Infectious diseases caused by bacteria, viruses, or parasites are the major causes of mortality and economic losses in commercial aquaculture. Some pathologies, especially those of bacterial origin, can be treated with commercially available drugs, while others are poorly managed. In fact, despite having been recognized as a useful preventive measure, no effective vaccination against many economically relevant diseases exist yet, such as for viral and parasitic infections.
- adjuvants
- aquaculture
- experimental challenge
- fish immunology
- fish welfare
- vaccines
- infectious diseases
1. Introduction
Aquaculture has experienced an enormous growth in productive terms, accounting to >527% in the 1990–2018 time frame. In 2018, aquaculture contributed to approximately 46% of the global total production of aquatic organisms (179 M tons) and 52% of seafood for human consumption (fish, crustaceans, mollusks, and other aquatic animals, excluding aquatic mammals, reptiles, seaweeds, and other aquatic plants) [1]. Capture-wise, any further increment in global productions will have to strictly ensure the preservation of natural resources, the 59.6% of which is currently being maximally sustainably fished, and avoid overfishing practices, also because of the severe ecological problems they are linked to (e.g., damages to coastal and marine ecosystems, alteration of multiple trophic levels, and algal blooms) [2][3]. Because of the increasing world population and per capita consumption [4], aquaculture is expected to continue growing, with conservative projections estimating 186 M tons production by year 2030 [5].
Commercial aquaculture is impacted by infectious diseases caused primarily by bacteria, viruses, parasites, and, to a lesser extent, fungi. Bacterial diseases can inflict significant biological, thus economic losses [6][7][8]. While these are usually controllable with antibiotics, the indiscriminate use of these pharmaceuticals is ultimately a threat to human health because of the development and transfer of resistance mechanisms among bacterial species, some of which are also human pathogens. Their employment is therefore strongly regulated in many countries [9].
Various prevention strategies are currently used such as (i) biocontainment measures (e.g., quarantine and disease screening of newly introduced fishes) [10], (ii) water treatment systems (e.g., magnetic, ultraviolet, and ozone sterilization, all practically applicable only in recirculating systems) [11][12], and (iii) probiotics/prebiotics supplementation for immune system stimulation and growth promotion [13].
Fish vaccination can prevent or mitigate disease spreading with proven effectiveness against many relevant pathogens. The vaccine against enteric redmouth disease (caused by Yersinia ruckeri) developed in 1970s was the first to become commercially available [14], later followed by vaccines against cold water vibriosis (caused by Aliivibrio salmonicida) [15]. Since then, various vaccines have been developed, commercialized, successfully employed and reviewed [16][17]. Still, because of their high development and production costs and general lower efficacy than bacterins, few vaccines exist against viral diseases, and no commercial vaccines at all are available to date against parasitic diseases [18].
2. The factors affecting the efficacy of vaccines
This entry discusses the most promising and updated state-of-the-art vaccine research on three economically relevant aquaculture commodities chosen because of their distinct biological traits and geographical distribution as well as for being representative of different culture systems: European sea bass (Dicentrarchus labrax), Nile tilapia (Oreochromis niloticus), and Atlantic salmon (Salmo salar). From here on, the term “vaccine” is used to describe any substance used to stimulate the immune response or protect fish from pathogens, regardless of their classification (i.e., bacterial, viral, and parasitic). A compilation of mainly experimental formulations against bacterial, viral and parasitic infections is presented for each species (Tables 1–3; Figures 1–3). Commercial vaccines were considered only in particular cases (e.g., when a commercial product was adjuvanted with a recombinant molecule, when the study was of particular interest because of its large scale or analytical methods, or when commercial and experimental vaccines were compared). Because it is quite difficult to determine the exact variables affecting vaccine efficacy [19], multiple factors such as (i) antigen dose, exposure and uptake, (ii) boost immunization strategy, (iii) adjuvant inclusion, type and performance, (iv) water temperature, (v) fish size, (vi) type, virulence, and route of experimental challenge need to be considered prior to being able to extrapolate fundamental scientific observations. For this reason, we herein provide readers with the essential procedural elements and findings from the available literature with the aim of delivering the most comprehensive understanding on the features and performances of protective vaccines and immunostimulants/adjuvants and, ultimately, on the fish immune response, a crucial end-point for further science-based vaccine developments.
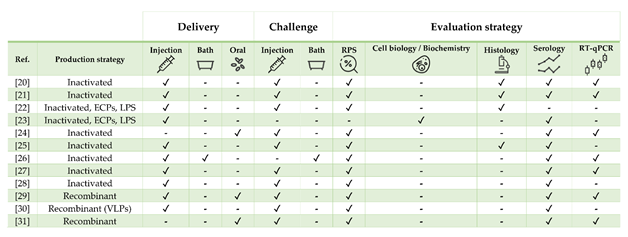
Table 1. Literature regarding experimental and commercial vaccines presented and discussed for European sea bass Dicentrarchus labrax. Approximate size refers to the fish body weight (BW) at the time of challenge or relative percentage of survival (RPS) calculation, as stated in or inferred from references. In case of commercial vaccines, the product description was linked. Challenges must be intended as homologous except when stated otherwise. List of abbreviations: ECPs—extracellular products; LPS—lipopolysaccharide; rTNFα—recombinant tumor necrosis factor alpha.
|
Pathogen |
Vaccine Status |
Adjuvant |
Approx. Size (g) |
Challenge |
Ref. |
|
Mycobacterium marinum |
Experimental |
50 |
Yes |
[20] |
|
|
M. marinum |
Experimental |
No |
20 |
Yes |
[21] |
|
Tenacibaculum maritimum |
Experimental |
No |
30 |
Yes |
[22] |
|
T. maritimum |
Experimental |
No |
5 |
No |
[23] |
|
Vibrio anguillarum + Vibrio ordalii |
Commercial (AquaVac Vibrio Oral) |
rTNFα |
30 |
Yes |
[24] |
|
V. anguillarum + Photobacterium damselae |
Commercial (AlphaJect 2000™ and AquaVac™ Vibrio-Pasteurella) |
Non-mineral |
35 |
Yes |
[25] |
|
Betanodavirus |
Experimental |
No |
2 and 6 |
Yes (only one exp. group) |
[26] |
|
Betanodavirus |
Experimental |
No |
11 |
Yes |
[27] |
|
Betanodavirus |
Experimental |
No |
6 |
Yes |
[28] |
|
Betanodavirus |
Experimental |
No |
11 |
Yes |
[29] |
|
Betanodavirus |
Experimental |
No |
30 |
Yes |
[30] |
|
Betanodavirus |
Experimental |
No |
6 |
Yes |
[31] |
Figure 1. Strategies for vaccine development, administration, and evaluation applied by referenced studies on European sea bass Dicentrarchus labrax.
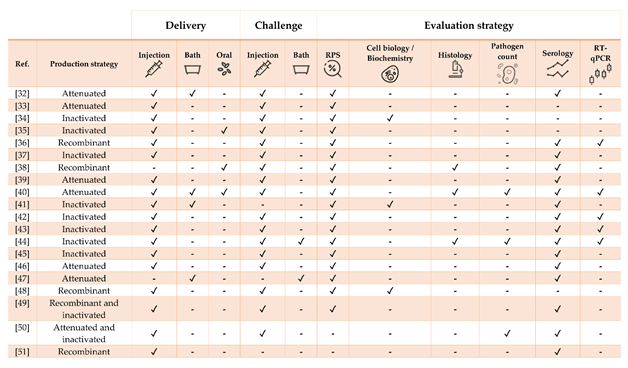
Table 2. Literature regarding experimental and commercial vaccines herein presented and discussed for Nile tilapia Oreochromis niloticus. Approximate size refers to the fish BW at the time of challenge or RPS calculation, as stated in or inferred from references. In case of commercial vaccines, the product description was linked. Challenges must be intended as homologous except when stated otherwise.
|
Pathogen |
Vaccine Status |
Adjuvant |
Approx. Size (g) |
Challenge |
Ref. |
|
Streptococcus iniae |
Experimental |
No |
10 |
Yes (homologous and heterologous) |
[32] |
|
S. iniae |
Experimental |
No |
40 |
Yes |
[33] |
|
S. iniae |
Experimental |
No |
5 |
Yes (heterologous) |
[34] |
|
S. iniae |
Experimental |
Oralject™ |
13 |
Yes |
[35] |
|
S. iniae |
Experimental |
No |
25 |
Yes |
[36] |
|
S. iniae |
Experimental |
No |
3 and 16 |
Yes |
[37] |
|
Streptococcus agalactiae |
Experimental |
No |
100 |
Yes |
[38] |
|
S. agalactiae |
Experimental |
No |
30 |
Yes (heterologous) |
[39] |
|
S. agalactiae |
Experimental |
No |
30 |
Yes |
[40] |
|
Polyvalent (S. agalactiae, S. iniae, Lactococcus garvieae and Enterococcus faecalis) |
Commercial (Mevac Aquastrept) |
500 and 1-month-old fry |
Yes |
[41] |
|
|
Francisella orientalis |
Experimental |
10 |
Yes |
[42] |
|
|
F. orientalis |
Experimental |
15 |
Yes (heterologous) |
[43] |
|
|
F. orientalis |
Experimental |
35 |
Yes |
[44] |
|
|
Aeromonas hydrophila |
Experimental |
No |
55 |
Yes |
[45] |
|
A. Hydrophila |
Experimental |
No |
10 |
Yes |
[46] |
|
Flavobacterium columnare |
Experimental |
No |
9 |
Yes (heterologous) |
[47] |
|
Vibrio anguillarum |
Experimental |
No |
3.5 |
Yes |
[48] |
|
Edwardsiella tarda |
Experimental |
102 |
Yes |
[49] |
|
|
E. tarda |
Experimental |
No |
42 |
Yes |
[50] |
|
Caligus rogercresseyi |
Experimental |
80 |
No |
[51] |
Figure 2. Strategies for vaccine development, administration, and evaluation applied by referenced studies on Nile tilapia Oreochromis niloticus.
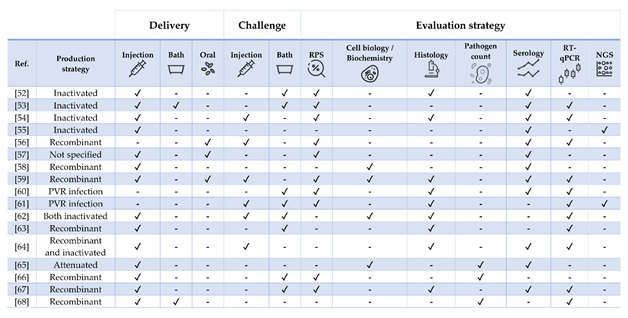
Table 3. Literature regarding experimental and commercial vaccines presented and discussed for Atlantic salmon Salmo salar. Approximate size refers to the fish BW at the time of challenge or RPS calculation, as stated in or inferred from references. In case of commercial vaccines, the product description was linked. Challenges must be intended as homologous except when stated otherwise. List of abbreviations: IFN—interferon; ISAV—infectious salmon anemia virus; IPNV—Infectious pancreatic necrosis virus; IHNV—infectious hematopoietic necrosis virus; SAV—salmonid alphavirus; PRV—piscine orthoreovirus; FCA—Freund’s complete adjuvant; FIA—Freund’s incomplete adjuvant.
|
Pathogen |
Vaccine Status |
Adjuvant |
Approx. Size (g) |
Challenge |
Ref. |
|
Tenacibaculum finnmarkense |
Experimental |
Mineral oil |
26 |
Yes (homologous and heterologous) |
[52] |
|
Yersinia ruckeri |
Experimental |
No |
9 |
Yes |
[53] |
|
Flavobacterium psychrophilum |
Experimental |
Squalene/alum or MontanideTM ISA 760 VG |
23 |
Yes |
[54] |
|
Polyvalent |
Commercial (Aquavac® PD7) |
Paraffin |
40 |
No |
[55] |
|
ISAV |
Experimental |
No |
40 |
Yes |
[56] |
|
ISAV and Piscirickettsia salmonis |
Commercial (Virbac-Centrovet) |
Oil |
40 |
No |
[57] |
|
ISAV |
Experimental |
IFNa- or IFNc |
40 |
No |
[58] |
|
IPNV |
Experimental |
No |
0.5 and 20 |
Yes |
[59] |
|
IHNV |
NA |
No |
5 g |
Yes (heterologous) |
[60] |
|
SAV |
NA |
No |
Post-smolt |
Yes (heterologous) |
[61] |
|
PRV |
Experimental and commercial (ALPHA JECT micro® 6) |
Paraffin |
55 |
Yes |
[62] |
|
PRV |
Experimental |
No |
35 |
Yes |
[63] |
|
SAV |
Experimental and commercial (Norvax® Compact PD) |
Montanide ISA 763A VG (only in the latter) |
30 |
Yes |
[64] |
|
Cryptobia salmositica |
Experimental |
No |
300 |
No |
[65] |
|
Caligus rogercressey |
Experimental |
80 |
Yes |
[66] |
|
|
Neoparamoeba perurans |
Experimental |
FCA (first immunization) and FIA (booster) |
100 |
Yes (two, 5-week apart) |
[67] |
|
Lepeophtheirus salmonis |
Experimental |
90 |
Yes |
[68] |
Figure 3. Strategies for vaccine development, administration, and evaluation applied by referenced studies on Atlantic salmon Salmo salar. For readability purposes, the bath and NGS columns also include cohabitation challenges and microarray experiments, respectively.
This entry is adapted from the peer-reviewed paper 10.3390/vaccines9020140
References
- FAO. The State of World Fisheries and Aquaculture 2020; FAO: Roma, Italy, 2020; ISBN 978-92-5-132692-3.
- Jackson, J.B.C.; Kirby, M.X.; Berger, W.H.; Bjorndal, K.A.; Botsford, L.W.; Bourque, B.J.; Bradbury, R.H.; Cooke, R.; Erland-son, J.; Estes, J.A.; et al. Historical overfishing and the recent collapse of coastal ecosystems. Science 2001, 293, 629–637, doi:10.1126/science.1059199.
- Daskalov, G.M.; Grishin, A.N.; Rodionov, S.; Mihneva, V. Trophic cascades triggered by overfishing reveal possible mecha-nisms of ecosystem regime shifts. Proc. Natl. Acad. Sci. USA 2007, 104, 10518–10523, doi:10.1073/pnas.0701100104.
- United Nations. World Population Prospects; United Nations: New York, NY, USA, 2019.
- Kobayashi, M.; Msangi, S.; Batka, M.; Vannuccini, S.; Dey, M.M.; Anderson, J.L. Fish to 2030: The Role and Opportunity for Aquaculture. Aquac. Econ. Manag. 2015, 19, 282–300, doi:10.1080/13657305.2015.994240.
- Leung, T.L.F.; Bates, A.E. More rapid and severe disease outbreaks for aquaculture at the tropics: Implications for food secu-rity. J. Appl. Ecol. 2013, 50, 215–222, doi:10.1111/1365-2644.12017.
- Raja, R.A.; Jithendran, K.P. Aquaculture disease diagnosis and health management. Adv. Mar. Brac. Aquac. 2015, February, 247–254, doi:10.1007/978-81-322-2271-2_23.
- Sharma, M.; Shrivastav, A.B.; Sahni, Y.P.; Pandey, G. Overviews of the Treatment and Control of Common Fish Diseases. Int. Res. J. Pharm. 2012, 3, 123–127.
- Lulijwa, R.; Rupia, E.J.; Alfaro, A.C. Antibiotic use in aquaculture, policies and regulation, health and environmental risks: A review of the top 15 major producers. Rev. Aquac. 2019, 12, 640–663, doi:10.1111/raq.12344.
- Jia, B.; St-Hilaire, S.; Singh, K.; Gardner, I.A. Biosecurity knowledge, attitudes and practices of farmers culturing yellow cat-fish (Pelteobagrus fulvidraco) in Guangdong and Zhejiang provinces, China. Aquaculture 2017, 471, 146–156, doi:10.1016/j.aquaculture.2017.01.016.
- Sharrer, M.J.; Summerfelt, S.T. Ozonation followed by ultraviolet irradiation provides effective bacteria inactivation in a freshwater recirculating system. Aquac. Eng. 2007, 37, 180–191, doi:10.1016/j.aquaeng.2007.05.001.
- Irhayyim, T.; Beliczky, G.; Havasi, M.; Bercsényi, M. Impacts of magnetic water treatment on water quality, feeding effi-ciency and growth performance of common carp in integrated recirculating aquaculture systems. J. Cent. Eur. Agric. 2020, 21, 246–255, doi:10.5513/JCEA01/21.2.2672.
- Amenyogbe, E.; Chen, G.; Wang, Z.; Huang, J.S.; Huang, B.; Li, H. The exploitation of probiotics, prebiotics and synbiotics in aquaculture: Present study, limitations and future directions: A review. Aquac. Int. 2020, 28, 1017–1041.
- Gudding, R.; Van Muiswinkel, W.B. A history of fish vaccination: Science-based disease prevention in aquaculture. Fish Shellfish Immunol. 2013, 35, 1683–1688, doi:10.1016/j.fsi.2013.09.031.
- Sommerset, I.; Krossøy, B.; Biering, E.; Frost, P. Vaccines for fish in aquaculture. Expert Rev. Vaccines 2005, 4, 89–101, doi:10.1586/14760584.4.1.89.
- Miccoli, A.; Saraceni, P.R.; Scapigliati, G. Vaccines and immune protection of principal Mediterranean marine fish species. Fish Shellfish Immunol. 2019, 94, 800–809, doi:10.1016/j.fsi.2019.09.065.
- Ma, J.; Bruce, T.J.; Jones, E.M.; Cain, K.D. A Review of Fish Vaccine Development Strategies: Conventional Methods and Modern Biotechnological Approaches. Microorganisms 2019, 7, 569, doi:10.3390/microorganisms7110569.
- Brudeseth, B.E.; Wiulsrød, R.; Fredriksen, B.N.; Lindmo, K.; Løkling, K.E.; Bordevik, M.; Steine, N.; Klevan, A.; Gravningen, K. Status and future perspectives of vaccines for industrialised fin-fish farming. Fish Shellfish Immunol. 2013, 35, 1759–1768, doi:10.1016/j.fsi.2013.05.029.
- Bøgwald, J.; Dalmo, R.A. Review on immersion vaccines for fish: An update 2019. Microorganisms 2019, 7, 1–28, doi:10.3390/microorganisms7120627.
- Ravid-Peretz, S.; Colorni, A.; Sharon, G.; Ucko, M. Vaccination of European sea bass Dicentrarchus labrax with avirulent My-cobacterium marinum (iipA::kan mutant). Fish Shellfish Immunol. 2019, 90, 317–327, doi:10.1016/j.fsi.2019.04.057.
- Ziklo, N.; Colorni, A.; Gao, L.Y.; Du, S.J.; Ucko, M. Humoral and Cellular Immune Response of European Seabass Dicentrar-chus labrax Vaccinated with Heat-Killed Mycobacterium marinum (iipA::kan Mutant). J. Aquat. Anim. Health 2018, 30, 312–324, doi:10.1002/aah.10042.
- Khalil, R.H.; Diab, A.M.; Shakweer, M.S.; Ghetas, H.A.; Khallaf, M.M.; Omar, A.A.E.D. New perspective to control of tenaci-baculosis in sea bass Dicentrarchus labrax L. Aquac. Res. 2018, 49, 2357–2365, doi:10.1111/are.13689.
- Salati, F.; Cubadda, C.; Viale, I.; Kusuda, R. Immune response of sea bass Dicentrarchus labrax to Tenacibaculum maritimum antigens. Fish. Sci. 2005, 71, 563–567, doi:10.1111/j.1444-2906.2005.01000.x.
- Galindo-Villegas, J.; Mulero, I.; García-Alcazar, A.; Muñoz, I.; Peñalver-Mellado, M.; Streitenberger, S.; Scapigliati, G.; Me-seguer, J.; Mulero, V. Recombinant TNFα as oral vaccine adjuvant protects European sea bass against vibriosis: Insights into the role of the CCL25/CCR9 axis. Fish Shellfish Immunol. 2013, 35, 1260–1271, doi:10.1016/j.fsi.2013.07.046.
- Spinos, E.; Kokkoris, G.D.; Bakopoulos, V. Prevention of sea bass (Dicentrarchus labrax, L. 1758) photobacteriosis and vibri-osis. Long term efficacy study of intraperitoneally administered bivalent commercial vaccines. Aquaculture 2017, 471, 172–184, doi:10.1016/j.aquaculture.2017.01.017.
- Nuñez-Ortiz, N.; Pascoli, F.; Picchietti, S.; Buonocore, F.; Bernini, C.; Toson, M.; Scapigliati, G.; Toffan, A. A forma-lin-inactivated immunogen against viral encephalopathy and retinopathy (VER) disease in European sea bass (Dicentrarchus labrax): Immunological and protection effects. Vet. Res. 2016, 47, 1–11, doi:10.1186/s13567-016-0376-3.
- Valero, Y.; Mokrani, D.; Chaves-Pozo, E.; Arizcun, M.; Oumouna, M.; Meseguer, J.; Esteban, M.Á.; Cuesta, A. Vaccination with UV-inactivated nodavirus partly protects European sea bass against infection, while inducing few changes in immun-ity. Dev. Comp. Immunol. 2018, 86, 171–179, doi:10.1016/j.dci.2018.05.013.
- Pascoli, F.; Guazzo, A.; Buratin, A.; Toson, M.; Buonocore, F.; Scapigliati, G.; Toffan, A. Lack of in vivo cross-protection of two different betanodavirus species RGNNV and SJNNV in European sea bass Dicentrachus labrax. Fish Shellfish Immunol. 2019, 85, 85–89, doi:10.1016/j.fsi.2017.10.033.
- Gonzalez-Silvera, D.; Guardiola, F.A.; Espinosa, C.; Chaves-Pozo, E.; Esteban, M.Á.; Cuesta, A. Recombinant nodavirus vac-cine produced in bacteria and administered without purification elicits humoral immunity and protects European sea bass against infection. Fish Shellfish Immunol. 2019, 88, 458–463, doi:10.1016/j.fsi.2019.03.013.
- Marsian, J.; Hurdiss, D.L.; Ranson, N.A.; Ritala, A.; Paley, R.; Cano, I.; Lomonossoff, G.P. Plant-made nervous necrosis vi-rus-like particles protect fish against disease. Front. Plant Sci. 2019, 10, 1–11, doi:10.3389/fpls.2019.00880.
- Valero, Y.; Awad, E.; Buonocore, F.; Arizcun, M.; Esteban, M.Á.; Meseguer, J.; Chaves-Pozo, E.; Cuesta, A. An oral chitosan DNA vaccine against nodavirus improves transcription of cell-mediated cytotoxicity and interferon genes in the European sea bass juveniles gut and survival upon infection. Dev. Comp. Immunol. 2016, 65, 64–72, doi:10.1016/j.dci.2016.06.021.
- Pridgeon, J.W.; Klesius, P.H. Development and efficacy of a novobiocin-resistant Streptococcus iniae as a novel vaccine in Nile tilapia (Oreochromis niloticus). Vaccine 2011, 29, 5986–5993, doi:10.1016/j.vaccine.2011.06.036.
- Wang, J.; Zou, L.L.; Li, A.X. Construction of a Streptococcus iniae sortase A mutant and evaluation of its potential as an atten-uated modified live vaccine in Nile tilapia (Oreochromis niloticus). Fish Shellfish Immunol. 2014, 40, 392–398, doi:10.1016/j.fsi.2014.07.028.
- Shoemaker, C.A.; LaFrentz, B.R.; Klesius, P.H.; Evans, J.J. Protection against heterologous Streptococcus iniae isolates using a modified bacterin vaccine in Nile tilapia, Oreochromis niloticus (L.). J. Fish Dis. 2010, 33, 537–544, doi:10.1111/j.1365-2761.2010.01148.x.
- Shoemaker, C.A.; Vandenberg, G.W.; Désormeaux, A.; Klesius, P.H.; Evans, J.J. Efficacy of a Streptococcus iniae modified bac-terin delivered using OraljectTM technology in Nile tilapia (Oreochromis niloticus). Aquaculture 2006, 255, 151–156, doi:10.1016/j.aquaculture.2005.12.016.
- Kayansamruaj, P.; Dong, H.T.; Pirarat, N.; Nilubol, D.; Rodkhum, C. Efficacy of α-enolase-based DNA vaccine against path-ogenic Streptococcus iniae in Nile tilapia (Oreochromis niloticus). Aquaculture 2017, 468, 102–106, doi:10.1016/j.aquaculture.2016.10.001.
- Shelby, R.A.; Klesius, P.H.; Shoemaker, C.A.; Evans, J.J. Passive immunization of tilapia, Oreochromis niloticus (L.), with an-ti-Streptococcus iniae whole sera. J. Fish Dis. 2002, 25, 1–6, doi:10.1046/j.1365-2761.2002.00327.x.
- Huang, L.Y.; Wang, K.Y.; Xiao, D.; Chen, D.F.; Geng, Y.; Wang, J.; He, Y.; Wang, E.L.; Huang, J.L.; Xiao, G.Y. Safety and im-munogenicity of an oral DNA vaccine encoding Sip of Streptococcus agalactiae from Nile tilapia Oreochromis niloticus deliv-ered by live attenuated Salmonella typhimurium. Fish Shellfish Immunol. 2014, 38, 34–41, doi:10.1016/j.fsi.2014.02.017.
- Liu, L.; Lu, D.Q.; Xu, J.; Luo, H.L.; Li, A.X. Development of attenuated erythromycin-resistant Streptococcus agalactiae vaccine for tilapia (Oreochromis niloticus) culture. J. Fish Dis. 2019, 42, 693–701, doi:10.1111/jfd.12977.
- Li, L.P.; Wang, R.; Liang, W.W.; Huang, T.; Huang, Y.; Luo, F.G.; Lei, A.Y.; Chen, M.; Gan, X. Development of live attenuated Streptococcus agalactiae vaccine for tilapia via continuous passage in vitro. Fish Shellfish Immunol. 2015, 45, 955–963, doi:10.1016/j.fsi.2015.06.014.
- Abu-Elala, N.M.; Samir, A.; Wasfy, M.; Elsayed, M. Efficacy of Injectable and Immersion Polyvalent Vaccine against Strep-tococcal Infections in Broodstock and Offspring of Nile tilapia (Oreochromis niloticus). Fish Shellfish Immunol. 2019, 88, 293–300, doi:10.1016/j.fsi.2019.02.042.
- Ramírez-Paredes, J.G.; Mendoza-Roldan, M.A.; Lopez-Jimena, B.; Shahin, K.; Metselaar, M.; Thompson, K.D.; Penman, D.J.; Richards, R.H.; Adams, A. Whole cell inactivated autogenous vaccine effectively protects red Nile tilapia (Oreochromis nilot-icus) against francisellosis via intraperitoneal injection. J. Fish Dis. 2019, 42, 1191–1200, doi:10.1111/jfd.13041.
- Shahin, K.; Shinn, A.P.; Metselaar, M.; Ramirez-Paredes, J.G.; Monaghan, S.J.; Thompson, K.D.; Hoare, R.; Adams, A. Efficacy of an inactivated whole-cell injection vaccine for nile tilapia, Oreochromis niloticus (L), against multiple isolates of Francisella noatunensis subsp. orientalis from diverse geographical regions. Fish Shellfish Immunol. 2019, 89, 217–227, doi:10.1016/j.fsi.2019.03.071.
- Pulpipat, T.; Maekawa, S.; Wang, P.C.; Chen, S.C. Immune responses and protective efficacy of a formalin-killed Francisella noatunensis subsp. orientalis vaccine evaluated through intraperitoneal and immersion challenge methods in Oreochromis ni-loticus. Vaccines 2020, 8, doi:10.3390/vaccines8020163.
- Bactol, I.D.C.; Padilla, L.V.; Hilario, A.L. Immune response of tilapia (Oreochromis niloticus) after vaccination with auto-clavekilled, heat-killed, and formalin-killed whole cell Aeromonas hydrophila vaccines as possible serotype-independent vac-cines. Int. J. Agric. Biol. 2018, 20, 846–850, doi:10.17957/IJAB/15.0575.
- Pridgeon, J.W.; Klesius, P.H. Development and efficacy of novobiocin and rifampicin-resistant Aeromonas hydrophila as novel vaccines in channel catfish and Nile tilapia. Vaccine 2011, 29, 7896–7904, doi:10.1016/j.vaccine.2011.08.082.
- Mohammed, H.; Olivares-Fuster, O.; LaFrentz, S.; Arias, C.R. New attenuated vaccine against columnaris disease in fish: Choosing the right parental strain is critical for vaccine efficacy. Vaccine 2013, 31, 5276–5280, doi:10.1016/j.vaccine.2013.08.052.
- Kwon, H.C.; Kang, Y.J. Effects of a subunit vaccine (FlaA) and immunostimulant (CpG-ODN 1668) against Vibrio anguillarum in tilapia (Oreochromis niloticus). Aquaculture 2016, 454, 125–129, doi:10.1016/j.aquaculture.2015.12.005.
- Cao, T.T.; Tsai, M.A.; Yang, C. Da; Wang, P.C.; Kuo, T.Y.; Chen, H.C.G.; Chen, S.C. Vaccine efficacy of glyceralde-hyde-3-phosphate dehydrogenase (Gapdh) from Edwardsiella ictaluri against E. tarda in tilapia. J. Gen. Appl. Microbiol. 2015, 60, 241–250, doi:10.2323/jgam.60.241.
- Igarashi, A.; Iida, T. A vaccination trial using live cells of Edwardsiella tarda in tilapia. Fish Pathol. 2002, 37, 145–148, doi:10.3147/jsfp.37.145.
- Yield improvement of the sea lice MY32/Cr novel antigen production and IgM immune response characterization in Oreo-chromis niloticus as a model. Available online: http://scielo.sld.cu/scielo.php?script=sci_arttext&pid=S1027-28522014000100004 (accessed on 4 Feburary 2021).
- Småge, S.B.; Frisch, K.; Vold, V.; Duesund, H.; Brevik, Ø.J.; Olsen, R.H.; Sjaatil, S.T.; Klevan, A.; Brudeseth, B.; Watanabe, K.; et al. Induction of tenacibaculosis in Atlantic salmon smolts using Tenacibaculum finnmarkense and the evaluation of a whole cell inactivated vaccine. Aquaculture 2018, 495, 858–864, doi:10.1016/j.aquaculture.2018.06.063.
- Nguyen, T.D.; Crosbie, P.B.B.; Nowak, B.F.; Bridle, A.R. The effects of inactivation methods of Yersinia ruckeri on the efficacy of single dip vaccination in Atlantic salmon (Salmo salar). J. Fish Dis. 2018, 41, 1173–1176, doi:10.1111/jfd.12803.
- Hoare, R.; Jung, S.J.; Ngo, T.P.H.; Bartie, K.; Bailey, J.; Thompson, K.D.; Adams, A. Efficacy and safety of a non-mineral oil adjuvanted injectable vaccine for the protection of Atlantic salmon (Salmo salar L.) against Flavobacterium psychrophilum. Fish Shellfish Immunol. 2019, 85, 44–51, doi:10.1016/j.fsi.2017.10.005.
- Lund, H.; Bakke, A.F.; Sommerset, I.; Afanasyev, S.; Schriwer, G.; Thorisdottir, A.; Boysen, P.; Krasnov, A. A time-course study of gene expression and antibody repertoire at early time post vaccination of Atlantic salmon. Mol. Immunol. 2019, 106, 99–107, doi:10.1016/j.molimm.2018.12.018.
- Caruffo, M.; Maturana, C.; Kambalapally, S.; Larenas, J.; Tobar, J.A. Protective oral vaccination against infectious salmon anaemia virus in Salmo salar. Fish Shellfish Immunol. 2016, 54, 54–59, doi:10.1016/j.fsi.2016.03.009.
- Tobar, I.; Arancibia, S.; Torres, C.; Vera, V.; Soto, P.; Carrasco, C.; Alvarado, M.; Neira, E.; Arcos, S.; Tobar, J.A. Successive oral immunizations against Piscirickettsia salmonis and infectious salmon anemia virus are required to maintain a long-term protection in farmed salmonids. Front. Immunol. 2015, 6, 1–7, doi:10.3389/fimmu.2015.00244.
- Robertsen, B.; Chang, C.J.; Bratland, L. IFN-adjuvanted DNA vaccine against infectious salmon anemia virus: Antibody ki-netics and longevity of IFN expression. Fish Shellfish Immunol. 2016, 54, 328–332, doi:10.1016/j.fsi.2016.04.027.
- Reyes, M.; Ramírez, C.; Ñancucheo, I.; Villegas, R.; Schaffeld, G.; Kriman, L.; Gonzalez, J.; Oyarzun, P. A novel “in-feed” delivery platform applied for oral DNA vaccination against IPNV enables high protection in Atlantic salmon (Salmo salar). Vaccine 2017, 35, 626–632, doi:10.1016/j.vaccine.2016.12.013.
- Vendramin, N.; Alencar, A.L.F.; Iburg, T.M.; Dahle, M.K.; Wessel, Ø.; Olsen, A.B.; Rimstad, E.; Olesen, N.J. Piscine orthoreo-virus infection in atlantic salmon (Salmo salar) protects against subsequent challenge with infectious hematopoietic necrosis virus (ihnv). Vet. Res. 2018, 49, 1–12, doi:10.1186/s13567-018-0524-z.
- Lund, M.; Røsæg, M.V.; Krasnov, A.; Timmerhaus, G.; Nyman, I.B.; Aspehaug, V.; Rimstad, E.; Dahle, M.K. Experimental Piscine orthoreovirus infection mediates protection against pancreas disease in Atlantic salmon (Salmo salar). Vet. Res. 2016, doi:10.1186/s13567-016-0389-y.
- Wessel, Ø.; Haugland, Ø.; Rode, M.; Fredriksen, B.N.; Dahle, M.K.; Rimstad, E. Inactivated Piscine orthoreovirus vaccine protects against heart and skeletal muscle inflammation in Atlantic salmon. J. Fish Dis. 2018, 41, 1411–1419, doi:10.1111/jfd.12835.
- Haatveit, H.M.; Hodneland, K.; Braaen, S.; Hansen, E.F.; Nyman, I.B.; Dahle, M.K.; Frost, P.; Rimstad, E. DNA vaccine ex-pressing the non-structural proteins of Piscine orthoreovirus delay the kinetics of PRV infection and induces moderate pro-tection against heart -and skeletal muscle inflammation in Atlantic salmon (Salmo salar). Vaccine 2018, 36, 7599–7608, doi:10.1016/j.vaccine.2018.10.094.
- Chang, C.J.; Gu, J.; Robertsen, B. Protective effect and antibody response of DNA vaccine against salmonid alphavirus 3 (SAV3) in Atlantic salmon. J. Fish Dis. 2017, 40, 1775–1781, doi:10.1111/jfd.12644.
- Chin, A.; Woo, P.T.K. Innate cell-mediated immune response and peripheral leukocyte populations in Atlantic salmon, Salmo salar L., to a live Cryptobia salmositica vaccine. Parasitol. Res. 2005, 95, 299–304, doi:10.1007/s00436-004-1270-x.
- Carpio, Y.; Basabe, L.; Acosta, J.; Rodríguez, A.; Mendoza, A.; Lisperger, A.; Zamorano, E.; González, M.; Rivas, M.; Contreras, S.; et al. Novel gene isolated from Caligus rogercresseyi: A promising target for vaccine development against sea lice. Vaccine 2011, 29, 2810–2820, doi:10.1016/j.vaccine.2011.01.109.
- Valdenegro-Vega, V.A.; Cook, M.; Crosbie, P.; Bridle, A.R.; Nowak, B.F. Vaccination with recombinant protein (r22C03), a putative attachment factor of Neoparamoeba perurans, against AGD in Atlantic salmon (Salmo salar) and implications of a co-infection with Yersinia ruckeri. Fish Shellfish Immunol. 2015, 44, 592–602, doi:10.1016/j.fsi.2015.03.016.
- Swain, J.K.; Carpio, Y.; Johansen, L.-H.; Velazquez, J.; Hernandez, L.; Leal, Y.; Kumar, A.; Estrada, M.P. Impact of a candidate vaccine on the dynamics of salmon lice (Lepeophtheirus salmonis) infestation and immune response in Atlantic salmon (Salmo salar L.). PLoS ONE 2020, 15, e0239827, doi:10.1371/journal.pone.0239827.