Metal oxide nanoparticles (NPs) have received a great deal of attention as potential theranostic agents. Despite extensive work on a wide variety of metal oxide NPs, few chemically active metal oxide NPs have received Food and Drug Administration (FDA) clearance. The clinical translation of metal oxide NP activity, which often looks so promising in preclinical studies, has not progressed as rapidly as one might expect. The lack of FDA approval for metal oxide NPs appears to be a consequence of the complex transformation of NP chemistry as any given NP passes through multiple extra- and intracellular environments and interacts with a variety of proteins and transport processes that may degrade or transform the chemical properties of the metal oxide NP. Moreover, the translational models frequently used to study these materials do not represent the final therapeutic environment well, and studies in reduced preparations have, all too frequently, predicted fundamentally different physico-chemical properties from the biological activity observed in intact organisms. Understanding the evolving pharmacology of metal oxide NPs as they interact with biological systems is critical to establish translational test systems that effectively predict future theranostic activity.
- cell trafficking
- endocytosis
1. Introduction
2. The Origin of Biological Activity in the Structure of Nanoparticles
2.1. Redox Reactivity of Metal Oxides
 Ce3+. Cerium oxide demonstrates both superoxide dismutase and catalase mimetic activity [17,18,19]. In the reaction scheme shown below, the hydroxyl radical is the ‘seed’ for the balanced set of redox processes. Given the high oxidation potential of •OH (+2.31 V at pH 7.0) and a high rate constant, hydroxyl radical can react rapidly with many biomolecules and lead to oxidative damage. However, the superoxide anion is likely the reactant with ceria. Superoxide anion is continually produced as a result of aerobic respiration, and the production of this reactive oxygen species, which functions as an important biological signaling molecule, can be greatly up-regulated in disease states. Superoxide dismutase converts superoxide anion into peroxide, and this species is quickly converted to water and oxygen by catalase or by reacting with hydroxyl anions via Fenton reactions. Both of these potent oxidizing agents, O2− and H2O2, likely contribute to oxidative stress and damage of DNA, proteins, and lipids [20].
Ce3+. Cerium oxide demonstrates both superoxide dismutase and catalase mimetic activity [17,18,19]. In the reaction scheme shown below, the hydroxyl radical is the ‘seed’ for the balanced set of redox processes. Given the high oxidation potential of •OH (+2.31 V at pH 7.0) and a high rate constant, hydroxyl radical can react rapidly with many biomolecules and lead to oxidative damage. However, the superoxide anion is likely the reactant with ceria. Superoxide anion is continually produced as a result of aerobic respiration, and the production of this reactive oxygen species, which functions as an important biological signaling molecule, can be greatly up-regulated in disease states. Superoxide dismutase converts superoxide anion into peroxide, and this species is quickly converted to water and oxygen by catalase or by reacting with hydroxyl anions via Fenton reactions. Both of these potent oxidizing agents, O2− and H2O2, likely contribute to oxidative stress and damage of DNA, proteins, and lipids [20].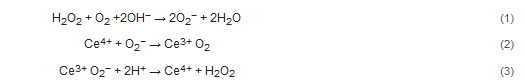

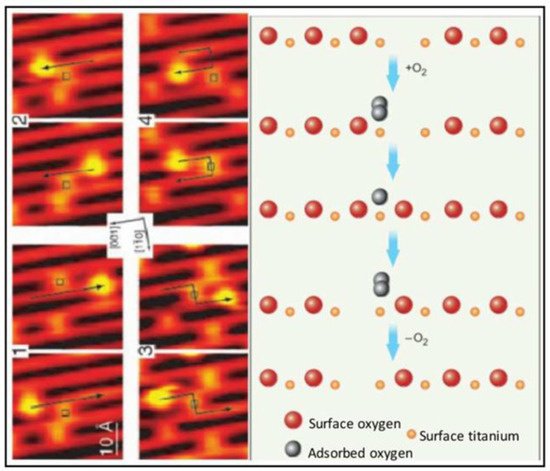
2.2. Intracellular pH Environments and Metal Oxide NP Activity
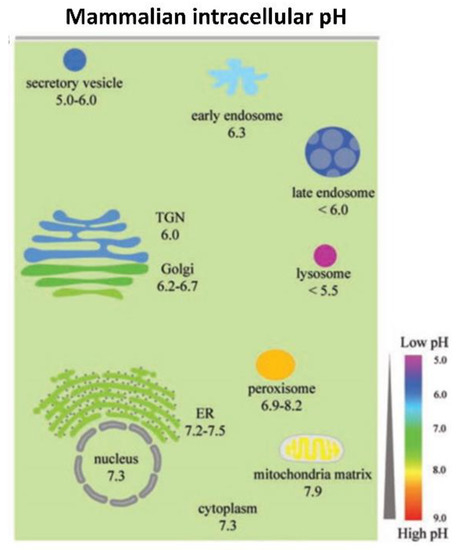
2.3. Model System Effects
This entry is adapted from the peer-reviewed paper 10.3390/antiox10040547
