Female infertility is mainly caused by ovulation disorders, which affect female reproduction and pregnancy worldwide, with polycystic ovary syndrome (PCOS) being the most prevalent of these. PCOS is a frequent endocrine disease that is associated with abnormal function of the female sex hormone estrogen and estrogen receptors (ERs). Estrogens mediate genomic effects through ERα and ERβ in target tissues. The G-protein-coupled estrogen receptor (GPER) has recently been described as mediating the non-genomic signaling of estrogen. Changes in estrogen receptor signaling pathways affect cellular activities, such as ovulation; cell cycle phase; and cell proliferation, migration, and invasion. Over the years, some selective estrogen receptor modulators (SERMs) have made substantial strides in clinical applications for subfertility with PCOS, such as tamoxifen and clomiphene, however the role of ER in PCOS still needs to be understood.
- estrogen
- estrogen receptor
- ovary
- polycystic ovary syndrome
1. Introduction
Polycystic ovary syndrome (PCOS) is a type of general disease in women that is associated with a variety of reproductive and metabolic disorders [1]. The symptom is characterized by arrested folliculogenesis, hyperandrogenism, and polycystic ovaries [2]. Women suffering from PCOS account for a large proportion of the world population, with the prevalence estimated at between 5% and 10% [3]. Since the National Institutes of Health established a more standardized diagnosis standard in 1990, and with the continuous revision of this standard, the prevalence of PCOS is now expected to be close to 18% in the United States [4]. People with PCOS have more adverse reproductive risks, such as increased incidence of implantation failure, recurrent abortion, spontaneous abortion, premature birth, and endometrial carcinoma [5,6]. For example, women with PCOS were shown to have a 2.7-fold increased risk of endometrial cancer [7]. In addition to reproductive abnormalities, PCOS is also closely related to large quantities of metabolic disorders, for instance increased risk of hyperlipidemia, diabetes mellitus type II (T2DM), and hypertension, as well as hepatic steatosis, glucose intolerance, and insulin resistance [8,9].
Normal ovulation is the result of synergy between follicle-stimulating hormone (FSH) and luteinizing hormone (LH). LH stimulates the theca cells of the ovarian follicle, leading to androgen synthesis. Some of these are bound to sex-hormone-binding globulin (SHBG), and some androgens spread to nearby granulosa cells (GCs), where they are converted to estrogen under the stimulation of FSH. This causes an increase in the level of estrogen hormones, which in turn generates positive feedback in the form of LH production, causing a surge in LH and triggering ovulation [10]. The proliferation and differentiation of theca cells and granulosa cells are regulated by locally produced growth factors. Therefore, ovarian follicular development is tightly regulated by many hormones and other growth factors [11]. After ovulation, the corpus luteum is formed, which secretes the steroid hormones progesterone and estrogen, and then makes the endometrium more receptive to implantation [12].
Research on women with PCOS indicates elevated production of LH and free testosterone. Under the influence of endocrine-disrupting chemicals (phytoestrogens or synthetic estrogenic compounds), hormone secretion is also further affected, resulting in the PCOS disease [13]. As its name implies, the disease is a state of chronic anovulation that involves many ovarian cysts, characterized by an increased number of immature cystic follicles [14]. The ovaries are the primary site of estrogen synthesis, mainly producing 17β-estradiol (E2), which exerts effects on target organs and cells via multiple estrogen receptors (ERs), ERα, ERβ, and the G-protein-coupled estrogen receptor (GPER, also known as GPR30) to maintain various stages of normal development in the human ovaries and uterus.
2. The Characteristics of Estrogen Receptors
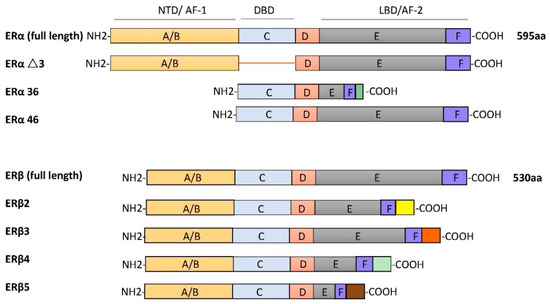
2.1. Estrogen Receptors: Expression, Structure
2.2. Estrogen Receptor Ligands
Natural endogenous estrogens are mostly produced in the ovaries, corpus luteum, and placenta, predominantly 17β-estradiol, which is the main ligand of ERs. Various natural and man-made chemicals also have estrogenic activity. Phytoestrogens are plant-derived compounds with structural similarity to 17β-oestradiol, including isoflavones, prenylflavonoids, coumestans, and lignans [33,34]. Synthetic estrogenic compounds (also known as environmental estrogens) include pesticides, dioxins, phthalates, bisphenol A, and diethylstilbestrol [35,36]. They are often widely dispersed in the environment and result in developmental and reproductive abnormalities in humans.
It is believed that most phytoestrogens and synthetic estrogenic compounds exert their physiological effects by regulating ERα and ERβ [37]. Many of these compounds can also activate GPER, including the soy isoflavone genistein, nonylphenol, and bisphenol A [38].
Other compounds are also widely used in clinical and therapeutic applications. For example, selective estrogen receptor modulators (SERMs) are synthetic non-steroidal drugs that act as both ER agonists and ER antagonists [39,40]. Common selective estrogen receptor modulators are tamoxifen, raloxifene, and clomiphene citrate (CC). By contrast, fulvestrant is a selective estrogen receptor downregulator (SERD) that results in ER degradation or downregulation and blocks the proliferation of breast cancer cells [41]. Apart from this, the non-steroidal ligand G-1 (1-[4-(6-bromobenzo[1,3] dioxol-5yl)-3a,4,5,9b-tetrahydro-3H-cyclopenta[c]quinolin-8-yl]-ethanone), which acts as a selective agonist for GPER, has been shown to induce the expression of genes by activation of GPER rather than the classical ERα or ERβ (as shown in Figure 2).
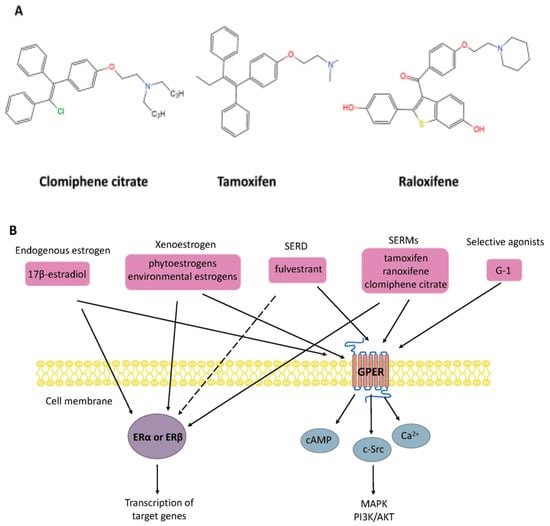
Figure 2. (A) Chemical structure of selective estrogen receptor modulators clomiphene citrate, tamoxifen, and raloxifene. (B) Nongenomic and genomic estrogen signal transduction pathways. Estrogen receptor ligands include 17β-estradiol and other compounds. Dashed lines indicate tissue-specific inhibition. SERD, selective estrogen receptor downregulator; SERM, selective estrogen receptor modulators; G-1, 1-[4-(6-bromobenzo[1,3] dioxol-5yl)-3a,4,5,9b-tetrahydro-3H-cyclopenta[c]quinolin-8-yl]-ethanone; ER, estrogen receptor; GPER, G-protein-coupled estrogen receptor; Ca2+, calcium ions; cAMP, cyclic adenosine monophosphate; MAPK, mitogen-activated protein kinase; PI3K/AKT, phosphatidylinositol 3-kinase/protein kinase B.
3. Functions of ERs in PCOS
3.1. ERs with Follicular Formation/Ovulation
3.2. ER Changes Associated with Endometrium
During normal menstrual cycles, the endometrium undergoes rapid cycling, proliferation, and growth in response to estrogen through the action of specific steroid receptors ERα and the ERβ [69]. During the follicular phase, E2 induces the rapid growth of the uterine endometrium and increases endometrial sensitivity to estrogen by increasing ERα levels. Conversely, at the luteal phase of the menstrual cycle, the corpus luteum continuously secretes progesterone and reduces estrogen to provide an appropriate uterine environment for maintaining the pregnancy. Thus, improper estrogen action affecting maximal uterine acceptance capability may change the normal expression of genes and reduce fertility in women with PCOS or increase the rate of spontaneous miscarriage.
ERα and the ERβ have different cellular localizations in the human endometrium. In general, ER (ERα and ERβ) expression reaches a maximum at the late proliferative stage and decreases at the secretory phase of the menstrual cycle [70,71]. For example, ERα mRNA is expressed in both the endometrial epithelial and stromal cells during the menstrual cycle, whereas ERβ mRNA is found predominantly in glandular epithelial cells [72]. In addition, the expression level of ERα mRNA in the uterus is more prominent than ERβ mRNA. GPER is localized in the plasma membrane and in the endoplasmic reticulum, which is considered to regulate the growth and proliferation of endometrial cells through its interaction with ERα [73,74,75].
There is growing clinical and experimental evidence showing that ER is an endometrial marker in patients with PCOS. Estrogen-induced uterine hyperplasia occurs by binding uterine epithelial ERα in adult mice to actively inhibit epithelial apoptosis in the uterus [76]. The same scenario is observed for ovulatory PCOS, where high levels of ERα expression are observed. In women with PCOS, ERα expression in the endometrium continues to enter the secretion stage, showing high expression in the stroma and luminal epithelium [77]. One woman with a homozygous ERα mutation and rats lacking ERα showed similar phenotypes and infertility similar to what has been observed in patients with PCOS [61,64].
In women with ovulatory dysfunction, the endometrium, as a target tissue for estrogen, is prone to hyperplasia and cancer [78,79,80]. PCOS symptoms are significantly related to endometrial cancer risk. Women under age 50 with PCOS have four times higher incidence of endometrial cancer than women without PCOS [81]. Based on previous research, increased ERα expression in the endometrium of PCOS women at both gene and protein expression levels has been observed, suggesting that the endometrium is more sensitive to estrogen, possibly explaining the significant increased incidence of hyperplasia and endometrial cancer, as well as the decreased ability to continue pregnancy [82,83]. Moreover, an ERβ polymorphism (+1730 G/A) was associated with the development of susceptibility to PCOS in humans [84]. Polymorphisms in identified ER-α were found to have an association with the development of endometrial cancer [85]. Furthermore, changes in ERα expression levels and the ERα/ERβ ratio in PCOS patients were higher than in the normal group, indicating outstanding ERα-mediated actions in the endometrium, which may be related to endometrial hyperplasia and endometrial cancer [86]. Previous studies have shown that p160 steroid receptor coactivator is increased in the secretory-phase endometria of women with PCOS, which further increases endometrial ERα expression to stimulate endometrial proliferation [86,87]. Abnormal estrogenic environments may alter endometrial receptivity in women, resulting in blastocyst implantation failure or a malformed abortion after implantation (the functions of ERs in PCOS are shown in Figure 3).
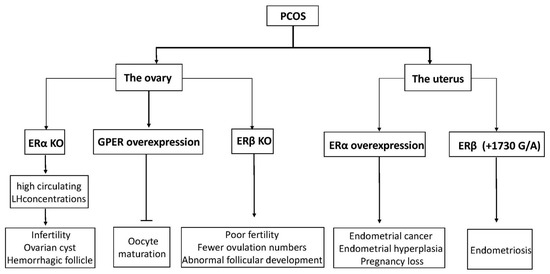
Figure 3. A diagram of physiological phenomena to explain disordered expression in the ER family. Arrows indicate direct effects, while no arrows indicate inhibition.
This entry is adapted from the peer-reviewed paper 10.3390/cells10020459
