In recent studies, the issue of limited publication has been identified in determining the rate of NP migration from food packaging to food and beverage (A). Metals usually migrate when foods come in contact with packaging materials. The phenomenon of migration comprises three stages: migrant (NP) diffusion, dissolution, and dispersion in food. Choi and his colleagues reported that even babies’ health can be negatively affected by the migration of embedded NPs, such as AgNPs from baby products (B) [
60]. Understanding NP migration is essential to assess the potential health impact of these substances in contact with foods [
61]. In particular, the degree of NP migration may be affected by the inherent effects of physicochemical properties of other components in foods [
62]. Many studies on the migration of NPs have avoided addressing the problem of analyzing them in real food matrices; only a few methods have been employed to assess the mass transfer process and estimate the specific and overall migration of substances into food simulants.
The migration of materials from packaging to food is highly complex and is expressed by two mechanisms of diffusion and adsorption. Emission is the transfer of matter by the random motion of molecules from a high-concentration region to a low-concentration region until equilibrium is reached. The degree of influence according to Fick’s second law is equal to the following relation.
where
CP is the concentration of the migrating material in the polymeric material,
D is the diffusion coefficient,
t is the penetration duration,
x is the distance between the package and the food. Here, “
D” as the most significant factor influences the migration rate. The diffusion process lies in several factors including food contact, packaging material, the nature of foods, storage time, and storage conditions. In addition, it was reported that by opening the packaging, the rate of diffusion will alter [
63].
3.1. Migration Experiments
The European Commission (EC) has established regulations and restrictions specifying the conditions in which migration tests are mandatory. It also proposed several food simulants, including water, acetic acid (3% v/v), ethanol (10% v/v and 50% v/v), and vegetable oil based on the food’s type in contact with the packaging. Thus, most migration studies employing such simulants and evaluation of the NP migration into actual foods are lacking.
Efforts to promote instrumentation and strategies through the analysis of foods experienced a lack of analytical tools. This field will entail more investigation. Regulatory authorities can cause limitations on NP usage in foods. The invention of instruments, which includes an apparatus to assess engineered NPs, has been deemed mandatory. It is important to be able to perform NP identification, size measurement, and its distribution in ways that must be noninvasive and unrestrictive [
65]. In this approach, a substrate covered by NPs is considered as a reference material, and it is used in combination with analytical and imaging systems that focus on screening techniques such as mass spectroscopy (test material composition), dynamic light scattering (DLS; particle size distribution), positron emission tomography (radio-tracing), spectroscopy (NP size), and optical emission spectrometry (trace level elemental analysis) for monitoring and collecting adequate information. Another technique for food evaluation in the viewpoint of harmful substances employs a sensor-based system that included NPs in solution or nanostructured surfaces that are able to respond to changes in the adsorbate electronic states (chemical signal enhancement) and changes in metal roughness (electromagnetic signal enhancement), where the chemisorption of the analysis at the NP surface of the biosensor is the main mechanism [
66]. In another technique, a system to detect food safety named Raman Nano Chip™ [
67] utilizes nanorods to adsorb or collect test samples under the adsorption of molecules onto the surface of the nanostructure. For more utility and applicability, sensors may be covered or coated with NPs in the form of a colloidal suspension of polymeric, metallic, or oxide NPs.
To measure nanomaterials in complex matrices, analysis techniques must make an explicit difference between NPs and other components in the matrix, and the employed methods must be sensitive enough to monitor the matters in low concentrations [
68]. On the other hand, a single analysis cannot guarantee the determination of NP migration in all foods and food simulants, at all temperatures, and therefore it is important to utilize several tests to obtain accurate results. shows a brief report of the test methods used to study different aspects of nanobased systems, including food packaging (as discussed by dos Santos [
69]). These methods also provide sufficient information about the composition, concentration, and physicochemical properties of the nanomaterials in the sample; however, it is not possible to determine the true amount of NPs in the food texture. According to the complexity of the networks, combined methods may give more reliable results in quantifying and identifying NPs, whereas independent methods are likely unable to determine all the information [
70]. During the engineering of novel FPSs, migration testing is one of the main steps. Tracing NPs and the validation of their responsibilities are important for FPS designing.
Table 1. A list of common tests employed in food packaging studies.
Common chromatographic methods for polymer additives are limited and unsuitable because they cannot measure the physicochemical properties of NPs. Therefore, only a limited number of methods work efficiently for detecting and characterizing NPs.
High resolution imaging such as electron microscopy (EM) is one of the most suitable methods for identifying and imaging the shape and structure as well as the size and density of NPs in the food matrix, including scanning electron microscope (SEM), transmission electron microscope (TEM), and atomic force microscopy (AFM) (A). Particle count is very time-consuming, and since these methods have destructive effects on the samples, the sample cannot be re-evaluated by other methods [
71]. The use of TEM is also appropriate when the matrix is made of polymers, but in case of other methods, it is necessary to detect migration in complex environments like food or quasi-food [
72].
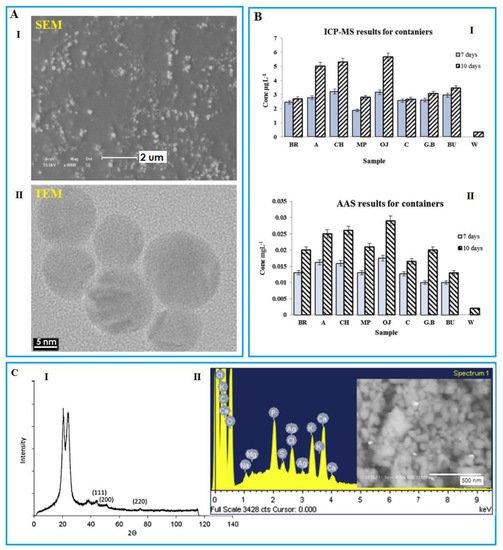
Figure 4. (
A) I: SEM illustration of the AgNPs synthesized by the modified polysaccharide. Reprinted with permission from [
82]. Copyright 2017 Elsevier; II: TEM image shows the migration of AgNPs detected from an orange juice sample under storage by a nano-silver coated film packaging at 40 °C for 10 days. Reprinted with permission from [
81]. Copyright 2015 Elsevier. (
B) I: Inductively coupled plasma mass spectrometry (ICP-MS); II: Atomic absorption determination of Ag from different food samples stored using nanosilver containers for 7 and 10 days at 40 °C. (
C) I: XRD spectrum of nylon fabric treated by copper NPs. Reprinted with permission from [
57]; II: EDS and SEM images indicate silver NPs as white spots detected inside the milk powder stored using nanosilver coated film packaging for 10 days at 40 °C. Reprinted with permission from [
81]. Copyright 2015 Elsevier.
Inductively coupled plasma with mass spectrometry (ICP-MS), atomic emission (ICP-AES), and optical emission spectroscopy (ICP-OES) as quantitative analysis methods are among the most important and practical methods for the determination of metal NPs (B-I). ICP-MS is employed to determine the quantification and elemental composition characterization of NPs in a single but fast analysis, i.e., SP-ICP-MS (SP stands for single particle). The reason is its high sensitivity and potential for identifying and quantifying metals and its better selectivity than other methods in this category [
73]. ICP-MS showed that it provides more reliable results when in incorporation with other techniques. Laser ablation (LA) ICP-MS is used for the qualitative determination of Ag content [
74]. According to the results, the regulation limits for both Ag and Cu were defined as <0.08 mg/kg and <4 mg/kg, respectively, while the limits of determination in ICP-MS were 0.03 mg/kg and 0.01 mg/kg, respectively [
75]. Atomic absorption spectroscopy (AAS) is an alternative with a higher speed and sensitivity than ICP methods, but this method is not suitable for multielement analysis (B-II).
Spectroscopic methods including X-ray diffraction (XRD) give information such as the elemental composition or aggregation and structure of nanomaterials (C-I) [
76,
77]. These methods have a wide range of applications due to their nondestructive nature. Spectroscopy is used as a backup method due to its low cost and ease. Energy dispersive X-ray spectroscopy (noted as EDS/EDX) is known as a chemical microanalysis technique employed in conjunction with SEM, conducted to identify the component, size, and shape of NPs (C-II).
NP tracking analysis (NTA) and dynamic light scattering (DLS) use the properties of both Brownian motion and light scattering in order to measure the NP size distribution of the samples in suspension. The technique is known as a nondestructive test, and if required, the sample can be recovered [
78]. DLS has been widely employed in studying the size distribution of NPs in colloidal suspensions in the nano- or submicrometer ranges before embedding the NPs in films or coating. The suspension concentration, particle shape, colloidal stability, and surface coating of NPs influence the size value obtained by DLS measurements. DLS is much more sensitive to the presence of aggregates.
The titration method has a higher sensitivity in terms of measuring the amount of NPs released. This method also provides more accurate information than the migration method and allows for the determination of the number of NPs released from the coating of polymer packages that were exposed to heat [
79]. The migration method is an old method and is used for other coatings, while according to the US National Standards Organization EN-1186 and 11737-3, this method is the best option for nylon, polystyrene, PVC, and poly ethylene terephthalate (PET), and it is unsuitable for NP coatings. However, the titration method is an up-to-date approach and is a commonly used method all over the world [
80].
Using ICP-MS and AAS, commercial nanocomposite food packaging in the form of nanosilver coated films and containers were evaluated in real-food matrices to measure the degree of NPs incorporation [
81]. Migration testing was done for a range of food materials packaged in various packaging materials at 40 °C for ten days. Water and 3% acetic acid were also used for identifying the factors that have an impact on migration.
3.2. Cu/Ag NPs Migration
As the use of NPs in various packages may have adverse effects on animal and human cells, legislators have established rules to measure and study the migration of NPs to food (). The diffusion, dissolution, and abrasion of the packaging surface can be considered as reasons for the migration of nanomaterials from packaging to food [
83]. Scientists have suggested that AgNPs are mandatory for achieving antimicrobial activity by using AgNP in reinforced thermoplastic polymers, such as polyethylene (PE), polypropylene (PP), polystyrene (PS), or nylon [
84].
Table 2. Applications of Ag/Cu NP systems in food packaging, their effect under study, and analytical methods for their determination.
Although the benefits of nanotechnology have led to the use of industry and the public to make greater usage of nanobased materials, careful research is needed to determine the likelihood of NPs migrating from any concentration to packaged materials, as toxic effects for high concentrations of nanomaterials were proven. Research shows that there are migrants such as silver and copper [
85]. Due to the expensive instruments, difficult analysis, and food matrix disturbances, few studies have been carried out on NP migration [
86].
Determining the maximum migration of NPs to food is one of the most important concerns in the food industry. Migration is defined as a mass transfer for low molecular weight particles. General and specific migration tests have limitations for each food, and these limits are set by the EFSA (European Food Safety Authority) and the FDA (Food and Drug Administration). According to the EFSA definition, a migration test is the measurement of the amount of transport and release of packaging components (such as NPs) into food or food stimulants [
87].
It is very important to perform a migration test during the production process of a material to provide new packaging for foods. Although numerous papers have covered the migration of other NPs, such as nanosilver [
73], nanoclay [
88], and nano-TiO
2 [
89], few studies have been conducted on CuNPs migration from packaging into food or food simulant, and researchers are mainly studying the way to increase the barrier properties, mechanical features, antimicrobial activities of those copper nanocomposites which are potentials for food packaging, and ultraviolet light stability.
3.2.1. AgNP Migration
Although numerous studies have been carried out on the synthesis of AgNPs and their application in packaging systems, few studies have been conducted on the detection and migration of harmful substances in AgNPs-based FPS. Additionally, the previous studies about AgNP migration from packaging resulted in contradictory conclusions on AgNP detection. Several researchers have reported the presence of NPs in food [
90,
91], while other studies have provided no evidence of NP migration and supported these results with physicochemical modeling [
92,
93].
Amal M. Metak et al. [
81] focused on the migration of AgNPs from coated nanosilver films and AgNP-based and commercial food packaging in juice as real-food matrices. Conversely, samples exposed to nanosilver coated films revealed higher levels of migration (≤0.03 mg L
−1), which is cautionary whether in the form of a NP or ionic metal. No chemical or biochemical changes were observed in the tested food samples.
Cushen and colleagues studied AgNP migration from synthesized nanocomposite-based PVC (polyvinyl chloride) in contact with chicken breasts (at temperatures between 5 and 20 °C) and carried out migration tests. The results depicted the levels of migration equaling 0.03–8.4 mg·kg
−1 [
94]. Based on the studies conducted by Song et al. [
73] on the migration of AgNPs from polyethylene packaging to the food web, results from the ICP-MS analysis showed that the rate of silver migration in acetic acid 3‰ is time-independent and temperature-dependent, while in ethanol 95‰ the amount of nanosilver migration was time-dependent, and temperature changes showed no significant effect. In addition, Lin et al. [
89] studied AgNP migration by ICP-MS and ICP-AES methods and found that the most important step in these techniques is sample digestion. It was also reported that the ICP-MS method is more accurate than the ICP-AES method for detecting the migration of silver NPs.
In a study done by Hosseini et al. [
79], titration and migration methods showed different results for silver NP residues in prepared coatings containing 5–8% NPs. Although no significant difference was reported between the emitted NPs in coatings containing 1–3% of NPs, a statistically significant difference was reported between the two methods at 5% and 8%. Findings from another study revealed that just very small concentrations of silver migrate from food packaging when in contact with acidic and aqueous food simulators in the form of ionic silver (Ag +). It was also found that the migration of silver from this type of food packaging seems to be mainly a surface process, and any conditions that lead to significant changes in surface properties may affect the silver concentration in the simulator [
95].
In a study by Carsten Engelhard [
96], by employing capillary electrophoresis coupled to ICP-MS for measuring AgNPs, it was suggested that AgNPs with polymeric coatings are more chemically stable compared with citrate coatings, and therefore they may reduce the reactivity and evolution of NPs in the environment, but more studies are needed to optimize their antibacterial impact while controlling their environmental hazardous (due to their migration). In another study done by Ahari and his colleagues, the migration of AgNPs into the caviar samples was also studied by the titration method and the application of tetrazole with concentrated sulfuric acid. No silver residual was reported from packages with various concentrations of AgNPs [
59].
In research done by Yolanda Echegoyen, three distinct commercialized food AgNP-based containers were studied. The migration of silver in the form of dissolved silver and silver NPs was conducted as a key aspect of the toxicity. Based on the results, migration values were very low, although the EU (European Union) provided no specific limit for nanomaterials. The size (10–60 nm) and morphology of the AgNPs were distinct for the samples. They also confirmed the migration of other nanoscale materials [
91].
The antibacterial effect of the migrants needs to be evaluated during storage. In this regard, Raul and his colleagues surveyed the Ag release from commercially food packaging to simulants (water, acetic acid, olive and ethanol-water) and also the antimicrobial capacity of the migrated silver in meats (turkey, tuna, and ham). Silver also showed its potential in migration in the form of Ag
+ ion and AgNPs. Nevertheless, a significant antibacterial performance of the migrated silver to the turkeys was observed for gram-negative bacteria. In addition, in comparing various packaging including food touch papers, cutting boards, containers, and bags, the higher level of release belonged to paper (25 ± 11 mg/kg) [
97]. It was also reported that the silver ion release rates decrease with enhancing pH and NP size while they increase with temperature [
98]. The oxidative attack of the AgNPs is known as a time-dependent chemical reaction and is the reason why for all samples under study, the level of the migrated silver is higher for the longer essays (even at low temperatures).
3.2.2. CuNP Migration
Unlike silver, there is a low percentage of studies on the migration of CuNPs from packaging coatings to food. In one study, a migration experiment was conducted to assess the impact of time and temperature on the CuNP/AgNP migration from polyethylene nanocomposites to chicken breasts [
58]. Copper and silver migration ranged from 0.024 to 0.049 mg/dm
2 and 0.003 to 0.005 mg/dm
2, respectively. Based on the results, time and temperature did not have any significant effect on migration. These results were verified by the proposed equation of Williams–Landel–Ferry for time-temperature superposition. Compared to the AgNPs, CuNPs showed an enhanced dissolution rate from the coating as a result of their smaller size, which was reported as one of the vital reasons for higher migration [
99].
Liu and her colleagues studied the migration of CuNPs from copper-coated films via simulants including acetic acid (3%) and ethanol (10%) by GFAAS and ICP-MS. Based on the results, copper migration was significantly lower in 10% ethanol, and the migration ratio was lower than limitations from FDA. It was concluded that copper migration from nanocomposite films is affected by the temperature, time, initial concentration of CuNPs in the films as well as contact media [
85]. Surprisingly, these results were not in agreement with those of the study done by Cushen [
58]. It was also shown that the addition of an additive with no chemical bonds can promote copper migration. The addition of CuNPs alters the morphology of the film surface, which results in more surface area. The more surface area, the higher the contact between the food and the film, which accelerates and increases copper migration [
85]. In a similar study, based on migration tests, if the initial concentration of CuNPs that is added into the polypropylene film reached 1% (
w/
w) whereas the real concentration of CuNPs within the film was 0.79%, then the concentration of copper migrating from the film to the acetic food simulant (3.0%) would reach to 4.5 μg/g and would damage the liver cell [
100].