Ferrofluids (FFs) are stable and quite homogeneous colloidal dispersions of ferromagnetic nanoparticles in a solvent referred to as the carrier.
- ferrofluids
- extraction
- sample preparation
- analytical chemistry
1. Introduction
Ferrofluids (FFs) are stable and quite homogeneous colloidal dispersions of ferromagnetic nanoparticles in a solvent referred to as the carrier. These materials have the properties of both of their components: (i) the magnetic nanoparticles (MNPs), which confer a paramagnetic behavior to the suspension; and (ii) the carrier, which is responsible for the fluidic nature of the material [1]. It is important to highlight that a liquid dispersion of MNPs can be considered as FF only if it presents enough uniformity to be a magnetic liquid by itself, and without observing the separation of those particles from the carrier when an external magnetic field is applied [2].
MNPs used to prepare FFs are generally made of magnetite (Fe3O4), maghemite (γ-Fe2O3), or either Fe (II)/Fe (III) compounds, the magnetic dipolar moment of which can be aligned parallelly to the dipolar moment of an external magnetic field [3]. Traditionally, MNPs used for the preparation of FFs are loaded at low content in the carrier, between 3% and 10% of the FF. Besides, MNPs with a particle size around 10 nm are preferred to form FFs [2]. Nevertheless, the size of the particles should be taken simply as an illustrative parameter, as MNPs with higher diameters have also been used to prepare FFs [4]. Regarding the carrier, it is the main component in the FF (90–97%), providing its liquid nature (despite the presence of small amounts of solid particles in suspension). It is also responsible for most FFs properties. The proper selection of the carrier allows the preparation of tunable FFs. A wide variety of solvents have been utilized as FFs carriers, either conventional solvents (organic solvents—mainly alcohols—and water) [5,6] and non-conventional solvents, including ionic liquids (ILs) [7], deep eutectic solvents (DESs) [8], and supramolecular solvents (SUPRASs) [9].
The stability of FFs can be clearly affected by the agglomeration of the MNPs, due to Van Der Waals interactions and magnetic attractions, among others. Thus, FFs need to incorporate a stabilizer or a dispersing agent to prevent such aggregation [10]. The stabilizer can be directly incorporated into the initial mixture of MNPs plus carrier, or the stabilization of MNPs can be accomplished by their surface modification through functionalization. The stabilization can take place through two different mechanisms of repulsion: (i) ionic repulsion and (ii) steric repulsion. Ionic stabilizers form a charged layer over the surface of MNPs, causing repulsion forces between the MNPs as they have the same electrostatic charge. On the other hand, steric repulsion is produced by the formation of a thicker layer that moves MNPs away from each other due to the steric hindrance. This layer is formed into two steps: the first stabilizer layer is attached by chemisorption and followed by a second layer that is attached by physisorption on the surface of MNPs [1]. Surfactants are the most widely used stabilizers in FFs due to their high molecular weight, which, in most cases, favors the establishment of steric repulsions, and, in the case of ionic surfactants, through the incorporation of the ionic repulsion. Oleic acid, lauric acid, palmitic acid, and stearic acid are some of the most representative surfactants used as stabilizer agents [11]. Some of the solvents used as FFs carriers have also been explored as ionic stabilizers, with ILs being the most popular solvents for this role [12].
The main preparation procedure of FFs is quite simple: the direct mixing of the precursors (stabilized MNPs and carrier, or MNPs, stabilizer, and carrier) for a certain time, to ensure the formation of a quite homogeneous colloidal suspension. Ultrasonic energy is the most common assistance method used to ensure such uniform and energic mixing [5,9,13,14,15,16,17,18,19,20,21,22,23,24,25,26,27,28]. In any case, the preparation of any FF clearly requires the synthesis of the MNPs, and, in several cases, the surface modification of the MNPs, and/or the synthesis of the carrier solvent if it is not commercially available.
If considering green issues, water-based FFs are undoubtedly the easiest and most direct and environmental-friendly types, while being (in general) biocompatible. In any case, the green label cannot be exclusively linked to their water-based nature, and indeed green aspects related to the synthetic procedure of the MNPs need to be also considered. Thus, it is important to consider if hazardous chemicals have been used as precursors, or if an inert atmosphere and/or thermal treatments at high temperatures for long times have been also required [29].
The most important feature of FFs is their magnetic susceptibility, which gives them an impressive magnetic-responsive behavior. The application of an external magnetic field can induce several modifications in the properties of FFs, making them smart and tunable materials with great potential in a wide variety of disciplines. Indeed, the application of magnetic fluids is not a recent trend in magnetochemistry [30] and biomedicine [31], among other fields. However, the use of FFs as extraction materials in sample preparation is a relatively new topic.
2. FFs in Analytical Sample Preparation Methods
The use of FFs within analytical chemistry has not been extensively consolidated, and indeed there is still controversy concerning mechanisms of FF-based extractions. To sum up, there is no consensus on the classification of the technique into the different existing analytical extraction methods [32]. From an operational point of view, in general, the extraction procedure based on FFs requires a dispersion of the as-prepared FF into the sample, followed by the application of an external force to ensure a strong mixing sample-FF. Once the analytes have been partitioned from the sample to the FF, the FF is decanted using an external magnet. Finally, as it is not possible to directly inject the FF into a chromatographic system (the most common technique in the reported applications) without damaging it, the FF is dispersed in a few µL of a solvent to perform a desorption step of the retained analytes. Figure 1 shows a general scheme of the most common extraction procedure followed when using FFs.
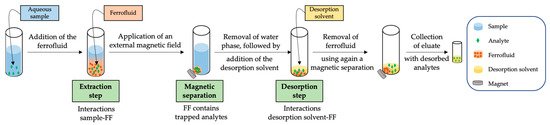
This extraction procedure has been identified by several authors as magnetic micro-dispersive solid-phase extraction (m-µ-dSPE), because the operational procedure resembles that of m-µ-dSPE, including both extraction and desorption steps [6,15,21,22,23,24,25,26,33,34,35,36], while other studies have included FF-based extractions within magnetic dispersive liquid–liquid microextraction (MDLLME) methods [5,8,9,13,16,17,18,27,37,38,39,40]. These two different classifications rely on the following considerations: (i) the main component is a liquid and the content of solid MNPs is almost negligible, and, thus, it can be classified as liquid-based; and (ii) the partition of the analytes in the extraction step takes place from the liquid sample to the solid magnetic micro-extractant phase, and, thus, it can be classified as solid-based. Independent of the classification, it is clear that the extraction mechanism comprises a synergetic effect between the role of the MNPs, which confer the magnetic nature to the FF (allowing the magnetic separation), and the role of the liquid carrier as the main promoter of the interactions between the analytes and the FF. Assuming that this important role of the carrier and the solvation effects of the FF given its (mostly) liquid nature, FF-based extraction procedures would be better classified as magnetic liquid-phase microextraction (MLPME).
The first FF used with extraction purposes was reported in 2010 by Shi et al. [5]. It was prepared by simple dispersion of 10 mg of silica-coated Fe3O4-based MNPs in 100 µL of 1-octanol as the carrier, under sonication. In this study, 28 mg of FF were added to 20 mL of an aqueous sample under agitation. After 20 min of extraction time, the FF containing the trapped analytes was separated by using an external magnet. Finally, the extracted analytes were desorbed using 100 µL of acetonitrile under sonication for 2 min. Then, the eluate was directly injected in a gas chromatograph (GC) equipped with a mass spectrometer (MS) for the determination of polycyclic aromatic hydrocarbons (PAHs). The incorporation of the FF reduced the extraction time needed, as any magnetic-based extraction method, avoiding centrifugation steps, and permitting a solvent-saving sample preparation approach.
Since then, a wide variety of analytical applications have arisen using FFs with different solvents as carriers. Undoubtedly, the chemical and physical properties of the carrier solvent exert an important influence on the extraction performance. Thus, the proper selection of the carrier is decisive for the design of the FF and its intended application in analytical sample preparation procedures.
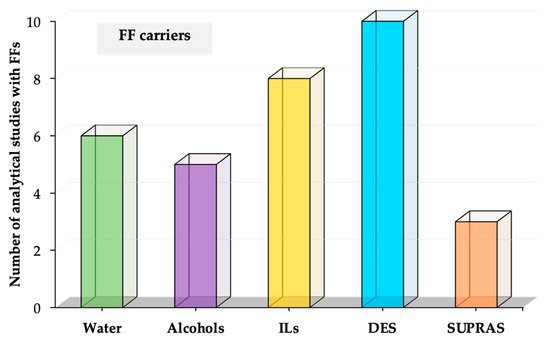
In this section, we propose to classify the FFs as a function of the nature of the carrier solvent. The main solvents used as carriers of FFs in sample preparation applications are (i) alcohol and water, (ii) ILs, (iii) DESs, and (iv) SUPRAS. Figure 2 shows the number of studies for each type of FF according to this classification. It is important to highlight that this review article focuses on those articles that clearly incorporate FFs in analytical studies. Table 1 contains a summary of those analytical applications using FFs [5,6,7,8,9,13,14,15,16,17,18,19,20,21,22,23,24,25,26,28,33,34,35,36,37,38,39,40,41,42,43,44]. As it can be observed, different analytes have been determined in a wide variety of samples, using all the types of FFs. Thus, the intended analytical application is not the main criterium to select a specific type of FF in the studies reported. In general, FFs are selected by their carrier, and each study benefits from the features of the carrier solvent, as is described in detail in this section for each type of FF.
| Nanoparticle/Carrier | Amount of Ferrofluid (µL) | Analytes (Number) | Sample/Volume (mL) | Analytical Technique | LOQ | RSDmax * (%) | Recovery (%) | Ref. |
|---|---|---|---|---|---|---|---|---|
| Water-based FFs | ||||||||
| γ-Fe2O3@citrate/water | – | metals (4) | water (–) | ICP-AES | – | – | – | [35] |
| Fe3O4@decanoic acid/water | 1000 | drugs (2) | water, plasma and urine (5) | HPLC-UV | 10 µg·L−1 | 8.4 | 89.6–97.4 | [6] |
| Fe3O4@oleic acid/water | 750 | aflatoxins (4) | MeOH/water extract of food (18) | spectrofluorimetry | 21 ng·L−1 ** | 4.6 | 91.6–99.6 | [41] |
| Fe3O4@oleic acid/water | 500 | aflatoxin (1) | MeOH/water extract of food (18) | spectrofluorimetry | 13 ng·L−1 ** | 4.3 | 91.3–99.5 | [42] |
| Fe3O4@decanoic acid/water | 1000 | drugs (3) | water and urine (20) | GC-FID | 10 µg·L−1 | 9.4 | 90.0–104 | [36] |
| Fe3O4@oleic acid/water | 500 | drug (1) | plasma (5) | spectrofluorimetry | 0.21 µg·L−1 | 1.5 | 93.5–102 | [43] |
| Alcohol-based FFs | ||||||||
| Fe3O4@oleic acid/1-heptanol | 100 | phenolic compounds (4) | beverages (20) | HPLC-DAD | 0.66 µg·L−1 ** | 7.3 | 83.7–106 | [13] |
| Fe3O4@SiO2/1-octanol | 28 (mg) | PAHs (16) | water (20) | GC-MS | 0.19 µg·L−1 | 12 | 59.2–93.3 | [5] |
| Fe3O4@SiO2/1-octanol | 200 | metal (1) | soil and road dust after acid digestion (50) | FAAS | 0.35 µg·L−1 ** | 3.3 | 95.2–103 | [33] |
| Fe3O4@SiO2/1-octanol | – | metal (1) | nail, hair, cabbage and iron after acid digestion (150) | FAAS | 0.21 µg·L−1 ** | 3.2 | 95.0–105 | [14] |
| Fe3O4@poly(β-CD-IL)/1-octanol | – | PAHs (7) | water, beverages and ACN extract of food (15) | GC-FID | 0.21 µg·L−1 | 8.9 | 80.5–116 | [37] |
| IL-based FFs | ||||||||
| Fe3O4@SiO2/[C4MIm]+[BF4]− | 25 mg | metal (1) | water and food after wet digestion (150) | FAAS | 0.11 µg·L−1 ** | 4.0 | 97.5–103 | [34] |
| Fe3O4@GO/[C4MIm]+[BF4]− | – | metal (1) | water, and vegetable and tobacco after acid digestion (250) | FAAS | 0.12 µg·L−1 ** | 1.4 | 98.2–101 | [21] |
| Fe3O4@GO/[C4MIm]+[BF4]− | – | metals (2) | water (100) | GFAAS | 26 ng·L−1 | 6.5 | 83–117 | [22] |
| Fe3O4@SiO2/[C6MIm]+[BF4]− | – | metal (1) | waters, and road dust and food after acid digestion (200) | FAAS | 1.7 µg·L−1 ** | 1.34 | 96.0–118 | [23] |
| Fe3O4@SiO2/[C6MIm]+[BF4]− | – | metal (1) | water, beverages, and food after acid digestion (200) | FAAS | 0.32 µg·L−1 ** | 2.6 | 95.5–105 | [24] |
| Fe3O4@SDS/[C6MIm]+[BF4]− | – | dye (1) | water and EtOH extract of food (175) | UV-Vis | 2.5 µg·L−1 ** | 2.9 | 99.0–109 | [25] |
| BaFe/[C6MIm]+[NTF2]− | 50 | pesticides (5) | beverages (10) | HPLC-DAD | 0.53 µg·L−1 ** | 5.3 | 85.1–99.6 | [7] |
| Fe3O4@CQD/[C8MIm]+[PF6]− | – | phenolic compounds (4) | water and beverage (20) | HPLC-DAD | 0.17 µg·L−1 ** | 4.1 | 94.5–102 | [26] |
| DES-based FFs | ||||||||
| Fe3O4@SiO2/ChCl:EG | 250 | drug (1) | plasma and urine (10) | HPLC-UV | 2.5 µg·L−1 ** | 6.2 | 92.8–98.4 | [15] |
| Fe3O4@oleic acid/menthol:HAc | 60 | drug (1) | urine (10) | HPLC-UV | 4.6 µg·L−1 | 4.6 | 80.3–97.4 | [16] |
| Fe3O4@oleic acid/menthol:octanoic acid | 150 | drug (1) | urine, blood plasma, and beverage (10) | HPLC-UV | 8.5 µg·L−1 | 5.7 | 86.7–97.5 | [17] |
| Fe3O4@SiO2/PCHCl:menthol:decanoic acid | 90 | PAHs (16) | ACN extract of food (6) | GC-MS | 0.27 µg·kg−1 | 10 | 73–92 | [18] |
| Fe3O4@Aliquat336/ChCl:stearic acid | 76 | PAHs (16) | saliva and urine (5) | GC-MS | 0.22 µg·L−1 | 9 | – | [19] |
| Fe3O4@Aliquat336/ChCl:stearic acid | 85 | pesticides (6) | beverages (30) | GC-MS | 40 ng·L−1 | 5.3 | 89.0–105 | [20] |
| Fe3O4@Aliquat336/ChCl:stearic acid | 225 | pesticides (7) | beverages (30) | GC-MS | 24 ng·L−1 | 7.9 | 82–94 | [38] |
| Fe3O4@SiO2/Et4N+:EG | 150 | flavonoids (1) | beverages (10) | HPLC-UV | 3.0 µg·L−1 | 3.8 | 82–98 | [8] |
| Fe3O4@clay/menthol:decanoic acid | 50 | explosives (11) | water and ACN extract of soil (10) | HPLC-UV | 0.91 µg·L−1 ** | 8 | 99–104 | [44] |
| Fe3O4@SDBS/menthol:decanoic acid | 200 | metal (1) | water (50) | FAAS | 2.2 µg·L−1 | 2.6 | 96–99 | [39] |
| SUPRAS-based FFs | ||||||||
| Fe3O4@CLDH(Zn-Fe)/1-dodecanol:toluene | 8 | pesticides (2) | beverages (–) | GC-FID | 2 µg·L−1 | 6.5 | 85.0–96.6 | [28] |
| Fe3O4@oleic acid/decanoid acid:Bu4N+ | 2000 | drug (1) | plasma and urine (30) | spectrofluorimetry | 0.2 µg·L−1 ** | 2.9 | 94.0–106 | [9] |
| Fe3O4@oleic acid/decanoid acid:Bu4N+ | 2000 | pesticides (3) | water and beverages (10) | HPLC-UV | 0.35 µg·L−1 ** | 5.7 | 92.2–110 | [40] |
* RSDmax as maximum relative standard deviation value reported in the study (in %). ** Limit of detection. For the definition of the abbreviations, please refer to the list of abbreviations at the end of the article.
2.1. Alcohols and Water-Based FFs
2.2. IL-Based FFs
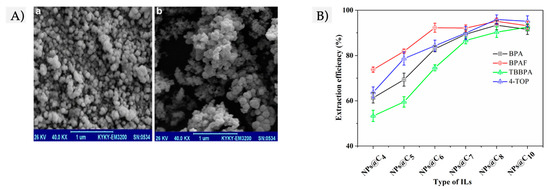
2.3. DES-Based FFs
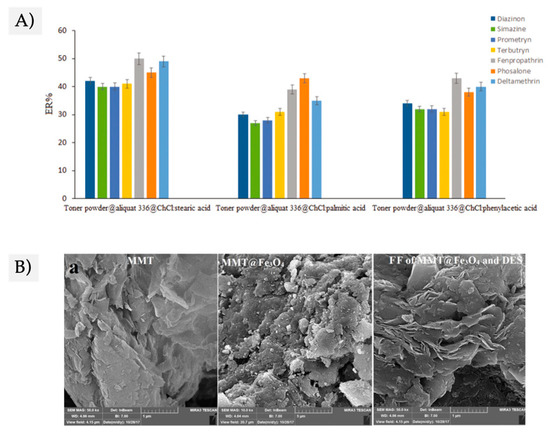
2.4. SUPRAS-Based FFs
SUPRASs are nanostructured liquids produced in colloidal suspensions of amphiphiles, by a self-assembly and coacervation phenomenon [52,53]. These solvents are characterized by presenting different polarity microenvironments, multiple binding sites, while having the possibility of tailoring their properties. Their preparation is divided into two steps: (i) preparation of a colloidal suspension above the critical aggregation concentration, and (ii) modification of the environmental conditions by introducing a coacervation-inducing agent to increase the size of the aggregates, thus forming droplets with different densities. This leads to the formation of a new liquid phase where the droplets are kept as individual entities [52,54]. Although the greenness and outstanding properties of these solvents, their use is still being not widely extended. To the best of our knowledge, only three studies have been reported on the use of SUPRAS-based FFs [9,28,40]. The main SUPRA used is the one prepared with decanoic acid and tetrabutylammonium salt [9,40]. The previously synthesized MNPs are mixed with the SUPRAS under sonication forming the colloidal suspension of the SUPRA-based FF. The loading charge of nanoparticles used is ~0.5%, and the amount of SUPRAS-based FFs used in the analytical applications reported are between 30 µL [40] and 70 µL [9]. These FFs have been used for the extraction of pesticides in water and fruit juice [28,40] and antibiotics in body fluids [9], showing, in all cases, adequate analytical performance.
This entry is adapted from the peer-reviewed paper 10.3390/separations8040047
