Overwhelming evidence suggests an association of cardiovascular disease (CVD) with non-alcoholic fatty liver disease (NAFLD); however, the underlying mechanisms remain largely speculative.
- diabetes
- metabolic syndrome
- lifestyle
- atherosclerosis
- NASH
- liver
- exercise
- nutrition
1. Introduction
During the last decade, strong evidence has demonstrated a significant interplay and multifaceted relationship between non-alcoholic fatty liver disease (NAFLD) and cardiovascular disease (CVD). Pathophysiological mechanisms associating NAFLD with CVD are incompletely understood, and the current literature on the role of NAFLD as an independent risk factor for CVD and excess CV mortality in NAFLD patients has yielded conflicting results and should therefore be interpreted with caution [1][2][3][4][5]. Nevertheless, NAFLD should be regarded as a systemic metabolic disease affecting extrahepatic tissues throughout the body via complex mechanisms, including diet-induced alterations of the gastrointestinal tract, adipose tissue inflammation and lipotoxicity.
2. Epidemiological Data Linking NAFLD to Cardiovascular Disease (CVD)
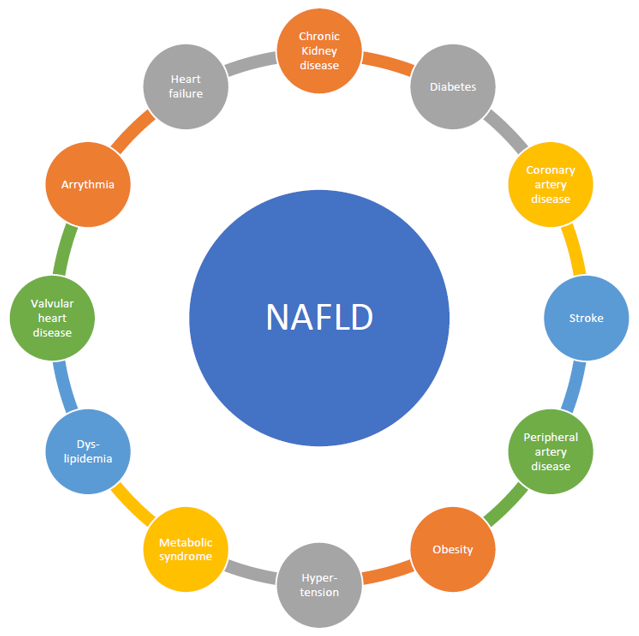
There are substantial epidemiological data that link NAFLD to CVD. We discuss vascular and metabolic disease but also other CV conditions (Figure 1).
Figure 1. Substantial epidemiological data that link non-alcoholic fatty liver disease (NAFLD) to cardiovascular disease (CVD).
2.1. Vascular Disease: Atherosclerosis including Stroke, Peripheral Artery Disease, and Coronary Artery Disease
Although the understanding, prevention, and therapy of non-communicable diseases (NCDs) have improved during recent decades, NCDs constitute the primary cause (around 70%) of death worldwide [6]. Among NCDs, cardiovascular disease (CVD) is responsible for about half of the observed mortality [6]. Besides mortality, CVD leads to high morbidity and costs in Western societies [7][8][9]. CVD can be considered a phenotype with multiple interlapping mechanisms, which include arterial hypertension, obesity, diabetes, socioeconomic factors, and behavioral factors such as physical inactivity and poor nutrition as well as genetic factors, age, and sex [10][11].
NAFLD and CVD share several risk factors. Like CVD, NAFLD is highly prevalent in the total population of developed countries with a prevalence between 20% and 30% [12]. However, in obese patients and patients with diabetes, these proportions increase to up to 90% [13]. Both NAFLD and CVD are more common in men than women, although NAFLD progresses faster in women [13]. More than half of the patients diagnosed with NAFLD suffer from hyperlipidemia, an established independent risk factor for cardiovascular disease [10][11][14]. Furthermore, low-grade systemic inflammation was reported in both NAFLD and CVD [15]. Some experts consider NAFLD as the hepatic manifestation of metabolic syndrome, insulin resistance, and type 2 diabetes mellitus (T2DM) [16].
As both NAFLD and CVD are diseases with high prevalence, sharing several risk factors, it comes as no surprise that epidemiological data supporting NAFLD and CVD coincidence are abundant [1]. Although traditionally considered a hepatic disease, a growing body of evidence suggests that cardiovascular disease in patients with NAFLD determines the outcome rather than liver disease progression [17][18][19].
Observational studies suggest a link between NAFLD diagnosis and an increased risk for CVD and cardiovascular events, including death [4][20][21]. Markers of endothelial dysfunction such as increased intima media thickness and lower flow-mediated dilatation are more common in NAFLD patients [15][22][23]. The risk of stroke was higher in subjects with NAFLD than the general population [15]. Elevated brachial ankle pulse wave velocity and increased arterial stiffness are other surrogate parameters for subclinical atherosclerosis described in NAFLD [24][25].
NAFLD patients evidence higher cardiovascular risk according to the Framingham risk score [26]. Several meta-analyses support the association of NAFLD with increased risk for CVD, although the link between NAFLD and CVD is less robust than between NAFLD and metabolic syndrome [3][16][27]. While some studies have found associations between NAFLD surrogates and CVD, many of these analyses are limited by insufficient adjustment for established cardiovascular risk factors [28][29]. The crucial question of whether NAFLD is a risk factor for CVD independent of metabolic syndrome is, therefore, not yet completely clear. However, since the inclusion of NAFLD in risk calculations for CVD has so far brought no added value compared to models that only consider classical metabolic risk factors, it is, based on the available evidence, currently more likely that NAFLD has no independent impact on cardiovascular outcome beyond their common metabolic background [30][31][32].
2.2. Metabolic Disease: Metabolic Syndrome, Dyslipidemia, Diabetes, and Obesity
NAFLD is closely embedded into metabolic syndrome (MetS), which describes the typical common occurrence of established cardiovascular risk factors such as overweight/obesity, dyslipidemia, and disordered glucose homeostasis, as indicated by insulin resistance (IR) or T2DM and arterial hypertension [33]. The prevalence of NAFLD increases with the number of manifestations of metabolic syndrome components [34]. Most NAFLD subjects are obese, and the prevalence in the general population increases with progressive degrees of obesity, approximating almost 95% in subjects undergoing metabolic surgery [34][35]. Likewise, it approaches 60–70% in subjects with T2DM [36]. However, increasing BMI as an indicator of excess adipose tissue appears not to be the sole determining or sufficient factor for NAFLD development as 10% of lean subjects also have NAFLD, and a small proportion of even morbidly obese subjects maintain a healthy liver [37][38][39]. The finding of NAFLD in the absence of excess body weight is commonly referred to as “lean NAFLD” [40]. Further studies have elucidated the central relevance of adipose tissue distribution to NAFLD development as particularly in lean subjects or subjects with a low degree of overweight, substantial visceral fat accumulation is present and directly linked to the presence of a fatty liver [41][42], A meta-analysis by Young et al. demonstrated that lean NAFLD versus healthy subjects or lean controls had higher odds for abnormalities on their metabolic profile, including metabolic syndrome and its components, glycemic dysregulation, renal and liver function, and patatin-like phospholipase domain-containing protein 3 (PNPLA3) G allele, and their inflammatory profile, including uric acid and C-reactive protein [43]. Moreover, Golabi et al. have already demonstrated that lean NAFLD subjects have a higher risk of cardiovascular mortality and overall mortality than normal subjects [44][44]. In our own experience, lean NAFLD is associated with metabolic syndrome and increased cardiovascular risk as assessed by the Framingham Risk Score (Semmler et al., under review). Hence, limited healthy expandability of subcutaneous adipose tissue appears to represent a pivotal mechanism that triggers fat deposition in ectopic regions such as visceral fat depots and the liver. IR is the unifying underlying mechanism of metabolic complications such as CVD and NAFLD [45]. The importance of IR as a driver for metabolic complications is exemplified by the increasing morbidity and mortality risk with the progression of IR to T2DM [46][47]. This increase in morbidity and mortality in T2DM is true for liver-related complications (i.e., end-stage liver disease and hepatocellular carcinoma (HCC)) as well as for CVD (i.e., heart failure and ischemic events) [48][49]. Thus, the close interrelations are well confirmed epidemiologically between the various manifestations of insulin resistance or MetS on one side and NAFLD on the other; however, these frequently reciprocal relationships make it difficult to ascertain a specific contribution of NAFLD to CVD, as the biological co-evolution allows interpretation as shared risk factors, mutual augmentation, and confounding and may even be due to reverse causation, as genetic analyses indicate [50] (e.g., the possibility of increased prevalence of NAFLD in subjects with CVD due to less physical activity).
2.3. Other Cardiovascular Disease Manifestations
Several studies have reported epidemiological associations of NAFLD and valvular heart disease besides vascular and metabolic disease, i.e., mitral annulus calcification and aortic sclerosis. Subsequently, mitral regurgitation and aortic stenosis might develop, which increase the risk of heart failure on the basis of left ventricular dilation (in mitral regurgitation) and left ventricular hypertrophy (in aortic stenosis). Furthermore, arrhythmias have been reported in NAFLD, mainly atrial fibrillation (due to left atrial enlargement secondary to increased filling pressures and mitral regurgitation), increased QTc interval (corrected QT interval on electrocardiogram), and degenerative cardiac conduction disease (mainly first-degree atrioventricular block and left anterior hemiblock) [51][52]. It was recently suggested that NAFLD and non-alcoholic steatohepatitis (NASH) are associated with expansion of epicardial adipose tissue and the release of pro-inflammatory adipocytokines that cause microcirculatory dysfunction and fibrosis of the adjoining myocardium, resulting in atrial fibrillation as well as heart failure with a preserved ejection fraction (HFpEF). Inflammatory changes in the left atrium lead to electroanatomical remodeling; thus, NAFLD and NASH markedly increase the risk of atrial fibrillation. Simultaneously, patients with NAFLD or NASH commonly show diastolic dysfunction or latent HFpEF [53].
Finally, the prevalence of chronic kidney disease (CKD, estimated glomerular filtration rate (eGFR) < 60 mL/min/1.73 m2, abnormal albuminuria, or overt proteinuria) is markedly increased in patients with NAFLD. The prevalence of CKD ranged from approximately 20% to 55% in patients with NAFLD as compared to 5% to 30% in patients without NAFLD, and the significant association of CKD and NAFLD mostly persisted after adjusting for confounding factors such as hypertension and T2DM [54][55].
This entry is adapted from the peer-reviewed paper 10.3390/jcm10030467
References
- Targher, G.; Day, C.P.; Bonora, E. Risk of Cardiovascular Disease in Patients with Nonalcoholic Fatty Liver Disease. New Engl. J. Med. 2010, 363, 1341–1350.
- Ekstedt, M.; Hagström, H.; Nasr, P.; Fredrikson, M.; Stål, P.; Kechagias, S.; Hultcrantz, R. Fibrosis stage is the strongest predictor for disease-specific mortality in NAFLD after up to 33 years of follow-up. Hepatology 2015, 61, 1547–1554.
- Wu, S.; Wu, F.; Ding, Y.; Hou, J.; Bi, J.; Zhang, Z. Association of non-alcoholic fatty liver disease with major adverse cardiovascular events: A systematic review and meta-analysis. Rep. 2016, 6, 33386.
- Söderberg, C.; Stål, P.; Askling, J.; Glaumann, H.; Lindberg, G.; Marmur, J.; Hultcrantz, R. Decreased survival of subjects with elevated liver function tests during a 28-year follow-up. Hepatology 2010, 51, 595–602.
- Stepanova, M.; Younossi, Z.M. Independent Association Between Nonalcoholic Fatty Liver Disease and Cardiovascular Disease in the US Population. Gastroenterol. Hepatol. 2012, 10, 646–650.
- World Health Organization. Global Health Estimates 2016: Disease Burden by Cause, Age, Sex, by Country and by Region, 2000–2016; WHO: Geneva, Switzerland, 2018.
- Arnett, D.K.; Blumenthal, R.S.; Albert, M.A.; Buroker, A.B.; Goldberger, Z.D.; Hahn, E.J.; Himmelfarb, C.D.; Khera, A.; Lloyd-Jones, D.; McEvoy, J.W.; et al. 2019 ACC/AHA Guideline on the Primary Prevention of Cardiovascular Disease. Circulation 2019, 74, 1376–1414.
- Benjamin, E.J.; Muntner, P.; Alonso, A.; Bittencourt, M.S.; Callaway, C.W.; Carson, A.P.; Chamberlain, A.M.; Chang, A.R.; Cheng, S.; Das, S.R.; et al. Heart disease and stroke statistics—2019 update: A report from the American heart association. Circulation 2019, 139, e56–e528.
- Herrington, W.; Lacey, B.; Sherliker, P.; Armitage, J.; Lewington, S. Epidemiology of Atherosclerosis and the Potential to Reduce the Global Burden of Atherothrombotic Disease. Res. 2016, 118, 535–546.
- Knuuti, J.; Wijns, W.; Saraste, A.; Capodanno, D.; Barbato, E.; Funck-Brentano, C.; Prescott, E.; Storey, R.F.; Deaton, C.; Cuisset, T.; et al. 2019 ESC Guidelines for the diagnosis and management of chronic coronary syndromes. Heart J. 2020, 41, 407–477.
- Cosentino, F.; Grant, P.J.; Aboyans, V.; Bailey, C.J.; Ceriello, A.; Delgado, V.; Federici, M.; Filippatos, G.; Grobbee, D.E.; Hansen, T.B.; et al. 2019 ESC Guidelines on diabetes, pre-diabetes, and cardiovascular diseases developed in collaboration with the EASD. Heart J. 2020, 41, 255–323.
- Bellentani, S.; Marino, M. Epidemiology and natural history of non-alcoholic fatty liver disease (NAFLD). Hepatol. 2009, 8, S4–S8.
- Scorletti, E.; Calder, P.C.; Byrne, C.D. Non-alcoholic fatty liver disease and cardiovascular risk: Metabolic aspects and novel treatments. Endocrinology 2011, 40, 332–343.
- Wanless, I.R.; Lentz, J.S. Fatty liver hepatitis (steatohepatitis) and obesity: An autopsy study with analysis of risk factors. Hepatology 1990, 12, 1106–1110.
- Wojcik-Cichy, K.; Koslinska-Berkan, E.; Piekarska, A. The influence of NAFLD on the risk of atherosclerosis and cardiovascular diseases. Exp. Hepatol. 2018, 4, 1–6.
- Morrison, A.E.; Zaccardi, F.; Khunti, K.; Davies, M.J. Causality between non-alcoholic fatty liver disease and risk of cardiovascular disease and type 2 diabetes: A meta-analysis with bias analysis. Liver Int. 2019, 39, 557–567.
- Marchesini, G.; Moscatiello, S.; Di Domizio, S.; Forlani, G. Obesity-associated liver disease. Clin. Endocrinol. Metab. 2008. 93, S74–S80.
- Kotronen, A.; Yki-Jarvinen, H. Fatty liver: A novel component of the metabolic syndrome. Thromb. Vasc. Biol. 2008, 28, 27–38.
- De Alwis, N.M.; Day, C.P. Non-alcoholic fatty liver disease: The mist gradually clears. Hepatol. 2008, 48, S104–S112.
- Wong, V.W.; Wong, G.L.; Yeung, J.C.; Fung, C.Y.; Chan, J.K.; Chang, Z.H.; Kwan, C.T.; Lam, H.W.; Limquiaco, J.; Chim, A.M.; et al. Long-term clinical outcomes after fatty liver screening in patients undergoing coronary angiogram: A prospective cohort study. Hepatology 2016, 63, 754–763.
- Treeprasertsuk, S.; Leverage, S.; Adams, L.A.; Lindor, K.D.; St Sauver, J.; Angulo, P. The Framingham risk score and heart disease in nonalcoholic fatty liver disease. Liver Int. 2012, 32, 945–950.
- Kim, H.C.; Kim, D.J.; Huh, K.B. Association between nonalcoholic fatty liver disease and carotid intima-media thickness according to the presence of metabolic syndrome. Atherosclerosis 2009, 204, 521–525.
- Sookoian, S.; Pirola, C.J. Non-alcoholic fatty liver disease is strongly associated with carotid atherosclerosis: A systematic review. Hepatol. 2008, 49, 600–607.
- Salvi, P.; Ruffini, R.; Agnoletti, D.; Magnani, E.; Pagliarani, G.; Comandini, G.; Praticò, A.; Borghi, C.; Benetos, A.; Pazzi, P. Increased arterial stiffness in nonalcoholic fatty liver disease: The Cardio-GOOSE study. Hypertens. 2010, 28, 1699–1707.
- Shiotani, A.; Motoyama, M.; Matsuda, T.; Miyanishi, T. Brachial-ankle Pulse Wave Velocity in Japanese University Students. Med. 2005, 44, 696–701.
- Motamed, N.; Rabiee, B.; Poustchi, H.; Dehestani, B.; Hemasi, G.R.; Khonsari, M.R.; Maadi, M.; Saeedian, F.S.; Zamani, F.Non-alcoholic fatty liver disease (NAFLD) and 10-year risk of cardiovascular diseases. Res. Hepatol. Gastroenterol. 2017, 41, 31–38.
- Ampuero, J.; Gallego-Duran, R.; Romero-Gomez, M. Association of NAFLD with subclinical atherosclerosis and coronary-artery disease: Meta-analysis. Esp. Enferm. Dig. 2015, 107, 10–16.
- Ekstedt, M.; Franzén, L.E.; Mathiesen, U.L.; Thorelius, L.; Holmqvist, M.; Bodemar, G.; Kechagias, S. Long-term follow-up of patients with NAFLD and elevated liver enzymes. Hepatology 2006, 44, 865–873.
- Ghouri, N.; Preiss, D.; Sattar, N. Liver enzymes, nonalcoholic fatty liver disease, and incident cardiovascular disease: A narrative review and clinical perspective of prospective data. Hepatology 2010, 52, 1156–1161.
- Hippisley-Cox, J.; Coupland, C.; Robson, J.; Brindle, P. Derivation, validation, and evaluation of a new QRISK model to estimate lifetime risk of cardiovascular disease: Cohort study using QResearch database. BMJ 2010, 341, c6624.
- Alexander, M.; Loomis, A.K.; van der Lei, J.; Duarte-Salles, T.; Prieto-Alhambra, D.; Ansell, D.; Pasqua, A.; Lapi, F.; Rijnbeek, P.; Mosseveld, M.; et al. Non-alcoholic fatty liver disease and risk of incident acute myocardial infarction and stroke: Findings from matched cohort study of 18 million European adults. BMJ 2019, 367, l5367.
- Sattar, N.; Forrest, E.; Preiss, D. Non-alcoholic fatty liver disease. BMJ 2014, 349, g4596.
- Marchesini, G.; Forlani, G.; Cerrelli, F.; Manini, R.; Natale, S.; Baraldi, L.; Ermini, G.; Savorani, G.; Zocchi, D.; Melchionda, N. WHO and ATPIII proposals for the definition of the metabolic syndrome in patients with Type 2 diabetes. Diabetes Med. 2004, 21, 383–387.
- Dietrich, P.; Hellerbrand, C. Non-alcoholic fatty liver disease, obesity and the metabolic syndrome. Best Pr. Res. Clin. Gastroenterol. 2014, 28, 637–653.
- Yki-Järvinen, H. Non-alcoholic fatty liver disease as a cause and a consequence of metabolic syndrome. Lancet Diabetes Endocrinol. 2014, 2, 901–910.
- Younossi, Z.M.; Koenig, A.B.; Abdelatif, D.; Fazel, Y.; Henry, L.; Wymer, M. Global epidemiology of nonalcoholic fatty liver disease-Meta-analytic assessment of prevalence, incidence, and outcomes. Hepatology 2016, 64, 73–84.
- Chang, Y.; Jung, H.S.; Cho, J.; Zhang, Y.; Yun, K.E.; Lazo, M.; Pastor-Barriuso, R.; Ahn, J.; Kim, C.W.; Rampal, S.; et al. Metabolically Healthy Obesity and the Development of Nonalcoholic Fatty Liver Disease. J. Gastroenterol. 2016, 111, 1133–1140.
- Chen, F.; Esmaili, S.; Rogers, G.B.; Bugianesi, E.; Petta, S.; Marchesini, G.; Bayoumi, A.; Metwally, M.; Azardaryany, M.K.; Coulter, S.; et al. Lean NAFLD: A Distinct Entity Shaped by Differential Metabolic Adaptation. Hepatology 2020, 71, 1213–1227.
- Iacobini, C.; Pugliese, G.; Blasetti Fantauzzi, C.; Federici, M.; Menini, S. Metabolically healthy versus metabolically unhealthy obesity. Metabolism 2019, 92, 51–60.
- Younes, R.; Bugianesi, E. NASH in Lean Individuals. Liver Dis. 2019, 39, 086–095.
- Gentile, C.L.; Weir, T.L.; Cox-York, K.A.; Wei, Y.; Wang, D.; Reese, L.; Moran, G.; Estrada, A.; Mulligan, C.; Pagliassotti, M.J.; Foster, M.T. The role of visceral and subcutaneous adipose tissue fatty acid composition in liver pathophysiology associated with NAFLD. Adipocyte 2015, 4, 101–112.
- Graffy, P.M.; Pickhardt, P.J. Quantification of hepatic and visceral fat by CT and MR imaging: Relevance to the obesity epidemic, metabolic syndrome and NAFLD. J. Radiol. 2016, 89, 20151024.
- Young, S.; Tariq, R.; Provenza, J.; Satapathy, S.K.; Faisal, K.; Choudhry, A.; Friedman, S.L.; Singal, A.K. Prevalence and Profile of Nonalcoholic Fatty Liver Disease in Lean Adults: Systematic Review and Meta-Analysis. Commun. 2020, 4, 953–972.
- Golabi, P.; Paik, J.; Fukui, N.; Locklear, C.T.; de Avilla, L.; Younossi, Z.M. Patients with Lean Nonalcoholic Fatty Liver Disease Are Metabolically Abnormal and Have a Higher Risk for Mortality. Diabetes 2018, 37, 65–72.
- Stols-Gonçalves, D.; Hovingh, G.K.; Nieuwdorp, M.; Holleboom, A.G. NAFLD and Atherosclerosis: Two Sides of the Same Dysmetabolic Coin? Trends Endocrinol. Metab. 2019, 30, 891–902.
- Patel, T.P.; Rawal, K.; Bagchi, A.K.; Akolkar, G.; Bernardes, N.; Dias, D.D.S.; Gupta, S.; Singal, P.K. Insulin resistance: An additional risk factor in the pathogenesis of cardiovascular disease in type 2 diabetes. Fail. Rev. 2016, 21, 11–23.
- Balakumar, P.; Maung-U, K.; Jagadeesh, G. Prevalence and prevention of cardiovascular disease and diabetes mellitus. Res. 2016, 113, 600–609.
- Lai, S.-W.; Lin, C.-L.; Liao, K.-F. Association between diabetes mellitus and hepatocellular carcinoma. J. Gastroenterol. Hepatol. 2019, 31, 898–899.
- Anstee, Q.M.; Targher, G.; Day, C.P. Progression of NAFLD to diabetes mellitus, cardiovascular disease or cirrhosis. Rev. Gastroenterol. Hepatol. 2013, 10, 330–344.
- Lauridsen, B.K.; Stender, S.; Kristensen, T.S.; Kofoed, K.F.; Køber, L.; Nordestgaard, B.G.; Tybjærg-Hansen, A. Liver fat content, non-alcoholic fatty liver disease, and ischaemic heart disease: Mendelian randomization and meta-analysis of 279 013 individuals. Heart J. 2018, 39, 385–393.
- Anstee, Q.M.; Mantovani, A.; Tilg, H.; Targher, G. Risk of cardiomyopathy and cardiac arrhythmias in patients with nonalcoholic fatty liver disease. Rev. Gastroenterol. Hepatol. 2018, 15, 425–439.
- Mantovani, A.; Dauriz, M.; Sandri, D.; Bonapace, S.; Zoppini, G.; Tilg, H.; Byrne, C.D.; Targher, G. Association between non-alcoholic fatty liver disease and risk of atrial fibrillation in adult individuals: An updated meta-analysis. Liver Int. 2019, 39, 758–769.
- Packer, M. Atrial Fibrillation and Heart Failure with Preserved Ejection Fraction in Patients With Nonalcoholic Fatty Liver Disease. J. Med. 2020, 133, 170–177.
- Musso, G.; Cassader, M.; Cohney, S.; De Michieli, F.; Pinach, S.; Saba, F.; Gambino, R. Fatty Liver and Chronic Kidney Disease: Novel Mechanistic Insights and Therapeutic Opportunities. Diabetes Care 2016, 39, 1830–1845.
- Targher, G.; Chonchol, M.; Byrne, C.D. CKD and Nonalcoholic Fatty Liver Disease. J. Kidney Dis. 2014, 64, 638–652.