In the development of inflammatory bowel disease (IBD), the gut microbiota has been established as a key factor. Recently, metabolomics has become important for understanding the functional relevance of gut microbial changes in disease. Animal models for IBD enable the study of factors involved in disease development. However, results from animal studies may not represent the human situation. The aim of this study was to investigate whether results from metabolomics studies on animal models for IBD were similar to those from studies on IBD patients. Medline and Embase were searched for relevant studies up to May 2017. The Covidence systematic review software was used for study screening, and quality assessment was conducted for all included studies. Data showed a convergence of ~17% for metabolites differentiated between IBD and controls in human and animal studies with amino acids being the most differentiated metabolite subclass. The acute dextran sodium sulfate model appeared as a good model for analysis of systemic metabolites in IBD, but analytical platform, age, and biological sample type did not show clear correlations with any significant metabolites. In conclusion, this systematic review highlights the variation in metabolomics results, and emphasizes the importance of expanding the applied detection methods to ensure greater coverage and convergence between the various different patient phenotypes and animal models of inflammatory bowel disease.
Study Characteristics
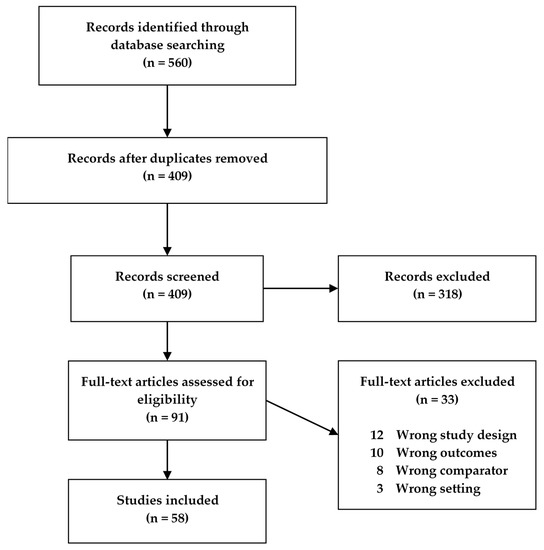
Fifty-eight studies met our search criteria and were included in this review (), of which 32 were human studies, 25 were animal model studies, and one study presented data from both humans and an animal model. The human studies were categorized according to disease (CD, UC, IBD) and age, while the animal model studies were categorized according to model type and age of the animals (). If animals in a study were grouped spanning more than one age group, the study was characterized according to the older age group. Descriptive characteristics for all studies were extracted, with different tables for the human and animal studies, respectively (
Supplementary Tables S1 and S2).
Figure 1. Flowchart of the study screening process for original studies in metabolomics for inflammatory bowel disease (IBD) patients and IBD animal models.
Table 1. Age categories for mouse studies (a) and human studies (b) in the systematic review on metabolomics in inflammatory bowel disease (IBD) patients and IBD animal models.
Quality Assessment
Two sets of quality criteria were used to assess the quality of the human and animal studies, respectively (
Supplementary Tables S3 and S4). Each study was assigned as being of “good”, “medium”, or “poor” quality, based on the amount of quality criteria fulfilled, as presented in . The majority of studies (75%) were of medium quality, while only 9% of all studies were considered good.
Table 2. Quality assessment of studies included in the systematic review on metabolomics in inflammatory bowel disease (IBD) patients and IBD animal models.
Metabolites Differentiated in Inflammatory Bowel Disease (IBD) Cases Versus Healthy Controls in Both Humans and Animal Models
A total of 200 different metabolites were reported as being increased in IBD across all included human studies, while 218 were decreased (). The numbers were higher for the animal studies with a total of 280 different metabolites reported as being increased in IBD, while 253 were decreased. Some metabolites were reported as both increased and decreased in each study type, but the majority was exclusively reported as increased or decreased. Results for human and animal model studies, respectively, are presented in separate tables for metabolites that are increased and decreased in each type of study (
Supplementary Tables S5–S8).
Table 3. Number of differentiated metabolites detected across study types included in the systematic review on metabolomics in inflammatory bowel disease (IBD) patients and IBD animal models.
To assess the similarities in metabolomics findings between study types, metabolites increased or decreased in IBD in both human and animal studies were identified and are presented in ; . Forty-eight metabolites were found to be increased in both types of studies, while 41 metabolites were decreased. This corresponds to 17% of metabolites found increased and 16% of metabolites found decreased in IBD in animal studies also being reported as increased and decreased, respectively, in human IBD studies. Of this subgroup of metabolites, 21 were reported as both increased and decreased, respectively, in IBD including several amino acids, and this overlap can largely be explained by the variation in study details. This leaves 27 metabolites exclusively increased, and 20 metabolites exclusively decreased in IBD in both human and animal studies (in bold in and ).
Table 4. Metabolites significantly increased in inflammatory bowel disease (IBD) vs healthy controls in both humans and animals in the systematic review.
Table 5. Metabolites significantly decreased in inflammatory bowel disease (IBD) vs healthy controls in both humans and animals in the systematic review.
Metabolites of Special Interest
Several tryptophan metabolites were found to be regulated in human studies, animal studies, or both. Kynurenine and quinolinic acid were increased in UC and CD patients, respectively (
Supplementary Table S5). Kynurenine was also found to be increased in DSS (dextran sodium sulfate) and IL-10
-/- mouse models (
Supplementary Table S7), while quinolinic acid was decreased in IL-10
-/- mice along with kynurenic acid and 5-hydroxyindoleacetic acid (
Supplementary Table S8). Additionally, 5-hydroxytryptophan and 3-hydroxykynurenine were also increased in DSS and IL-10
-/- mouse models, respectively (
Supplementary Table S7). Conflicting observations were made for tryptophan itself, which was reported to be both increased and decreased in human studies as well as the DSS mouse model (see and ). SCFAs were reported to be regulated in numerous human IBD studies, although some results were conflicting. Formic acid and acetic acid were thus observed to be both increased and decreased in CD and UC patients, depending on the study (
Supplementary Tables S5 and S6). However, propionic acid, butanoic acid, isobutyric acid, and pentanoic acid were all observed to be decreased in CD and UC patients (
Supplementary Table S6). Interestingly, only animal studies using the acute DSS mouse model or the TNBS (2,4,6-trinitrobenzenesulfonic acid) rat model reported differentiated levels of SCFAs (
Supplementary Tables S7 and S8). Acetic acid was decreased in the DSS model, while butanoic acid was decreased in the TNBS model (
Supplementary Table S8). Dong et al. [
13] also observed butanoic acid to be decreased, but only on the first day of DSS, after which it was increased throughout the experiment.
Included Studies Are Characterized by Great Variation in the Key Experimental Elements
A metabolomics study consists of several different key experimental elements that can vary between studies. Here, these elements are the experimental subjects (disease subtype for the human studies and species, strain, and type of model for the animal studies), biological sample type, analysis methodology, and age of experimental subjects/study population. Large variations in these elements can make it difficult to compare results across the different studies and thereby difficult to draw any overall assumptions on the topic in question.
To clearly elucidate the large variation between the different studies included in this review, we tallied up the number of studies containing the different variants of each key experimental element in animal studies and human studies, respectively (see and ). Looking at and , it becomes immediately clear that there could be a very high degree of variation between studies as a result of the different elements applied in the studies. For the animal studies (), three different species with a total of 11 different mouse and rat strains were used along with eight different IBD animal models, three main analytical platforms, 13 different sample types, and four different age groups across the 26 studies. The variation in study population and sample type was less for the human studies (), however seven different analytical platforms were applied, giving rise to a considerable heterogeneity across the human studies.
Table 6. Overview of the variation in key experimental elements in animal model studies and the number of studies containing the different versions of each element.
Table 7. Overview of the variation in key experimental elements in human studies and the number of studies containing the different versions of each element in the systematic review on metabolomics in inflammatory bowel disease (IBD) patients and IBD animal models.
A few studies did, however, share a high degree of similarity in experimental factors. Animal studies by Shiomi et al., Gu et al., and Wang et al. all used C57BL/6J mice from the same age group for a 3% DSS model as well as using gas chromatography-mass spectrometry (GC-MS) to detect metabolites in serum and colon samples (see
Supplementary Table S2) [
17,
37,
55], although it is worth noting that Gu et al. and Wang et al. belong to the same department at Kobe University, Japan. Equally, two studies by the same first author also shared a similar degree of similarity using an IL10
-/- model [
33,
40]. For the human studies, two studies used proton nuclear magnetic resonance (
1H-NMR) to detect metabolites in serum samples from CD and UC patients of 18-60+ years of age [
15,
43], while two other studies detected metabolites in serum samples from CD and UC patients in the >1–60+ age groups using GC-MS [
20,
24]. The authors of the latter two studies are also from the same department and even co-authors the other study, again underlining the difficulties at present comparing studies from different research groups.
Differentiation of Metabolites According to Key Experimental Elements
We found that in both human and animal studies, the vast majority of the metabolites were detected by more than one analytical platform (
Supplementary Table S9). The study subjects in most of the human studies spanned all age groups from very early onset and young to old, making it difficult to differentiate metabolite detection between age groups in the human studies. However, most metabolites were generally detected in more than one age group in the animal studies, suggesting that age is not a deciding factor when it comes to the metabolome. Nevertheless the amino acid isoleucine stood out, as it was increased only in human subjects above 18 years of age and in mice of >8–24 weeks. One of the animal studies that reported increased levels of isoleucine also included animals of 1 week, but the amino acid was not significantly altered in this group [
26].
The subgroup of metabolites differentiated in both study types was sorted according to the biological sample types in which they were detected (
Supplementary Table S9). This allowed us to examine any parallels between human and animal studies. Many metabolites were found in several different sample types in both humans and animals, but not necessarily the same. For example, alanine was increased in serum [
24] and feces [
4,
25] from humans and in colon [
17] and plasma [
26] from mice, but it was decreased in urine [
43] and colon [
35,
36] in humans and serum [
16] and urine [
38] in animals, illustrating the differences observed for many metabolites (
Supplementary Table S9). The highest similarity to human studies was observed with the acute DSS mouse model (
Supplementary Table S9). Since this model was used in almost half of the included animal studies, this finding is not surprising. However, only five of the acute DSS mouse model studies analyzed serum samples, but still 11 of the increased and 11 of the decreased metabolites were detected in serum samples from both humans and the DSS mouse model. A total of 34 and 29 different metabolites were reported as increased and decreased in IBD, respectively, in serum samples from the acute DSS mouse model. This means that 32% of the increased metabolites and 38% of the decreased metabolites in serum samples from the acute DSS mouse model were reported to be correspondingly differentiated in the human studies. Conversely, the acute DSS mouse model could account for 16% (22 out of 136 metabolites) of the overall metabolite changes observed in serum of IBD patients. This could suggest serum samples from the acute DSS mouse model as having good translational potential when analyzing systemic metabolites in IBD.
Correlation between Animal Models and IBD Subtypes
For all the metabolites significantly differing in both human and animal studies, it was investigated if some animal models were specifically good models for CD or UC when it comes to metabolomics (
Supplementary Table S9). Most of the models had similarities with both CD and UC. For instance, regarding metabolites decreased in the IL10
−/– mouse model, glucose was also decreased in CD, while leucine was decreased in UC, and trimethylamine in both CD and UC. The TNF
ΔARE/WT model only had similarities with UC, but this could easily be due to the fact that only one study with this model was included. Overall, this indicates that the metabolomes of the animal models included in this review are not correlated specifically to CD or UC.
Metabolite Classifications
All metabolites differentiated between IBD cases and controls in either humans or animals were sorted into metabolite subclasses according to the classification system used in The Human Metabolome Database (
www.hmdb.ca) (
Supplementary Tables S10 and S11). The most differentiated subclass was “amino acids, peptides, and analogues” in both human and animal studies, representing approximately 16% of all differentiated metabolites reported. “Fatty acids and conjugates” as well as “carbohydrates and carbohydrate conjugates” were also among the most differentiated in both human and animal study types. “Glycerophosphocholines” were also differentiated in both, but to a much larger extent in animal studies. In general, different kinds of lipids were reported more frequently as differentiated in IBD in animal studies compared to human studies. Metabolites from 142 different subclasses were reported as differentiated between IBD and controls overall. Of these, 47 were differentiated in both human and animal studies, while 48 and 47 differentiated subclasses were unique to human and animal studies, respectively. This shows a large gap between the type of metabolites that are investigated and detected in the two study types, as only a third of the total amount of differentiated subclasses are reported in both.
When focusing on the metabolites differentiated in IBD in both human and animal studies, they represented a total of 25 subclasses overall. Metabolites from nine different subclasses were present among both the increased and decreased metabolites, while eight subclasses were exclusively increased and decreased, respectively.
- Lin, H.M.; Edmunds, S.J.; Helsby, N.A.; Ferguson, L.R.; Rowan, D.D. Nontargeted urinary metabolite profiling of a mouse model of crohn’s disease. J. Proteome Res. 2009, 8, 2045–2057. [Google Scholar] [CrossRef] [PubMed]
- Le Gall, G.; Noor, S.O.; Ridgway, K.; Scovell, L.; Jamieson, C.; Johnson, I.T.; Colquhoun, I.J.; Kemsley, E.K.; Narbad, A. Metabolomics of fecal extracts detects altered metabolic activity of gut microbiota in ulcerative colitis and irritable bowel syndrome. J. Proteome Res. 2011, 10, 4208–4218. [Google Scholar] [CrossRef] [PubMed]
- Balasubramanian, K.; Kumar, S.; Singh, R.R.; Sharma, U.; Ahuja, V.; Makharia, G.K.; Jagannathan, N.R. Metabolism of the colonic mucosa in patients with inflammatory bowel diseases: An in vitro proton magnetic resonance spectroscopy study. Magn. Reson. Imaging 2009, 27, 79–86. [Google Scholar] [CrossRef] [PubMed]
- Sharma, U.; Singh, R.R.; Ahuja, V.; Makharia, G.K.; Jagannathan, N.R. Similarity in the metabolic profile in macroscopically involved and un-involved colonic mucosa in patients with inflammatory bowel disease: An in vitro proton (1H) MR spectroscopy study. Magn. Reson. Imaging 2010, 28, 1022–1029. [Google Scholar] [CrossRef]
- Gu, X.; Song, Y.; Chai, Y.; Lu, F.; Gonzalez, F.J.; Fan, G.; Qi, Y. GC-MS metabolomics on PPARalpha-dependent exacerbation of colitis. Mol. Biosyst. 2015, 11, 1329–1337. [Google Scholar] [CrossRef]
- Martin, F.P.J.; Lichti, P.; Bosco, N.; Brahmbhatt, V.; Oliveira, M.; Haller, D.; Benyacoub, J. Metabolic phenotyping of an adoptive transfer mouse model of experimental colitis and impact of dietary fish oil intake. J. Proteome Res. 2015, 14, 1911–1919. [Google Scholar] [CrossRef]
- Fathi, F.; Majari-Kasmaee, L.; Mani-Varnosfaderani, A.; Kyani, A.; Rostami-Nejad, M.; Sohrabzadeh, K.; Naderi, N.; Zali, M.R.; Rezaei-Tavirani, M.; Tafazzoli, M.; et al. 1H NMR based metabolic profiling in Crohn’s disease by random forest methodology. Magn. Reson. Chem. MRC 2014, 52, 370–376. [Google Scholar] [CrossRef]
- Lin, H.M.; Barnett, M.P.G.; Roy, N.C.; Joyce, N.I.; Zhu, S.; Armstrong, K.; Helsby, N.A.; Ferguson, L.R.; Rowan, D.D. Metabolomic analysis identifies inflammatory and noninflammatory metabolic effects of genetic modification in a mouse model of Crohn’s disease. J. Proteome Res. 2010, 9, 1965–1975. [Google Scholar] [CrossRef]
- Qu, C.; Yuan, Z.W.; Yu, X.T.; Huang, Y.F.; Yang, G.H.; Chen, J.N.; Lai, X.P.; Su, Z.R.; Zeng, H.F.; Xie, Y.; et al. Patchouli alcohol ameliorates dextran sodium sulfate-induced experimental colitis and suppresses tryptophan catabolism. Pharmacol. Res. 2017, 121, 70–82. [Google Scholar] [CrossRef]
- Kominsky, D.J.; Keely, S.; MacManus, C.F.; Glover, L.E.; Scully, M.; Collins, C.B.; Bowers, B.E.; Campbell, E.L.; Colgan, S.P. An endogenously anti-inflammatory role for methylation in mucosal inflammation identified through metabolite profiling. J. Immunol. 2011, 186, 6505–6514. [Google Scholar] [CrossRef] [PubMed]
- Alonso, A.; Julia, A.; Vinaixa, M.; Domenech, E.; Fernandez-Nebro, A.; Canete, J.D.; Ferrandiz, C.; Tornero, J.; Gisbert, J.P.; Nos, P.; et al. Urine metabolome profiling of immune-mediated inflammatory diseases. BMC Med. 2016, 14, 133. [Google Scholar] [CrossRef] [PubMed]
- Murdoch, T.B.; Fu, H.; MacFarlane, S.; Sydora, B.C.; Fedorak, R.N.; Slupsky, C.M. Urinary metabolic profiles of inflammatory bowel disease in interleukin-10 gene-deficient mice. Anal. Chem. 2008, 80, 5524–5531. [Google Scholar] [CrossRef] [PubMed]
- Lu, K.; Knutson, C.G.; Wishnok, J.S.; Fox, J.G.; Tannenbaum, S.R. Serum metabolomics in a helicobacter hepaticus mouse model of inflammatory bowel disease reveal important changes in the microbiome, serum peptides, and intermediary metabolism. J. Proteome Res. 2012, 11, 4916–4926. [Google Scholar] [CrossRef] [PubMed]
- Johnson, J.C.; Schmidt, C.R.; Shrubsole, M.J.; Billheimer, D.D.; Joshi, P.R.; Morrow, J.D.; Heslin, M.J.; Washington, M.K.; Ness, R.M.; Zheng, W.; et al. Urine PGE-M: A Metabolite of Prostaglandin E2 as a Potential Biomarker of Advanced Colorectal Neoplasia. Clin. Gastroenterol. Hepatol. 2006, 4, 1358–1365. [Google Scholar] [CrossRef]
- Willenberg, I.; Ostermann, A.I.; Giovannini, S.; Kershaw, O.; Von Keutz, A.; Steinberg, P.; Schebb, N.H. Effect of acute and chronic DSS induced colitis on plasma eicosanoid and oxylipin levels in the rat. Prostaglandins Other Lipid Mediat. 2015, 120, 155–160. [Google Scholar] [CrossRef] [PubMed]
- Williams, H.R.T.; Cox, I.J.; Walker, D.G.; North, B.V.; Patel, V.M.; Marshall, S.E.; Jewell, D.P.; Ghosh, S.; Thomas, H.J.W.; Teare, J.P.; et al. Characterization of inflammatory bowel disease with urinary metabolic profiling. Am. J. Gastroenterol. 2009, 104, 1435–1444. [Google Scholar] [CrossRef]
- Ahmed, I.; Greenwood, R.; Costello, B.; Ratcliffe, N.; Probert, C.S. Investigation of faecal volatile organic metabolites as novel diagnostic biomarkers in inflammatory bowel disease. Aliment. Pharmacol. Ther. 2016, 43, 596–611. [Google Scholar] [CrossRef]
- Stephens, N.S.; Siffledeen, J.; Su, X.; Murdoch, T.B.; Fedorak, R.N.; Slupsky, C.M. Urinary NMR metabolomic profiles discriminate inflammatory bowel disease from healthy. J. Crohn’s Colitis 2013, 7, e42–e48. [Google Scholar] [CrossRef]
- Hong, Y.S.; Ahn, Y.T.; Park, J.C.; Lee, J.H.; Lee, H.; Huh, C.S.; Kim, D.H.; Ryu, D.H.; Hwang, G.S. 1H NMR-based metabonomic assessment of probiotic effects in a colitis mouse model. Arch. Pharmacal Res. 2010, 33, 1091–1101. [Google Scholar] [CrossRef]
- De Preter, V.; Joossens, M.; Ballet, V.; Shkedy, Z.; Rutgeerts, P.; Vermeire, S.; Verbeke Phd, K. Metabolic profiling of the impact of oligofructose-enriched inulin in Crohn’s disease patients: A double-blinded randomized controlled trial. Clin. Transl. Gastroenterol. 2013, 4, e30. [Google Scholar] [CrossRef] [PubMed]
- Williams, H.R.; Cox, I.J.; Walker, D.G.; Cobbold, J.F.; Taylor-Robinson, S.D.; Marshall, S.E.; Orchard, T. Differences in gut microbial metabolism are responsible for reduced hippurate synthesis in Crohn’s disease. Gastroenterology 2010, 138, S579. [Google Scholar] [CrossRef]
- Yau, Y.Y.; Leong, R.W.L.; Shin, S.; Bustamante, S.; Pickford, R.; Hejazi, L.; Campbell, B.; Wasinger, V.C. Bimodal plasma metabolomics strategy identifies novel inflammatory metabolites in inflammatory bowel diseases. Discov. Med. 2014, 18, 113–124. [Google Scholar] [PubMed]
- Wang, R.; Gu, X.; Dai, W.; Ye, J.; Lu, F.; Chai, Y.; Fan, G.; Gonzalez, F.J.; Duan, G.; Qi, Y. A lipidomics investigation into the intervention of celastrol in experimental colitis. Mol. Biosyst. 2016, 12, 1436–1444. [Google Scholar] [CrossRef] [PubMed]
- Kiesler, P.; Fuss, I.J.; Strober, W. Experimental Models of Inflammatory Bowel Diseases. Cell. Mol. Gastroenterol. Hepatol. 2015, 1, 154–170. [Google Scholar] [CrossRef] [PubMed]
- Bamias, G.; Arseneau, K.O.; Cominelli, F. Mouse models of inflammatory bowel disease for investigating mucosal immunity in the intestine. Curr. Opin. Gastroenterol. 2017, 33, 411–416. [Google Scholar] [CrossRef]
This entry is adapted from the peer-reviewed paper 10.3390/ijms21113856