhanced antiviral defense in rice [93,94]. It is reasonable to hypothesize that certain rice MYC homologs also interact with JAMYB.
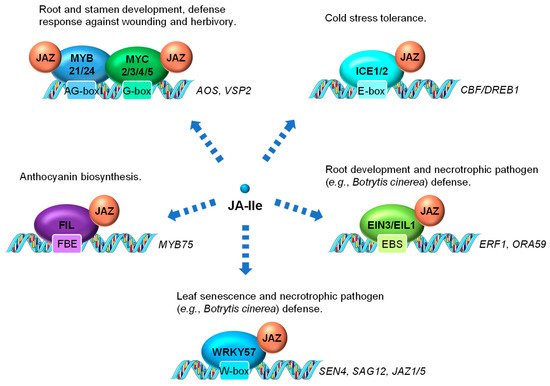
Figure 3. Examples of JA-regulated processes control through the interaction between JAZ and transcription factors (TFs). The MYC TFs, several R2R3-MYB family members (including MYB21 and MYB24), and other TFs (e.g., ICE1, EIN3, EIL1, and FIL) are direct targets of JAZ repressors. These TFs are activated by JA-mediated JAZ degradation and positively regulate JA responses. WRKY57 is also a direct target of JAZ but acts as a negative regulator of JA regulated leaf senescence and defense against necrotrophs. ICE1, INDUCER OF CBF EXPRESSION1; EIN3, ETHYLENE INSENSITIVE3; EIL1, EIN3-LIKE1; FIL, FILAMENTOUS FLOWER; EBS, EIN3 binding site; FBE, FIL DNA binding element.
INDUCER OF CBF EXPRESSION1 (ICE1) and ICE2 are two MYC-like bHLH transcriptional activators playing critical roles in modulating cold stress responses [1,95]. Both are repressed by JAZ repressors (e.g., JAZ1 and JAZ4) through physical interaction [96,97] (Figure 3). Cold stress-induced endogenous JA production triggers the turnover of JAZ repressors and the activation of ICE1 and ICE2, which further activate the C-REPEAT BINDING FACTOR/DRE BINDING FACTOR1 (CBF/DREB1) transcriptional cascade for cold stress tolerance [96,97].
Several members in the APETALA2/ETHYLENE RESPONSE FACTOR (AP2/ERF) TF family, such as ETHYLENE INSENSITIVE3 (EIN3), EIN3-LIKE 1 (EIL1), ERF1 and ORA59 comprise the classical ERF branch of JA signaling, which is marked by the activation of the downstream defense gene PLANT DEFENSIN1.2 (PDF1.2) [98]. Transcription of ERF1 and ORA59 is directly controlled by EIN3 and its homolog EIL1, which are identified as direct targets of JAZ proteins and activated by JA (Figure 3). EIN3 and EIL1 have been shown to play important roles in mediating JA-induced processes, such as root development and defense responses to necrotrophic fungi [66]. EIN3 and EIL1 are two well-recognized TFs essential for the activation of ethylene (ET) responses [99] and thus are likely to be key components necessary for integrating JA and ET signaling.
WRKY57, a WRKY TF involved in both JA-induced leaf senescence and necrotrophic pathogen response in Arabidopsis, is repressed by JAZ4 and JAZ8 through physical interaction [85,86]. WRKY57 directly binds to the promoters of SENESCENCE4 (SEN4) and SENESCENCE-ASSOCIATED GENE12 (SAG12), as well as another two JAZ repressor genes (JAZ1 and JAZ5) to regulate their expression (Figure 3). Intriguingly, genetic evidence shows that the loss-of-function of WRKY57 enhances JA-mediated leaf senescence and resistance against Botrytis cinerea (B. cinerea) infection, suggesting it a negative regulator of JA signaling [85,86].
A YABBY (YAB) TF family member FILAMENTOUS FLOWER (FIL)/YAB1 has been found as a direct target of JAZ3 (Figure 3). Via JA-triggered degradation of JAZ3, FIL/YAB1 promotes anthocyanin biosynthesis through, at least in part, direct transcriptional activation of MYB75 [100], a key component of the WD-repeat/bHLH/MYB transcription complex that is also repressed by several JAZ repressors [101,102].
Whether these JAZ-regulated TFs also share other components of the regulatory mechanism (e.g., MED25, TPL, HAD, and HAC), and if they act synergistically or independently in response to different types of stimuli upstream of JA, are certainly questions for future research.
3.3. Negative Feedbacks and Termination of JA Signal
Since JA is a stress signal that generally leads to growth inhibition, proper desensitization and termination of the JA signal is undoubtedly as important as its activation for overall plant growth and fitness. In fact, the JA signal is elaborately controlled at multiple levels to ensure that each response only lasts for an appropriate period at an appropriate amplitude [30,80,103].
Cytosolic JA-Ile dynamics are shaped by at least two JA-inducible catabolic pathways in Arabidopsis. The first pathway is the direct oxidation of JA-Ile by members of the cytochrome P450 subfamily 94 (CYP94) enzymes, CYP94B1, CYP94B3 and CYP94C1, which turn JA-Ile into bio-inactive 12OH-JA-Ile and 12COOH-JA-Ile [104,105] (Figure 1). The second pathway is the deconjugation of JA-Ile mediated by two amidohydrolases, IAR3 and ILL6, which hydrolyze both JA-Ile and 12OH-JA-Ile [106] (Figure 1). Both pathways have been shown to contribute additively for the turnover of JA-Ile but act differently for JA responses and tolerance to related stress conditions [103,107]. In crop plants (e.g., rice and corn), JA catabolism has also been proven to be crucial to both the development, such as sexual determination [108,109], and stress tolerances, such as salt and cold [110,111].
JA-Ile stimulates rapid activation of JAZ gene expression while most JAZ genes in Arabidopsis can produce truncated JAZ splice variants that can still bind to the MYC proteins but have little capability of forming complexes with JA-Ile and COI1 for proteasomal degradation. Overexpression of certain JAZ splice variants, such as JAZ10, result in dominant repression of JA responses [112,113]. Crystal structure reveals that the JAZ10 splice variant tightly binds to MYC3 and blocks the interaction between MYC3 and MED25, which is crucial for the transcriptional activation of MYC3 target genes [114]. These findings indicate that the alternative splicing of JAZ genes serve as a general feedback mechanism to desensitize JA signaling. Intriguingly, JA also induces the recruitment of two splicing factors, PRE-mRNA-PROCESSING PROTEIN 39a (PRP39a) and PRP40a to JAZ loci by MED25. PRP39a and PRP40a, in turn, facilitate the full splicing of JAZ transcripts to produce the full-length JAZ proteins, thus preventing the excessive desensitization of JA responses caused by the overaccumulation of JAZ splice variants [115]. These data suggest that the JA-induced negative feedback mechanism by the alternative splicing of JAZ genes is under exquisite modulation.
Several bHLH family subgroup IIId members (e.g., the JASMONATE-ASSOCIATED MYC2-LIKE proteins (JAMs) in Arabidopsis and the MYC2-TARGETED BHLHs (MTBs) in tomato) have been identified as negative regulators of JA responses [116,117,118,119]. In Arabidopsis, JAM1/bHLH17, JAM2/bHLH13 and JAM3/bHLH3 interact with JAZs and function as transcriptional repressors by competing with MYCs for G-box binding [116,117,118]. Likewise, the tomato MTB1, MTB2 and MTB3 are activated by MYC2 but act in turn to negatively regulate JA responses by competing with MYC2 for the target promoter binding site (i.e., the G-box motif), impeding the formation of the MYC2-MED25 complex [119]. In addition to the activation of JAMs or MTBs, JA also stabilizes BTB/POZ-MATH3 (BPM3), one of the BPM proteins that function as adaptors of Cullin3-based E3 ubiquitin ligases [120]. Several BPMs are found to directly target MYC2, MYC3 and MYC4 for polyubiquitination and degradation. Thus, the stabilities of JA-activated MYCs are negatively controlled by BPMs, especially BPM3, whose stability is greatly enhanced by JA [120].
Taken together, these groundbreaking discoveries suggest that plant cells orchestrate a complex and autoregulatory negative feedback circuit to desensitize and terminate JA signals at multiple layers.
This entry is adapted from the peer-reviewed paper 10.3390/ijms22062914
This entry is offline, you can click here to edit this entry!
