1. Introduction
Polycystic ovary syndrome (PCOS) is a wide-spread endocrine disorder affecting 5–15% of women in their reproductive age worldwide and is a frequent cause of infertility [
1]. Diagnosis according to current international guidelines that apply the diagnostic “Rotterdam” criteria require the presence of at least two of the following features: hyperandrogenism, ovulatory dysfunction or/and polycystic ovary in ultrasonic scans, while other pathologies must be excluded [
2]. PCOS is further characterized by an elevated LH/FSH ratio and often associated with obesity and insulin resistance leading to an hyperinsulinemic state in 80% of the obese women and 30–40% of the lean ones [
3]. The etiology of the syndrome has not been clarified completely yet. Certain genetic patterns that impact for example synthesis, regulation and action of sex hormones and the insulin receptor, but also gestational factors like high maternal levels of androgens or AMH (anti-Müllerian hormone) seem to contribute to the onset of the disease [
4,
5,
6]. Postnatal lifestyle factors like inadequate nutrition accompanied by a lack of physical exercise promote the development of the disease as they often result in obesity and disturbances of the glucose metabolism. In fact, hyperinsulinemia, independent of BMI, is a key contributor to PCOS pathogenesis [
7,
8] as it results in augmented androgen production in the adrenal cortex and follicles via stimulation of LH secretion, while concomitantly reducing SHBG (sex hormone binding globulin) synthesis [
8]. Resulting elevated androgene levels interfere with follicular maturation [
9] and may lead to the characteristic clinical manifestations like acne and hirsutism. Obesity on the other hand, may not only reinforce insulin resistance [
10], but is a pathogenicity factor itself contributing to the endocrinologic disorder due to the altered hormone metabolism of adipocytes [
11]. Furthermore, adipose tissue contributes to constant low-level systemic inflammation as will be described further below in this review. PCOS itself nowadays is considered a condition of chronic inflammation with elevated levels of leukocytes, pro-inflammatory cytokines, elevated white blood count and markers such as the C-reactive protein being detectable [
12] and also affects women with a normal BMI [
13].
Inflammatory processes are crucial for the survival of human beings and occur as a reaction to stimuli like injury or infections. Potential pathogens may enter a host due to a trauma, barrier breakage or microbial invasion and in order to regain the integrity of the organism, the invaders must be eliminated, removed and the functional state of affected tissue must be restored. During the acute inflammatory response, eicosanoid lipid mediator (LM) molecules that derive from the ω-6 poly-unsaturated fatty acid (PUFA) arachidonic acid (AA) are rapidly synthesized by cells of the innate immune system, such as granulocytes and macrophages that are recruited to the sited of the event [
14]. For this step, the enzymes cyclooxagenases 1 and 2 (Cox-1/2) are utilized and prostanoids including prostaglandins (PG), leukotrienes (LTs) and thromboxanes (TXs) are synthesized from AA [
15,
16]. Additionally, mast cells and the classically activated M1 macrophages secrete different kinds of inflammatory cytokines including TNFα, IL-1 and Il-6—all of them being highly inflammatory substances that drive the generation of the classical inflammatory symptoms: redness, heat, swelling, pain, and loss of function [
14,
15,
16]. Guided by the inflammatory eicosanoids and cytokines present at the site of infection, neutrophiles and monocytes leave the blood vessels and migrate into the tissue, thereby contributing to the formation of inflammatory exudates and progression of the inflammation process [
14,
15,
16]. An effective initiation of the inflammatory response is essential for survival, but its self-limitation is equally important. Exceeding inflammation may lead to the phenomenon of a cytokine storm and subsequent life-threatening sepsis [
17]. The failure to temporally limit inflammation results in chronic inflammatory diseases including cardiovascular and neurological disorders, auto-immune diseases, diabetes, and cancer [
18,
19]. For a long time, the resolution of inflammation had been considered a passive process with mere dissolution of inflammatory mediators and inflammation divided into initiation and resolution [
20]. This view was changed by the discovery of distinct lipid mediator molecules that can “switch on” resolution processes in animal models and actively drive the resolution processes. These mediators comprise four different subgroups: the resolvins (Rvs), lipoxins (LXs), protectins (PDs) and maresins (Masr) and are depicted as SPMs: specialized pro-resolving mediators [
21,
22,
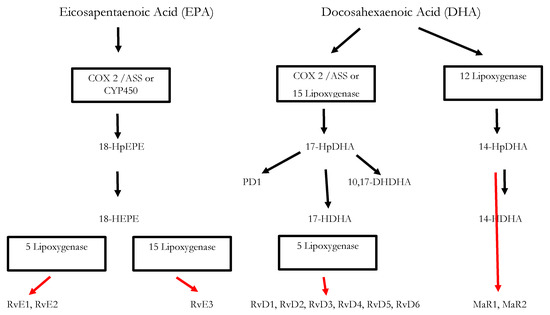
23]. Resolvins, protectins, and maresins are synthesized from the ω-3 PUFAs EPA (Eicosapentanenoic acid) and DHA (docosahexaenoic acid) via certain intermediates (18-HpETE, 17-HpDHA and 14-HpDHA). Their biosynthetic pathways (see for an overview) involve certain lipoxygenases as well as the Cox-enzymes that take part both in the eicosanoid synthesis as well as in SPM production. The synthesis of LX in contrast starts from ω-6 PUFA AA [
24,
25]. Interestingly, Aspirin as an irreversible inhibitor of Cox-enzymes blocks prostanoid synthesis by modification of their catalytic domain. However, these enzymes remain active and are triggered to synthesize the SPM precursors 18-HpETE and 17-HpDHA. Resulting SPMS are called aspirin-triggered resolvins, -maresins, or protectins (AT-SPMs) and are potent mediators of resolution that are widely used in experimental approaches [
21,
24,
26,
27].