Malaria is an infectious disease caused by protozoan parasites of the Plasmodium genus through the bite of female Anopheles mosquitoes, affecting 228 million people and causing 415 thousand deaths in 2018. Artemisinin-based combination therapies (ACTs) are the most recommended treatment for malaria; however, the emergence of multidrug resistance has unfortunately limited their effects and challenged the field.
- Plasmodium
- malaria
- sponge
- resistance
- antimalarial
1. Introduction
Human malaria is an infectious disease caused by single-celled protozoan parasites of the Plasmodium genus (P. falciparum, P. vivax, P. ovale, P. malariae, and P. knowlesi) through the bite of female Anopheles mosquitoes [1]. It affected 228 million people in 2018, and nearly half of the world’s population is still at risk for this disease [2]. Symptoms can range from being mild to very severe, causing chronic illness, physical disability, death and a huge health burden, especially to the most vulnerable populations.
Antimalarials based in quinolines scaffolds (i.e., chloroquine, mefloquine, amodiaquine, and piperaquine) possess a complex mechanism of action. One well-studied mechanism involves compromising the detoxification of hemoglobin degradation with heme polymerization for hemozoin crystal formation in digestive vacuole by protonated forms of quinolones [3]. It was noted that some strains of P. falciparum triggered resistance to protonated drugs due to a genetic mutation in the transporter (PfCRT) and could lead to antimalarial drug extrusion from the organelle [3].
Artemisinin-based combination therapies (ACTs) are the most recommended treatment for uncomplicated P. falciparum malaria, while artesunate is considered the most effective antimalarial drug for severe cases [4], with several biochemical processes reported as targets in parasite cells [3][5]. Despite the safety and efficiency that have been proven for the use of these drugs, the emergence of multidrug resistance has unfortunately limited their effects and challenged the field [6]. The resistance to ACTs is already spreading from Southeast Asia, as reported in 2008 [7], giving rise to a danger alert to other high-poverty regions in the world, and the identified resistance phenotype is associated with mutation of kelch domain protein gene (k13), which is postulated to be involved in protein trafficking organelles in the parasite during intraerythrocytic cycle [3][8].
In this context, the ocean, with its rich biodiversity, has been emerging as a very promising resource of bioactive compounds and secondary metabolites from different marine organisms (bacteria, fungi, micro-algae, mollusks and other invertebrates) with multiple pharmacological properties [9][10][11]. Among them, the phylum Porifera (sponges) is the most promising for providing raw material for the development of biotechnological products for multiple human health problems [12][13][14]. Marine sponges are very primitive sessile animals with origins dated at least from the late Proterozoic over 580 million years ago [15]. Being considered representatives of the first multicellular animals, these filter-feeding organisms evolutionarily developed morphological and chemical defense mechanisms constituted mainly by secondary metabolites, compounds with a wide range of effects such as antitumor, antiviral, anti-inflammatory and antibiotic effects, which have been investigated for the treatment of human health problems [15][16]. Additionally, some authors have demonstrated the antimalarial effects of the secondary metabolites of marine sponges and have shown that these components present inhibitory activity against the malaria parasite Plasmodium falciparum [6][17].
2. Results and Discussion
2.1. Study Selection and Analysis
The flow diagram (Figure 1) demonstrated the search strategy (identification, inclusion and exclusion) used in the present study. A total of 77 articles were retrieved from the databases (PubMed, Web of Science and Scopus). Then, the duplicated records were excluded (n = 14). Thus, 66 full-text articles were assessed for eligibility, and 30 studies were excluded for different reasons, such as the following: some studies reported only the extraction of compounds and did not report the antiplasmodial activity; others described only the mechanism of the compounds; some studies were only computational. Finally, 36 studies were included and analyzed in this systematic review (Figure 1).
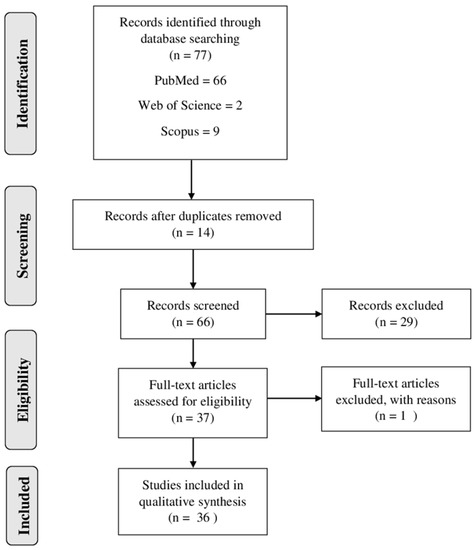
Figure 1. Flow diagram of literature search and selection criteria used in the present review adapted from PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analysis).
A summary of the studies is presented in Table 1. The articles analyzed were published from 1992 to 2019 in different countries. The antimalarial activity was assessed in vitro using Plasmodium falciparum culture [18] for quantification of cell viability over 24-96 h. For the in vitro assays, different lab strains were used (such as 3D7, W2, DD2, NF54), and a wide variety of methods were used for assessing P. falciparum viability ([3H] hypoxanthine, LDH, Microscopy, SYBR Green) presenting as IC50 values instead of the option of XC50. The Demospongiae sponge class was the most explored, where 30 studies evaluated their antiplasmodial activity. Among the genera in Table 1, most belong to the Demospongiae class except for Plakortis (Plakortis simplexs, Plakortis lita, Plakortis halichondrioides), which is from the Homoscleromorpha class. In addition, a great geographical variety was observed, which shows that sponges from different regions of the globe have this potential antiplasmodial activity. The inhibitory concentration for 50% of the parasites (IC50) varied from low micromolar to low nanomolar range, and the species Xestospongia sp showed the best bioactive potential, from which the compound Saringosterol was extracted, which had an IC50 of 0.25 nM. The individual IC50 for each extracted compound is reported in Table 1. The IC50 value units in µg/mL and ng/mL were converted to µM and nM for data comparison, and then some of compounds in Table 1, which IC50 was below 10 µg/mL became higher than 10 µM, as were compounds 10 and 11 [19], 52 [20], 2 and 3 [21], 99 [22].
Table 1. Summary of descriptions of characteristics of included articles.
| Author | Sponge Genus | Material Collection Location | Extracted Material (P. falciparum Strain and IC50 Value) |
|---|---|---|---|
| Campos et al., (2019) [19] | Fascaplysinopsis reticulata | Mayotte (Indian Ocean) | 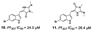 |
| Jeong., et al. (2019) [23] | Coscinoderma sp. | Chuuk Island, Federated States of Micronesia | 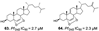 |
| Ju et al., (2019) [23] | Hyrtios erectus | Chuuk Island, Federated States of Micronesia |  |
| Murtihapsari. et al., (2019) [24] | Xestospongia sp | Kaimana, West Papua, Indonesia | 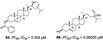 |
| Parra et al., (2018) [25] | Tedania Brasiliensis | Brazil |  |
| Campos et al., (2017) [26] | Monanchora unguiculata | Mitsio islands, Madagascar |  |
| Yang et al., (2016) [27] |
Diacarnus megaspinorhabdosa | SouthChina Sea Sponge |  |
| Gros et al., (2015) [28] |
Biemna laboutei | Madagascar | 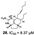 |
| Chianese et al., (2014) [29] | Plakortis simplexs | South China Sea | 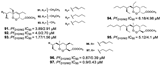 |
| Gros et al., (2014) [30] | Biemna laboutei | Madagascar at Salary Ba |  |
| Yang et al., (2014) [17] | Diacarnus megaspinorhabdosa | South China Sea |  |
| Alvarado et al., (2013) [31] |
Spongosorites sp | Not reported | 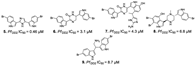 |
| Davis et al., (2013) [32] |
Plakortis lita | Not reported | 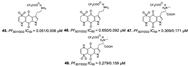 |
| Farokhi et al., (2013) [33] |
Axinyssa djiferi | Senegalese coasts |  |
| Sirirak et al., (2013) [34] |
Pachastrissa nuxs | Thailand | 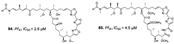 |
| Chanthathamrongsiri et al., (2012) [35] |
Stylissacf. massa | Not reported | 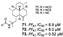 |
| Davis et al., (2012) [36] |
Zyzzya sp | Not reported |  |
| Ilias et al., (2012) [37] |
Petrosia | Eastern Fields north of Australia | 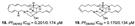 |
| Mudianta et al., (2012) [38] |
Aplysinella strongylata | Tulamben, Bali, Indonesia |  |
| El Sayed et al., (2011) [20] | Diacarnus erythraeanus | Red Sea |  |
| Galeano et al., (2011) [39] |
Verongula rigida | Urabá Gulf is located in the Southwestern Caribbean |  |
| Sirirak et al., (2011) [40] |
Pachastrissa nux | Koh-Tao, Surat-Thani ProvinceChumphon IslandsNational Park, Chumphon Province, |  |
| Xu et al., (2011) [41] |
Pseudoceratina sp | Australian biota |  |
| Jiménez-Romero et al., (2010) [42] |
Plakortis halichondrioides | Puerto Rico | 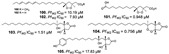 |
| Samoylenko et al., (2009) [43] |
Acanthostrongylophora ingens | Pacific |  |
| Ueoka et al., (2009) [44] |
Agelas gracilis | southern Japan |  |
| Wright et al., (2009) [45] |
Cymbastela hooperi | Not reported |  |
| Appenzeller et al., (2008) [46] |
Agelas cf. mauritiana | Solomon Islands |  |
| Desoubzdanne et al., (2008) [47] |
New Caledonian | Norfolk Rise (New Caledonia) |  |
| Tasdemir et al., (2007) [21] | Agelas oroides | Northern Aegean Sea, Turkey | - fractions: fatty acid mixtures FAME (3.4 μg/mL) and FAMF (8.7 μg/mL)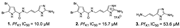 |
| Laurent et al., (2006) [48] | Xestospongia | Vanuatu |  |
| Mancini et al., (2004) [49] | Oceanapia fistulosa | New Caledonia Main Island | -crude mixture (0.98 μM) -N-methyl derivatives from the crude mixture (8 μM) |
| Fattorusso et al., (2002) [50] | Plakortis simplex | Berry Island (Bahamas) |  |
| Gochfeld et al., (2001) [22] | Plakortis sp. | Jamaica |  |
| Kirsch et al., (2000) [51] | Hyrtios cf. erecta | Fiji |  |
| Angerhofer et al., (1992) [52] | Acanthella klethra | Australia |  |
To assess the study quality, we used the GRADE method [53]. The 36 studies analyzed were categorized as moderate quality (17) because (i) there were no controls in the experiments; (ii) the toxicity of the compounds was not assessed in parallel, which made it impossible to determine the selectivity of compounds; (iii) all compounds analyzed presented a high cytotoxicity, which demonstrates the unspecified use against P. falciparum; (iv) the methods used to measure the antiplasmodial activity were not described. A total of 19 studies were classified as being of high quality.
After this detailed review of the articles reporting the activity of compounds from marine sponges, we made a brief survey of data in the literature to compare the number of articles published reporting the activity of marine organisms with the number of articles published reporting the activity of extracts from plants. To do so, the following combinations of keywords were used: "new antimalarials and plants" or "new antimalarials and marine" and selected the works published in the last 10 years. Figure 2 represents the number of studies reporting antiplasmodial activity of new compounds found. The search for new compounds from marine sources is still uncommon compared to the search for natural products from plants. Other recent reviews have also reported this comparison, which reinforces the importance of seeking new products from marine sources, especially considering that the diverse nature of metabolites produced by these alternative sources presents a compelling case for intensive exploration [54].
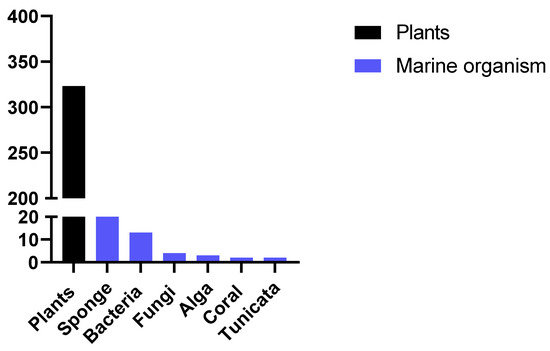
Figure 2. Number of published papers reporting the antimalarial activity of new compounds from marine sources or plants in the past 10 years.
This entry is adapted from the peer-reviewed paper 10.3390/md19030134
References
- Crompton, P.D.; Moebius, J.; Portugal, S.; Waisberg, M.; Hart, G.; Garver, L.S.; Miller, L.H.; Barillas, C.; Pierce, S.K. Malaria immunity in man and mosquito: Insights into unsolved mysteries of a deadly infectious disease. Annu. Rev. Immunol. 2014, 32, 157–187.
- World Health Organization. WHO Malaria Report 2017; World Health Organization: Geneva, Switzerland, 2017.
- Wicht, K.J.; Mok, S.; Fidock, D.A. Molecular Mechanisms of Drug Resistance in Plasmodium falciparum Malaria. Annu. Rev. Microbiol. 2020, 74, 431–454.
- Roussel, C.; Caumes, E.; Thellier, M.; Ndour, P.A.; Buffet, P.A.; Jauréguiberry, S. Artesunate to treat severe malaria in travellers: Review of efficacy and safety and practical implications. J. Travel Med. 2017, 24, taw093.
- Bridgford, J.L.; Xie, S.C.; Cobbold, S.A.; Pasaje, C.F.A.; Herrmann, S.; Yang, T.; Gillett, D.L.; Dick, L.R.; Ralph, S.A.; Dogovski, C.; et al. Artemisinin kills malaria parasites by damaging proteins and inhibiting the proteasome. Nat. Commun. 2018, 9, 1–9.
- Nieves, K.; Prudhomme, J.; Le Roch, K.G.; Franzblau, S.G.; Rodríguez, A.D. Natural product-based synthesis of novel anti-infective isothiocyanate- and isoselenocyanate-functionalized amphilectane diterpenes. Bioorganic Med. Chem. Lett. 2016, 26.
- Noedl, H.; Se, Y.; Schaecher, K.; Smith, B.L.; Socheat, D.; Fukuda, M.M. Evidence of Artemisinin-Resistant Malaria in Western Cambodia. N. Engl. J. Med. 2008, 359, 2619–2620.
- Ariey, F.; Witkowski, B.; Amaratunga, C.; Beghain, J.; Langlois, A.C.; Khim, N.; Kim, S.; Duru, V.; Bouchier, C.; Ma, L.; et al. A molecular marker of artemisinin-resistant Plasmodium falciparum malaria. Nature 2014, 505, 50–55.
- Voultsiadou Eleni, E. Therapeutic properties and uses of marine invertebrates in the ancient Greek world and early Byzantium. J. Ethnopharmacol. 2010, 130.
- Mayer, A.M.S.; Avilés, E.; Rodríguez, A.D. Marine sponge Hymeniacidon sp. amphilectane metabolites potently inhibit rat brain microglia thromboxane B 2 generation. Bioorganic Med. Chem. 2012, 20, 279–282.
- De Alencar, D.B.; Da Silva, S.R.; Pires-Cavalcante, K.M.S.; De Lima, R.L.; Pereira, F.N.; De Sousa, M.B.; Viana, F.A.; Nagano, C.S.; Do Nascimento, K.S.; Cavada, B.S.; et al. Antioxidant potential and cytotoxic activity of two red seaweed species, amansia multifida and meristiella echinocarpa, from the coast of Northeastern Brazil. An. Acad. Bras. Cienc. 2014, 86, 251–263.
- Leys, S.P.; Hill, A. The Physiology and Molecular Biology of Sponge Tissues. Adv. Marine Biol. 2012, 62, 1–56.
- Mehbub, M.F.; Lei, J.; Franco, C.; Zhang, W. Marine sponge derived natural products between 2001 and 2010: Trends and opportunities for discovery of bioactives. Mar. Drugs 2014, 12, 4539–4577.
- Amina, M.; Musayeib, N.M. Al Biological and Medicinal Importance of Sponge. In Biological Resources of Water; IntechOpen: London, UK, 2018.
- Feuda, R.; Dohrmann, M.; Pett, W.; Philippe, H.; Rota-Stabelli, O.; Lartillot, N.; Wörheide, G.; Pisani, D. Improved Modeling of Compositional Heterogeneity Supports Sponges as Sister to All Other Animals. Curr. Biol. 2017, 27, 3864–3870.
- Wang, B.; Dong, J.; Zhou, X.; Huang, R.; Zhang, S.; Liu, Y.; Lee, K.J. Nucleosides from the Marine Sponge Haliclona sp. Z. Fur Nat.-Sect. C J. Biosci. 2009, 64.
- Yang, F.; Zou, Y.; Wang, R.P.; Hamann, M.T.; Zhang, H.J.; Jiao, W.H.; Han, B.N.; Song, S.J.; Lin, H.W. Relative and absolute stereochemistry of diacarperoxides: Antimalarial norditerpene endoperoxides from marine sponge Diacarnus megaspinorhabdosa. Mar. Drugs 2014, 12, 4399–4416.
- Trager, W.; Jensen, J.B. Human malaria parasites in continuous culture. Science 1976, 193, 673–675.
- Campos, P.E.; Pichon, E.; Moriou, C.; Clerc, P.; Trépos, R.; Frederich, M.; De Voogd, N.; Hellio, C.; Gauvin-Bialecki, A.; Al-Mourabit, A. New antimalarial and antimicrobial tryptamine derivatives from the marine sponge fascaplysinopsis reticulata. Mar. Drugs 2019, 17, 167.
- El Sayed, K.A.; Hamann, M.T.; Hashish, N.E.; Shier, W.T.; Kelly, M.; Khan, A.A. Antimalarial, antiviral, and antitoxoplasmosis norsesterterpene peroxide acids from the red sea sponge Diacarnus erythraeanus. J. Nat. Prod. 2001, 64, 522–524.
- Tasdemir, D.; Topaloglu, B.; Perozzo, R.; Brun, R.; O’Neill, R.; Carballeira, N.M.; Zhang, X.; Tonge, P.J.; Linden, A.; Rüedi, P. Marine natural products from the Turkish sponge Agelas oroides that inhibit the enoyl reductases from Plasmodium falciparum, Mycobacterium tuberculosis and Escherichia coli. Bioorganic Med. Chem. 2007, 15, 6834–6845.
- Gochfeld, D.J.; Hamann, M.T. Isolation and biological evaluation of filiformin, plakortide F, and plakortone G from the Caribbean sponge Plakortis sp. J. Nat. Prod. 2001, 64.
- Jeong, H.; Latif, A.; Kong, C.S.; Seo, Y.; Lee, Y.J.; Dalal, S.R.; Cassera, M.B.; Kingston, D.G.I. Isolation and characterization of antiplasmodial constituents from the marine sponge Coscinoderma sp. Z. Fur Nat.-Sect. C J. Biosci. 2019.
- Murtihapsari, M.; Salam, S.; Kurnia, D.; Darwati, D.; Kadarusman, K.; Abdullah, F.F.; Herlina, T.; Husna, M.H.; Awang, K.; Shiono, Y.; et al. A new antiplasmodial sterol from Indonesian marine sponge, Xestospongia sp. Nat. Prod. Res. 2019.
- Parra, L.L.L.; Bertonha, A.F.; Severo, I.R.M.; Aguiar, A.C.C.; De Souza, G.E.; Oliva, G.; Guido, R.V.C.; Grazzia, N.; Costa, T.R.; Miguel, D.C.; et al. Isolation, Derivative Synthesis, and Structure-Activity Relationships of Antiparasitic Bromopyrrole Alkaloids from the Marine Sponge Tedania brasiliensis. J. Nat. Prod. 2018, 81.
- Campos, P.E.; Wolfender, J.L.; Queiroz, E.F.; Marcourt, L.; Al-Mourabit, A.; Frederich, M.; Bordignon, A.; De Voogd, N.; Illien, B.; Gauvin-Bialecki, A. Unguiculin A and Ptilomycalins E-H, Antimalarial Guanidine Alkaloids from the Marine Sponge Monanchora unguiculata. J. Nat. Prod. 2017, 80.
- Yang, F.; Wang, R.P.; Xu, B.; Yu, H.B.; Ma, G.Y.; Wang, G.F.; Dai, S.W.; Zhang, W.; Jiao, W.H.; Song, S.J.; et al. New antimalarial norterpene cyclic peroxides from Xisha Islands sponge Diacarnus megaspinorhabdosa. Bioorganic Med. Chem. Lett. 2016, 26, 2084–2087.
- Gros, E.; Martin, M.T.; Sorres, J.; Moriou, C.; Vacelet, J.; Frederich, M.; Aknin, M.; Kashman, Y.; Gauvin-Bialecki, A.; Al-Mourabit, A. Netamines O-S, Five New Tricyclic Guanidine Alkaloids from the Madagascar Sponge Biemna laboutei, and Their Antimalarial Activities. Chem. Biodivers. 2015, 12.
- Chianese, G.; Persico, M.; Yang, F.; Lin, H.W.; Guo, Y.W.; Basilico, N.; Parapini, S.; Taramelli, D.; Taglialatela-Scafati, O.; Fattorusso, C. Endoperoxide polyketides from a Chinese Plakortis simplex: Further evidence of the impact of stereochemistry on antimalarial activity of simple 1,2-dioxanes. Bioorganic Med. Chem. 2014, 22.
- Gros, E.; Al-Mourabit, A.; Martin, M.T.; Sorres, J.; Vacelet, J.; Frederich, M.; Aknin, M.; Kashman, Y.; Gauvin-Bialecki, A. Netamines H-N, tricyclic alkaloids from the marine sponge biemna laboutei and their antimalarial activity. J. Nat. Prod. 2014, 77.
- Alvarado, S.; Roberts, B.F.; Wright, A.E.; Chakrabarti, D. The bis(Indolyl)imidazole alkaloid nortopsentin a exhibits antiplasmodial activity. Antimicrob. Agents Chemother. 2013, 57.
- Davis, R.A.; Duffy, S.; Fletcher, S.; Avery, V.M.; Quinn, R.J. Thiaplakortones A-D: Antimalarial thiazine alkaloids from the Australian marine sponge plakortis lita. J. Org. Chem. 2013, 78.
- Farokhi, F.; Grellier, P.; Clément, M.; Roussakis, C.; Loiseau, P.M.; Genin-Seward, E.; Kornprobst, J.M.; Barnathan, G.; Wielgosz-Collin, G. Antimalarial activity of axidjiferosides, new β-galactosylceramides from the African sponge Axinyssa djiferi. Mar. Drugs 2013, 11.
- Sirirak, T.; Brecker, L.; Plubrukarn, A. Kabiramide L, a new antiplasmodial trisoxazole macrolide from the sponge Pachastrissa nux. Nat. Prod. Res. 2013, 27.
- Chanthathamrongsiri, N.; Yuenyongsawad, S.; Wattanapiromsakul, C.; Plubrukarn, A. Bifunctionalized amphilectane diterpenes from the sponge Stylissa cf. massa. J. Nat. Prod. 2012, 75.
- Davis, R.A.; Buchanan, M.S.; Duffy, S.; Avery, V.M.; Charman, S.A.; Charman, W.N.; White, K.L.; Shackleford, D.M.; Edstein, M.D.; Andrews, K.T.; et al. Antimalarial activity of pyrroloiminoquinones from the Australian marine sponge zyzzya sp. J. Med. Chem. 2012, 55.
- Ilias, M.; Ibrahim, M.A.; Khan, S.I.; Jacob, M.R.; Tekwani, B.L.; Walker, L.A.; Samoylenko, V. Pentacyclic ingamine alkaloids, a new antiplasmodial pharmacophore from the marine sponge Petrosid Ng5 Sp5. Planta Med. 2012, 78.
- Mudianta, I.W.; Skinner-Adams, T.; Andrews, K.T.; Davis, R.A.; Hadi, T.A.; Hayes, P.Y.; Garson, M.J. Psammaplysin derivatives from the balinese marine sponge Aplysinella strongylata. J. Nat. Prod. 2012, 75.
- Galeano, E.; Thomas, O.P.; Robledo, S.; Munoz, D.; Martinez, A. Antiparasitic Bromotyrosine derivatives from the marine sponge Verongula rigida. Mar. Drugs 2011, 9.
- Sirirak, T.; Kittiwisut, S.; Janma, C.; Yuenyongsawad, S.; Suwanborirux, K.; Plubrukarn, A. Kabiramides J and K, trisoxazole macrolides from the sponge Pachastrissa nux. J. Nat. Prod. 2011, 74.
- Xu, M.; Andrews, K.T.; Birrell, G.W.; Tran, T.L.; Camp, D.; Davis, R.A.; Quinn, R.J. Psammaplysin H, a new antimalarial bromotyrosine alkaloid from a marine sponge of the genus Pseudoceratina. Bioorganic Med. Chem. Lett. 2011, 21.
- Jiménez-Romero, C.; Ortiz, I.; Vicente, J.; Vera, B.; Rodríguez, A.D.; Nam, S.; Jove, R. Bioactive cycloperoxides isolated from the Puerto Rican sponge Plakortis halichondrioides. J. Nat. Prod. 2010, 73.
- Samoylenko, V.; Khan, S.I.; Jacob, M.R.; Tekwani, B.L.; Walker, L.A.; Hufford, C.D.; Muhammad, I. Bioactive (+)-manzamine A and (+)-8-hydroxymanzamine A tertiary bases and salts from Acanthostrongylophora ingens and their preparations. Nat. Prod. Commun. 2009, 4.
- Ueoka, R.; Nakao, Y.; Kawatsu, S.; Yaegashi, J.; Matsumoto, Y.; Matsunaga, S.; Furihata, K.; Van Soest, R.W.M.; Fusetani, N. Gracilioethers A-C, antimalarial metabolites from the marine sponge Agelas gracilis. J. Org. Chem. 2009, 74.
- Wright, A.D.; Lang-Unnasch, N. Diterpene formamides from the tropical marine sponge cymbastela hooperi and their antimalarial activity in vitro. J. Nat. Prod. 2009, 72.
- Appenzeller, J.; Mihci, G.; Martin, M.T.; Gallard, J.F.; Menou, J.L.; Boury-Esnault, N.; Hooper, J.; Petek, S.; Chevalley, S.; Valentin, A.; et al. Agelasines J, K, and L from the Solomon Islands marine sponge Agelas cf. mauritiana. J. Nat. Prod. 2008, 71.
- Desoubzdanne, D.; Marcourt, L.; Raux, R.; Chevalley, S.; Dorin, D.; Doerig, C.; Valentin, A.; Ausseil, F.; Debitus, C. Alisiaquinones and Alisiaquinol, dual inhibitors of Plasmodium falciparum enzyme targets from a new caledonian deep water sponge. J. Nat. Prod. 2008, 71.
- Laurent, D.; Jullian, V.; Parenty, A.; Knibiehler, M.; Dorin, D.; Schmitt, S.; Lozach, O.; Lebouvier, N.; Frostin, M.; Alby, F.; et al. Antimalarial potential of xestoquinone, a protein kinase inhibitor isolated from a Vanuatu marine sponge Xestospongia sp. Bioorganic Med. Chem. 2006, 14.
- Mancini, I.; Guella, G.; Sauvain, M.; Debitus, C.; Duigou, A.G.; Ausseil, F.; Menou, J.L.; Pietra, F. New 1,2,3,4-tetrahydropyrrolo [1,2-a]pyrimidinium alkaloids (phloeodictynes) from the New Caledonian shallow-water haplosclerid sponge Oceanapia fistulosa. Structural elucidation from mainly LC-tandem-MS-soft-ionization techniques and discovery of antiplasmodial activity. Org. Biomol. Chem. 2004, 2, 783–787.
- Fattorusso, E.; Parapini, S.; Campagnuolo, C.; Basilico, N.; Taglialatela-Scafati, O.; Taramelli, D. Activity against Plasmodium falciparum of cycloperoxide compounds obtained from the sponge Plakortis simplex. J. Antimicrob. Chemother. 2002, 50, 883–888.
- Kirsch, G.; Köng, G.M.; Wright, A.D.; Kaminsky, R. A new bioactive sesterterpene and antiplasmodial alkaloids from the marine sponge Hyrtios cf. erecta. J. Nat. Prod. 2000, 63.
- Angerhofer, C.K.; Pezzuto, J.M.; König, G.M.; Wright, A.D.; Sticher, O. Antimalarial activity of sesquiterpenes from the marine sponge acanthella klethra. J. Nat. Prod. 1992, 55.
- Guyatt, G.; Oxman, A.D.; Akl, E.A.; Kunz, R.; Vist, G.; Brozek, J.; Norris, S.; Falck-Ytter, Y.; Glasziou, P.; Debeer, H.; et al. GRADE guidelines: 1. Introduction-GRADE evidence profiles and summary of findings tables. J. Clin. Epidemiol. 2011, 64.
- Tajuddeen, N.; Van Heerden, F.R. Antiplasmodial natural products: An update. Malar. J. 2019, 18, 1–62.
