Human skin is continuously subjected to environmental stresses, as well as extrinsic and intrinsic noxious agents. Although skin adopts various molecular mechanisms to maintain homeostasis, excessive and repeated stresses can overwhelm these systems, leading to serious cutaneous damage, including both melanoma and non-melanoma skin cancers. Phytochemicals present in the diet possess the desirable effects of protecting the skin from damaging free radicals as well as other benefits. Dietary phytochemicals appear to be effective in preventing skin cancer and are inexpensive, widely available, and well tolerated. Multiple in vitro and in vivo studies have demonstrated the significant anti-inflammatory, antioxidant, and anti-angiogenic characteristics of dietary phytochemicals against skin malignancy. Moreover, dietary phytochemicals affect multiple important cellular processes including cell cycle, angiogenesis, and metastasis to control skin cancer progression. Herein, we discuss the advantages of key dietary phytochemicals in whole fruits and vegetables, their bioavailability, and underlying molecular mechanisms for preventing skin cancer. Current challenges and future prospects for research are also reviewed. To date, most of the chemoprevention investigations have been conducted preclinically, and additional clinical trials are required to conform and validate the preclinical results in humans.
- Skin carcinogenesis
- Dietary phytochemicals
- Chemoprevention
1. Introduction
1.1. Structure of Skin
Skin represents the largest barrier of the human body to external insults. Histologically, skin is divided into three distinct layers, namely, epidermis, dermis, and hypodermis [1] (Figure 1). In general, skin provides protection from external pressures, serves as a source of percutaneous absorption, and has a significant role in thermoregulation. It is also responsible for the synthesis of macromolecules and aesthetics. The hypodermis consists of subcutaneous fatty tissue along with connective tissue. The dermis exhibits vascularity, neuronal ending, receptors (touch, nociception, and proprioception), hair, and glands. The extracellular matrix of the dermis contains connective tissue rich in collagen and elastin. The epidermis is the outer skin layer which is in direct contact with the atmosphere [1][2]. Approximately 80% of epidermal cells are keratinocytes. Due to its contact with the external environment, these cells undergo continuous wear and tear with constant replacement by newer keratinocytes from the basal epidermal layer. The separation between the epidermis and dermis is through the basement membrane [3]. Other specialized cells in the epidermis include melanocytes (melanin producing), Merkel cells (sensory), and Langerhans cells (immune role). All the cells of the skin perform complex functions, working to achieve and maintain constant homeostasis against various internal and external stimuli. In the case of damage to the skin cells, connective tissue, and other macromolecules, normal processes are dysregulated and give rise to various diseases, including mycoses, inflammatory diseases, and neoplasms [1][4][5].
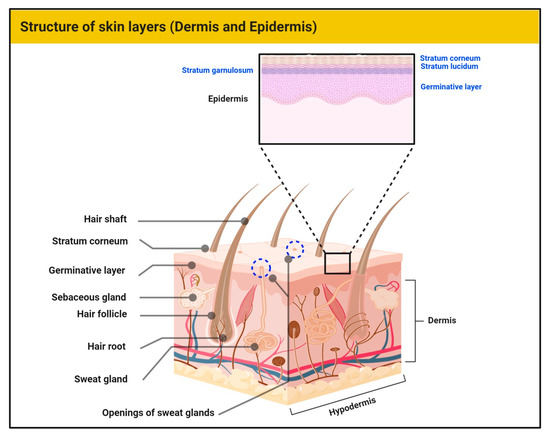
Figure 1. The human skin showing the layers of the epidermis and main structures of the dermis.
1.2. Skin Carcinogenesis
Neoplastic development and progression in the skin is a process consisting of three phases: initiation, followed by promotion and progression of tumor cells (Figure 2). The etiology of initiation may be either exogenous (chemicals, virus, ultraviolet (UV) radiation) or endogenous factors (inflammation). Damage caused by these factors to skin cells is irreparable when cells that cannot regain normal function advance to neoplastic progression [6]. Most ultraviolet B (UVB) radiation induces DNA damage that occurs as a transition from C to T or CC to TT in various genes including tumor suppressor gene p53 [7]. Ultraviolet A (UVA) radiation causes mutations via 8-oxodG. These are present as transitions from AT to CG in various genes that regulate cell cycle, apoptosis, and genomic stability (p53) [7][8]. Genetic mutation and continuous exposure to UV radiation leads to the advancement of initiated cells into the promotion stage [9]. Cells in the initiation stage replicate more vigorously and have less of a tendency to undergo apoptosis [9]. Actinic keratosis (AK) is an example of a precancerous lesion in humans that exhibits extensive mutations when compared with normal human skin cells [10].
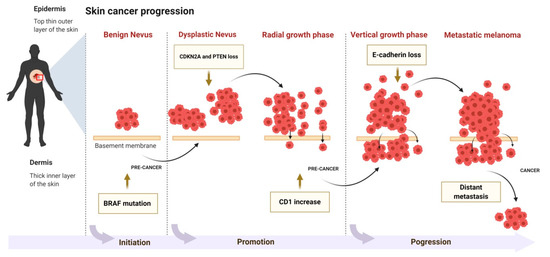
Figure 2. The molecular mechanism of skin cancer progression.
Basal cell carcinoma (BCC), as well as squamous cell carcinoma (SCC), constitute the majority of non-melanoma skin cancers (NMSCs), with Caucasians being affected the most. Both arise from the cancerous transformation of normal keratinocytes [4][8]. Rogers et al. have estimated that approximately two million procedures in about 1.3 million individuals were carried out in 2012, and has increased in recent years [11]. Individuals usually develop invasive NMSCs in later stages of life due to its long latent period, even if the tumor-initiating mutations occurred during childhood. The incidence is less common at a younger age [11][12] and it is more frequent in men [13]. As the fifth most expensive cancer, NMSCs account for the expenditure of billions of dollars in health care and treatment [14]. Usually, patients diagnosed with NMSC die from secondary tumors in the lungs, prostate, or colon resulting from distant metastasis [15]. Therefore, early diagnosis and prompt treatment is highly recommended.
In the United States, most of the reported cases of BCC account for >3 million patients annually [11][12]. BCCs exhibit a slower growth rate with a rare chance of metastasis [16], though it does have a tendency to invade surrounding tissues [17]. In the US, SCC remains the second most prevalent skin cancer, accounting for 0.2–0.4 million cases each year. SCC has more of a tendency to metastasize, exhibiting a rate of approximately 4%. The annual number of deaths from metastatic disease is approximately 3000 [15][18][19]. The mortality rate increases in certain cases such as that of immunocompromised individuals [20]. Several studies have explored the transformation mechanism from normal keratinocytes to AK and finally, to SCC. It is believed that a greater period of latency occurs in SCC because AK is resistant to increases in harmful mutations that lead to SCC [10].
The current treatment for NMSCs includes complete eradication of the lesion while preserving the structural and functional architecture of the skin. Current treatment modalities involve surgical intervention including excision, curettage and electrodessication, cryosurgery, micrographic surgery, radiotherapy, and photodynamic therapy. Surgical procedures, such as the Mohs procedure, are preferred to other methods in cases of low-risk lesions [21]. However, the chance of recurrence is high with surgical procedures lacking a complete histological evaluation of the tumor to ensure its entire excision [22].
2. Dietary Phytochemicals for Skin Cancer Therapy
Dietary phytochemicals are present in plant-based food and exhibit certain nutritional and medicinal value, particularly in preventing various ailments and promotion of human health. Dietary phytochemicals are usually plant secondary metabolites and help proper functioning of human body. Studies have reported approximately 25,000 phytonutrients belonging to different classes of phytochemicals like polyphenols, phenolic acids, flavonoids, diarylalkanoids, carotenoids, lignans, anthocyanins, coumarins, terpenes, and sterols. In addition to various commonly-consumed vegetables and fruits, food items like nuts, beans, tea, and whole grains also contain significant amounts of phytonutrients. Owing to their inherent antioxidative characteristics, these phytonutrients contribute to enhance cardiovascular health, manage the diabetes, and offer cancer chemoprevention [23][24][25]. Although most dietary phytochemicals do not act like drugs for treating diseases, researchers suggests that their intake can be correlated to a number of positive health outcomes including reduced risk of cancer [24][26]. In this review we have covered various classes of dietary phytochemicals mainly in the context of their beneficial role against skin cancer. Our aim is to briefly introduce various aspects of dietary phytochemicals, highlighting their major sources, chemical classes, and major benefits in skin malignancy.
3. Role of Whole Fruits and Vegetables in Skin Cancer Prevention
Fruits and vegetables have high nutrient content. Whole fruits can be fresh, dried, canned, or frozen. Dieticians recommend that fruits should be ingested whole or in 100% pure juice form, which preserves their nutrient-dense nature [27]. Studies have shown that fruits contain various phytonutrients with anti-carcinogenic, anti-mutagenic, antioxidant, and anti-inflammatory properties [28][29][30][31]. Here, we discuss the benefits whole fruits and vegetables in the context of skin cancer. This section includes multiple in vitro and in vivo studies on the effects of whole fruit and fruit extracts against skin cancer (Figure 5).
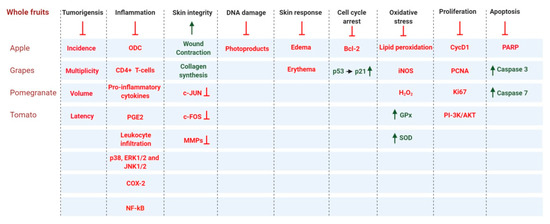
Figure 5. Whole fruit and vegetable products contribute to the prevention of skin carcinogenesis by regulating a broad range of cell signaling pathways. Arrows and dark green text indicate activation and/or upregulation, and red lines with a blunt end and red text indicate inhibition and/or downregulation.
4. Limitation, Safety Consideration, and Future Prospects of Dietary Phytochemicals
Dietary phytochemicals have experienced many challenges including low bioavailability. In our daily diet, we consume a significant amount of these phytochemicals. They are readily digested and eliminated by our body, resulting in a short-lived pharmacologic window [32]. Researchers are striving to develop new approaches that will increase the stability of dietary phytochemicals. Noteworthy strategies include the development of a stable dosage form of dietary phytochemicals, such as microparticles or nanoparticles, exhibiting increased stability and antioxidant properties [33]. Iqbal et al. demonstrated that EGCG showed increased stability and high absorption in the intestine when green tea extract was coated with chitosan [34]. Yadav et al. observed enhanced stability and antioxidant activity of bovine serum albumin-coated catechin and epicatechin nanoparticles [35].
The absence of target specificity is another important challenge for dietary phytochemicals in cancer treatment. It has been shown that phytochemicals exert pleiotropic effects at the cellular level, whereas cancer cells activate other cell signals resulting from the failure of targeted therapy [36]. Scientists strive to utilize alternative strategies for managing these obstacles, including novel formulations for targeted delivery of phytochemicals, formulating semi-synthetic derivatives and analogs of phytochemicals, and development of novel drug delivery systems to enhance the effectiveness, protective characteristics, bioavailability, and pharmacokinetics of phytochemicals in humans [37][38]. Further studies are needed to meet the challenges of topical skin cancer formulations which include skin penetration, optimum drug concentration, stability, dosing strategy, and sustained drug release following topical application.
5. Conclusions
Dietary phytochemicals have several advantages for skin cancer prevention because they are readily available, cost-effective, and well tolerated. The use of dietary phytochemicals have an inverse relationship with skin cancer [39][40]. They are natural antioxidants, elevating the levels of antioxidant enzymes, CDKs, cyclins, p53, p21, and Bax. Moreover, they scavenge ROS, and decrease various molecular targets including EGFR, Notch-1, ERK, MAPK, NF-kB, STAT, β-catenin, PI3K, AKt, and mTOR [41][42][40]. Dietary phytochemicals can also inhibit the proliferation of established skin cancer cells by arresting the cell cycle, preventing metastasis and angiogenesis, suppressing EMT, regulating epigenetic alterations, and downregulating MMPs and COX-2 enzymes. Further investigation, including short-term human studies, may be beneficial in assessing the human relevance of the preclinical data. Moreover, skin cancer chemoprevention investigations involving whole fruits and vegetables are required in the humans who are at high risk, such as individuals with compromised immunity. Preclinical studies in models of high-risk skin carcinogenesis may show beneficial effects. Furthermore, whole fruits and vegetables may also be combined with existing therapeutic strategies for the better management of skin cancer.
This entry is adapted from the peer-reviewed paper 10.3390/antiox9100916
References
- Brohem, C.A.; da Silva Cardeal, L.B.; Tiago, M.; Soengas, M.S.; de Moraes Barros, S.B.; Maria-Engler, S.S. Artificial skin in perspective: Concepts and applications. Pigment. Cell Melanoma Res. 2011, 24, 35–50.
- Kumari, S.; Pasparakis, M. Epithelial cell death and inflammation in skin. In Apoptotic and Non-Apoptotic Cell Death; Springer: Cham, Switzerland, 2015; pp. 77–93.
- Fuchs, E.; Raghavan, S. Getting under the skin of epidermal morphogenesis. Nat. Rev. Genet. 2002, 3, 199–209.
- Apalla, Z.; Nashan, D.; Weller, R.B.; Castellsagué, X. Skin cancer: Epidemiology, disease burden, pathophysiology, diagnosis, and therapeutic approaches. Dermatol. Ther. 2017, 7, 5–19.
- Bickers, D.R.; Athar, M. Oxidative stress in the pathogenesis of skin disease. J. Investig. Dermatol. 2006, 126, 2565–2575.
- Pitot, H.C.; Dragan, Y.P. Facts and theories concerning the mechanisms of carcinogenesis. FASEB J. 1991, 5, 2280–2286.
- Benjamin, C.L.; Ananthaswamy, H.N. p53 and the pathogenesis of skin cancer. Toxicol. Appl. Pharmacol. 2007, 224, 241–248.
- Agar, N.S.; Halliday, G.M.; Barnetson, R.S.; Ananthaswamy, H.N.; Wheeler, M.; Jones, A.M. The basal layer in human squamous tumors harbors more UVA than UVB fingerprint mutations: A role for UVA in human skin carcinogenesis. Proc. Natl. Acad. Sci. USA 2004, 101, 4954–4959.
- Rebel, H.; Mosnier, L.O.; Berg, R.J.; Westerman-de Vries, A.; van Steeg, H.; van Kranen, H.J.; de Gruijl, F.R. Early p53-positive foci as indicators of tumor risk in ultraviolet-exposed hairless mice: Kinetics of induction, effects of DNA repair deficiency, and p53 heterozygosity. Cancer Res. 2001, 61, 977–983.
- Ra, S.H.; Li, X.; Binder, S. Molecular discrimination of cutaneous squamous cell carcinoma from actinic keratosis and normal skin. Mod. Pathol. 2011, 24, 963–973.
- Rogers, H.W.; Weinstock, M.A.; Feldman, S.R.; Coldiron, B.M. Incidence estimate of nonmelanoma skin cancer (keratinocyte carcinomas) in the US population, 2012. JAMA Dermatol. 2015, 151, 1081–1086.
- Christenson, L.J.; Borrowman, T.A.; Vachon, C.M.; Tollefson, M.M.; Otley, C.C.; Weaver, A.L.; Roenigk, R.K. Incidence of basal cell and squamous cell carcinomas in a population younger than 40 years. JAMA 2005, 294, 681–690.
- Wu, S.; Han, J.; Li, W.-Q.; Li, T.; Qureshi, A.A. Basal-cell carcinoma incidence and associated risk factors in US women and men. Am. J. Epidemiol. 2013, 178, 890–897.
- Guy, G.P., Jr.; Machlin, S.R.; Ekwueme, D.U.; Yabroff, K.R. Prevalence and costs of skin cancer treatment in the US, 2002−2006 and 2007−2011. Am. J. Prev. Med. 2015, 48, 183–187.
- Rees, J.R.; Zens, M.S.; Celaya, M.O.; Riddle, B.L.; Karagas, M.R.; Peacock, J.L. Survival after squamous cell and basal cell carcinoma of the skin: A retrospective cohort analysis. Int. J. Cancer 2015, 137, 878–884.
- Jensen, A.Ø.; Lamberg, A.L.; Jacobsen, J.B.; Olesen, A.B.; Sørensen, H.T. Non-melanoma skin cancer and ten-year all-cause mortality: A population-based cohort study. Acta Derm. Venereol. 2010, 90, 362–367.
- Epstein, E.H. Basal cell carcinomas: Attack of the hedgehog. Nat. Rev. Cancer 2008, 8, 743–754.
- Alam, M.; Armstrong, A.; Baum, C.; Bordeaux, J.S.; Brown, M.; Busam, K.J.; Eisen, D.B.; Iyengar, V.; Lober, C.; Margolis, D.J. Guidelines of care for the management of cutaneous squamous cell carcinoma. J. Am. Acad. Dermatol. 2018, 78, 560–578.
- Brantsch, K.D.; Meisner, C.; Schönfisch, B.; Trilling, B.; Wehner-Caroli, J.; Röcken, M.; Breuninger, H. Analysis of risk factors determining prognosis of cutaneous squamous-cell carcinoma: A prospective study. Lancet Oncol. 2008, 9, 713–720.
- Cooper, J.Z.; Brown, M.D. Special concern about squamous cell carcinoma of the scalp in organ transplant recipients. Arch. Dermatol. 2006, 142, 755–758.
- Madan, V.; Lear, J.T.; Szeimies, R.-M. Non-melanoma skin cancer. Lancet 2010, 375, 673–685.
- Werlinger, K.D.; Upton, G.; Moore, A.Y. Recurrence rates of primary nonmelanoma skin cancers treated by surgical excision compared to electrodesiccation-curettage in a private dermatological practice. Dermatol. Surg. 2002, 28, 1138–1142.
- Krzyzanowska, J.; Czubacka, A.; Oleszek, W. Dietary phytochemicals and human health. In Bio-Farms for Nutraceuticals; Springer: Boston, MA, USA, 2010; pp. 74–98.
- Probst, Y.C.; Guan, V.X.; Kent, K. Dietary phytochemical intake from foods and health outcomes: A systematic review protocol and preliminary scoping. BMJ Open 2017, 7.
- Bansal, M.; Singh, N.; Pal, S.; Dev, I.; Ansari, K.M. Chemopreventive role of dietary phytochemicals in colorectal cancer. In Advances in Molecular Toxicology; Fishbein, J.C., Heilman, J.M., Eds.; Elsevier: Amsterdam, The Netherlands, 2018; Volume 12, pp. 69–121.
- Forni, C.; Facchiano, F.; Bartoli, M.; Pieretti, S.; Facchiano, A.; D’Arcangelo, D.; Norelli, S.; Valle, G.; Nisini, R.; Beninati, S. Beneficial role of phytochemicals on oxidative stress and age-related diseases. BioMed Res. Int. 2019, 2019.
- DeSalvo, K.B.; Olson, R.; Casavale, K.O. Dietary guidelines for Americans. JAMA 2016, 315, 457–458.
- Hussain, F.; Rana, Z.; Shafique, H.; Malik, A.; Hussain, Z. Phytopharmacological potential of different species of Morus alba and their bioactive phytochemicals: A review. Asian Pac. J. Trop. Biomed. 2017, 7, 950–956.
- Chaudhary, P.; Sharma, A.; Singh, B.; Nagpal, A.K. Bioactivities of phytochemicals present in tomato. J. Food Sci. Technol. 2018, 55, 2833–2849.
- Cavazos-Garduño, A.; Serrano-Niño, J.C.; García-Varela, R.; García, H.S. Anticarcinogenic phytochemicals. Fruit Veg. Phytochem. Chem. Hum. Health 2017, 2, 53–66.
- Singh, C.K.; Siddiqui, I.A.; El-Abd, S.; Mukhtar, H.; Ahmad, N. Combination chemoprevention with grape antioxidants. Mol. Nutr. Food Res. 2016, 60, 1406–1415.
- Stylos, E.; Chatziathanasiadou, M.V.; Syriopoulou, A.; Tzakos, A.G. Liquid chromatography coupled with tandem mass spectrometry (LC–MS/MS) based bioavailability determination of the major classes of phytochemicals. J. Chromatogr. B 2017, 1047, 15–38.
- Krupkova, O.; Ferguson, S.J.; Wuertz-Kozak, K. Stability of (−)-epigallocatechin gallate and its activity in liquid formulations and delivery systems. J. Nutr. Biochem. 2016, 37, 1–12.
- Iqbal, J.; Abbasi, B.A.; Ahmad, R.; Mahmood, T.; Ali, B.; Khalil, A.T.; Kanwal, S.; Shah, S.A.; Alam, M.M.; Badshah, H. Nanomedicines for developing cancer nanotherapeutics: From benchtop to bedside and beyond. Appl. Microbiol. Biotechnol. 2018, 102, 9449–9470.
- Yadav, R.; Kumar, D.; Kumari, A.; Yadav, S.K. Encapsulation of catechin and epicatechin on BSA NPs improved their stability and antioxidant potential. EXCLI J. 2014, 13, 331.
- Ahmad, A.; Li, Y.; Sarkar, F.H. The bounty of nature for changing the cancer landscape. Mol. Nutr. Food Res. 2016, 60, 1251–1263.
- Islam, S.U.; Ahmed, M.B.; Ul-Islam, M.; Shehzad, A.; Lee, Y.S. Switching from Conventional to Nano-natural Phytochemicals to Prevent and Treat Cancers: Special Emphasis on Resveratrol. Curr. Pharm. Des. 2019, 25, 3620–3632.
- Subramanian, A.; Jaganathan, S.; Manikandan, A.; Pandiaraj, K.; Gomathi, N.; Supriyanto, E. Recent trends in nano-based drug delivery systems for efficient delivery of phytochemicals in chemotherapy. RSC Adv. 2016, 6, 48294–48314.
- Chikara, S.; Nagaprashantha, L.D.; Singhal, J.; Horne, D.; Awasthi, S.; Singhal, S.S. Oxidative stress and dietary phytochemicals: Role in cancer chemoprevention and treatment. Cancer Lett. 2018, 413, 122–134.
- Afrin, S.; Giampieri, F.; Gasparrini, M.; Forbes-Hernández, T.Y.; Cianciosi, D.; Reboredo-Rodriguez, P.; Zhang, J.; Manna, P.P.; Daglia, M.; Atanasov, A.G. Dietary phytochemicals in colorectal cancer prevention and treatment: A focus on the molecular mechanisms involved. Biotechnol. Adv. 2020, 38, 107322.
- Iqbal, J.; Abbasi, B.A.; Ahmad, R.; Batool, R.; Mahmood, T.; Ali, B.; Khalil, A.T.; Kanwal, S.; Shah, S.A.; Alam, M.M. Potential phytochemicals in the fight against skin cancer: Current landscape and future perspectives. Biomed. Pharmacother. 2019, 109, 1381–1393.
- Montes de Oca, M.K.; Pearlman, R.L.; McClees, S.F.; Strickland, R.; Afaq, F. Phytochemicals for the prevention of photocarcinogenesis. Photochem. Photobiol. 2017, 93, 956–974.
