Methylxanthines (MTX) are purine derived xanthine derivatives. Methylxanthines are also known to have anti-inflammatory and anti-oxidative properties, mediate changes in lipid homeostasis and have neuroprotective effects.
- methylxanthines
- Parkinson´s disease
1. Introduction
These multifactorial disorders share several common histopathological hallmarks and mechanisms like neuronal cell loss linked to gliosis, misfolding and accumulation of proteins, oxidative stress and neuroinflammation. In case of AD, disease progression is strongly associated with Aβ peptide generation and aggregation, associated with pathological extracellular and intracellular filamentous deposits, hyperphosphorylated tau proteins, neuroinflammation and synaptic loss. PD is characterized by dopaminergic neurodegeneration and accumulation of α-synuclein in Lewy bodies. Concerning MS, neuroinflammatory processes leading to demyelination of neurons are in the focus of the disease.
Numerous studies have been performed to address the question of whether xanthine derivatives like caffeine and theobromine have beneficial properties in respect to the characteristic histopathological changes that occur in the above-mentioned diseases and to cognitive decline. Although the outcome of the clinical studies, especially for AD, were heterogeneous [1], some mechanisms were identified, showing how methylated xanthine derivatives could protect against neuronal damage.
In general, in physiological concentrations, achieved for example by coffee consumption or by intake of methylxanthine containing beverages, methylxanthines act as antagonists of the adenosine receptor (AR), histone deacetylase activator or antioxidant. Affecting these pathways, xanthine derivatives are able to modulate molecular mechanisms associated with neurodegenerative diseases like accumulation of misfolded proteins, oxidative stress and neuroinflammation. Interestingly, in particular the beneficial role of adenosine receptor antagonists in the treatment of neurodegenerative diseases has become more and more apparent in the last years [2,3,4,5].
2. Parkinson’s Disease
2.1. Epidemiological and Clinical Studies
| Author | Year | Type of Study/n | Substance | Outcome |
|---|---|---|---|---|
| Bakshi et al. [59] | 2020 | cross sectional, case-control/197 healthy control vs. 369 idiopathic PD patients | caffeine, urate | the authors found a robust inverse association between idiopathic PD and caffeine intake and urate plasma levels |
| Fujimaki et al. [60] | 2018 | clinical trial/31 healthy control vs. 108 PD patients without dementia1 | caffeine, theophylline, theobromine, paraxanthine and other downstream metabolites | absolute lower levels of caffeine and metabolites were found to be a promising biomarker for early PD |
| Crotty et al. [62] |
2020 | clinical trial/samples from “23andMe” study, LRRK2 longitudinal study and LRRK2 cross-sectional study (n = 380) | caffeine, theophylline, paraxanthine and other downstream metabolites, trigonelline (non-xanthine constituent of coffee) | significantly lower plasma and CSF levels of caffeine and downstream metabolites in PD patients, even more in LRRK2 mutation carriers |
| Ohmichi et al. [63] | 2018 | clinical trial/31 PD patients vs. 33 age-matched controls | theophylline | theophylline serum levels were significantly lower in PD patients compared to control |
| Hong et al. [64] |
2020 | meta-analysis/13 studies (9 healthy cohort, 4 PD cohort) | caffeine | caffeine consumption resulted in a significantly lower rate of PD |
| Maclagan et al. [65] | 2020 | computational & pharmacoepidemiologic study ranking 620 drugs, case-control study/14,866 PD and 74,330 controls | pentoxifylline, theophylline, dexamethasone | the authors state, that corticosteroids and the found methylxanthines should be investigated as disease-modifying drugs |
| Sako et al. [67] |
2017 | meta-analysis/six studies | istradefylline | 20 and 40 mg/day of istradefylline revealed significantly decreased durations of “off episodes” in PD patients |
2.2. Animal Studies/Molecular Pathways
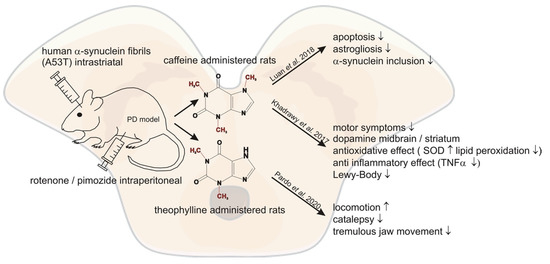
| Author | Year | Used Model | Substance | Outcome |
|---|---|---|---|---|
| Luan et al. [69] | 2018 | Injected α-synuclein fibrils intra-striatal in mice | caffeine | reduced inclusion of α-synuclein, apoptosis, microglial activation and astrogliosis after caffeine treatment |
| Khadrawy et al. [70] | 2017 | rotenenoe induced PD mice model | caffeine | recovering dopamine levels in midbrain and striatum ameliorating motor symptoms, antioxidative and anti-inflammatory effect of caffeine, prevention of neurodegeneration through less lewy bodies |
| Pardo et al. [71] | 2020 | pimozide induced PD mice model | theophylline | reversed locomotion, catalepsy and tremulous jaw movement |
| Rohilla et al. [72] | 2019 | perphenazine induced catatonia in rats | newly synthesized xanthine derivatives | most xanthines significantly lowered catatonia score, most potent MTX shows a similar response as L-DOPA |
This entry is adapted from the peer-reviewed paper 10.3390/nu13030803
