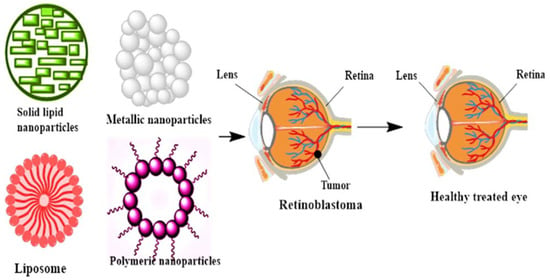
1. Background
Retinoblastoma (RB) is an aggressive ophthalmological cancer found during childhood and infancy but an uncommon malignancy of older children and young adults, with a worldwide prevalence of 1/15,000 to 1/20,000 live births [
1]. Strabismus and leukocoria are two major signs of RB [
2]. If not treated in the early stages, it often causes devastating consequences, such as the loss of vision, secondary nonocular tumors, and even death [
3,
4]. As a highly malignant tumor, RB usually manifests in the first three years of life and represents the prototypic pattern for inherited tumors, with the tumor initiated by the somatic inactivation of both alleles of the RB gene (
RB1) [
5,
6]. RB tumors, deriving from the immature cells of the retina, have a large amount of heterogeneous vasculature and depend on the vascular supply for their proliferation [
7].
The traditional treatments of children with RB used to be external beam radiotherapy, episcleral plaque radiotherapy, enucleation, cryotherapy, and photocoagulation [
8]. Over the last decade, RB treatment has changed enormously. In this respect, with changing attitudes towards concerns over radiotherapy, intravenous and intra-arterial chemotherapies became the cornerstone of RB treatment, since they have been shown to effectively decrease the tumor’s size, prevent the spread of the disease, and preserve vision [
9]. Yet, their clinical application is limited due to possible systematic toxicity, drug resistance, and rapid blood clearance [
10]. New therapeutic strategies, which are more intensive and demand an integrative approach, were put into practice to avoid deleterious complications of the aforementioned modalities, including cataracts, radiation retinopathies, and facial deformities [
3,
11].
The conventional administration of chemotherapeutic drugs decreases their clinical efficacy, particularly for water-insoluble drugs and for delivering such medications to the eye’s posterior segment [
12,
13,
14,
15,
16,
17,
18]. In order to overcome this therapeutic obstacle, the local delivery of these drugs—explicitly, alkylating agents—to the eye has gained much attention as a beneficial strategy for minimizing systematic complications, such as ischemic necrosis, orbital fat necrosis, atrophy of the optic nerve, and changes in ocular motility [
19,
20]. These unfavorable effects are probably caused by prompt dispersion of the aqueous solution of alkylating drugs to the orbital, periorbital, and surrounding tissues [
21]. However, the delivery of eye drugs continues to pose a serious challenge due to the clearance of conjunctival vessels. Besides, the existence of physiological and anatomical barriers of the eye, comprised of ocular surface epithelium, blood–retina barriers, and blood–aqueous barriers, might limit its efficiency [
12]. From this perspective, the establishment of productive delivery transporters is therefore essential for RB therapy.
In terms of the diagnosis, clinicians routinely confirm RB by the appearance of retinal tumors using needle biopsy, fluorescein angiography, commutated tomography and ultrasonography, and magnetic resonance imaging (MRI) [
6]. Despite the availability of these ophthalmic imaging modalities, there is still an urgent need, particularly for ocular molecular imaging, that further enables the early detection of eye disorders prior to the appearance of grossly visible morphological alterations [
22]. In those cases, ophthalmic tumors are typically diagnosed when they can no longer be regarded as malignant cells but malignant tissue. On the other hand, biopsy sampling can provide us with invaluable information concerning the histological type of the ophthalmic tumor, and sampling errors might cause falsely negative specimens [
23].
Recently, the use of nanotechnologies has been experiencing exponential growth in the diagnosis and treatment of tumors and eye disorders. Nanoparticles (NPs), nanocages, nanocapsules, nanoliposomes, nanohydrogels, nanodendrimers, and nanomicelles are amongst the most applicable nanotechnology-based ocular delivery systems providing several advantages over routine diagnostics/therapies [
24,
25,
26,
27,
28,
29,
30]. Owing to their unique characteristics and potential applications in medicine and biology, nanomaterials (NMs) were developed to revolutionize disease diagnoses, treatments, and therapies [
31,
32,
33,
34,
35,
36]. Currently, NPs are extensively used for the effective delivery of drugs, small molecules, peptides, nucleic acids, and even vaccines [
37]. Thanks to their controlled release, nanometer-scale dimensions, and desirable therapeutic toxicity, NPs yield promising outcomes even at very low concentrations and have fewer side effects than traditional chemotherapeutics [
38,
39]. This makes NPs an object of even broader interest for being used in implants, NP-contained contact lenses, films, nanofabricated devices, and designed nanocarriers for ocular drug delivery [
39].
Different NPs can be administrated into the eye by different routes, including topical, periocular, systemic, intravitreal, and suprachoroidal. In this regard, suprachoroidal, intravitreal, and periocular administration is recommended for either slow-release NPs or stimuli-responsive NPs. Simultaneously, bioadhesive or rapid uptake NPs are better for being topically injected [
40]. Functionalizing NPs with peptides and protein ligands, such as transferrin, could facilitate their conjunctival entry and transport [
41]. Another example is applying magnetic NPs loaded with a drug payload to enhance the cellular uptake of the payload [
42]. These functionalized NPs can be utilized in ocular delivery systems and serve as novel promising contrast agents for MRIs [
40]. Furthermore, NPs provide many advantages for delivering nucleic acids (i.e., RNA, short interfering RNS, and microRNAs) [
43]. The advantages of gene transfection using NPs include (i) enhancement of the cell entry of nucleic acids, (ii) protection of nucleic acids from degradation by nucleases in the body and increasing the duration of gene transfection, (iii) avoiding nucleic acids to bind to specific cell surface receptors and reduce their off-target effects [
44,
45]. Therefore, functionalized NPs could be regarded as desirable tools for retinal gene therapy and nucleic acid delivery for treating other ophthalmic pathologies—specifically, RB [
46,
47].
Nanoparticle drug delivery is a novel approach to the treatment of eye disorders. Still, there are several challenges in using NPs for such purposes [
48,
49,
50,
51,
52,
53,
54,
55,
56]. For one thing, as a drug or gene carrier, NPs do not affect the retina and cornea [
12]. Moreover, for using some NPs, long incubation periods are needed [
57], and NPs might release insoluble particles and factors that interact with biological systems [
58].
2. The Role of Nanotechnology in the Diagnosis of Retinoblastoma
The risk of intraocular cancer problems and metastatic potential needs to be controlled in the same way as other cancers [
9]. The early detection of intraocular cancer is important for maintaining vision because of its proximity to vital ocular tissues [
59]. There are two types of intraocular tumors that can be classified according to the average age of incidence: childhood RB and adult ocular melanoma [
60].
In children under five years of age, RB, often an inherited disease, occurs and is induced by down regulation of the RB gene. In developing countries, the prevalence of this type of intraocular cancer is higher. The silencing of this gene eliminates the cell cycle regulation restrictions that lead to the uncontrolled proliferation of cells [
61,
62]. Ocular inflammation due to extra ocular colonization of the tumor can be seen in severe instances. RB can spread into the pleural cavity and to the brain and spinal cord. The choroid vasculature may also be infiltrated and distributed to the bone and stem cells [
1].
An RB diagnosis is usually performed by an ophthalmologist’s evaluation and imaging of the eye. Fundoscopy identification usually reveals a large white-to-creamy-colored tumor with retinal and vitreous space lesions. Ultrasonography is used to classify and evaluate intraocular tumors, because CT scans are not advised for young kids [
63,
64,
65]. Additionally, the extra ocular extension of the tumor is studied through magnetic resonance imaging (MRI) of the brain and the orbits [
66]. Traditional optical imaging and ultrasound imaging, considering the flexibility of the ophthalmic imaging methods, are not effective at identifying the early pathologies of eye diseases until morphological changes are apparent.
On the other hand, in order to improve health outcomes, several diagnostic methods have been developed. However, when using the existing techniques, there are restrictions and limitations [
67]. Additionally, several studies have recently established new RB biomarkers that can be used as prognostic factors for diagnosis and can contribute to the understanding of RB pathogenesis and help address potential treatments and diagnosis approaches [
68].
The early detection of RB is a key feature in successful treatment [
69]. Nanotechnology offers new molecular contrasting agents and nanomaterials for earlier and more reliable initial detection and continuous monitoring of the treatment of cancer patients [
15,
16,
25,
27,
70]. Recently, several nanoplatforms have been developed to enhance the image quality of the traditional imaging techniques (See ) [
49,
50,
54,
71]. Amidst this progress, inadequate studies have been conducted to enhance the performance of traditional ocular imaging techniques such as MRI, ultrasound imaging, and optical coherence tomography through the use of nanoplatforms [
72]. Despite inadequate research efforts in this area, these nanoplatforms have demonstrated tremendous potential for enhancing the imaging and diagnosis quality of retinal diseases.
Table 1. Summary of several nanostructures in the diagnosis of retinoblastoma (RB). [NPs: nanoparticles, QDs: quantum dots, MT: metallothionein, PT-OCT: photothermal optical coherence tomography, and Au: gold].
Quantum dots (QDs) have been evaluated for their applications in ocular imagery. They have excellent optical durability and can make multimodal detection easier [
80,
81]. The injection of cultured human corneal endothelial cells (cHCECs) into the anterior chamber is a newly developed modality for the successful treatment of corneal endothelium dysfunction. For instance, to monitor injected cHCECs, Toda et al. investigated cultured human corneal endothelial cells (cHCECs) labeled by semiconductor QDs. They explored the efficacy of in vivo fluorescence imaging in a corneal endothelial dysfunction mouse model to study the dynamics and aggregation of QD-labeled injected cHCECs [
77]. In this study, no morphological alteration in the cHCECs or the expression of functional markers of cHCECs were induced by QD-labeling. The injected cHCECs QDs were quantified. The retention of cHCECs QDs was obvious in the cryogenically injured corneal endothelium mouse model eyes from 3 to 48 h post-cell injection on the posterior surface but not in the non-injured healthy control eyes. QDs may be good contrast agents if the toxicity of the dots is considered. Some researchers have proposed AuNPs as an alternative.
AuNPs can act as perfect contrast agents for imaging, in addition to QDs, and several authors have used AuNPs for imaging eye cancers over the past few years [
82]. For example, Cruz-Alonso et al. described an immunohistochemical approach for visualizing the distribution of metallothionein 3 (MT3) and metallothionein 1/2 (MT 1/2) in human ocular tissue [
74]. In this methodology, Au nanocluster (AuNC)-connected antibodies are used as markers and can be coupled with ICP-MS. Water-soluble fluorescent AuNCs with an average size of 2.7 nm were prepared by carbodiimide coupling and then covalently linked to antibodies. To prevent nonspecific contact with biological tissue, the surfaces of the modified AuNCs were then blocked with hydroxylamine. The signal enhancement by >500 Au atoms in each nanocluster enabled LA-ICP-MS to identify the antigens (MT 1/2 and MT 3) using a laser spot size as small as 4 μm. In this study, the picture patterns found in the retina were in good agreement with those obtained by the traditional immunohistochemistry of fluorescence.
In another study, for the first time, Lapierre-Landry et al. examined in vivo photothermal optical coherence tomography (PT-OCT) in the eye for endogenous (melanin) and exogenous (Au nanorods) absorbers [
75]. In retinal imaging, OCT has become a quality of treatment. OCT facilitates noninvasive tissue architecture mapping but lacks the specificity of contrasting agents that could be used for in vivo molecular imaging. PT-OCT is a practical technique based on OCT that was produced in a sample to identify the absorbers. To separate the photothermal signal from melanin in the retina, pigmented mice and albino mice were used. After the systemic injection of Au nanorods to investigate their passive aggregation in the retina, pigmented mice with laser-induced choroidal neovascularization lesions were also visualized. The current research has demonstrated the capacity of combining the PT-OCT method with Au nanorods to image the distribution of both endogenous and exogenous absorbers in mouse eyes.
In another study, Kim et al. showed the medicinal use of fucoidan-coated Au NPs and those encapsulated by doxorubicin (DOX) for the in vivo and in vitro dual photothermal therapy (PTT) and chemotherapy of eye tumors [
73]. Marine-derived fucoidan was used to obtain a higher photostability for AuNPs as a capping agent, and DOX was loaded to stimulate a chemotherapy anticancer drug. The prepared DOX-fucoidan@AuNPs demonstrated high tumor cell cytotoxicity and good light absorption for in vitro temperature rises. Following an intratumoral injection of DOX-fucoidan@AuNPs into rabbit eye tumors, PTT-assisted NPs resulted in the complete and nonrecurrent elimination of eye tumors for 14 days after the procedure. Due to responsive light absorption by the administered NPs, the photoacoustic image contrast from the tumor tissues was improved dramatically. Interestingly, the use of marine-derived fucoidan, along with AuNPs, can improve the ability of AuNPs for better photothermal therapy.
Altundal et al. also explored the dosimetric possibility of using AuNPs or carboplatin-loaded AuNPs to increase the effectiveness of radiotherapy for ocular cancers (choroidal melanoma) and RB during kV energy external and internal beam radiotherapy [
83]. The data predicted that using AuNPs or carboplatin-loaded AuNPs combined with radiation therapy for ocular cancer utilizing kV energy photon beams could achieve major dose improvements. In the kV energy range, brachytherapy sources produce higher dose improvements than an external beam. The external beam, however, has the benefit of being noninvasive.
The effectiveness of brachytherapy with ultrasonic hyperthermia modality in the existence of AuNPs on ocular RB tumors was tested by Moradi et al. in arabbit model [
84]. The tumor area was assessed at day zero and the end of the third week using a B-mode ultrasound imaging approach. For a histopathological analysis of the tumor necrosis, the groups were investigated. A high difference between the relative tumor area changes in the combination group and the other study groups was observed. The necrosis of living RB cells was supported by the findings of the histological examination. Once again, Au NPs demonstrated a high ability in different imaging techniques, such as ultrasounds. Therefore, AuNPs can be a better alternative to quantum dots.
Due to their structural differences and their wide range of functionally based electrical and chemical characteristics, carbon nanomaterials have received much interest these days [
85]. Researchers have concentrated on electroanalysis using carbon materials for biomolecules, because electrochemical techniques give the benefits of flexibility and responsiveness in constructing a sensor design [
86]. Goto et al., for example, described the direct electrochemical identification of DNA methylation using a nanocarbon film electrode in relatively long sequences. The film was developed using the sputtering method of electron cyclotron resonance and had a mixed bond structure of nanocrystalline sp(2) and sp(3) [
78]. Their strategy of methylation identification calculated the variations between both the 5-methylcytosine and cytosine oxidation currents without a bisulfite reaction or labeling. The film electrode enabled the quantitative identification of DNA methylation ratios under optimized conditions and sensor measure methylated 5’-cytosine-phosphoguanosine (CpG) repetition oligonucleotides (60 mers) with different methylation ratios. Despite the high ability and low toxicity of carbon nanostructures, there are a few papers for the diagnosis of RB.
In an in vitro setting, magnetic NPs that can provide great contrast for MRIs have, so far, been effective [
87]. Previous studies have shown that human serum albumin-coated iron oxide (IO) NPs (HSA and IO/HSA NPs) increase the half-lives of cross-linked therapeutic factors, implying that they can be used for the controlled delivery of therapeutics [
88]. To follow other applications, Tzameret et al. evaluated the in vivo monitoring by MRI and the long-term protection of IO/HSA NP delivery into the suprachoroid of a rat retinalmodel [
88]. Jaidev et al. synthesized NPs of fluorescent iron oxide in another study and tested their effectiveness against RB cell imaging [
76]. Using oleic acid, the iron oxide NPs were prepared and stabilized. Sulforhodamine B was adsorbed onto albumin over NPs of oleic acid-capped iron oxide. In MRI studies, the nanomaterials exhibit a great negative contrast to natural, as well as cancer, cells without cytotoxicity, suggesting their bioavailability. Until now, iron oxide (IO) NPs have been the most used NPs in MRIs. The coating process can decrease some of the stability and toxicity issues.
The combination of nanotechnological strategies in cancer imaging makes it important for their application in ocular diagnostics. Multi-functional nanostructures enable the intraocular tumor responses to different localized chemotherapeutic drugs to be monitored simultaneously in the eye. In this light, a multi-functional nanostructure for multimodal low-intensity centered ultrasound (LIFU)/immune synergistic RB therapy driven by imagery was reported by Wang et al. In order to encapsulate perfluoropentane (PFP) and muramyl dipeptide (MDP), magnetic hollow mesoporous Au nanocages (AuNCs) connected to Fe3O4 NPs(AuNCs-Fe3O4) were prepared. The multi-functional magnetic NPs improved the in vivo and in vitro photoacoustic, ultrasound, and magnetic resonance imaging, which was effective for the treatment and efficacy imaging. Upon accumulation in tumors via a magnetic field, the NPs underwent phase transition under LIFU irradiation, and MDP was released. AuNCs-Fe3O4/MDP/PFP strengthened LIFU’s therapeutic effect and led to direct tumor apoptosis/necrosis, while MDP facilitated dendritic cell (DC) maturation and activation and allowed DCs to recognize and clear tumor cells. The multi-functional AuNC-Fe3O4/MDP/PFP NPs showed great potential for multimodal imaging-guided LIFU/immune synergistic therapy of RB by improving photoacoustic, ultrasound, and magnetic resonance imaging and inhibiting tumor development.
In the efficient diagnosis of RB, we believe that the combination of different NPs with different abilities can be the best approach.
5. Lipid Nanoparticles (LNPs)
Lipid nanoparticles (LNPs) are valuable aspects of nanotechnology, being utilized in pharmaceutics and nutraceuticals, as well as cosmetics. Most lipid-based bioactive compounds, i.e., fatty acids, flavonoids, tocopherols, polyphenols, carotenoids, and preservatives, possess a hydrophobic nature [
115]. The encapsulation of all these mentioned lipids in the form of colloidal dispersions in the aqueous environment of the oil-in-water (o/w) type is an utmost requirement to ensure the stability of the formulations [
116]. LNPs have gained much importance in treating cancers and infectious diseases, as well as the adsorption of heavy metals. Melphalan is the drug of choice as a chemotherapeutic agent for treating RB. However, the risk of immunogenicity and devastating healthy cells is unavoidable [
117]. To overcome the disadvantages and to ensure ideal delivery and treatment, Tabatabaei et al. (2019) developed 171-nm switchable LNPs for the codelivery of melphalan and miR-181 with 93% encapsulation efficiency [
106]. To prepare melphalan-loaded LNPs (LNP/melphalan), a melphalan and ethanol mixture was added into the lipid mixture to form 10% of the total lipids. Ethanol was then evaporated to form a thin lipid film and again hydrated with 5% dextrose in water for 30 min at 40 °C followed by incubation to develop the LNPs. Next, melphalan was quantified, and the encapsulation of miR-181a was executed. The encapsulation efficiency of miR-181 was determined indirectly via the fluorescence displacement assay. Various characterization techniques have been utilized to assess formulated NPs, and the results showed that LNPs increased the expression of apoptotic genes and the highest uptake and targeted killing of RB cells [
106].
5.1. Solid Lipid Nanoparticles (SLNs)
SLNs are versatile lipid-based nanocarriers systems enriched with the synergistic qualities of polymeric particles, liposomes, and emulsions. SLNs are synthesized from solid lipid blends via containing lipid droplets that are crystalline in a highly ordered structure and composed of bioactive compounds in the lipid matrix part. The bioactive compounds mobility can be controlled via controlling the physical state of the SLN lipid matrix. The advantages related to SLNs include controlled drug release, drug targeting, encapsulation efficiency, and drug stability [
118,
119,
120]. Ahmad et al. (2019) synthesized SLNs for the safe and targeted delivery of etoposide against RB [
121]. SLNs were synthesized via the techniques of melt-emulsification and ultrasonication. Optimization of the novel SLNs was done through a three-factor levels Box-Behnken design for establishing the functional relationship between the response variables of the particle size, surface morphology, and entrapment efficiency (EE). Moreover, the SLNs were characterized for size, surface morphology, entrapment efficiency, and in-vitro drug release. However, pharmacokinetic studies were carried out after the intravitreal administration of SLN formulation in Wister rats. Furthermore, a gamma scintigraphic analysis was performed to check the deposition of SLNs in the ocular tissues of albino rabbits. Gamma scintigraphy involves the injection of radioisotopes (called radiopharmaceuticals) into the bloodstream that actively seek out bone that is irritated or destroyed or rebuilt or tissues that are inflamed or necrotic. Later on, histological studies were performed to assess the toxicity and morphological changes after treatment. However, it was concluded from the results that the particle size, PDI, and EE of the optimized formulation were 239.43 nm, 0.261 ± 0.001, and 80.96% ± 2.21%, respectively. The most advantageous aspect of this formulation was its sustained drug release for seven days with only a single intravitreal administration. The sustained drug release for seven days was also confirmed and supported by the results of the gamma scintigraphy study. The histological studies confirmed the nontoxic nature of the SLNs, as the posterior tissues of eyes did not exhibit detrimental effects. Therefore, it can be obvious that etoposide-loaded SLNs are efficacious and safe in treating RB [
121].
5.2. Nanoliposomes
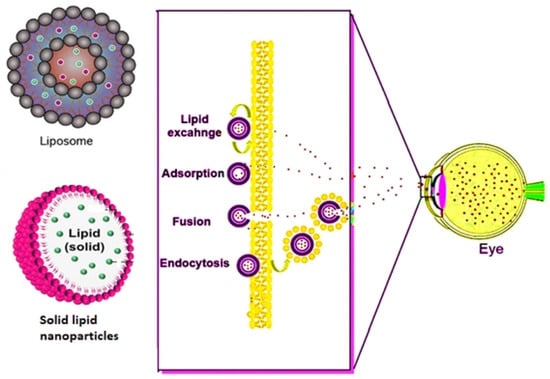
Lipids, when placed in contact with water, the hydrophobic system of the molecule interacts with water, leading to the self-assembly of lipids via forming liposomes () [
50]. Liposomes consist of an aqueous core encapsulated by a lipid bilayer and often functionalize via ligand attachments [
122,
123]. Zhao et al. (2020) synthesized cisplatin nanoliposomes to determine the apoptosis regarding the RB cell lines in vitro, as well as in vivo [
124]. Y-79 cells were cultured, and their exposure with Annexin V/propidium iodide (PI) was tested for determining apoptosis. In order to detected cell death, Annexin V/PI double staining kit are used in flow cytofluorimetric analyses. The Annexin V corresponding signal provides a very sensitive method for detecting cellular apoptosis, while propidium iodide (PI) is used to detect necrotic or late apoptotic cells, characterized by the loss of the integrity of the plasma and nuclear membranes. Y-79 cells were also evaluated for the determination of caspase-3 in order to assess any change in inflammatory caspase-3 as well, as it was also determined based on Western blotting for testing various expressions of Bcl-2 and Bax expression proteins. The Y-79-transplanted tumor model in nude mice was done and divided into three groups (
n = 5). The control group of nude mice was injected with cisplatin, and the blank group of mice was administered with saline. After injecting, the nude mice were slaughtered, and the tumors were removed. After removing of the tumors, the total volumes and weights of the tumors were compared. Furthermore, nucleic acid extraction was done with magnetic beads for the extraction of DNA and RT-PCR, and an in-situ cell death assay kit was applied in testing the apoptotic cells. Furthermore, after comparing the reduction rate of the tumors, the cisplatin liposome group showed a higher Y-79 apoptotic rate, caspase-3, lower volume and weight of the tumors, and Bax protein expression as compared to the cisplatin solution and dimethyl sulfoxide (DMSO) groups, with a significance of
p < 0.05 [
124].
Figure 3. Mechanism followed by lipid and polymeric nanocarriers in overcoming the ophthalmic barrier.
In this research, researchers utilized LNPs to treat RB, and they discovered that LNPs are the most promising for encapsulating hydrophobic drugs via improving the oral bioavailability. Various LNP (SLNs, liposomes, and core-shell nanostructures)-based techniques have been utilized in the past for the successful loading of anticancer drugs and resulted in several limitations of limited drug loading, instability, high cost, poor industrial scaling, and the use of organic solvents. In our opinion, to mitigate the side effects of various lipid formulations, self-emulsifying carriers should be introduced in the targeted killing of RB. Self-emulsifying drug delivery systems (SEDDS) have drawn innumerable attention in the field of pharmaceutical technology and drug development owing to their thermodynamic and kinetic stability and easy manufacturing, as well as a distinct feature of solubilizing both hydrophilic and hydrophobic drugs.