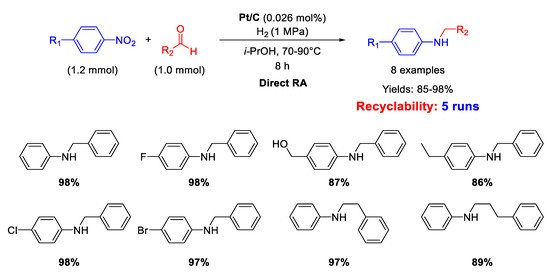
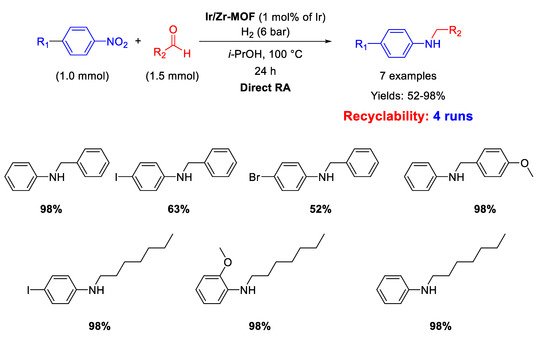
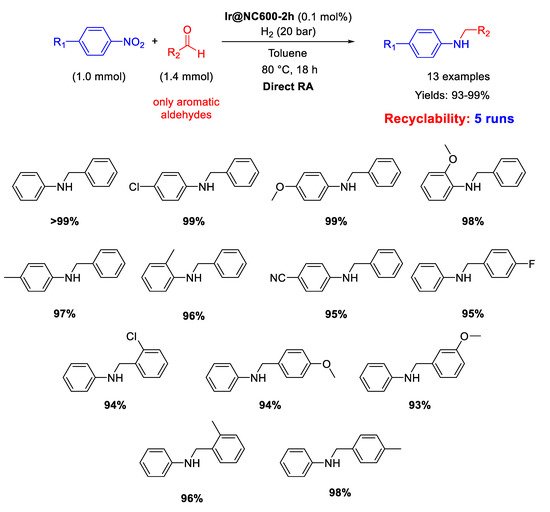
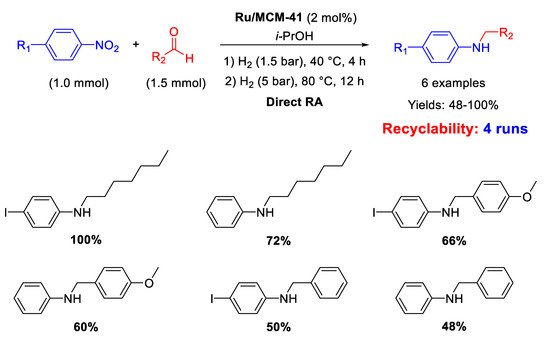
Palladium is by far the most versatile and widely used metal among transition metals employed in catalysis. In all cases, for this kind of catalysts the catalytically active species have proven to be Pd nanoparticles (NPs) supported on various insoluble matrices. Generally, the catalysts kept their activity and selectivity over several subsequent runs and the reported studies differ from each other for the employed reductant and/or the dimension distribution of the metal NPs.
As recently highlighted by Sukhorukov [
22], the RA of carbonyl derivatives with nitro compounds can viewed as an old reaction because more than 80 years ago Major [
39] and Emerson et al. [
40,
41] reported on the coupling of nitroarenes with aldehydes under catalytic hydrogenation over platinum oxide (PtO
2: Adams’ catalyst [
42]) and Raney Nickel catalyst, respectively. The proposed protocols were generally applicable to both aromatic and aliphatic aldehydes but in most cases low or moderate yields in secondary amines were achieved.
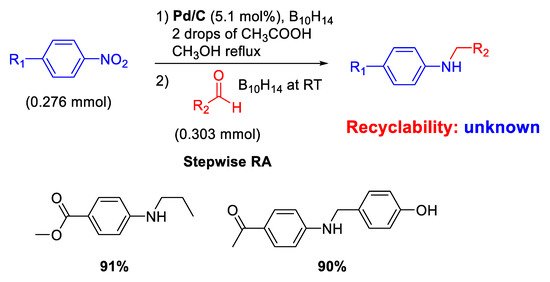
The modern revaluation for the employment of heterogeneous catalysis for carrying out RA of aldehydes with nitroarenes started only in the 2000 s with the use of commercial source of palladium catalyst (Pd/C). In fact, Yoon and co-workers reported that Pd/C promoted the conversion of nitroaromatics and aldehydes into
N-alkylaminoaromatics employing decaborane (B
10H
14) as a reducing agent (
Scheme 4) [
43].
Scheme 4. Stepwise RA catalyzed by Pd/C in the presence of decaborane.
A simple and efficient stepwise RA was achieved throughout reduction of nitroarenes followed by reductive amination. Although the proposed protocol resulted chemoselective and could also be extended to ketones, neither investigations of the scope of reaction nor data on recyclability of catalyst were reported. However, in a follow-up paper [
44], the same group was able to extend their protocol under excess of formaldehyde for the synthesis of tertiary amines in high yields.
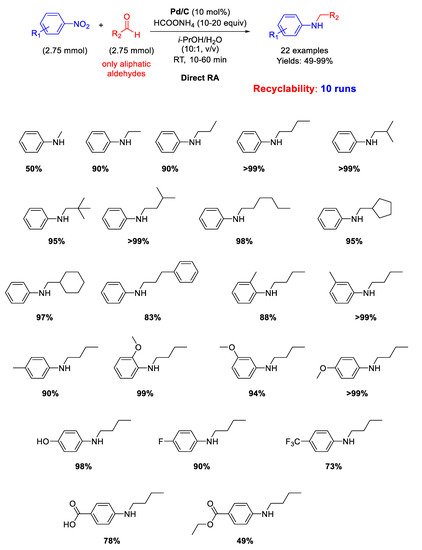
In 2007, Rhee and co-workers proposed another protocol for DRA accelerated by Pd/C [
45]. Twenty-two mono-
N-alkylation of anilines in moderate to excellent yield (49%–99%) were obtained at room temperature by starting from nitroarenes and aliphatic aldehydes in aqueous 2-propanol solvent with ammonium formate (HCOONH
4), as in situ hydrogen donor (
Scheme 5). Moreover, it was demonstrated that Pd/C catalyst could be reused for 10 cycles. The proposed protocol, although facile and environmentally benign, is unfortunately not applicable to aromatic aldehydes.
Scheme 5. DRA catalyzed by Pd/C in the presence of ammonium formate.
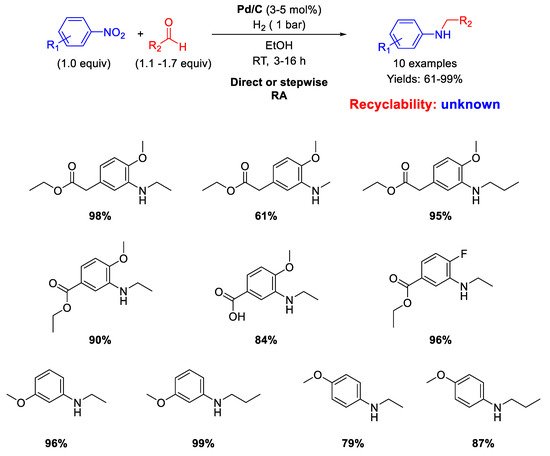
Another RA of nitroarenes with aliphatic aldehydes catalyzed by Pd/C which furnished good to excellent isolated yields (61%−99%) was proposed by Sydnes and Isobe in 2008 (
Scheme 6) [
46].
Scheme 6. RA catalyzed by Pd/C under H2.
However, when formaldehyde was used, formation of substantial amounts of the corresponding tertiary amine was observed, but in this case the authors found out that a stepwise RA could be applied with satisfactory yield. No data on the recyclability of the catalyst were given.
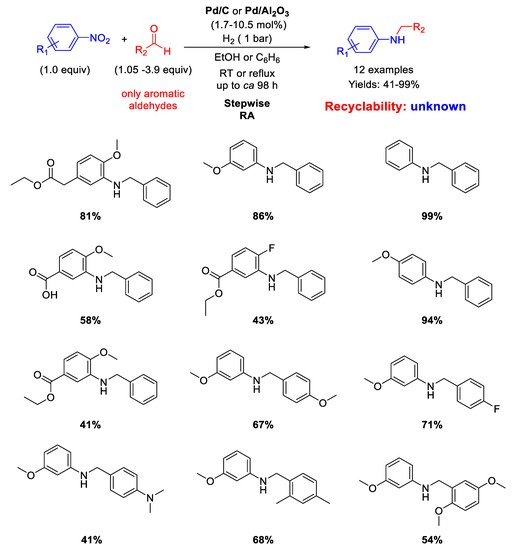
Sydnes and co-workers, in a follow-up paper [
47], extended the scope of their stepwise RA to aromatic aldehydes (
Scheme 7). Isopropanol was used as solvent in place of ethanol. However, for the substrates giving a particularly slow imine-forming step, benzene (or benzene/ethanol) under refluxing conditions was employed as solvent for improving the total yields. In these cases, Pd/C was replaced with Pd/Al
2O
3 due to the greater stability of the latter catalyst at high temperatures with respect to Pd/C [
48]. Sometimes, it was found beneficial to add 0.5 equiv (with respect to nitroarene) of triethylamine before the final imine reduction. Twelve benzylated anilines in yields ranging from 41% to 99% were obtained and some case of low yield was attributed to the substitution patterns associated with the aromatic aldehydes. Once again, no investigations on the recyclability of the catalyst were made.
Scheme 7. Stepwise RA catalyzed by Pd/C or Pd/Al2O3 under H2.
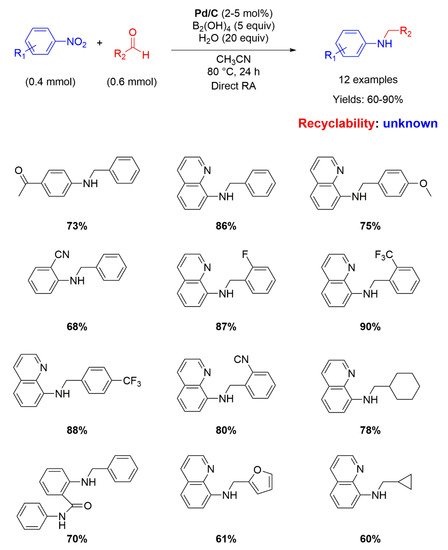
Nine years later, Pd/C has been employed again as catalyst for RA, but this time in the direct (not stepwise) fashion. Indeed, Zhou and co-workers used water (20 equiv.) as the hydrogen source mediated by diboronic acid (B
2(OH)
4 5 equiv.) in acetonitrile at 80° for 24 h for reacting various nitroarenes with different aromatic and aliphatic aldehydes, affording yields ranging from 60 to 90% (
Scheme 8) [
49]. The yields were higher when the substituent in the benzaldehyde ring was electron withdrawing. The role of water as hydrogen donor was verified by using D
2O as transfer agent. No recyclability studies on the catalytic system have been carried out.
Scheme 8. DRA catalyzed by Pd/C with B2(OH)4 and H2O.
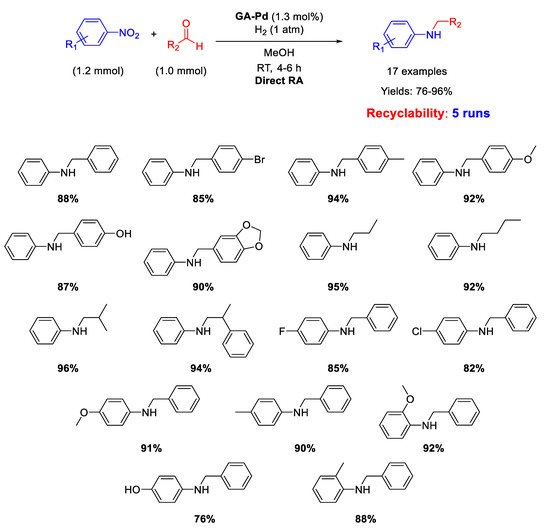
Another important kind of palladium-based heterogeneous catalysts effective for RA is constituted by Pd NPs supported onto organic naturally derived matrices. Pd NPs supported on Gum-Acacia colloids (GA-Pd) have been reported by Sreedhar and co-workers for successful Pd-catalysed DRA applied to both aromatic and aliphatic aldehydes [
50]. The supported Pd NPs with a narrow size distribution (ranging from 6 to 12 nm) resulted active and selective towards the formation of secondary anilines for at least five subsequent cycles at room temperature, in methanol solvent, using 1 atm dihydrogen as the reductant (
Scheme 9). The yields were quite high (76–96%).
Scheme 9. DRA catalyzed by Gum-Acacia supported Pd NPs.
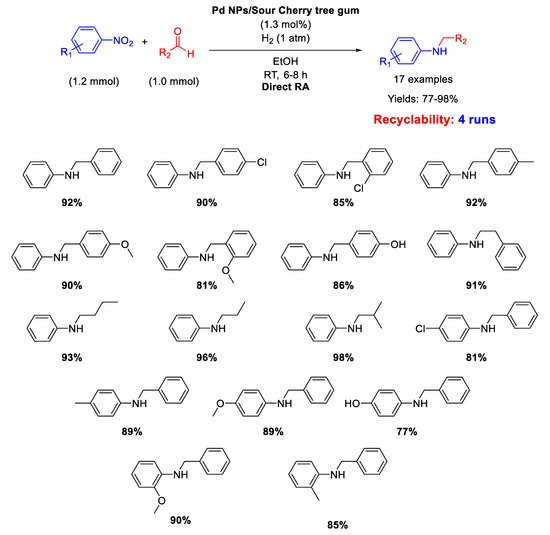
A similar catalyst has been prepared by Nasrollahzadeh, that reported on the synthesis of Pd nanoparticles, with size ranging from 2.5 to 15 nm, stabilized on the surface of Sour Cherry tree gum and their use in catalysis for the direct one-pot RA in ethanol at room temperature under 1 atm H
2 [
51]. The catalytic system was active, selective, and recyclable for up to four subsequent runs (
Scheme 10).
Scheme 10. DRA catalyzed by Pd NPs supported onto Sour Cherry tree gum.
Pd NPs stabilised by naturally occurring cinchona-based alkaloids, such as cinchonidine, cinchonine, quinine and quinidine, have been reported as catalysts for the one-pot three-step sequential process for the synthesis of
N-substituted anilines under 3 bar dihydrogen in glycerol at 100 °C [
52]. The yields of secondary anilines ranged from 62 to 89%. No recyclability experiments have been carried out on the RA with this catalytic system.
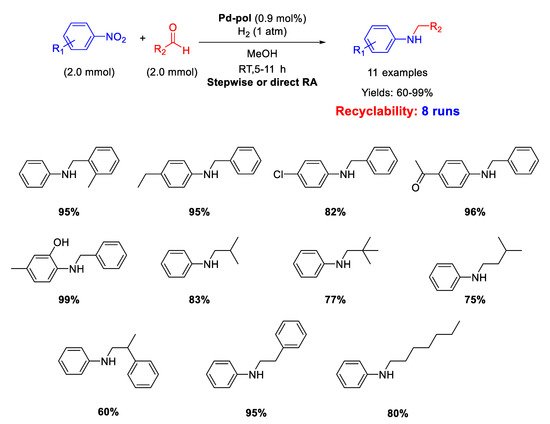
Dell’Anna and co-workers reported the catalytic activity and recyclability of polymer supported Pd nanoparticles (Pd-pol) obtained by copolymerization of Pd(AAEMA)
2 [AAEMA
− = deprotonated form of 2-(acetoacetoxy) ethyl methacrylate] with methacrylic comonomers, followed by reduction under reaction conditions [
53]. Pd-pol also proved to accelerate other kinds of chemical reactions such as reductions, oxidations and esterification. [
54,
55,
56,
57,
58,
59] However, the Pd-pol catalysed reductive amination was successful in one step by using aliphatic aldehydes only. In fact, when benzaldehydes were employed, the one-pot reaction was carried out in three steps to avoid benzyl alcohol formation (
Scheme 11). The catalytically active species were in situ formed Pd NPs with a narrow size distribution centred at 5 nm in diameter, whose morphology remained almost unchanged during reaction. The catalyst resulted recyclable for at least eight subsequent runs without loss of activity and selectivity. The catalytic reactions were performed under mild conditions (1 atm H
2, room temperature, methanol as the solvent) by using equimolar amounts of nitroarene and aldehyde.
Scheme 11. RA catalyzed by Pd-pol.
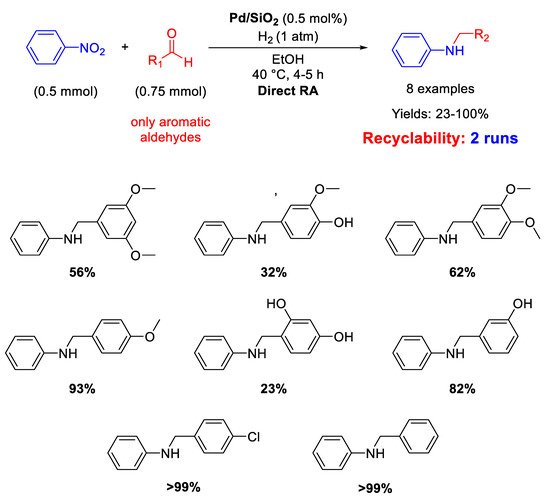
Pd NPs dispersed onto inorganic supports, such as silica and zeolite, have been successfully used as catalysts for DRA. In details, Ma and co-workers reported that SiO
2 supported Pd nanoparticles with a mean diameter of 5 ± 2 nm were active and selective in the one-pot DRA starting from benzaldehydes and nitroarenes under H
2, at 40 °C in ethanol (
Scheme 12) [
60]. The catalytic system gave poor yield (23%) in the desired product, when the benzaldehyde had a substituent in
ortho position, due to steric hindrance. The catalyst was recyclable for at least two subsequent runs.
Scheme 12. DRA catalyzed by Pd/SiO2.
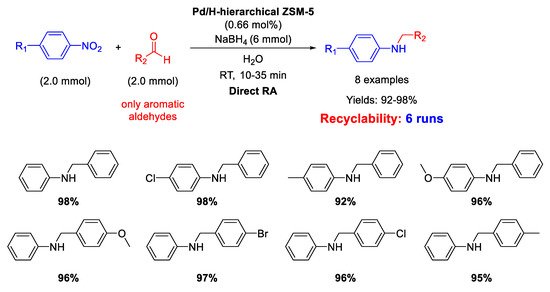
On the other hand, in 2016, Pd NPs incorporated in ZSM-5 zeolite (ZSM-5 = Zeolite Socony Mobil–5) with a hierarchical structure (Pd/H-hierarchical ZSM-5) have been found effective catalyst for DRA starting from nitroarene compounds, using aldehyde in the presence of NaBH
4 as a reducing agent (molar ratio of reductant/substrate = 3) and water as the solvent at room temperature (
Scheme 13) [
61]. Transmission Electron Microscopy (TEM) images showed the formation of an ordered mesostructure in H-hierarchical ZSM-5, supporting Pd NPs with an average diameter of 3–6 nm dispersed in the matrix. The activity of the catalyst was quite high, reaching excellent yields (92%–98%) in short time (10–35 min). Recyclability tests demonstrated that after 6 runs with the same catalyst, the yield into the secondary amine passed from 98% (1
st run) to 82% (6
th run), and a slight palladium leaching into solution was observed. However, the very interesting aspect of this catalytic system stands in both the use of water as the solvent (which notoriously inhibits the imine formation via condensation of aniline and carbonyl group) and the employing of NaBH
4 as the reductant, that normally quickly reduces the C=O polar double bond of the aldehyde, even in the absence of metal catalyst, thus subtracting aldehyde for the condensation step reaction. The acid sites and the mesoporous structure of the hierarchical zeolite allowed to overcome all problems associated with the use of water and NaBH
4. In addition, the authors observed that the reaction rate increased when an electron-withdrawing group was located on the nitroaromatic ring (because the tendency of nitro groups to accept hydride ions increased), or when an electron-donating group was placed on the benzaldehyde ring (because the electron donating group stabilized the protonated carbonyl bond intermediate).
Scheme 13. DRA catalyzed by Pd/H- hierarchical ZSM-5.
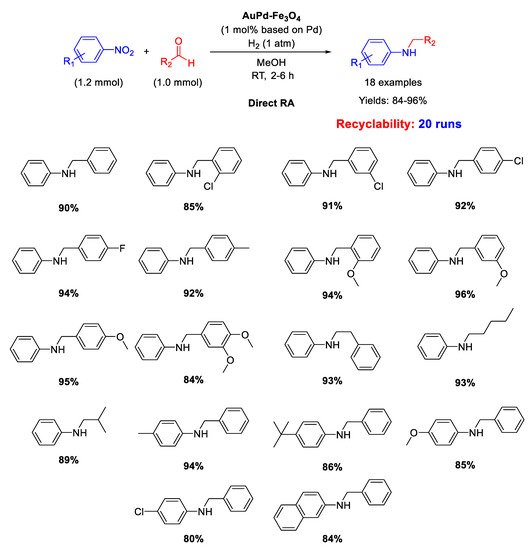
Several research groups synthetized suitable Pd catalysts active in the RDA, by supporting Pd NPs onto Fe3O4, very often using also SiO2 or other inorganic or organic functionalized supports, aiming at taking advantage of the magnetic features of magnetite for recyclability purposes. In all these cases, nitroarene was always used in higher equivalent with respect to aldehyde.
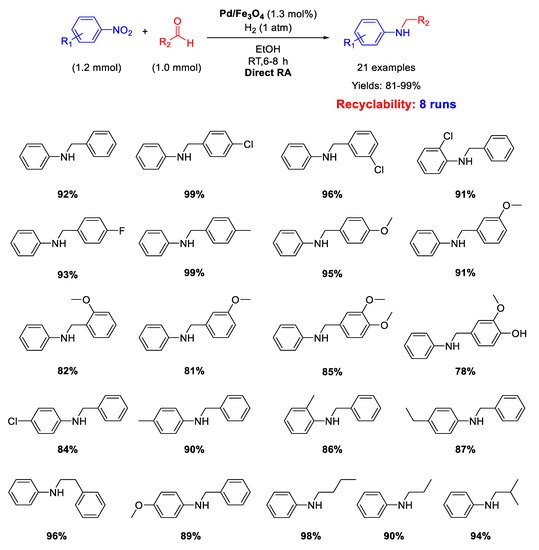
Ma research group reported on the synthesis of Pd/Fe
3O
4 nanocomposites constituted by Pd NPs of 3 nm ca. in size dispersed onto magnetite NPs with an average size of 20 nm [
62]. These interesting nano-catalysts were able to catalyze the DRA between benzaldehyde and various nitrobenzenes under hydrogen (1 atm) at room temperature (
Scheme 14). Ethanol and methanol were both good solvents, but EtOH was preferred to MeOH from the environmental point of view. The catalyst was easily removed from the reaction mixture thanks to its magnetic features, and then recycled for up eight times without loss of activity and selectivity.
Scheme 14. DRA catalyzed by Pd/Fe3O4.
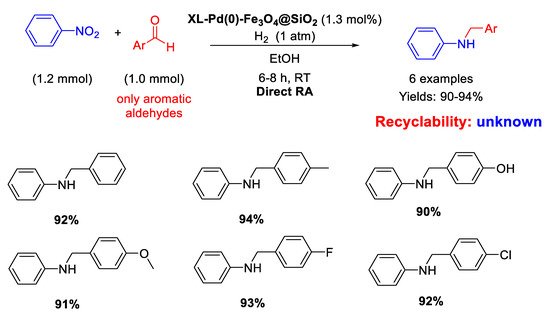
In 2014, following their study on Pd nanoparticles supported onto magnetite seeds, Li and Ma and co-workers synthesized with the reverse microemulsion method uncommon nano-catalysts constituted by Fe
3O
4@SiO
2 core-shell nanospheres of 35 nm in size, which were functionalized with ethylenediamine groups in turn reacted with succinyl chloride to support Pd NPs of 5 nm in diameter, homogeneously distributed onto the external matrix surface (XL-Pd(0)–Fe
3O
4@SiO
2) [
63]. These nano-catalysts resulted active and selective in the DRA at room temperature in ethanol under 1 atm H
2 starting from nitrobenzene with various benzaldehydes (
Scheme 15). The catalyst was easily recovered at the end of the reaction for further re-uses. The authors noticed that the presence of an electron withdrawing group in
para- position in the aldehyde ring caused a reduction in the reaction time, while the presence of an electron donating group led to increased reaction times, according to the mechanism proposed in
Scheme 3.
Scheme 15. DRA catalyzed by XL-Pd(0)–Fe3O4@SiO2.
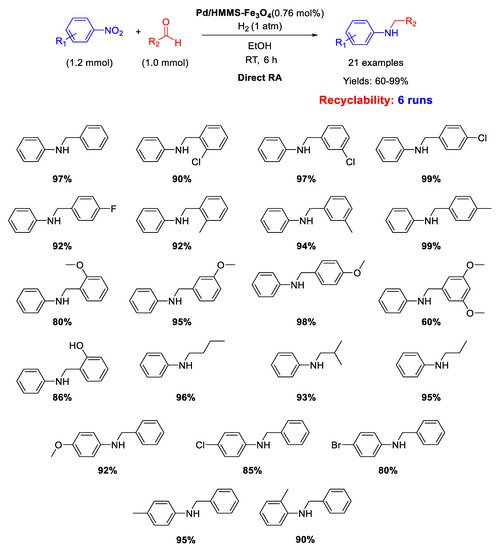
Similar catalytic systems were developed by Li and co-workers for DRA catalyzed by Pd NPs supported on hollow magnetic mesoporous spheres containing Fe
3O
4 nanoparticles embedded in a mesoporous silica shell (Pd/HMMS-Fe
3O
4) [
64]. The Pd NPs with a mean diameter of 7–8 nm resulted homogeneously dispersed onto the surface of the magnetite coated hollow silica sphere. The catalyst could be easily recovered at the end of the reaction, due to its magnetic features, and was reused for six times, without loss of activity. The catalytic tests were carried out in ethanol under dihydrogen atmosphere at room temperature (
Scheme 16) and the obtained yields were quite high (60%–99%).
Scheme 16. DRA catalyzed by Pd/HMMS-Fe3O4.
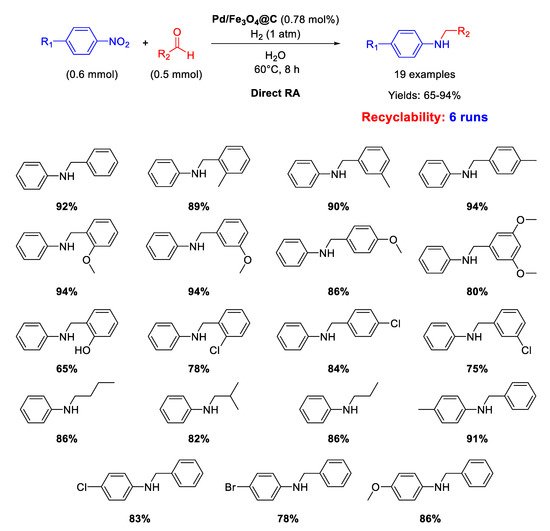
Li’s research group prepared other core–shell Fe
3O
4@C nanoparticles (of 160 nm in diameter) having a carbon layer coating of 10 nm in thickness onto which Pd NPs of 3–8 nm in size were homogeneously dispersed (Pd/Fe
3O
4@C) [
65]. These nano materials represent a rare example of catalysts active for DRA performed in water. Since it was proven the existence of carboxyl groups in the outer carbon layer of these nanocomposites, in our opinion, this occurrence was beneficial for the imine formation step, being the latter favored by acid environment [
36], thus allowing the use of water as the solvent. The catalytic experiments were carried out in water at room temperature or at 60 °C under 1 atm dihydrogen. The catalyst could be recycled for up to six subsequent runs (
Scheme 17).
Scheme 17. DRA catalyzed by Pd/Fe3O4@C.
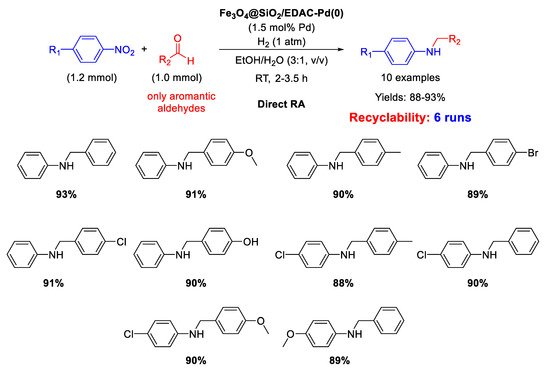
Another example of DRA catalysed by Pd NPs supported onto magnetite based nanocomposites has been reported by Paul’s group, that synthesized in 2016 Pd(0) nanoparticles dispersed onto ethylene diamine functionalized inorganic/organic magnetic composite [
66]. In details, Fe
3O
4 nanoparticles were prepared with the co-precipitation method, and were coated with silica spheres with the sol-gel process. The final inorganic/organic hybrid magnetite nanoparticles were arranged via formation of covalent bonds between ethylene diamine functionalized cellulose (EDAC) and silica coated Fe
3O
4, and then treated with PdCl
2 and in situ reduced using NaBH
4 to get Fe
3O
4@SiO
2/EDAC–Pd(0). TEM analyses showed that Pd NPs diameters ranged from 10 to 15 nm and the nano catalysts resulted active, selective, and recyclable for at least six runs in the DRA under 1 atm H
2, using ethanol/water (3/1,
v/
v) as the solvent (
Scheme 18). This is another rare example of metal catalysed DRA performed in aqueous medium.
Scheme 18. DRA catalyzed by Fe3O4@SiO2/EDAC–Pd(0).
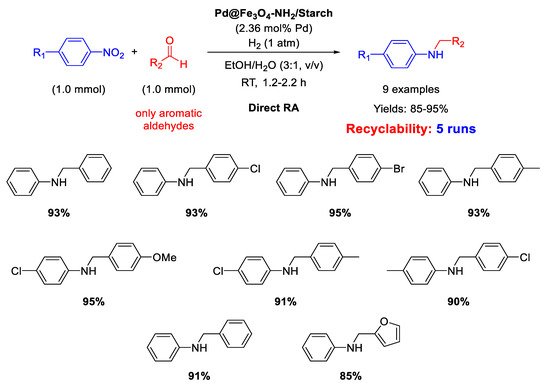
In 2017, Paul’s group synthesized another magnetite-based core-shell nanocomposite, useful for catalyzing DRA reaction, namely Pd@Fe
3O
4-NH
2/Starch, which was slightly different from the previously discussed one, because starch was used in the place of cellulose and the coating of silica layer was avoided [
67]. Pd NPs deposited onto the amine functionalized starch surface had a mean dimension centered at 8.5 nm in diameter. As for Fe
3O
4@SiO
2/EDAC–Pd(0), Pd@Fe
3O
4-NH
2/Starch was active for the DRA performed in ethanol/water (3/1,
v/
v), obtaining good to excellent yield in the desired product (
Scheme 19). Recyclability test demonstrated that the catalyst recovered at the end of reaction with an external magnet could be re-used for at least five runs without losing its activity and selectivity.
Scheme 19. DRA catalyzed by Pd@Fe3O4-NH2/Starch.
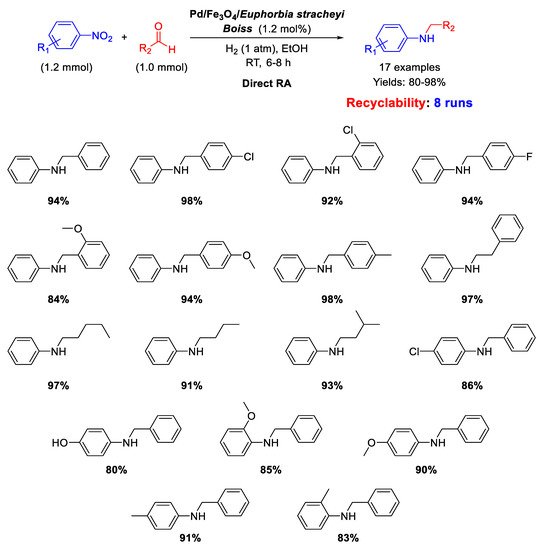
In the framework of Pd/Fe
3O
4 nanocomposites as efficient catalysts for DRA reaction, Nasrollahzadeh’s group reported on the synthesis, characterization, and catalytic activity of palladium and magnetite NPs supported on
Euphorbia stracheyi Boiss root extract [
68]. FESEM micrographs of the catalyst showed that Fe
3O
4 and Pd NPs were homogeneously distributed, and TEM images revealed that the obtained nanoparticles had spherical shape with an average diameter of 10 nm. The catalytic system was tested in the DRA of various nitroarenes with benzaldehyde and in the reaction between nitrobenzene with differently substituted benzaldehydes, in ethanol, at room temperature, under 1 atm H
2, affording good (80%) to excellent (98%) yields in the target product (
Scheme 20). Recyclability experiments demonstrated that the catalyst could be easily recovered at the end of reaction thanks to its magnetic properties and could be re-used for at least 8 runs without loss of activity.
Scheme 20. DRA catalyzed by Pd/Fe3O4/Euphorbia stracheyi Boiss root extract.
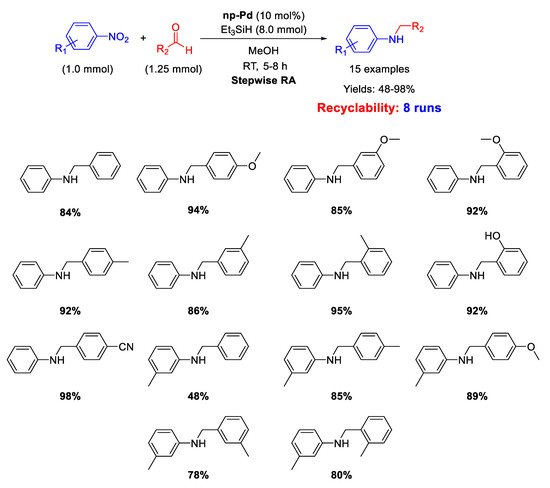
Very recently, in 2020 Cao et al. described the catalytic activity of nanoporous palladium (np-Pd) in the stepwise RA [
69]. This nanoporous palladium was obtained by dealloying an aluminum-palladium alloy by treatment with a concentrated solution of sodium hydroxide, that caused the leaching of aluminum from the alloy. The catalytic system was constituted by np-Pd (pore size of 5 nm), using triethyl silane (Et
3SiH) as the hydrogen donor in methanol. The overall reductive amination reaction occurred in one-pot three-step way (
Scheme 21). In the first step, the nitroarene was reduced to aniline with the addition of Et
3SiH; in the second step, benzaldehyde was added to the reaction mixture for the formation of the imine; in the last step, fresh Et
3SiH was added for the reduction of imine to amine. The catalytic system resulted recyclable for at least 8 runs without losing activity and selectivity. The amount of catalyst employed was, however, unusually high (10 mol% of Pd).
Scheme 21. Stepwise RA catalyzed by np-Pd.
6. Gold
Between 2007 and 2009, Corma’s group was the first to understand the potential of gold nanoparticles for promoting several cascade-type reactions for the synthesis of different nitrogen-based compounds [
79,
80] but they reported only one case of DRA starting from nitroarenes and aldehydes catalyzed by TiO
2 supported Au NPs. In this framework, in 2009 Tokunaga and co-workers [
81] employed Au NPs (2−3 nm in diameter) supported on Fe
2O
3 (Au/Fe
2O
3) under low hydrogen pressure for one-pot synthesis of aniline derivatives from nitroarenes and aliphatic or aromatic aldehydes in yields from 58 to 96% (
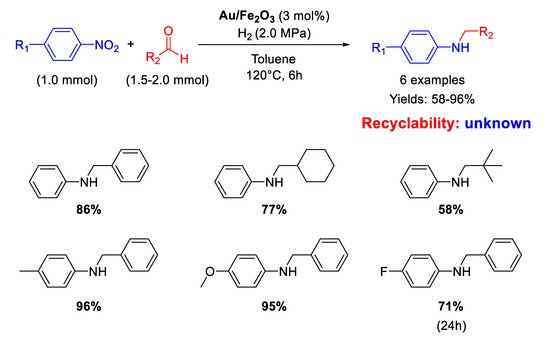
Scheme 29). Aliphatic aldehydes gave lower yields while nitroarenes with electron-withdrawing substituents needed longer reaction times. No attempts of recyclability were carried out with the catalyst because Au/Fe
2O
3 after the catalytic reaction showed a slightly increase of gold particle size as revealed by TEM investigations.
Scheme 29. DRA catalysed by Au/Fe2O3.
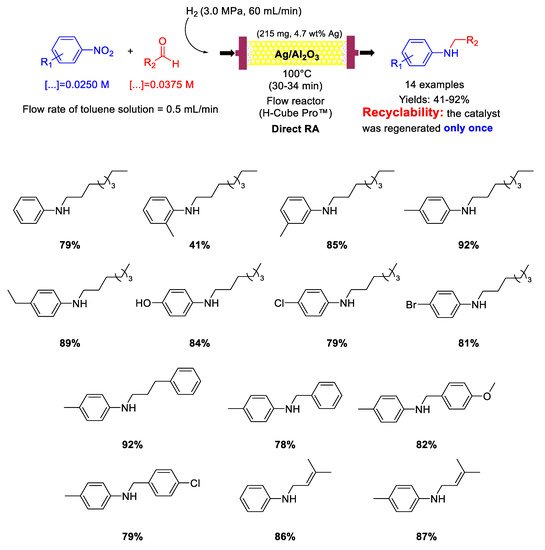
In 2015, Nuzhdin and co-workers [
82] published their first paper reporting on one-pot reductive amination of aromatic and aliphatic aldehydes with nitroarenes, promoted by Au/Al
2O
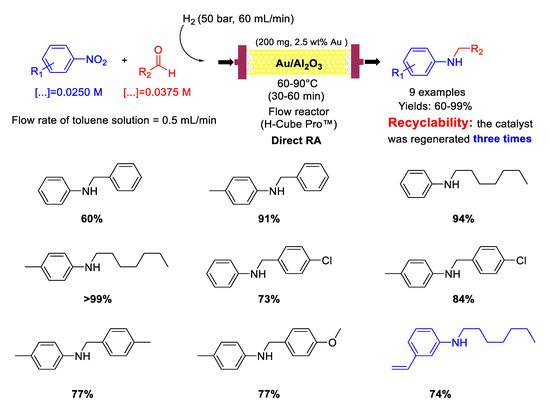
3 catalyst with a mean Au particle diameter of 3.4 nm in a continuous flow packed bed reactor using high pressure of molecular hydrogen as a reducing agent (
Scheme 30). Nine secondary aromatic amines were achieved in good to excellent yields and the catalyst showed remarkably high chemoselectivity towards the synthesis of
N-heptyl-3-vinylaniline starting from 3-nitrostyrene and
n-heptaldehyde. In the case of
N-heptylaniline, the catalyst was regenerated three times by washing every 135 min the catalyst cartridge with a flow of pure methanol (1 mL/min) for 30 min. Between any cycle (reaction and regeneration) only a slight decay in the yield of
N-heptylaniline was observed without leaching of Au from the Au/Al
2O
3 or increasing in gold particle size, but carbon deposition occurred at some extent, as ascertained by differential thermal analysis.
Scheme 30. DRA catalysed by Au/Al2O3 in flow reactor.
The high chemoselectivity shown by Au/Al
2O
3 paved the way to a follow-up article [
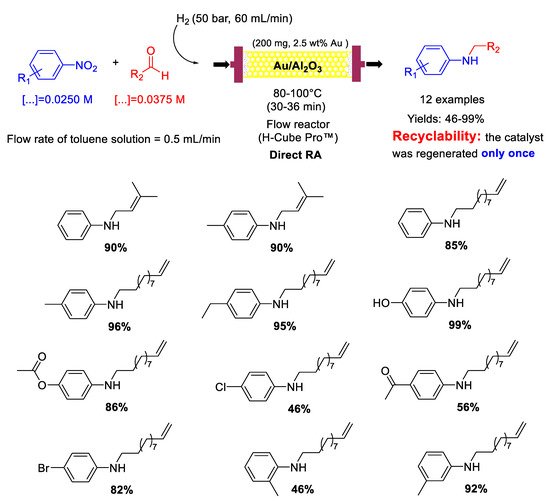
83] in which the same group reported that Au/Al
2O
3 could also catalyze in a continuous-flow the DRA of aliphatic unsaturated aldehydes with nitroarenes (
Scheme 31) obtaining unsaturated secondary amines in moderate to excellent yields (46−99%). Also in this case, the authors were able to regenerate the catalyst by washing it every 150 min with a flow of pure toluene (0.5 mL/min) for 30 min and again it was ascertained by thermal analysis that the formation of carbon-based deposits was the main reason for the slight catalyst deactivation.
Scheme 31. DRA catalyzed by Au/Al2O3 in flow reactor using unsaturated aliphatic aldehydes.
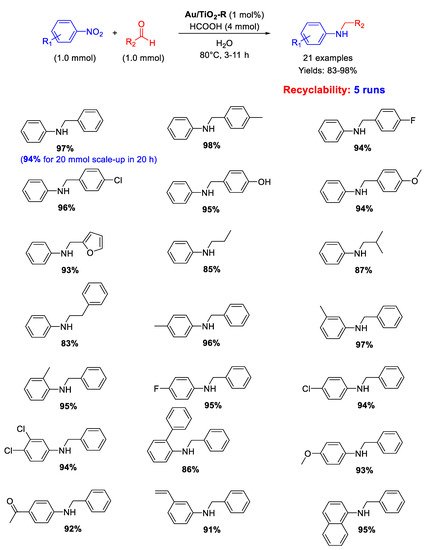
Cao and co-workers showed that Au/TiO
2-R, constituted by Au NPs with an average particle size of 2.2 nm supported on titania in the form of rutile (TiO
2-R), catalyzed under very mild reaction conditions the chemoselective transfer hydrogenative DRA of aldehydes with nitroarenes in neat water as solvent employing formic acid (FA) as hydrogen donor (
Scheme 32) [
84]. A broad scope reaction was demonstrated with twenty-one secondary aromatic amines being obtained in good to excellent yields (83–98%) minimizing
N-formyl derivatives and other unwanted by-products that usually form in appreciable levels with FA-based protocols. Moreover, raising the reaction time up to 20 h, a gram-scale reaction (up to 20 mmol scale-up) was accomplished for the synthesis of
N-benzylaniline in 94% yield. Au/TiO
2-R was reused five times without loss of activity and metal leaching. In addition, investigations by TEM, X-ray Photoelectron Spectroscopy (XPS) and X-Ray Diffraction (XRD) revealed almost no change in the dispersion of the Au NPs or metallic state of Au before and after each reuse.
Scheme 32. DRA catalyzed by Au/TiO2-R.
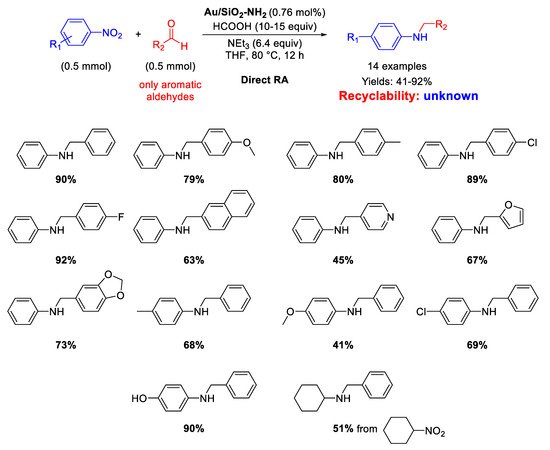
In 2020, Rossi and co-workers immobilized pre-synthesized Au NPs by sol-immobilization method on Stöber silica (microspheres of ca 400 nm) obtaining the Au/SiO
2-NH
2 catalyst [
85], which was employed for both reductions and reductive aminations using formic acid as hydrogen source (
Scheme 33). The active species were supported Au NPs with an average particle size of 2.2 nm that catalyzed the formation of fourteen secondary amines in moderate to very good yield (41%–92%). To achieve high reaction rates, it was necessary to employ an appropriate Brønsted base (triethylamine), which acted cooperatively with the gold surface for the activation of formic acid as reducing agent. Although the Au/SiO
2-NH
2 catalyst could be reused up to five times without any significant loss of activity and almost no agglomeration of the Au NPs in selective hydrogenation of alkynes, no details on the catalyst recyclability was furnished.
Scheme 33. DRA catalyzed by Au/SiO2-NH2.
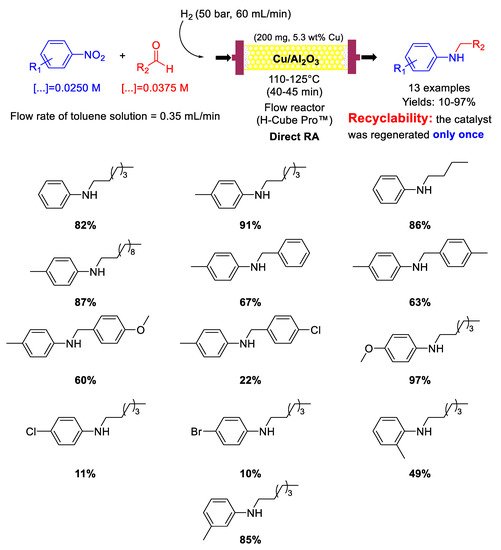
10. Cobalt
In 2013 Beller’s group synthesized the cobalt-based catalyst Co
3O
4/NGr@C (analogous to Fe
2O
3/NGr@C) by loading Vulcan XC72R carbon powder with a cobalt (II)-phenanthroline-chelate complex, and then pyrolyzing the obtained material at 800 °C for 2 h under Ar [
76]. Co
3O
4/NGr@C was a core-shell nanocomposite constituted by a core of Co NPs and a shell of Co
3O
4 NPs with size in the range of 2–20 nm, forming agglomerates sizing 20–200 nm, coated in turn by a N-doped graphene layer. The so obtained heterogeneous catalyst was employed for DRA in THF/H
2O under quite severe conditions: 110–125 °C, 50 bar of H
2, reaction time = 24 h (
Scheme 38) [
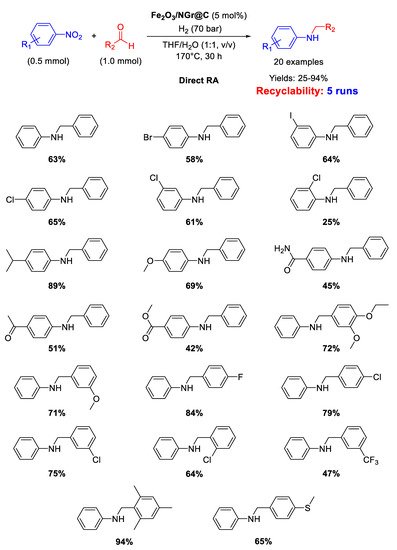
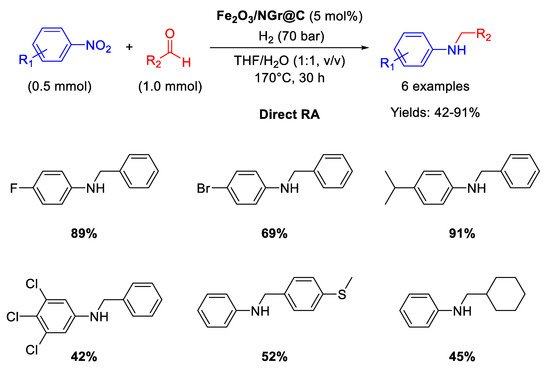
91]. However, the designed catalytic system had the great advantage to tolerate the presence of other reducible groups, such as C=O, or halogen substituents (resisting to dehydrohalogenation) in the starting nitroarene ring or/and in the used benzaldehyde. This valuable peculiarity is a characteristic common to all Co-based catalytic systems for DRA proposed by Beller in subsequent works. Recyclability tests demonstrated that on passing from the first to the second run with the same catalyst, the yield dropped from 95% to 70% ca., then remaining constant at around 60% until the sixth cycle.
Scheme 38. DRA catalyzed by Co3O4/NGr@C.
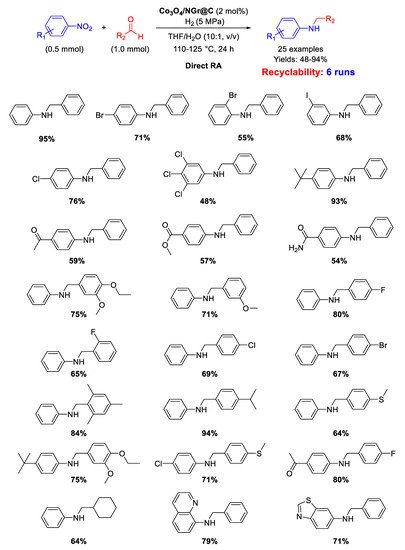
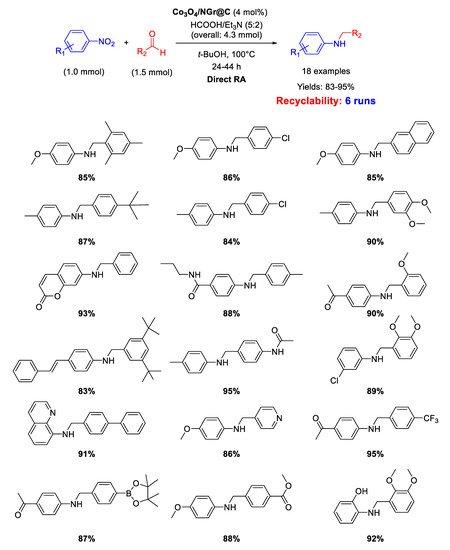
In 2018 Jagadeesh and co-workers employed the same Co
3O
4/NGr@C material as catalyst for DRA, using in this case the mixture formic acid/Et
3N (in 5/2 molar ratio), instead of H
2, as the hydrogen donor (
Scheme 39) [
92]. The good yields obtained in the catalytic tests remained mainly constant also performing gram-scale reactions. The catalyst could be recycled up to five times demonstrating stability and reusability. This catalytic system was highly chemoselective, tolerating the presence of many reducible groups.
Scheme 39. DRA catalyzed by Co3O4/NGr@C with HCOOH/Et3N.
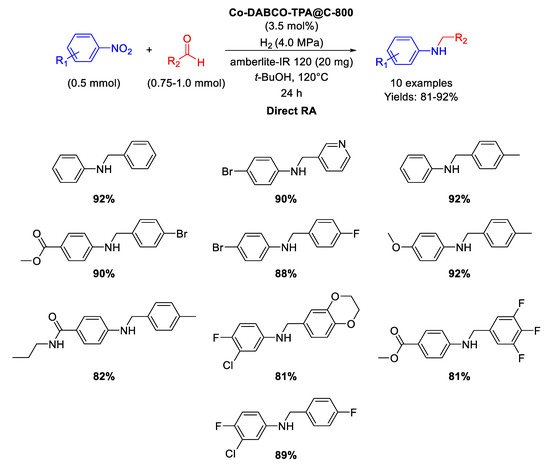
In 2017 Beller and co-workers synthesized MOF-derived cobalt nanoparticles catalytically active in direct RA reactions [
93]. The catalyst was obtained by preparing a Co-diamine-dicarboxylic acid MOF precursor (namely Co-DABCO-TPA MOF, DABCO = 1,4-diazabicyclo [2.2.2]octane and TPA = terephthalic acid), which was in turn pyrolyzed on Vulcan XC72R carbon at 800 °C for 2 h under Ar, to get the final active nanomaterial (Co-DABCO-TPA@C-800), constituted by Co nanoparticles with diameters ranging from <5 to 30 nm, encapsulated in N-doped graphitic shells, supporting also single Co atoms. As in the previously reported work [
91], some core-shell particles with cobalt oxide Co
3O
4 shells at metallic Co cores were also present. The new nanomaterial was tested as catalyst in the synthesis of a wide range of amines in good to excellent yields (
Scheme 40) through DRA starting from different aldehydes with nitro compounds under 40 bar of molecular hydrogen at 120 °C in
tert-BuOH in the presence of amberlite IR-120 for 24 h. The recyclability of the catalyst was tested in the synthesis of amphetamine, i.e. in the reaction between acetophenone and ammonia: the catalyst resulted recyclable for at least six runs without loss of activity and selectivity.
Scheme 40. DRA catalyzed by Co-DABCO-TPA@C-800.
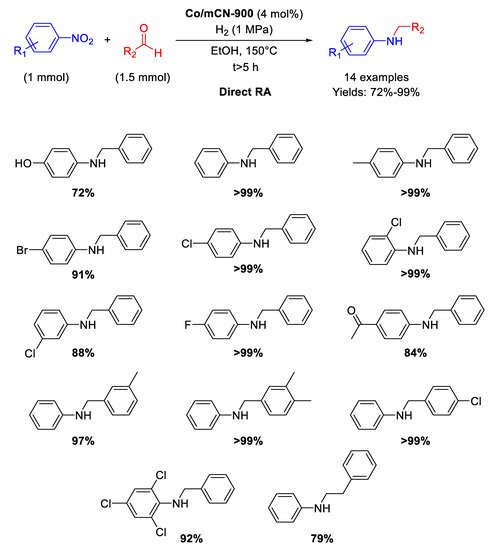
Following the above mentioned strategy of using N-doped carbon support for synthetizing Co-based catalysts, in 2017 Cui and co-workers have described the preparation of a new nanomaterial (Co/mCN-900) by pyrolysis under N
2 at 900 °C for 1 h of a mixture of melamine, polyacrylonitrile, and Co(NO
3)
2⋅6H
2O [
94]. In this way, a fluffy mesoporous carbon structure with high surface area and high nitrogen content was obtained. This N-doped carbon material supported dispersed accessible Co nanoparticle active sites (average size of 6 nm) and resulted an efficient catalyst in the N-alkylation of different nitrobenzenes with substituted benzaldehydes, allowing the synthesis of a wide range of aromatic amines with good yields under 1 MPa of H
2, at 150 °C in ethanol (
Scheme 41). The recyclability test was performed for the hydrogenation of nitrobenzene only (first step of the RDA) for six runs with the same catalyst. The catalytic system tolerated the presence of other reducible groups.
Scheme 41. DRA catalyzed by Co/mCN-900.
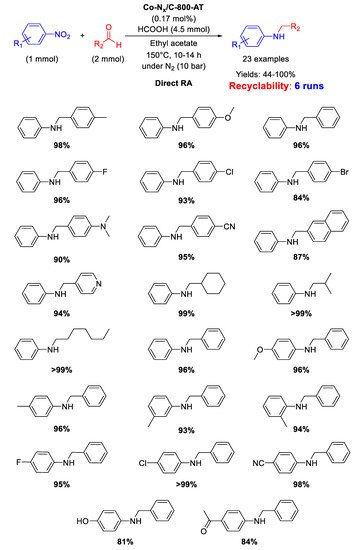
In 2017, Zhang and co-worker investigated the catalytic activity in DRA of a cobalt-based material, namely Co-Nx/C-800-AT [
95], generated from the pyrolysis of cobalt phthalocyanine/silica composite at 800 °C under N
2 and subsequent etching by HF to remove Co nanoparticles and silica template, affording a mesoporous polymer with mean pore size of 3.6 nm, containing Co-N
x catalytically active sites [
96]. This strategy of synthesis, differently from the above reported ones, aims to remove all metal (or eventually metal oxide) nanoparticles, leaving a carbon-based support retaining just Co-N
x fragments, considered responsible for catalytic activity. As shown in
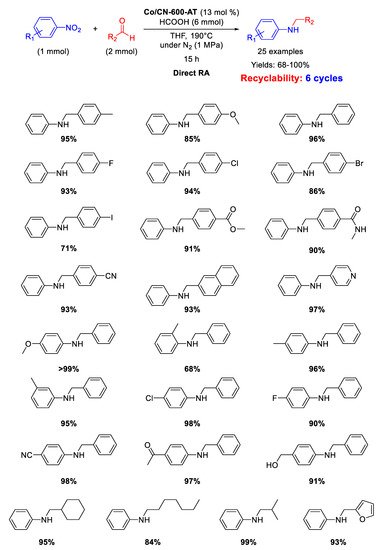
Scheme 42, DRA was carried out at 150 °C in ethyl acetate using formic acid as hydrogen donor and a combination of different aldehydes and nitro compounds was well tolerated under reaction conditions. Moreover, the catalyst was active and stable during six runs without leaching of Co from the system. The catalytic system was highly chemoselective tolerating the presence of C=O, CN, and halogen atoms in the nitroarene ring.
Scheme 42. DRA catalyzed by Co-Nx/C-800-AT.
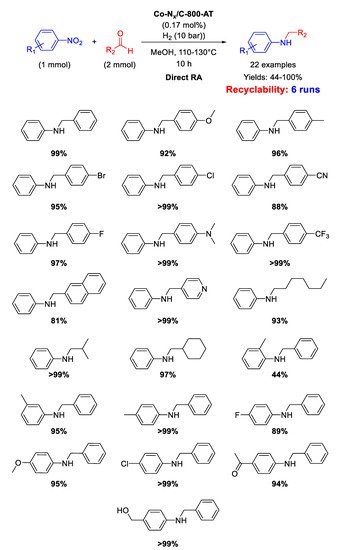
In addition, the catalyst Co-N
x/C-800-AT has been tested in the RDA reaction using H
2 (10 bar), as the reductant, in methanol at 110 °C for 10 h [
97]. Kinetic studies pointed out that, for the Co-N
x/C-800-AT catalytic system, the rate-determining step of the overall reaction was the hydrogenation of the imine intermediate. For this reason, the steric hindrance of the substituents located in
ortho-position in nitroarene and benzaldehyde substrates dramatically decreased the final yields towards the target molecule.
Scheme 43 summarizes the wide scope of substrates explored. The catalyst was successfully recycled for six runs
Scheme 43. DRA catalyzed by Co-Nx/C-800-AT under 10 bar H2.
On the basis of the above reported results, in the second part of 2017, Zhang’s group synthesized another catalyst by pyrolysis at 600 °C for 3 h under N
2 of cobalt supporting zeolitic imidazolate-frameworks (ZIF-67)/graphene oxide (GO) composite (ZIF-67/GO), followed by acid-etching with H
2SO
4, aiming to remove Co NPs formed under calcination [
98]. Differently from the Co-N
x/C-800-AT material, in this case the acid washing was able to remove just the surficial metal nanoparticles of the new material and the resulting catalyst (Co@CN-600-AT) still contained Co NPs with an average diameter of 8.2 nm, embedded into the reduced graphene oxide phase (graphene oxide became reduced graphene oxide during calcination). Co@CN-600-AT was active, stable, and selective in the direct RA using formic acid as hydrogen source, in THF at 190 °C for 15 h (
Scheme 44). The catalyst could be recycled for six times without losing its activity.
Scheme 44. DRA catalyzed by Co@CN-600-AT.
Following their studies on the synthesis of N-doped cobalt-based material, in 2017 Zhang’s group prepared another cobalt containing precursor, similar to the above reported ZiF-67/GO, but with silica in place of graphene oxide (ZIF-67/SiO
2 composite). This cobalt containing material was pyrolyzed at 800 °C for 8 h under N
2, and subsequently treated with hydrofluoric acidto remove surficial Co nanoparticles and silica template, yielding nitrogen-doped carbon-embedded Co nanoparticles with an average size of 6.3 nm (Co@CN-800) [
99].
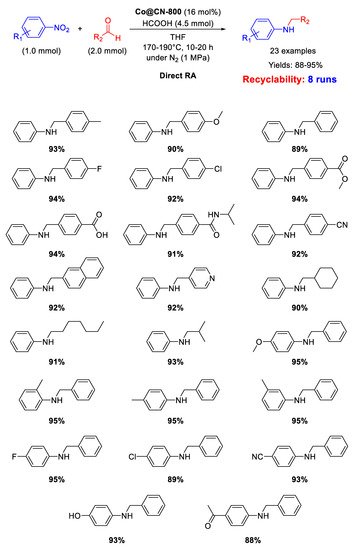
As shown in
Scheme 45, Co@CN-800 catalyst was active and recyclable (for at least eight runs) in the direct reductive amination of various substrates using formic acid, as hydrogen donor, at 170–190 °C in THF.
Scheme 45. DRA catalyzed by Co@CN-800.
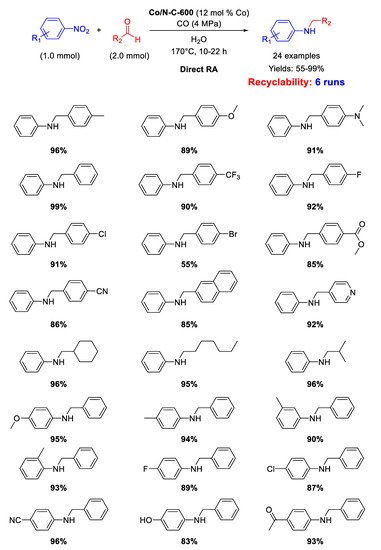
Pyrolysis of ZIF-67 under N
2 at 600 °C, followed by HF etching to remove Co NPs from the surface gave a new material (called Co/N-C-600) that was employed as catalyst for DRA. ZIF-67, as previously reported, consists of cobalt supported zeolitic imidazolate-frameworks, made up of cobalt cation and 2-methylimidazole ligand, obtained by simply mixing Co(NO
3)
2·6H
2O with 2-methylimidazole in water. Co/N-C-600 contained Co-nanoparticles (with average diameter of 14 nm) embedded in N-doped carbon support and was active in the direct RA using, for the first time in this reaction, CO/H
2O as hydrogen source [
100]. The proposed catalytic cycle envisaged the formation of formate ion and the resulting transfer hydrogenation mechanism resembled the pathway observed with the use of acid formic as hydrogen donor. The presence of nitrogen in the N-doped material was of crucial importance because doping N served as base site to capture the H
+ from water during the activation of CO/H
2O, to generate the active species (a proton and a metal hydride). Several aldehydes were tested in the reaction with nitro compounds under the conditions experienced without loss of activity. Noteworthy, the strong interaction between cobalt nanoparticles and nitrogen atoms warranted a high stability of the catalyst for 6 recycling runs (
Scheme 46).
Scheme 46. DRA catalyzed by Co/N-C-600 with CO/H2O.
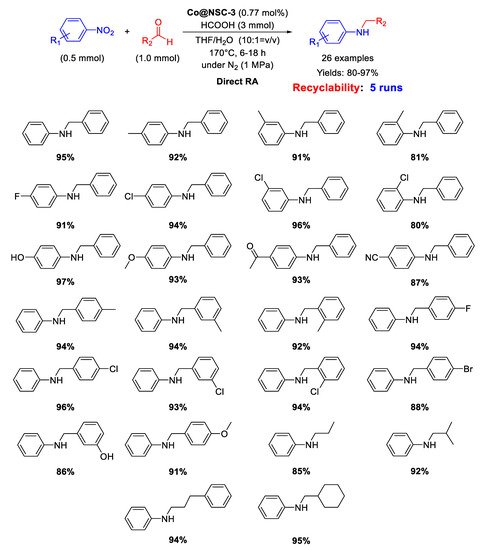
In 2019, Guo’s group developed a N,S-dual-doped carbon material embedding Co nanoparticles with an average size of 2.01 nm, useful for catalyzing DRA with formic acid [
101]. For the synthesis of the new catalyst, first the microwave assisted condensation of melamine and terephthalaldehyde was carried out in DMSO to give a Shiff base network (SNW-1, a porous organic polymer) incorporating sulfur impurities (due to the solvent used). Then, SNW-1 was impregnated with a cobalt salt and coated with glucose, and then pyrolyzed under inert atmosphere at 850 °C for 2 h. The resulting nanomaterial, Co@NSC-3 had a core-shell structure with cores of well-distributed cobalt NPs and N,S-doped carbon shells. Co@NSC-3 catalyzed the direct RA with formic acid as a hydrogen donor at 170 °C in THF/H
2O. The system worked without a base and showed interesting activity, selectivity, and stability for the synthesis of several kinds of secondary amines (
Scheme 47). Recyclability tests demonstrated that the catalytic system could be re-used for at least five runs without loss of activity and selectivity.
Scheme 47. DRA catalyzed by Co@NSC-3.
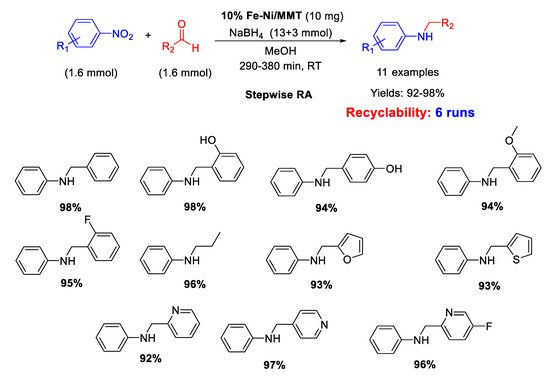
11. Nickel
In 2015 Kalbasi and Mazaheri [
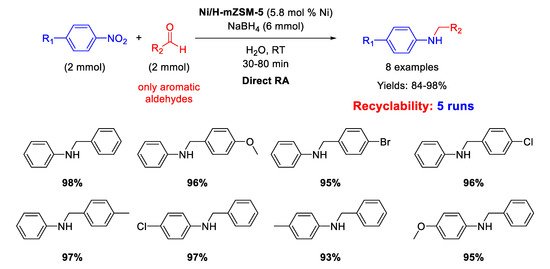
102] reported the synthesis of nickel nanoparticles (average size of 5 nm), supported on acid hierarchical zeolite (Ni/H-mZSM-5) with a microporous/mesoporous structure, as an acid-metal bifunctional catalyst. The catalytic activity of Ni/H-mZSM-5 was tested in one-pot RA of aromatic aldehydes with nitroarenes at room temperature, by using sodium borohydride as reducing agent and water as the solvent (
Scheme 48). The simultaneous presence of acid and metal in the catalytic structure promoted a high activity and reusability of the heterogeneous catalyst in water under mild conditions for at least five runs, with a slight reduction of activity (the yield dropped from 98% of the first run down to 73% of the fifth cycle). The efficiency of the Ni catalytic system prompted the same authors to replace Ni for Pd, preparing the analogous Pd/H-hierarcal ZSM-5 already discussed in [
61].
Scheme 48. DRA catalyzed by Ni/H-mZSM-5.
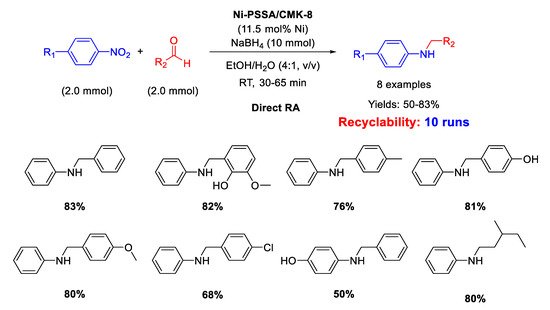
On the basis of the above reported results, Kalbasi and coworker [
103] developed a new heterogeneous catalyst constituted by ordered mesoporous carbon CMK-8 supporting both Ni nanoparticles (size ranging from 2 to 6 nm) and polystyrene sulfonic acid (PSSA) chains, thus having a dual (metal and acid) functionality. The new heterogeneous bi-functional catalyst, Ni-PSSA/CMK-8 was similar in the concept to the previously reported Ni/H-mZSM-5. The difference between the two catalysts was in the solid support: while hierarchical ZSM-5 is an inorganic solid acid with a relatively low surface area, PSSA/CMK-8 composite is an organic solid acid with a higher surface area, thus potentially more active. The new catalyst was tested in direct RA of aldehydes under mild reaction conditions and short reaction time in ethanol/water as solvent (
Scheme 49). Recyclability tests demonstrated that the catalyst could be used for at least ten runs with slight metal leaching into solution and small decrease of yield (83% in the first run; 68% in the 10
th cycle). For DRA carried out with Ni-PSSA/CMK-8 the authors observed the same substituent electronic effects registered with Pd/H-hierarchical ZSM-5 catalytic system [
61], that is: the reaction rate was improved by the presence of electron withdrawing and electron donating substituents in nitrobenzene and benzaldehyde rings, respectively. In addition, to get insights into the mechanism pathway, Kalbasi and coworker mixed aniline, benzaldehyde, and Ni-PSSA/CMK-8 in EtOH/H
2O in the absence of the reductant and observed 40% yield in imine (no secondary amine formation), but when they added to the mixture the appropriate amount of NaBH
4, they registered 83% yield in the target molecule (secondary amine), and not 40% as they thought to find. On the basis of this observation, the authors claimed for their catalytic system the presence of an additional mechanism for the imine formation, passing through the reaction between benzyl alcohol (eventually formed under reaction conditions, but not detected by the authors) and aniline, to give imine, subsequently reduced to secondary amine. However, this speculation is not convincing because the condensation between benzaldehyde and aniline is an equilibrium reaction [
36], whose balance is shifted towards the imine product once the latter is subtracted from the system, due to its hydrogenation. Therefore, it was not surprising that the yield into secondary amine increased when the imine was removed from the equilibrium.
Scheme 49. DRA catalyzed by Ni- PSSA/CMK-8.
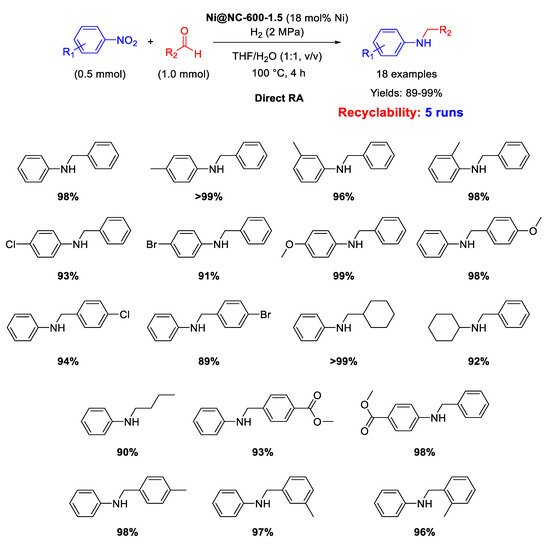
In 2019 Wang and Chen prepared a MOF-derived catalyst with nickel nanoparticles (6–7 nm in size) uniformly embedded in nitrogen doped carbon shells [
104], following roughly the strategy already introduced by Beller and discussed in the above reported paragraphs. In details, the new catalyst was obtained by mixing 2-aminoterephthalic acid and polyvinylpyrrolidone with a nickel(II) salt in DMF/water, thus obtaining a green solid precipitate (Ni-MOF-NH
2), which was pyrolyzed at 600 °C for 1.5 h under Ar to get the final Ni@NC-600-1.5 catalyst. The new catalyst resulted active and selective for at least five runs in direct RA under H
2 (2 MPa) at 100 °C in THF/H
2O (
Scheme 50). The catalyst possessed magnetic features which allowed its easy removal from the reaction mixture by an external magnet, and its subsequent recycling for five runs without loss of activity and recyclability. XRD analyses excluded the formation of NiO, while N-doping promoted the dispersion of Ni nanoparticles and enhanced their ability to activate H
2 through heterolytic cleavage, further facilitated by the presence of pyridinic N found in the carbon shells.
Scheme 50. DRA catalyzed by Ni@NC-600-1.5.
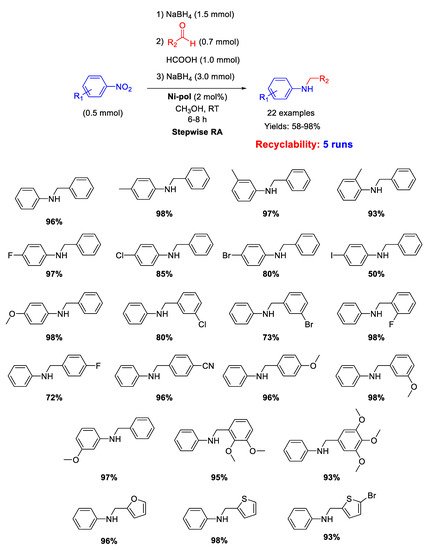
Another example of N-doped organic matrix supporting Ni NPs as efficient catalyst for RA reaction has been reported in 2019 by Dell’Anna and co-workers [
105]. Indeed, on the basis of the good results obtained with the already discussed Pd-pol [
53], the strategy of synthesis followed for Pd-pol was transferred for the synthesis of Ni-pol, wherein the noble metal Pd was replaced by the earth abundant nickel. The polymer has been prepared by co-polymerizing the metal containing monomer [
106] Ni(AAEMA)
2 [AAEMA
− = deprotonated form of 2-(acetoacetoxy) ethyl methacrylate] with
N,
N-dimethylacrylamide and
N,N’-methylenebisacrylamide [
107] and submitting it to calcination at 300 °C under N
2, obtaining a N-containing organic polymer (Ni-pol) with a homogeneous distribution of cubic nanocrystals of Ni of average cross section value of 35 nm [
108]. Ni-pol was tested in the stepwise RA of various benzaldehydes with different nitroarenes in methanol at room temperature, using NaBH
4 as the reductant (
Scheme 51). The addition of few drops of formic acid was necessary in the condensation step to promote the formation of the imine intermediate. Twenty-two secondary amines have been synthetized with Ni-pol catalytic system, some of them containing halogen substituents (no hydro-dehalogenation occurred) or reducible group (-CN). Ni-pol was recovered at the end of reaction and used for five subsequent runs without loss of activity and selectivity.
Scheme 51. Stepwise RA catalyzed by Ni-pol.
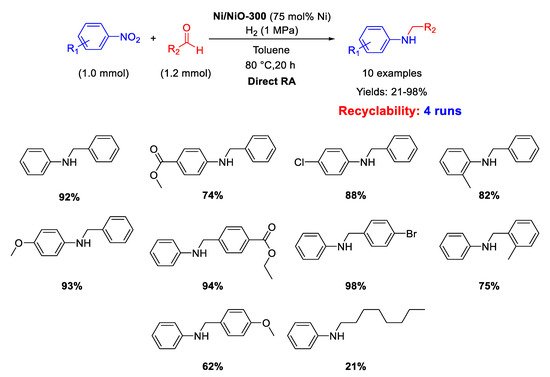
In 2020, Hara and co-workers prepared Ni/NiO-300 by partial reduction of NiO at 300 °C with H
2 and employed it as heterogeneous catalyst for one-pot reductive amination of benzaldehydes with different nitroarenes under dihydrogen (1 MPa) in toluene for 20 h (
Scheme 52) [
109]. The catalyst could be reused for four cycles without catalytic losses. Scanning Electron Microscopy (SEM) and XPS analyses showed that Ni/NiO-300 was constituted by Ni particles with size of about 0.5–3 μm coated with a layer of NiO, which remained the same after catalytic cycles. The metal amount used is very high for a catalytic reaction.
Scheme 52. DRA catalyzed by Ni/NiO-300.