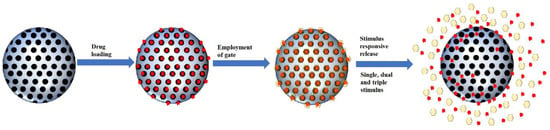
The development of MSNs as drug delivery agents evolved from passive targeting, active targeting and now stimulus responsive system. Based on our literature search, the general form of a stimulus-responsive system utilizes both passive targeting and active targeting simultaneously with an additional gate to trap the cargo. The weakness of both passive and active targeting is premature to release of cargo during circulation; therefore, a gate was employed to close the pores of MSNs. This gate will be open once it reaches the target sites through a specific stimulus (). The type of stimulus can be classified into two categories: internal and external stimulus. However, in this review, we will be categorizing the responsive stimulus system into the number of stimuli, which are single stimuli and multiple stimuli.
2.3.1. Single Stimulus
As was mention previously, there are two types of stimuli, which are internal, such as pH, redox and enzyme, and external stimuli, such as light and temperature. In this section, we will be covering the common stimulus for both types of stimuli with the focus on the single-stimulus-responsive-MSNs system. summarizes the research that utilized single-stimulus-responsive-MSNs for delivery of the anticancer drug. The gatekeeper and stimuli type are also indicated.
Table 2. Various stimulus-responsive-MSNs for controlled release.
|
Gatekeeper
|
Stimulus
|
Cargo/Drugs
|
Ref
|
|
Polyacrylic acid
|
pH
|
Umbelliferone
|
[150]
|
|
Poly-L-lysine
|
pH
|
Doxorubicin
|
[166]
|
|
Poly(tannic acid)
|
pH
|
Doxorubicin
|
[167]
|
|
Hyaluronic acid
|
pH
|
Doxorubicin
|
[157]
|
|
Polyethylene glycol and chitosan
|
pH
|
Doxorubicin
|
[168]
|
|
Zinc oxide
|
pH
|
Doxorubicin
|
[169]
|
|
Albumin
|
pH
|
Lamivudine
|
[170]
|
|
Ferritin
|
pH
|
Doxorubicin
|
[162]
|
|
Beta-cyclodextrin
|
pH
|
5-fluorouracil
|
[171]
|
|
Chitosan
|
Redox
|
Rhodamine 6G
|
[172]
|
|
Cucurbit[6]uril, cyclopentyl methylamine and polyacrylic acid
|
Redox
|
Doxorubicin
|
[173]
|
|
Gold
|
Redox
|
Doxorubicin
|
[90]
|
|
Zinc sulphide
|
Redox
|
Doxorubicin
|
[174]
|
|
Ferrocene-containing amphiphilic block copolymer
|
Redox
|
Doxorubicin
|
[175]
|
|
Organosilica
|
Redox
|
Doxorubicin
|
[176]
|
|
Graphene
|
Redox
|
Rhodamine B
|
[177]
|
|
Bovine serum albumin
|
Redox
|
Epirubicin
|
[178]
|
|
Chitosan
|
Enzyme
|
Doxorubicin
|
[179]
|
|
Peptide
|
Enzyme
|
Organotin
|
[180]
|
|
Guar gum
|
Enzyme
|
5-fluorouracil
|
[181]
|
|
Hyaluronic acid
|
Enzyme
|
5-fluorouracil
|
[182]
|
|
Hyaluronic acid
|
Enzyme
|
Doxorubicin
|
[183]
|
|
Hyaluronic acid and collagen I
|
Enzyme
|
Doxorubicin
|
[184]
|
|
Iron oxide
|
Enzyme
|
Doxorubicin
|
[185]
|
|
Gold
|
Enzyme
|
Doxorubicin
|
[186]
|
|
Ruthenium complex
|
Light
|
Sarafanin O
|
[187]
|
|
α-cyclodextrin
|
Light
|
Doxorubicin
|
[188]
|
|
β-cyclodextrin
|
Light
|
Doxorubicin
|
[189]
|
|
β-cyclodextrin
|
Light
|
Camptothecin
|
[190]
|
|
β-cyclodextrin and -diazo-1.2-napthoquinones
|
Light
|
Doxorubicin
|
[191]
|
|
Human serum albumin
|
Light
|
Doxorubicin
|
[192]
|
|
Supported lipid bilayer
|
Temperature
|
Doxorubicin
|
[193]
|
|
Poly(N-isopropylacrylamine)-co-poly(methacrylic acid) (PNINAM-co-PMAA)
|
Temperature
|
Doxorubicin
|
[194]
|
|
Poly(N-isopropylacrylamide)-co-(1-butyl-3-vinyl imidazolium bromide) (p-NIBIm)
|
Temperature
|
Cytochrome C
|
[195]
|
|
PNINAM
|
Temperature
|
Methylene Blue
|
[196]
|
pH-responsive MSNs is one of the promising vehicles to deliver anticancer drugs to the targeted region. This system was developed through the exploitation of pH values in most tumors, which is lower compared to normal cells. The lower pH (around 6.5) value is due to the Warburg effect, where the cancer cells produce energy through glycolysis with or without oxygen, and this leads to the production of acidic lactate [
197]. Furthermore, after the internalization of the carrier to the cancer cells, it will be exposed to even more acidic media in endosomal (pH 5.5–6.0) and lysosomal (pH 4.5–5.0). Kundu and colleagues reported on the delivery of umbelliferone, a plant-derived natural product, using pH-sensitive MSNs. The pores of MSNs were capped with polyacrylic acid, pH-sensitive polymer, and the surface was grafted with folic acid as the targeting agent. The drug was released under acidic pH due to the less electrostatic interaction and dissociation of the amide bond between the MSNs and the polymer [
150]. In another work, poly-
l-lysine gated MSNs were used to deliver doxorubicin. In a neutral environment, poly-
l-lysine shrunk, which formed a dense barrier on the pore entrance. As the pH reduced, the polymer is swollen, and the drug was released [
166]. One work reported on the usage of zinc oxide quantum dots as the gate to trap doxorubicin in the MSNs pores. The drug released at lower pH as the zinc oxide quantum dots dissolve in acidic condition and this lead to drug released [
169]. Chen and coworkers successfully produced pH-responsive MSNs with “release-stop-release” properties. Typical pH-responsive MSNs will be activated in acidic conditions, and the process is not reversible. In this work, they managed to produce pH-responsive MSNs with reversible function. They utilized poly(tannic acid) as the gatekeeper with tetrethylenepentamine (TEPA) as a crosslinker. In acidic conditions, TEPA was protonated, thus increasing its hydrophilicity. This resulted in poly(tannic acid) swelling and substantial drug release. As the pH increased, the TEPA deprotonated and poly(tannic acid) will become dense again, thus closed the pores of MSNs. The authors stated that through this system, the side effect of the leftover drug could be minimized as safely excreted out [
167].
Utilizing redox potential is another pathway to develop a stimulus responsive drug delivery agent. In a cancerous cell, the production of reactive oxygen species (ROS) is elevated due to genetic mutation, mitochondrial dysfunction and different metabolism. To overcome this, the production of ROS scavenger, the prominent being tripeptide glutathione (GSH), is also increased. It should be noted that GSH also presents in normal cells, but in cancerous cells, it contributes to the cancer progression and responsible for the resistance increment of radio- and chemoresistance of cancer cells [
136]. It was found that the concentration of GSH is significantly higher in cancer cells (approximately 2–20 mM) compared to the extracellular part (2–20 nM) [
198]. By utilizing the difference in the GSH concentration, the release of drugs from redox responsive MSNs upon cancer cell entrance will occur. One work reported on the utilization of redox potential to produce a redox responsive system with chitosan as the gate to trap the drug in the MSNs pore. Interestingly, this work used a fluorescent agent as the anchoring molecule which binds the chitosan with MSNs. This fluorescent agent was reported to be sensitive to GSH. Once the carrier entered the cancer cell, this fluorescence agent will be cleaved, and chitosan will be released from the carrier. Therefore, the drug will be released to treat the cancerous cell [
172]. Thiol-linker is one of the most utilize linker to develop redox responsive MSNs. This linker will be broken by GSH, which will lead to the drug release. Zhang and coworkers reported on the redox responsive system with gold (Au) nanoparticles as the gate to the MSNs. The Au was bonded to the MSNs surface using a thiol-linker and exposure of this carrier to high GSH concentration’s environment, the linker was broken, and the drug, doxorubicin, was released [
90]. In another work, the redox responsive MSNs was developed through the layer-by-layer assembly of curcubit-6-uril and cyclopentyl methylamine polymer, which served as the gate to cover the MSNs pores. This work studied the effect of layers’ numbers on the drug release activity with redox stimulus. In the presence of GSH, the percentage of drug released from the carrier decreased with increased layer number. They concluded that the cargo release rate could be manipulated through the variation of polymer layers [
173].
Another stimulus-responsive system that is popular is the enzyme responsive MSNs carrier. Utilization of enzyme as stimulus poses several advantages such as high chemical selectivity, substrate specificity and mild condition. For cancer treatment, there is a specific enzyme present in this tumor microenvironment. Therefore, by employing a carrier with a component that sensitive to the present enzyme, a control released system through enzymatic cleavage is obtained [
199]. Cai et al. reported on the production of enzyme responsive MSNs with chitosan as the gate responsible for trapping the drug in the MSNs pores. The component that was sensitive to the enzyme is the azo bond linked to the MSNs and chitosan. The results showed that the azo bond was broken in the presence of colon enzyme secreted by colonic microflora [
179]. In another work, the same enzyme was used as a response to MSNs with guar gum as the pores’ cap. The colon enzyme degraded the guar gum, and the drug, 5-fluorouracil, was released [
181]. Functionalization of MSNs with hyaluronic acid was found to be an enzyme responsive carrier in addition to active targeting [
183]. Hyaluronic acid as a targeting agent can bind with CD-44 receptor, which is found in most cancer cells, and degraded by the enzyme hyaluronidase. One work used hyaluronic acid as both the gate and targeting agent [
182]. Zhou and colleagues reported on the production of MSNs conjugated with hyaluronic acid and collagen I matrix for enzyme responsive system. Interestingly, this carrier was reported to be responsive to two enzymes, hyaluronidase and metalloproteinase-2. Through drug released studies, it was found that in the absence of enzymes, no drug was detected, while in the presence of an enzyme, up to 75% of the drug was released [
184].
Other than internal stimulus, as stated previously, an external stimulus such as light and temperature were found to be commonly applied in the development of a responsive delivery system using MSNs. To generate light responsive MSNs, functionalization using a photoactive group is a need. The trigger for drug release will be light with various wavelengths such as ultraviolet (UV), visible (Vis) and near-infrared (NIR) [
133]. Wang and coworkers reported on UV responsive MSNs carrier for anticancer treatment. The pore of MSNs was capped with α-cyclodextrin with hydrazone bond linking the azobenzene functionalized MSNs and α-cyclodextrin. Through UV irradiation, the azobenzene conformation changed, which led to the dissociation of α-cyclodextrin and the drug, doxorubicin, released [
188]. Although this work showed promising results, UV light as a trigger is not practical as it poses an adverse effect on the cell and tissue and has low tissue penetrability [
200]. Visible light has also been reported as a trigger to release the cargo from MSNs. One work reported on the usage of the ruthenium complex as the gate to cap the MSNs’ pores. The result indicated that in the absence of light, the carrier was stable, and when Vis light was irradiated, the rapidly released cargo was observed. Interestingly, the gate, the ruthenium complex, can be controlled. Through heating at elevated temperatures, the pores re-closed, which can be re-opened upon irradiation [
187]. Visible light can overcome the problem posed by UV, but it still has poor tissue penetration, which makes it difficult to be applied in cancer treatment. The best light wavelength to achieve good tissue penetration lies between 650–900 nm, which is in the NIR range. Therefore, the majority of light-responsive MSNs developed with NIR trigger [
200]. One work reported on Janus gold nanostar-MSNs as NIR responsive carrier. The gold surface was functionalized with a thiolated photolabile molecule to bind itself to MSNs and β-cyclodextrin as the gate. Through NIR irradiation, the photolabile molecule dissociated and form succinic acid and this succinic acid induced the opening of the gate [
189]. In another work, upconversion nanoparticles (UCNP) were coated with MSNs layer, and the gate employed was β-cyclodextrin. The β-cyclodextrin was linked to the MSNs layer with photo-cleavable pyrenemethyl ester. The principle of this light-responsive system as follows: 1) NIR light was irradiated to the carrier. Due to the presence of UCNP, the NIR was converted to Vis and UV light. 2) The pyrenemethyl ester bond was broken with higher energy light, and the β-cyclodextrin was dissociated, which led to the drug released [
190].
Temperature is another stimulus being used to develop stimulus responsive MSNs. The important criteria in producing thermo-responsive MSNs are it should be stable during the circulation (37 °C) and the drug released at the locally heated tumor (~40–45 °C) [
201]. One work reported on the production of thermo-responsive MSNs by incorporating a supported lipid bilayer (SBL) on the MSNs surface. This SBL also served as the gate to close the MSNs pores and trap the drug. At elevated temperature, the lipid bilayer became flexible and exhibited a liquid state. This caused it to be more permeable, which led to the drug released [
193]. Besides that, the thermo-responsive polymer was used to coat the MSNs surface to develop thermo-responsive MSNs. Copolymer consisting of poly(N-isopropylacrylamine) and poly(methacrylic acid) (PNINAM-co-PMAA) was used as the coating of MSNs surface to trap the drug, doxorubicin. This copolymer served as both the gate and thermo-responsive component of the carrier. At 37 °C, which is lower than the low critical solution temperature (LCST), the copolymer swelled and blocked the MSNs pores. As the temperature increased (>LCST), the copolymer collapsed, and the drug was released [
194]. Overall, through our literature search, we discovered that the articles on MSNs carriers that depend solely on temperature stimulus are lacking. Most articles report on the production of dual or more stimulus-responsive MSNs, which consists of temperature and any other stimulus.
2.2.2. Multiple Stimulus
In recent years, we discovered that the production of stimulus-responsive-MSNs as a drug delivery agent is no longer limited to single stimuli. Most articles reported on the utilization of dual-responsive systems, and some even reported up to three responsive systems. The most common stimulus being used simultaneously is pH and redox stimulus. Lu and colleague reported on the usage of hollow MONs with disulfide bridge in the silica walls and supramolecular interaction between α-cyclodextrin and anilino alkane group as the gate to produce pH and redox responsive carrier. The results showed that at low pH (mimicking the tumor environment), the percentage of doxorubicin released was about 58%. This was reported due to the dissociation of α-cyclodextrin from protonated aniline. As state previously, the disulfide bond is susceptible to GSH, which present at a high concentration in cancerous cells. GSH will cleave the disulfide bond resulted in the breaking of hollow MONs walls, and the drug was released [
114]. Through our literature search, there were three articles reported on the utilization of chitosan and disulfide bond to create pH and redox responsive MSNs. Chitosan is a pH-responsive biopolymer. In acidic conditions, chitosan may disassemble and lead to the pore opening and drug being released. By using a disulfide bond to link the MSNs and chitosan, a redox responsive system was created. With a high concentration of GSH, the disulfide bond was broken, which lead to chitosan gate opening and release of the drug [
202,
203,
204]. Besides simultaneous use of pH and redox stimulus, there have been reports on pH and ROS stimulus being used. Interestingly, Song et al. reported that polydopamine served as both the pH and ROS responsive component of the carrier. At low pH, the amine group of polydopamine was protonated, which broke the interaction between the polymer and curcumin (drug). In the presence of hydrogen peroxide (ROS stimulus), the hydrogen bond between polydopamine and curcumin was broken, and the drug was released [
205]. Dual responsive MSNs that are sensitive towards pH and temperature has also been developed. MSNs consist of carbon dots and poly(N-vinylcaprolactam) was produced as a pH and temperature-responsive carrier. The carbon dots and poly(N-vinylcaprolactam) were functionalized to the MSNs surfaces using a Schiff base bond. This Schiff base was reported to be acid sensitive and broken at acid atmosphere, which led to drug released. The polymer, poly(N-vinylcaprolactam), which served as a temperature-responsive molecule were at an elevated temperature; the polymer underwent phase transition and collapsed where the drug was then released from the carrier [
206]. There have been other dual responsive systems reported and are summarized in
Table 2. Multiple responsive MSNs with their responsive linker/moiety.
|
Responsive linker/moiety
|
Stimulus
|
Ref
|
|
pH
|
Redox
|
Enzyme
|
ROS
|
Temperature
|
Light
|
Other compounds
|
|
|
Dual stimulus
|
|
α-cyclodextrin and anilinoalkane
|
Disulfide bond
|
|
|
|
|
|
[114]
|
|
Polydopamine
|
Disulfide bond
|
|
|
|
|
|
[209]
|
|
Chitosan
|
Disulfide bond
|
|
|
|
|
|
[210]
|
|
Citraconic
|
Disulfide bond
|
|
|
|
|
|
[211]
|
|
Chitosan
|
Disulfide bond
|
|
|
|
|
|
[202] [203] [204]
|
|
Benzoic imine bonds
|
Disulfide bond
|
|
|
|
|
|
[158]
|
|
Zinc oxide quantum dots
|
Disulfide bond
|
|
|
|
|
|
[212]
|
|
Bull serum albumin
|
Disulfide bond
|
|
|
|
|
|
[213]
|
|
Sodium alginate
|
Disulfide bond
|
|
|
|
|
|
[214]
|
|
Polydopamine
|
|
|
Polydopamine
|
|
|
|
[205]
|
|
Carboxymethyl chitin
|
|
|
Thioketal bond
|
|
|
|
[215]
|
|
Schiff base bonds
|
|
|
|
Poly(N-vinylcaprolactam)
|
|
|
[206]
|
|
PEG-like Jeffamine M-2005
|
|
|
|
Polyphosphazene
|
|
|
[216]
|
|
Poly(N-isopropylacrylamide-co-methacrylic acid)
|
|
|
|
Poly(N-isopropylacrylamide-co-methacrylic acid)
|
|
|
[217]
|
|
Polydopamine
|
|
|
|
|
Gold
|
|
[139]
|
|
Hydroxyapatite
|
|
|
|
|
Gold
|
|
[218]
|
|
Carboxylic acid
|
|
|
|
|
|
Chloride ions
(Salt)
|
[219]
|
|
|
Disulfide bond
|
Cystine-dopamine
|
|
|
|
|
[220]
|
|
|
Dithiodipropionic
|
|
Selenocystine
|
|
|
|
[221]
|
|
|
Disulfide bond
|
|
|
Poly(γ-benzyl-l-glutamate)
|
|
|
[222]
|
|
|
Disulfide bond
|
|
|
|
Azobenzene / Galactose-grafted polymer
|
|
[223]
|
|
|
|
|
Ferrocene
|
Poly(N-isopropylacrylamide)
|
|
|
[224]
|
|
Triple stimulus
|
|
Amide bond
|
Gold-sulphur bond
|
Hyaluronic acid
|
|
|
|
|
[207]
|
|
Schiff base bond
|
Disulfide bond
|
|
|
poly(N-isopropylacrylamide-block-poly(2-(4-formylbenzoyloxy) ethyl methacrylate)
|
|
|
[208]
|
|
Polydopamine
|
Disulfide bond
|
|
|
|
Polydopamine
|
|
[225]
|
|
Electrostatic interaction
|
Disulfide bond
|
|
|
|
Carbon dots
|
|
[226]
|
|
Ester bond
|
Disulfide bond
|
|
|
|
|
Molecular interaction
(Glucose)
|
[227]
|
Besides dual responsive systems, there have been reports on tiple responsive systems. pH and redox the common stimuli being used while the third stimulus varied. Chen and coworkers reported on triple responsive carriers, which were sensitive to pH, redox and enzyme stimuli. The MSNs were functionalized with gold via a sulfide bond and hyaluronic acid via an amide bond. The gold–sulfur bond, amide bond and hyaluronic acid were reported to be responsive to the redox, pH and enzyme stimulus, respectively [
207]. In other work, a triply responsive system was developed to be sensitive towards pH, redox and temperature. The carrier consisted of multiwalled carbon nanotubes covered with mesoporous silica graft poly(
N-isopropylacrylamide-
block-poly(2-(4-formylbenzoyloxy) ethyl methacrylate) via disulfide linkages. As stated previously, the disulfide link is receptive towards redox stimulus. The drug was bonded to the polymer via Schiff base bond, and this bond was broken in acidic condition, which led to doxorubicin released. At elevated temperatures, the polymer collapsed. Thus, the drug was released [
208]. Other articles related to triple responsive MSNs are summarized in