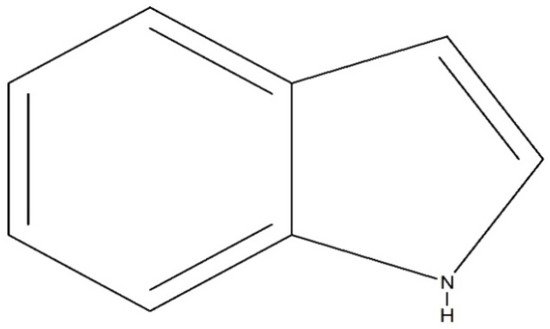
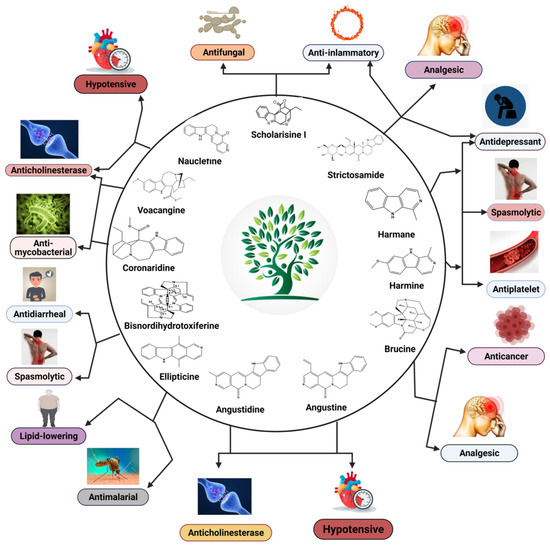
Indole (C8H7N) is a weakly basic molecule consisting of a pyrrole ring fused to a benzene nucleus, and ten π electrons move throughout the structure. The basic environment of indole alkaloids is thought to be caused by the delocalization of the lone pair of nitrogen electrons into the free circulation of the π electronic system. This results in indole becoming protonated at the C-3 position, which is thermodynamically more stable.Indole alkaloids have gained popularity due to their diverse pharmacological activities. Indole alkaloids have been identified in several prominent plant families, including Apocynaceae, Rubiaceae, Nyssaceae, and Loganiaceae, among others. Some of the identified indole alkaloid compounds have been highly effective in pre-clinical and clinical studies. Thousands of compounds containing the indole nucleus have been isolated from plant sources. Their pharmacological activities were assessed, with some now being examined in clinical trials and some already approved for therapeutic use in humans. Indole alkaloids are often characterized by their potent biological activities, which are relevant to the field of medicine, including anticancer, antibacterial, antiviral, antimalarial, antifungal, anti-inflammatory, antidepressant, analgesic, hypotensive, anticholinesterase, antiplatelet, antidiarrheal, spasmolytic, antileishmanial, lipid-lowering, antimycobacterial, and antidiabetic activities.
- indole
- alkaloids
- pharmacological activity
1. Indole Alkaloids
2. Pharmacological Activities
2.1. Antimicrobial Activity
2.2. Antiviral Activity
2.3. Antidepressant Activity
2.4. Anticancer Activity
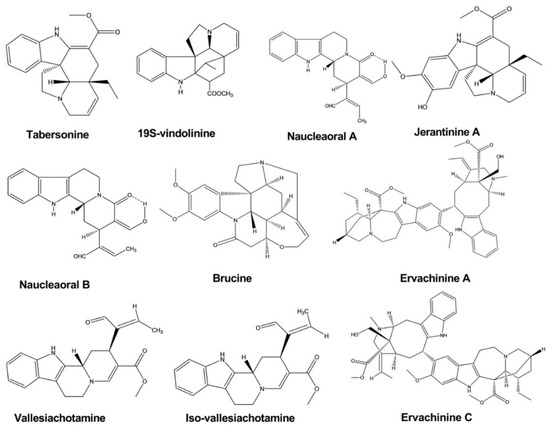
2.5. Anti-Inflammatory Activity
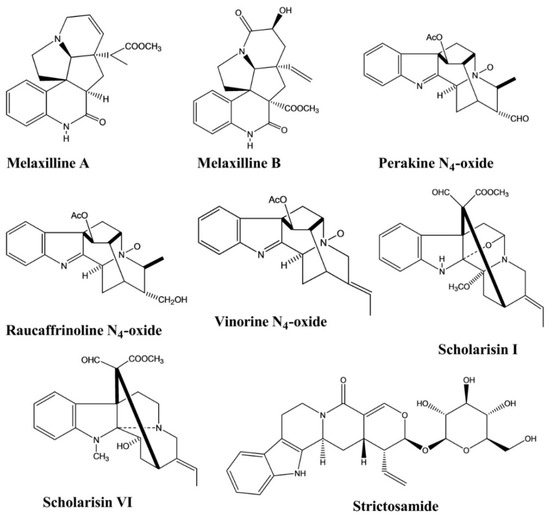
2.6. Analgesic Activity
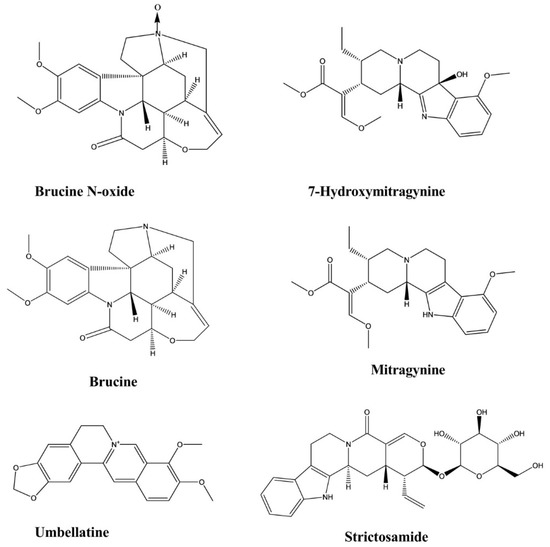
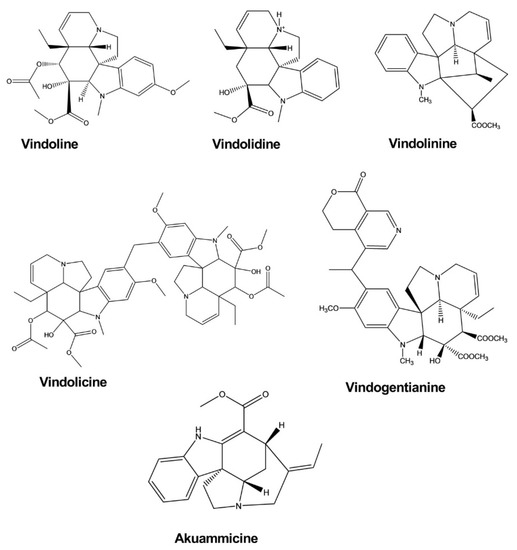
2.7. Antidiabetic Activity
2.8. Antimalarial Activity
2.9. Hypotensive Activity
2.10. Anticholinesterase Activity
2.11. Antiplatelet Activity
2.12. Antidiarrheal Activity
2.13. Spasmolytic Activity
2.14. Antileishmanial Activity
2.15. Lipid-Lowering Activity
2.16. Antimycobacterial Activity
| Compounds | Plant Source | Posology (Route, Dose) | Subject | Method | Identified Effect | References |
|---|---|---|---|---|---|---|
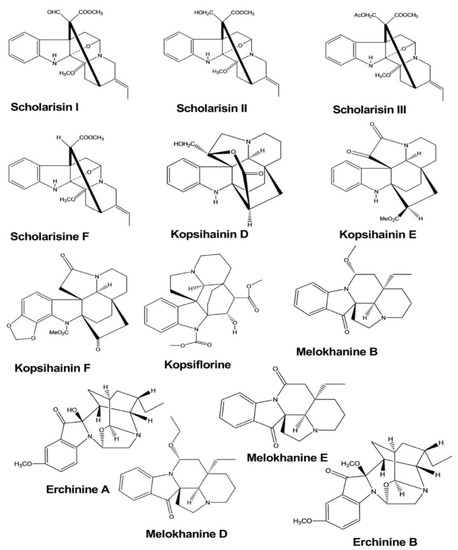
| Scholarisine I, II, III, F | Alstonia rupestris | - | Fungi | Disc diffusion method | Antifungal | [5] |
| Scholarisine I, VI | Alstonia rupestris | - | Enzyme | Enzyme inhibition assay | Anti-inflammatory | [5] |
| Kopsihainin D, E, F | Kopsia hainanensis | - | Bacteria | Disc agar diffusion method | Antibacterial | [6] |
| Kopsiflorine | Kopsia hainanensis | - | Bacteria | Disc agar diffusion method | Antibacterial | [6] |
| Erchinine A, B | Ervatamia chinensis | - | Bacteria Fungus |
Broth microdilution method | Antibacterial, Antifungal | [7] |
| Melokhanine B, D, E, F | Melodinus khasianus | - | Bacteria | Broth microdilution method | Antibacterial | [8] |
| Melokhanine B, D, E, F | Melodinus khasianus | - | Bacteria | Broth microdilution method | Antibacterial | [8] |
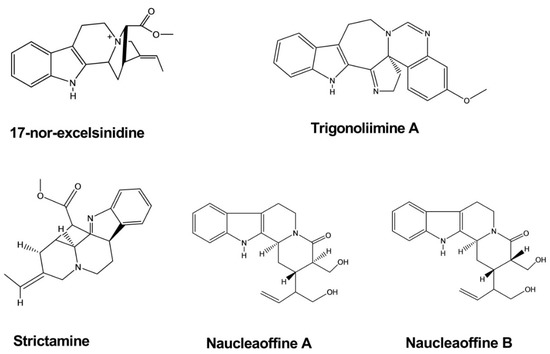
| Strictamine | Alstonia scholaris | - | Virus | - | Antiviral | [9] |
| 17-nor-excelsinidine | Alstonia scholaris | - | Virus | - | Antiviral | [9] |
| Trigonoliimine | Trigonostemon lii | - | Virus | Microtiter syncytium formation infectivity assay | Antiviral | [10] |
| Naucleaoffine A, B | Nauclea officinalis | Virus | Antiviral | [11] | ||
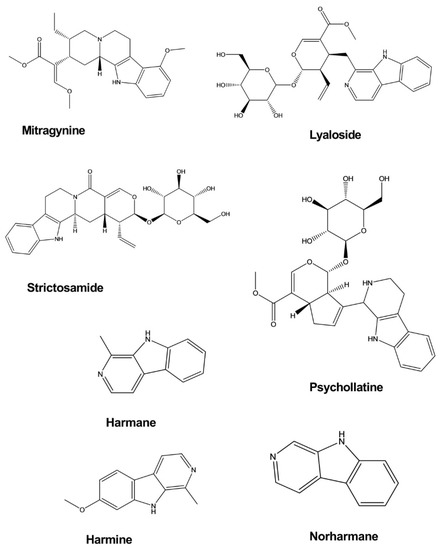
| Mitragynine | Mitragyna speciosa | Intraperitoneal, 10 and 30 mg/kg | Mice | Forced swim test, Tail suspension test | Antidepressant | [12] |
| Lyaloside | Psychotria suterella | - | Rat brain mitochondria | Enzymatic assay | Antidepressant | [13] |
| Strictosamide | Psychotria laciniata | - | Rat brain mitochondria | Enzymatic assay | Antidepressant | [13] |
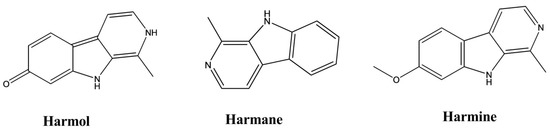
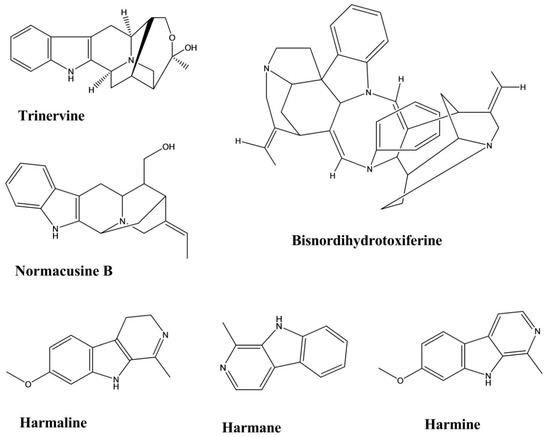
| Harmane | Peganum harmala | Intraperitoneal, 5–15 mg/kg | Mice | Forced swim test | Antidepressant | [14] |
| Norharmane | Peganum harmala | Intraperitoneal, 2.5–10 mg/kg | Mice | Forced swim test | Antidepressant | [14] |
| Harmine | Peganum harmala | Intraperitoneal, 5–15 mg/kg | Mice | Forced swim test | Antidepressant | [14] |
| Psychollatine | Psychotria umbellate | 3, 7.5, and 15 mg/kg | Mice | Hole-board test, Forced swim test | Antidepressant | [15] |
| Tabersonine | Melodinus fusiformis | - | Human tumor cell line | MTT assay | Anticancer | [16] |
| Brucine | Strychnos nux-vomica | - | Human hepatoma cell line | MTT-colorimetric assay | Anticancer | [17] |
| Naucleaoral A, B | Nauclea orientalis | - | Human cancer cell line | MTT-colorimetric assay | Anticancer | [18] |
| Vallesiachotamine | Anthocephalus cadamba | - | Human lung cancer cell line | MTT assay | Anticancer | [19] |
| Iso-vallesiachotamine | Anthocephalus cadamba | - | Human lung cancer cell line | MTT assay | Anticancer | [19] |
| Ervachinine A, C, D | Ervatamia chinensis | - | Human cancer cell line | MTT assay | Anticancer | [20] |
| Jerantinine A, B | Tabernaemontana corymbosa | Human cancer cell line | MTT assay | Anticancer | [21] | |
| Melaxilline A, B | Melodinus axillaris. | - | Rat | Platelet-activating factor induced inhibition assay | Anti-inflammatory | [22] |
| Perakine N4-oxide | Alstonia yunnanensis. | - | Enzyme | Enzyme inhibition assay | Anti-inflammatory | [23] |
| Raucaffrinoline N4-oxide | Alstonia yunnanensis. | - | Enzyme | Enzyme inhibition assay | Anti-inflammatory | [23] |
| Vinorine N4-oxide | Alstonia yunnanensis. | - | Enzyme | Enzyme inhibition assay | Anti-inflammatory | [23] |
| Strictosamide | Nauclea officinalis | Intraperitoneal, 20 and 40 mg/kg |
Mice | Hot plate test, Writhing test | Analgesic | [24] |
| Strictosamide | Psychotria laciniata | Intravenous, 20 and 40 mg/kg |
Mice | Acetic acid and TPA-induced assay | Anti-inflammatory | [24] |
| Brucine | Strychnos nux-vomica | Intraperitoneal, 30, 15, and 7.5 mg/kg | Mice | Formalin test | Analgesic | [25] |
| Brucine | Strychnos nux-vomica | Intraperitoneal, 30, 20, 14.7, and 10.3 mg/kg | Mice | Hot plate test | Analgesic | [25] |
| Brucine | Strychnos nux-vomica | Intraperitoneal, 30, 15, 7.5, and 3.75 mg/kg | Mice | Writhing test | Analgesic | [25] |
| Brucine N-oxide | Strychnos nux-vomica | Intraperitoneal, 200, 100 and 50 mg/kg | Mice | Formalin test | Analgesic | [25] |
| Brucine N-oxide | Strychnos nux-vomica | Intraperitoneal, 200, 140, 98, and 68 mg/kg | Mice | Hot plate test | Analgesic | [25] |
| Brucine N-oxide | Strychnos nux-vomica | Intraperitoneal, 200, 100, 50, and 25 mg/kg | Mice | Writhing test | Analgesic | [25] |
| Mitragynine | Mitragyna speciosa | Intraperitoneal, 3–35 mg/kg | Mice | Hot plate test | Analgesic | [26] |
| 7-hydroxymitragynine | Mitragyna speciosa | Subcutaneous, 2.5–10 mg/kg | Mice | Hot plate test, Tail flick test | Analgesic | [27] |
| Umbellatine | Psychotria umbellate | 100–300 mg/kg | Mice | Tail-flick test, Hot-plate test, Formalin test and Capsaicin-induced pain test | Analgesic | [28] |
| Vindoline | Catharanthus roseus | - | Enzyme, Myoblast cell |
Enzyme inhibition assay, Glucose uptake activity assay | Antidiabetic | [29] |
| Vindolidine | Catharanthus roseus | - | Enzyme, Myoblast cell |
Enzyme inhibition assay, Glucose uptake activity assay | Antidiabetic | [29] |
| Vindolicine | Catharanthus roseus | - | Enzyme, Myoblast cell |
Enzyme inhibition assay, Glucose uptake activity assay | Antidiabetic | [29] |
| Vindolinine | Catharanthus roseus | - | Enzyme, Myoblast cell |
Enzyme inhibition assay, Glucose uptake activity assay | Antidiabetic | [29] |
| Vindogentianine | Catharanthus roseus | - | Enzyme, Myoblast cell |
Enzyme inhibition assay, Glucose uptake activity assay | Antidiabetic | [30] |
| Akuammicine | Picralima nitida | - | Myoblast cell |
Glucose uptake activity assay | Antidiabetic | [31] |
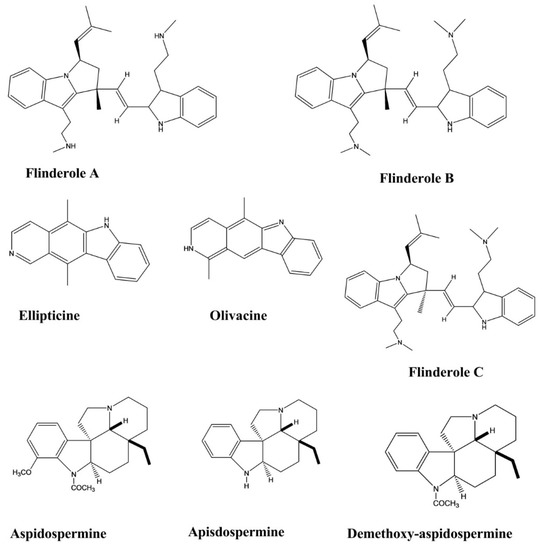
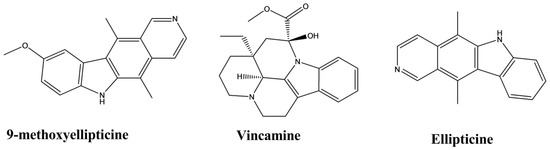
| Ellipticine | Aspidosperma vargasii | Oral, Subcutaneous, 50, 10 and 1 mg/kg/day | Mice | Suppressive test | Antimalarial | [32] |
| Olivacine | Aspidosperma olivaceum. | Oral, Subcutaneous, 100, 50, 10, and 1 mg/kg/day | Mice | Suppressive test | Antimalarial | [32] |
| Flinderole A, B, C | Flindersia acuminate F. amboinensis |
- | Malarial Parasite | Micrdilution method | Antimalarial | [33] |
| Apisdospermin | - | Malarial Parasite | Micrdilution method | Antimalarial | [34] | |
| Aspidospermine | Aspidosperma pyrifolium | - | Malarial Parasite | Micrdilution method | Antimalarial | [34] |
| Demethoxy-aspidospermine | Aspidosperma pyrifolium | - | Malarial Parasite | Micrdilution method | Antimalarial | [34] |
| Vallesine | Aspidosperma pyrifolium | - | Malarial Parasite | Micrdilution method | Antimalarial | [34] |
| Palosine | Aspidosperma pyrifolium | - | Malarial Parasite | Micrdilution method | Antimalarial | [34] |
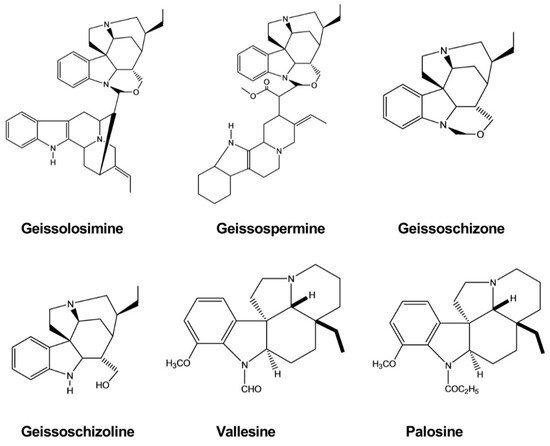
| Geissolosimine | Geissospermum vellosii | - | Malarial Parasite | Parasite lactate dehydrogenase assay | Antimalarial | [35] |
| Geissospermine | Geissospermum vellosii | - | Malarial Parasite | Parasite lactate dehydrogenase assay | Antimalarial | [35] |
| Geissoschizoline | Geissospermum vellosii | - | Malarial Parasite | Parasite lactate dehydrogenase assay | Antimalarial | [35] |
| Geissoschizone | Geissospermum vellosii | - | Malarial Parasite | Parasite lactate dehydrogenase assay | Antimalarial | [35] |
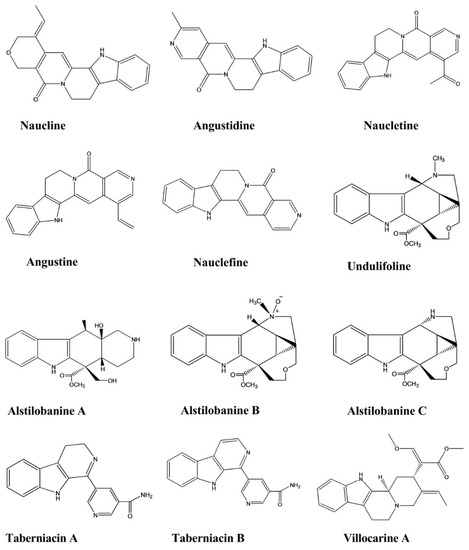
| Naucline | Nauclea officinalis |
Injection, 1 × 10−5 M |
Rat | Phenylephrine-induced vasodilation assay | Hypotensive | [36] |
| Angustine | Nauclea officinalis |
Injection, 1 × 10−5 M |
Rat | Phenylephrine-induced vasodilation assay | Hypotensive | [36] |
| Angustidine | Nauclea officinalis |
Injection, 1 × 10−5 M |
Rat | Phenylephrine-induced vasodilation assay | Hypotensive | [36] |
| Nauclefine | Nauclea officinalis |
Injection, 1 × 10−5 M |
Rat | Phenylephrine-induced vasodilation assay | Hypotensive | [36] |
| Naucletine | Nauclea officinalis |
Injection, 1 × 10−5 M |
Rat | Phenylephrine-induced vasodilation assay | Hypotensive | [36] |
| Alstilobanine A, B, C | Alstonia angustiloba | - | Rat | Phenylephrine-induced vasodilation assay | Hypotensive | [37] |
| Undulifoline | Alstonia undulifolia | - | Rat | Phenylephrine-induced vasodilation assay | Hypotensive | [37] |
| Taberniacin A, B | Tabernaemontana divaricata | - | Rat | Phenylephrine-induced vasodilation assay | Hypotensive | [38] |
| Villocarine A | Uncaria villosa | Injection, 30 μM |
Rat | Phenylephrine-induced vasodilation assay | Hypotensive | [39] |
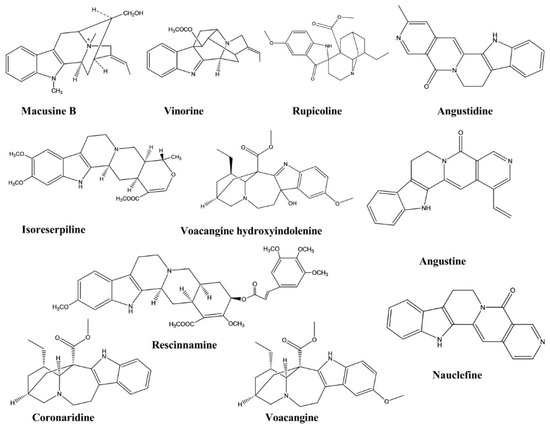
| Macusine B | Rauvolfia reflexa | - | Cholinesterase Enzyme | Cholinesterase Inhibition assay | Anticholinesterase | [40] |
| Vinorine | Rauvolfia reflexa | - | Cholinesterase Enzyme | Cholinesterase Inhibition assay | Anticholinesterase | [40] |
| Isoreserpiline | Rauvolfia reflexa | - | Cholinesterase Enzyme | Cholinesterase Inhibition assay | Anticholinesterase | [40] |
| Rescinnamine | Rauvolfia reflexa | - | Cholinesterase Enzyme | Cholinesterase Inhibition assay | Anticholinesterase | [40] |
| Voacangine hydroxyindolenine | Tabernaemontana australis | - | Cholinesterase Enzyme | Cholinesterase Inhibition assay | Anticholinesterase | [41] |
| Rupicoline | Tabernaemontana australis | - | Cholinesterase Enzyme | Cholinesterase Inhibition assay | Anticholinesterase | [41] |
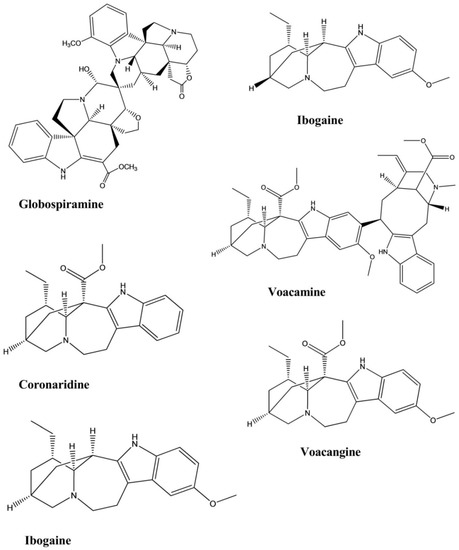
| Coronaridine | Ervatamia hainanensis | - | Cholinesterase Enzyme | Cholinesterase Inhibition assay | Anticholinesterase | [42] |
| Voacangine | Ervatamia hainanensis | - | Cholinesterase Enzyme | Cholinesterase Inhibition assay | Anticholinesterase | [42] |
| Angustidine | Nauclea officinalis | - | Cholinesterase Enzyme | Cholinesterase Inhibition assay | Anticholinesterase | [43] |
| Nauclefine | Nauclea officinalis | - | Cholinesterase Enzyme | Cholinesterase Inhibition assay | Anticholinesterase | [43] |
| Angustine | Nauclea officinalis | - | Cholinesterase Enzyme | Cholinesterase Inhibition assay | Anticholinesterase | [43] |
| Harmane | Perganum harmala | 100–200 µM | Rabbit Platelet | - | Antiplatelet | [44] |
| Harmine | Perganum harmala | 100–200 µM | Rabbit Platelet | - | Antiplatelet | [44] |
| Harmol | Perganum harmala | 100–200 µM | Rabbit Platelet | - | Antiplatelet | [44] |
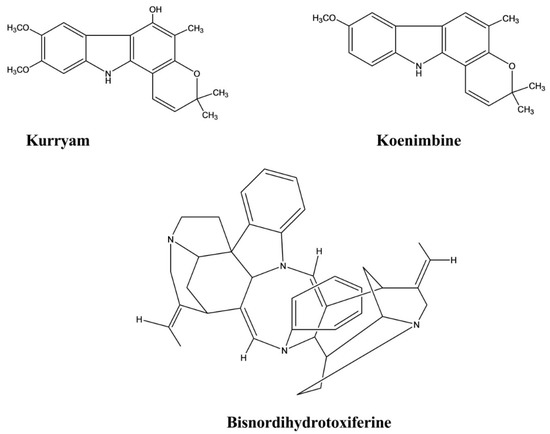
| Kurryam | Murraya koenigii | 10, 30, and 50 mg/kg | Rat | Castor oil-induced test | Antidiarrheal | [45] |
| Koenimbine | Murraya koenigii | 10, 30, and 50 mg/kg | Rat | Castor oil-induced test | Antidiarrheal | [45] |
| Bisnordihydrotoxiferine | Strychnos trinervis | Intraperitoneal, 3.12–25.00 mg/kg |
Rat, Mice | Castor oil, Magnesium sulfate, and Arachidonic acid-induced diarrhea test | Antidiarrheal | [46] |
| Trinervine | Strychnos trinervis | - | Rat fundic strip, Guinea-pig ileum | Arachidonic acid, 5-hydroxytryptamine, Histamine, and Carbachol mediated contractions | Spasmolytic | [47] |
| Bisnordihydrotoxiferine | Strychnos diuaricuns | - | Rat uterus Guinea-pig ileum | Acetylcholine, Oxytocin-induced contraction, and Histamine-induced contractions | Spasmolytic | [48] |
| Normacusine B | Strychnos atlantica | 0.1, 0.3, 1.0, and 3.0 μM | Rat aorta | Phenylephrine and Serotonin-induced contractions. | Spasmolytic | [49] |
| Harmine | Perganum harmala | 1–100 μM | Guinea-Pig Trachea | Histamine, Carbachol, and KCl-induced contractions | Spasmolytic | [50] |
| Harmane | Perganum harmala | 1–100 μM | Guinea-Pig Trachea | Histamine, Carbachol, and KCl-induced contractions | Spasmolytic | [50] |
| Harmaline | Perganum harmala | 1–100 Μm | Guinea-Pig Trachea | Histamine, Carbachol, and KCl-induced contractions | Spasmolytic | [50] |
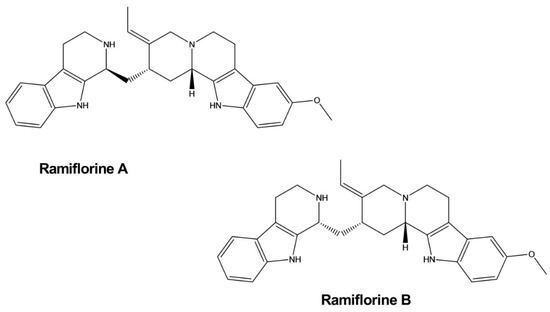
| Ramiflorine A, B | Aspidosperma ramiflorum | - | Parasite | Antileishmanial | [51] | |
| Ellipticine | Ochrosia borbonica | 0.01–10 µmol·L−1 | Mouse fibroblast cells | Triglyceride assay | Lipid-lowering | [52] |
| 9-methoxyellipticine | Ochrosia borbonica | 0.01–10 µmol·L−1 | Mouse fibroblast cells | Triglyceride assay | Lipid-lowering | [52] |
| Vincamine | Vinca minor | Oral, 20 and 30 mg/kg |
Rat | Lipid-lowering | [53,54] | |
| Coronaridine | Tabernaemontana ternifolia | - | Bacteria | Microplate Alamar blue assay | Antimycobacterial | [55] |
| Globospiramine | Voacanga globosa | - | Bacteria | Microplate Alamar blue assay and Low-oxygen recovery assay | Antimycobacterial | [56] |
| Ibogaine | Tabernaemontana citrifolia | - | Bacteria | Bactec radiometric methodology | Antimycobacterial | [57] |
| Voacangine | Tabernaemontana citrifolia | - | Bacteria | Bactec radiometric methodology | Antimycobacterial | [57] |
| Voacamine | Tabernaemontana arborea. | - | Bacteria | Resazurin microtiter assay | Antimycobacterial | [58] |
3. Pharmacokinetic Profile
| Parameters | Compound Name | ||||
|---|---|---|---|---|---|
| Brucine | Harmane | Vindoline | Mitragynine | 7-OH-mitragynine | |
| Administration Route | Oral, IV | Oral, IV | Oral, IV | Oral | Oral |
| Oral Bioavailability | 40.31%–47.15% | 19.41% | 5.4% | - | - |
| AUC | Ratio: 1:1.9:5.3 (at different doses) | - | Oral: 606.3 ng/mL h, IV: 2245.7 (first dose), 2258.0 (second dose) ng/mL h |
- | - |
| Cmax | 929.22–1451.58 μg/L | Oral: 1059.56 ± 91.06 ng/mL, IV: 583.19 ng/mL |
Oral: 606.6 ng/mL, IV: 1595.9 (first dose), 2913.9 ng/mL (second dose) |
0.63 ± 0.18 µg/mL | - |
| Tmax | 0.3–0.5 h | Oral: 0.23 ± 0.06 h, IV: 0.03 h |
Oral: 0.3 h | 1.83 ± 1.25 h | - |
| t1/2 | - | - | Oral: 0.5 h IV: 1.0 h (first dose), 1.4 h (second dose) |
- | 24 min |
| Vd | - | - | Oral; 21.6 L/kg IV: 2.0 L/kg (first dose), 3.3 L/kg (second dose) |
89.50 ± 30.30 L/kg | - |
| CL | - | - | Oral: 26.6 L/h/kg IV: 1.4 L/h/kg (first dose), 1.4 L/h/kg (second dose) |
- | 43.2 ± 3.5 mL/min/kg |
| References | [60] | [64] | [63] | [61] | [62] |
This entry is adapted from the peer-reviewed paper 10.3390/molecules26082297
