1. Introduction
Nowadays, mining is a critical industry for global economic and social development, and it will continue to be the main resources provider in the immediate future [
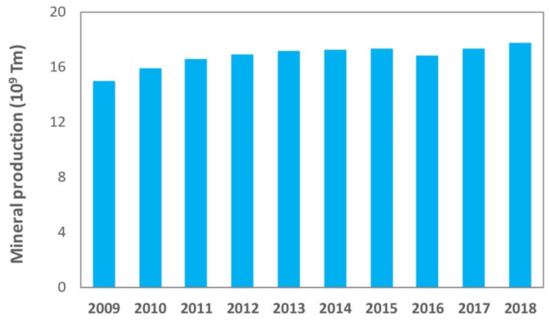
1]. However, just as there have been great technological advances in mining techniques that allow a larger recovery of minerals and therefore a continuous growth of world production, the impoverishment of deposits in terms of the grade, which requires larger scale, more complex and depth excavations, has an even higher impact on a major generation of waste. shows the evolution of world production of minerals in the 2009–2018 decade [
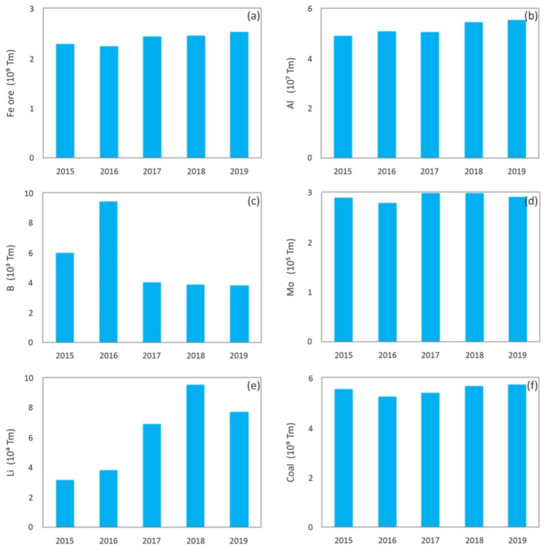
2], and shows global production, in the last years, of different metals or minerals reported in this paper, which include: iron [
3], aluminum [
4], boron [
5], molybdenum [
6], lithium [
7], coal [
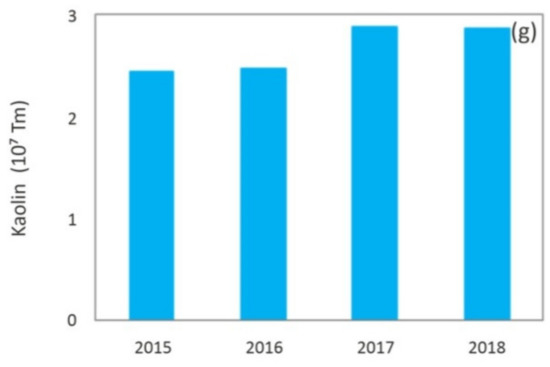
8] and kaolin [
9], among others.
Figure 1. Evolution of world mineral production [
2].
Figure 2. Worldwide production of: (
a) Iron ore mineral [
3]; (
b) Countries with the largest smelter production of aluminum [
4]; (
c) Boron [
5]; (
d) Molybdenum [
6]; (
e) Lithium [
7]; (
f) Coal [
8]; (
g) Kaolin [
9].
Thus, mining activities imply the generation of huge amounts of mining waste that produce great impacts on the environment, among others instability in waste dumps, acid drainage mainly due to oxidation processes, water pollution, air pollution and a long etcetera, which also includes the huge extents of territory occupied by the deposits of residues from the mineral washing and flotation plants [
10,
11].
Worldwide, there are approximately 3500 solid mining waste facilities, with an estimated generation of over 100 billion tons per year [
12]. An idea of the magnitude of waste generated is given by the fact that, for example, in the case of iron or aluminum mining, the amount of solid waste generated is several times greater than the mass of the metals extracted, but in the case of noble metals such as gold, this ratio is greater than millions of times [
13,
14].
Tayebi-Khorami et al. [
12] in an interesting study on the current management frameworks of mining waste, established the need for the mining industry to develop strategies in the medium and short term for proper management and use of waste aimed at achieving a lower environmental impact. The design and implementation of robust long-term solutions remains, to date, a challenge for the mining industry, due to the specificity of the sites where wastes are generated, the local weather conditions and the physicochemical heterogeneity of their wastes [
15]. Besides, the global megatrends (large, social, economic, political, environmental, or technological changes) as driving technology demands should be taking into consideration by the mining industry for its future economic growth [
16].
Solid mining waste is grouped into two main groups, viz mine waste rock (steriles) and tailings. Mine waste rock consists of low-grade excavated bedrock that has been transported to access profitable ore, and generally comprises of relatively coarse broken granular rock in the size range from sand to pebbles or boulders. They are mainly piled on high mounds which are porous, hydraulically unsaturated, and therefore highly exposed to atmospheric conditions (oxidation). Tailings are the main waste, both in volume and weight, generated in the process of concentrating minerals. Tailings are composed of process water and fines remaining after crushing and beneficiation of the ore, which may contain secondary materials from reactions with processing reagents, such as explosive agents or extraction chemicals [
17]. Tailings streams are pumped into tailings storage facilities, TSFs (pools or dams) where they are deposited with low atmospheric exposure, which can even originate reducing conditions. Tailings can also be dried and stacked with the consequent displacement of the fines by the wind over long distances. The volume of tailings generated in the extraction of ore is almost equal to the volume of ore processed. It is estimated that there are approximately 1900 controlled TSFs worldwide in which 56 × 10
9 m
3 of mine tailings are stored. This volume is expected to increase up to 69 × 10
9 m
3 by 2025 [
18]. The high volume of tailings stored at TSF gives rise to significant environmental problems resulting from the occupancy of large areas of land, including farmland and forests, and the consequent destruction of the landscape [
19].
Moreover, tailings disposal also involves serious impacts on human health. In dry seasons, water from the surface of the tailings dams evaporates, leaving a fine, powdery material on the surface that pollutes the air in the surrounding areas. On the other hand, in rainy seasons, tailings deposits can collapse, causing landslides, with the consequent risk for the inhabitants of neighboring towns [
20]. Over the past 40 years, numerous tailings dam collapses have occurred, resulting in significant impacts such as a prolonged suspension of mining activity, environmental degradation, damage to the image of the mining company, economic implications for the industry, legal implications and loss of human life [
21]. shows shocking images of the collapse of several tailings dams.
Figure 3. Images of several environmental disasters produced by collapse of tailing dams: (
a) collapse of tailings dam in Aználcollar, Spain, 1998 [
22]; (
b) Baia Mare disaster, Romania, 2000 [
23]; (
c) Mariana dam disaster, Brazil, 2015 [
24]; (
d) Brumadinho dam disaster, Brazil, 2019 [
25].
Definitive solutions to the problem of mining waste are still far from being reached and much research and technological development must continue to be carried out. Although both types of waste present different geochemical behaviors, due to the large granulometric differences between them, different physical-chemical characteristics, and above all to the different storage practices, they can be considered, a priori, as a source of new exploitable materials [
26].
In recent years, different studies have been carried out with the objective of recovering mining waste through their incorporation into different materials. Thus, nanostructured Mg(II)Al(III) layered double hydroxide (LDH) was manufactured from waste serpentine tailings by a chemical precipitation method [
27]. Hexagonally ordered mesoporous silica materials were prepared from iron tailings by an innovative non-hydrothermal process at room temperature. These materials can be applied to energy storage or environmental protection [
28]. Several studies have reported on the valorization of mining waste in building materials. In this way, iron ore waste was used as a complementary cementitious material to prepare colored composite cements more resistant to acid attack than Portland cement [
29]; gold ore tailings were used as siliceous raw material for cement production [
30]. For this application, the cementitious/pozzolanic properties of mineral tailings can be improved by activation methods [
31]; molybdenum tailings have been investigated as replacement of fine aggregates in structural concrete [
32]; and copper tailings were evaluated as raw material for geopolymer manufacture. Fluorite mines waste has been incorporated in the raw material mixture for the manufacture of glass [
33], which allows for the incorporation of the potentially toxic elements of the tailings into the glass structure [
34]. The valuation of mining waste as fillers has also been considered in some studies. For instance, lead-zinc mill waste was studied as paste backfilling for an underground metalliferous mine [
35]; and perlite wastes was used for the manufacture of lightweight materials by combining the geopolymerization technology with the foaming process [
36]. A further recovery route investigated for these wastes is the production of synthetic stone. Thus, an artificial arenite was developed from different particle size fractions of the sandstone industry waste mixed with an unsaturated polyester resin [
37]. Finally, as one of the potential recovery options, the manufacture of ceramic materials has been satisfactorily evaluated [
38,
39,
40].The studies conducted to incorporate mine tailings into mullite-based ceramic materials are described hereafter.
Under the conventional model of the linear economy, taking into account the lowering of the ore grade and consequently higher excavation rates, the problem of mining waste and its associated inherent risks would continue to aggravate. In contrast, a circular economy articulates the importance of closed-loop systems that reduce the need for extraction and processing of new resources. As such, within mining activities and following the 3Rs principle of waste reduction (reduces, reuse, recycle), it can make an important contribution [
41]. Thus, within the framework of the circular economy (which requires design for repair, reuse, recovery, refurbishment, restoration, and recycling) and the sustainable development objectives (ODS), many mining wastes could be considered, among others, as alternative materials in the construction sector instead of sources of pollution and risk. This solution will lead to the conservation of finite natural resources extensively used in the field of construction and to reduce the environmental impacts related to them [
10,
16,
42,
43]. Among construction materials, ceramic materials and, specifically those based on mullite, have been established as an appropriate alternative for the recovery of solid mining waste. The use of mining waste in the production of mullite-based ceramics contributes to various sustainable development objectives (ODS) of Agenda 2030, an action plan that resulted from the commitment of the member states of the United Nations, whose main objective is to ensure the protection of people, the planet and prosperity [
44]. Thus, as far as objective 7 (affordable and clean energy) is concerned, the use of mining waste as a substitute for pure raw materials allows for a reduction in energy consumption. With regard to objective 13 (climate action), the recovery of waste contributes significantly to the reduction of CO
2 and other greenhouse gas emissions. Furthermore, the appropriate management and use of waste avoids negative impacts on terrestrial ecosystems, which are covered by objective 15 (life on land). But undoubtedly, the valorization of mining waste is closely linked to objective 12 (responsible consumption and production), which includes the achievement of a sustainable management and efficient use of natural resources by 2030, while considerably reducing waste generation through prevention, reduction, recycling and reuse activities.
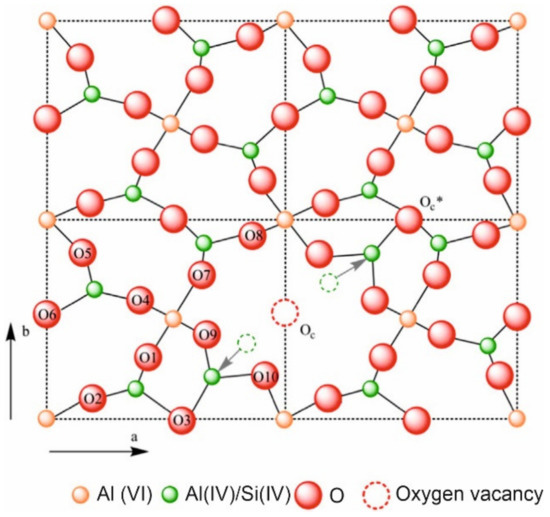
According to the Mindat database [
45], mullite (The name and chemical composition of the minerals and crystalline phases mentioned in this review are showed in
Appendix A) is a nesosilicate, a group of silicates characterized by un-polymerized SiO
4 tetrahedrons, linked to one another by ionic bonds through interstitial cations. The chemical formula is Al(
4+2x)Si(
2−2x)O(
10−x) where x = 0.17–0.59, and is composed of 38.00 wt.% aluminum, 13.18 wt.% silicon, and 48.82 wt.% oxygen, with the empiric formula Al
4.5Si
1.5O
9.75. shows the structure of mullite [
46]. The mullite etymology is related to the place where it was found the first time in 1924, the Isle of Mull, Scotland, UK. Mullite is a crystalline phase of great technological importance due to its excellent technical properties, such as: low expansion and thermal conductivity (~4.5 × 10
−6 °C
−1 and 6 kcal·m
−1·h
−1 °C
−1 at 20 °C, respectively), and appropriate fracture strength and toughness (~200 MPa y ~2.5 MPa m
1/2, respectively) [
47]. In addition, it features high creep resistance, corrosion stability and thermal stability. All these features currently make mullite to be, probably, one of the most important phases in traditional and advanced ceramic. Monolithic mullite ceramics find application in tableware [
48], porcelain stoneware [
49,
50], refractory [
51] or electronic devices [
52]. Mullite coatings have been used successfully as environmental barrier coatings for the protection against oxidizing agents, reducing agents or aggressive chemicals at high temperatures [
53,
54]. Besides ceramic or metallic surfaces, mullite composites, including composites prepared with mullite matrix or mullite fiber, find important applications as components in turbine engines, high-performance furnaces or heat shields for space vehicles [
55,
56,
57].
Figure 4. Crystal structure of mullite along c-axis [
46].
Mullite is a mineral not very abundant in nature since high temperature and low pressure conditions are required for its formation. Due to its high technological demand, mullite has to be manufactured by synthetic methods, rather than being mined. For the synthesis of mullite, there are three basic routes, namely: sintering, melting and chemical processing [
58].
-
Sinter-mullite. Through sintering processing, mullite is basically obtained by reactions in the solid state by interdiffusion of aluminum, silicon and oxygen atoms. The aluminum-bearing raw materials used for the synthesis of mullite are mainly clay minerals, basically kaolinite (Al
2Si
2O
5(OH)
4) and pirofilite (4SiO
2·Al
2O
3·H
2O); other minerals such as Al
2SiO
5 (sillimanite, cianite and andalusite); AlO(OH) (boehmite and diaspore); gibbsite (Al(OH)
3) and bauxite ((AlO
x(OH)
3−2x) with x = 0–1) are usually added as supplementary raw materials. Quartz (SiO
2) is used as Si source. During sintering at ~500 °C, kaolinite is transformed into metakaolinite (Al
2Si
2O
7) by the loss of structural molecules of water. Later, at higher temperature (~980 °C) metakaolinite is decomposed in Si-Al spinel and amorphous silica. The reaction between these two phases at~1200 °C origins the formation of mullite [
50]. For the complete transformation of kaolinite into mullite by the sintering process, very high temperatures are required (1600–1700 °C) due to the diffusion coefficient of mullite in the grain border being very low. To reduce the mullitization temperature, it is convenient to use systems to mix raw materials at the atomic level.
-
Fused-mullite. Mullite is synthesized by fusing of raw materials (alumina Bayer, quartz sand, rock crystal and fused silica) in an electric furnace at a temperature over 2000 °C until homogeneous molten is achieved. By controlled crystallization during the cooling, molten mullite is developed [
59].
-
Chemical-mullite. This method comprises the use of different chemical processes such as sol-gel technologies [
60], precipitation [
61], hydrolysis [
62], pyrolysis [
63] or cool vapor deposition techniques (CVD) [
64].
Accordingly, the synthesis of mullite is an expensive process due to the requirement of high temperature or complex synthesis methods. On the other hand, the kaolin world reserves are drastically decreasing, because their demand for the production of paper and ceramic building materials (principally ceramic tiles). For both reasons, during the two last decades, research efforts have been conducted to study the synthesis of mullite from less common and low cost raw materials such as inorganic waste from different sources and characteristics. Thus, waste from the agro-food industry (rice husk ash) [
65,
66], spent cracking catalysts [
67,
68,
69], aluminum buffing dust [
70], aluminum sludge generated in water treatment plants [
71], ceramic wastes from the manufacture of ceramic tiles [
72] or fly ash [
73,
74], among others have been investigated as raw materials for the synthesis of mullite.
Furthermore, the valorization of mining waste by the production of mullite-based ceramic materials has also been the object of scientific interest. The aim of this work is to carry out a review of the scientific production performed in recent years on the use of waste rock and tailings from the mining industry in the manufacture of ceramic materials in which mullite is developed as one of the main crystalline phases. In this sense, the review includes mining waste from the extraction of metals (iron, aluminum, boron, molybdenum and lithium) and minerals (coal gangue, kaolin and ornamental rocks).