3. Key Enzymes of De Novo Lipogenesis (DNL)
Cell requirement for fatty acids is normally met by the utilization of dietary fatty acids. The amount of fatty acids synthetizes by
DNL is of minor importance in most human tissues, except for the liver, mammary gland, and to a lesser extent, adipose tissue [
38]. However, the rate of
DNL and the expression of several lipogenic enzymes are increased in various cancer types.
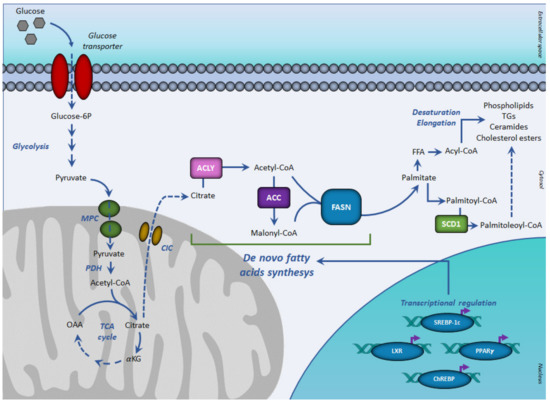
DNL is a metabolic process by which pyruvate, mainly derived from carbohydrate sources is converted into fatty acids () [
38].
In aerobic conditions, pyruvate, the end product of glycolysis is transformed in acetyl-CoA, which enters the TCA cycle by condensing with oxaloacetate to form citrate. When the energetic charge is high such as after a meal rich in carbohydrates, citrate can be transported from the mitochondria into the cytosol, where fatty acid synthesis occurs. Citrate efflux into the cytosol is catalyzed by the citrate carrier (CIC), an intrinsic protein of the inner mitochondrial membrane, which catalyzes an electroneutral exchange of citrate plus a proton with malate. In the cytosol, glucose-derived citrate is converted into oxaloacetate and acetyl-CoA by the ATP citrate lyase (ACLY). The obtained acetyl-CoA is required for lipid synthesis during membrane biogenesis as well as for histone acetylation reactions to regulate the expression of certain proteins in aberrantly proliferating cancer cells.
Key enzymes of DNL are acetyl-CoA carboxylase (ACC) and the multi enzymatic complex fatty acid synthase (FASN).
ACC represents the rate-limiting enzyme of
DNL, catalyzing the irreversible carboxylation of acetyl-CoA into malonyl-CoA. The reaction requires biotin and ATP. In humans and mammals, there are two isoforms of ACCs: ACC1 (or ACC-α) with 265 kDa and ACC2 (or ACC-β) with 280 kDa. ACC1 presents in the cytosol with a lipogenic role, so is particularly expressed in lipogenic tissues and ACC2 is mainly associated with mitochondria in oxidative tissues [
39,
40,
41]. Therefore, ACC1 is enriched in lipogenic tissues such as the liver, adipose, and lactating mammary gland, where it catalyzes the biosynthesis of long-chain fatty acids. In contrast, ACC2 is highly expressed in oxidative tissues such as skeletal muscle and heart, where it regulates fatty acid β-oxidation.
Cytosolic malonyl-CoA, produced by ACC, can be used for fatty acid biosynthesis. The reaction is catalyzed by FASN. After priming with acetyl-CoA, FASN uses malonyl-CoA as a carbon donor and NADPH as a reduced cofactor to produce palmitoyl-CoA.
By furnishing malonyl-CoA, ACC not only plays a key role in DNL, but also regulates mitochondrial fatty acid β-oxidation, considering that malonyl-CoA is an inhibitor of carnitine palmitoyl-transferase-1, the key enzyme of this metabolic process.
The de novo synthesized fatty acids can be used for the synthesis of complex lipids such as phospholipids, ceramides, cholesterol esters, and triacylglycerols and thereby play a major role in membrane structure, cell signaling, and energy storage. Following
DNL, the enzyme stearoyl-CoA desaturase (SCD) catalyzes the introduction of the first double bond in the cis-delta 9 position of saturated fatty acyl-CoA giving monounsaturated fatty acids, which are preferentially transformed into triacylglycerols for storage [
41]. Two isoforms of SCD have been reported in human cells, SCD1 and SCD5 [
42]. Both isoforms are overexpressed in luminal cancer models compared to the TNBC subtypes [
43].
4. Transcription Factors Regulating De Novo Lipogenesis (DNL)
DNL is a highly regulated metabolic pathway. Having common features at their promoter regions, lipogenic genes are coordinately regulated at the transcription level. The transcription factors sterol regulatory element-binding protein-1c (SREBP-1c), upstream stimulatory factor (USF), peroxisome-proliferation-activated receptors (PPARs), carbohydrate response element-binding protein (ChREBP), and liver X receptors (LXRs) play critical roles in regulating this process ().
SREBPs represent the master transcriptional factors regulating
DNL. SREBPs are members of the basic helix-loop-helix (bHLH)-leucine zipper transcription factors and can be classified into three types: SREBP-2, SREBP-1a, and SREBP-1c. Whereas SREBP-2 preferentially regulates genes involved in cholesterol metabolism, SREBP-1 regulates fatty acid synthesis enzymes. Expressions of ACC, FASN, and SCD1 are under the control of SREBP-1c [
44]. Moreover, SREBP-1c activates the expression of CIC both in hepatocytes [
45] and in the mammary epithelium [
46] and SREBP-1 overexpression increases the CIC transcript and protein levels. Moreover, SREBP-1 upregulates ACLY at the mRNA level via Akt signaling [
47].
Although SREBP-1 plays a pivotal role in regulating lipogenic gene expression, it is not the only one. In vitro studies have demonstrated that insulin effect on FASN promoter also requires the presence of the upstream stimulatory factors (USFs). USFs are bHLH-leucine zipper transcription factors able to bind the CANNTG sequence present in the promoter region of FASN. The effects of SREBP-1 and USFs on FASN are independent and additive [
48].
Lipogenic enzyme transcription may also be regulated by ChREBP [
49], a glucose-regulated bHLH transcription factor. In response to increased glucose levels, ChREBP undergoes dephosphorylation steps that allow translocation from the cytoplasm to the nucleus where, in association with its binding partner Max-like (MLX) interacting protein, it binds carbohydrate response elements of lipogenic genes [
50,
51,
52].
LXRs are members of the nuclear receptor superfamily that heterodimerize with retinoid X receptor (RXR) [
53]. Two isoforms of LXRs have been identified, LXRα and LXRβ [
53,
54]. It has been reported that LXRs perform an important role in the regulation of fatty acid synthesis. LXRs can activate lipogenic enzymes directly or by SREBP-1c. FASN is transcriptionally regulated by both LXRα and LXRβ [
54,
55,
56].
PPARs are members of the superfamily of nuclear hormone receptors that function as ligand-dependent transcription factors. Upon ligand activation, they regulate the expression of genes containing a specific response element, called the PPAR-responsive element (PPRE), which consists of a hexameric nucleotide direct repeat of the recognition motif (TGACCT) spaced by one nucleotide (DR-1). Three subtypes of PPARs termed α, δ (or β), and γ, have been identified [
57,
58]. These receptors heterodimerize with the retinoid X receptor (RXR) and alter the transcription of target genes after binding to PPRE.
Although PPARγ is considered to be the master regulator of adipocyte differentiation, an increase in PPARγ expression has been associated with accumulation in hepatic triacylglycerols. A study reports that PPARγ is capable of inducing lipid accumulation in hepatocytes in which an increase in SREBP-1 as well as ACC and FASN expression is also measured. These data suggest that PPARγ may play a role in stimulating lipogenesis [
57,
58,
59]. Heterozygous PPARγ mutant mice exhibit smaller fat stores upon a high-fat diet [
60,
61]. Recently, it has been reported that the overexpression of PPARα/RXRα and PPARγ/RXRα heterodimers enhances CIC promoter activity in BRL-3A and 3T3-L1 cells, respectively [
45].
5. Role of DNL Enzymes in Breast Cancer
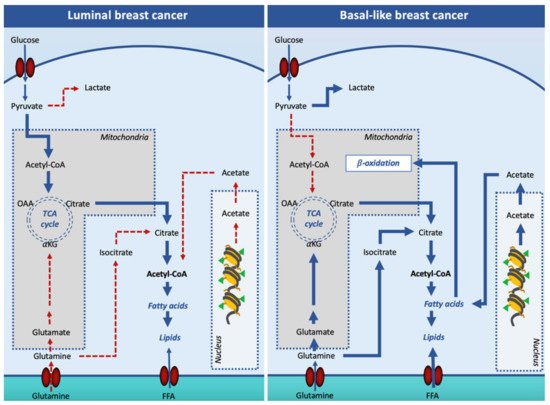
Oncogenic signaling has been reported to increase DNL in order to prepare the cell for invasion and metastasis. However, it appears that not all breast cancer subtypes depend on DNL for fatty acid supply. Indeed, while the luminal subtypes rely on DNL, the TNBC subtype overexpresses genes involved in the utilization of exogenous-derived fatty acids, in the synthesis of triacylglycerols and lipid droplets, and fatty acid oxidation ().
Figure 2. Lipogenesis in luminal and basal-like breast cancer cells. In luminal breast cancer cells, glucose-derived acetyl-CoA is the main source of citrate for the cytosolic synthesis of lipids. In basal-like, pyruvate is mainly converted into lactate in the cytosol, and other substrates such as glutamine and acetate are used to support cell lipid synthesis. Additionally, basal-like breast cancer cells increase free fatty acid entry in the cell to fulfill the β-oxidation pathway. Solid arrows signify the main reaction processes and dotted arrows signify processes with a minor relevance. Abbreviations: FFA, free (non-esterified) fatty acid; OAA, oxaloacetate; αKG, α-ketoglutarate; TCA, tricarboxylic acid cycle.
To support the high demand of acetyl-CoA for the increased
DNL, luminal breast cancer cells increase glucose entry and glycolytic flux [
62]. By transporting citrate into the cytosol, CIC plays an important role in
DNL. Thus, CIC inhibition can potentially limit cancer cell proliferation. Indeed, inhibition of CIC activity by BTA was reported to reduce breast xenograft tumor growth [
63].
Changes in ACLY expression have been found in diverse types of tumors including breast cancer, suggesting that this enzyme plays a crucial role in cancer metabolism [
64]. ACLY has been reported to have a strong expression in breast cancer tissue, with respect to adjacent normal tissues, and silencing ACLY expression in MCF-7 cell line suppressed cell viability and increased cell apoptosis [
65]. Accordingly, a study reported that genetic or chemical inhibition of ACLY reduces, both in vitro and in vivo, proliferation, and tumor growth [
66].
In recent work, Lucenay et al. demonstrated that cyclin E, an independent predictor of survival in patients with invasive breast cancer, upregulating ACLY activity leads to lipid droplet accumulation, a process positively correlated with tumor growth and development [
67]. ACLY mRNA has been reported to be mostly expressed in the HER2-enriched subtype with respect to TNBC, linking the expression of this enzyme to the EMT process [
65,
66].
Several studies have highlighted the association between ACC 1/2 and FASN expression and activity with invasion, proliferation, and EMT [
66,
67,
68,
69,
70]. Enhanced of both expression and activity of FASN are considered early events in breast cancer progression [
71] and blocking FASN can induce antitumor effects in TNBC [
68]. Additionally, inhibition of FASN by cerulenin can affect EMT [
72] and reverse the hyperglycemia-induced EMT phenotype [
73]. Fasnall, a selective FASN inhibitor, reduced the proliferation of breast cancer cells and modulated the lipidomic profile of these cells by increasing ceramide levels due to malonyl-CoA accumulation and consequent CPT-1 inhibition [
74]. More recently, CRISPR/Cas9 knockout of
FASN in MCF-7 cells demonstrated that FASN inhibition has a role in reducing proliferation, cell survival, cell size, cell cycle, migration, cell adhesion, and DNA replication [
75].
A study conducted by Alò and collaborators demonstrated that FASN overexpression is associated with the stage of progression of breast cancer and that FASN expression can be used as a prognostic indicator for disease-free survival and overall disease survival [
76]. In breast cancer stem cell sub-populations, high expression levels of ACC 1/2 and FASN have been correlated with increased cell survival and, in turn, with the formation of pre-malignant lesions [
77]. Moreover, a decreased level of palmitic acid, associated with
ACC1 and
FASN gene silencing, can induce apoptosis in human breast cancer cells [
70]. Interestingly, it has been reported that breast cancer susceptibility gene 1 (BRCA1) can exert its tumor suppressor function by preventing p-ACC1 dephosphorylation and, in turn, decreasing
DNL [
78].
ACC 1/2 and FASN expression in breast cancer cells is regulated by diverse growth factors and sex hormones through their corresponding receptors such as PR, ER, androgen receptor, and HER [
79]. Based on this responsiveness, lipogenic enzyme expression is associated with molecular subtypes, and then with the malignant phenotype of breast cancers. Data suggest that in breast cancer cell lines overexpressing HER2, both FASN and ACC1 levels increased compared with cells in which HER2 expression is relatively low (such as MDA-MB-231) [
80]. Indeed, induction of HER2 in MDA-MB-231 cells stimulates ACC1 expression via the PI3K/Akt pathway [
81]. FASN upregulation in HER2-positive cells occurs throughout an SREBP-1-mediated mechanism. More recently, HER2 has also been shown to directly phosphorylate and activate FASN activity [
82].
It must be pointed out that a metabolic transition that suppresses lipogenesis and promotes energy production is an essential component of metastasis in breast cancer. Indeed, Snail, a key inducer of EMT, has been related to ACC2 suppression and increased oxidation of mitochondrial fatty acids [
83] and TGFβ1, which induces EMT, suppresses ACC in MCF-7 cells [
84]. Furthermore, epithelial breast cancer cells with high expression of E-cadherin showed high expression of FASN, while mesenchymal cells with high expression of vimentin showed high expression of carnitine palmitoyltransferase-1 and therefore of β-oxidation [
84]. A recent work reports that in both human and murine breast cancers, ACC1 inhibition, by increasing the level of acetyl-CoA, can favor acetylation and activation of the transcription factor Smad2, and thus EMT and metastasis [
85].
It has been suggested that SCD1 may play a key role in the generation of the malignant phenotype as well as in the subsequent proliferation and survival of cancer cells [
86]. Accordingly, SCD1 expression is enhanced in breast cancer tissues in situ compared to normal tissue [
87,
88] and SCD1 expression was associated with shorter survival times in breast cancer patients [
89]. SCD1 was reported to be overexpressed in both HER2-enriched subtype [
90,
91] and in breast cancer cells that overexpress mucin-1 [
92]. Inhibition of SCD1 activity or silencing its expression leads to anti-proliferation effects in breast cancer cell lines [
93,
94,
95,
96,
97,
98]. Moreover, ERα regulates SCD1 expression. Indeed, in vitro treatment of MCF-7 and T47D cell lines with 17β-estradiol induces SCD1 expression and modulates the cellular monounsaturated/saturated fatty acid ratio [
99]. This was also observed in vivo, where the relative amounts of phosphatidylcholines (PC) (36:1) compared to PC (36:0) and that of PC (36:1) compared to lysoPC (18:0) were significantly higher in the cancerous areas characterized by higher levels of SCD1 expression compared to normal areas [
100].