The increase of public awareness on ocular conditions leads to an early diagnosis and treatment, as well as an increased demand for more effective and minimally invasive solutions for the treatment of both the anterior and posterior segments of the eye. Despite being the most common route of ophthalmic drug administration, eye drops are associated with compliance issues, drug wastage by lacrimation, and low bioavailability due to the ocular barriers. In order to overcome these problems, the design of drug-eluting ophthalmic lenses constitutes a non-invasive and patient-friendly approach for the sustained drug delivery to the eye. Several examples of therapeutic contact lenses and intraocular lenses have been developed, by means of different strategies of drug loading, leading to promising results.
- therapeutic contact lenses
- therapeutic intraocular lenses
- eye diseases
- glaucoma
- cataract
- corneal diseases
- posterior segment of the eye diseases
1. Drug Loading Methods
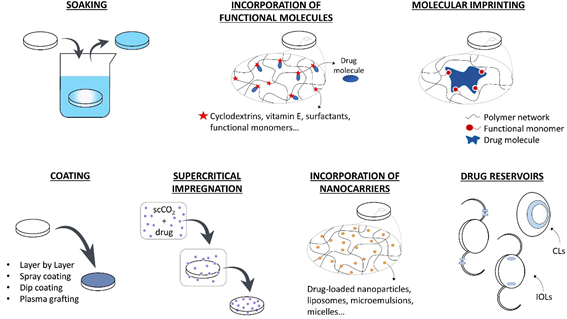
Several examples of therapeutic CLs and IOLs have been developed in the last decade with promising results. The use of drug-loaded CLs can substitute the topical application of eye drops increasing the drug residence time on the cornea, the drug bioavailability, and the patient compliance. Therapeutic IOLs, which are directly implanted in the anterior chamber, are expected to perform an efficient prophylaxis for post-surgical inflammation and infection or temporarily substitute intraocular injections in case of chronic diseases. In the following section, the various strategies that have been developed for loading drugs in therapeutic lenses are summarized. In Figure 1, a schematic description of these methods is presented.
1.1. Soaking
The simplest method for obtaining therapeutic lenses is soaking into a drug solution [24,30–45]. The amount of drug loaded and released depends on the material and the structure of the lens (e.g., porosity, swelling capacity), on the drug characteristics (e.g., molecular structure, molecular weight, charge), and on eventual interactions that may be established between the drugs and the lens material [22]. Besides, the soaking parameters also affects drug loading, i.e., the concentration of the solution [46], loading time [40], and environmental factors, such as temperature and pH [47]. The main issue of the soaking method still is the limited control over the drug release profile, which is usually characterized by a high initial release rate and a short delivery time after lens placement onto the eye [47,48]. Furthermore, economic and environmental concerns rise due to the waste of the drug present in the soaking solution [46]. Most drug-loaded lenses that are obtained by soaking are able to retain their therapeutic effect for a few hours or for some days [48], although a few examples of sustained release over weeks do exist [24]. This fast release kinetics could be suitable in the case of disposable CLs with a daily use, but it is not compatible with the development of IOLs with a long-term therapeutic purpose [49].
Figure 1. Strategies for the development of therapeutic ophthalmic lenses: soaking into a drug solution, incorporation of functional molecules with a high affinity to the drug, molecular imprinting, drug-eluting or drug-barrier coating, supercritical impregnation, incorporation of nanocarriers and incorporation of drug reservoirs.
1.2. Incorporation of Functional Molecules
The incorporation of functional molecules into the lens polymer (e.g., cyclodextrins, vitamin E, surfactants, functional monomers) proved to be a potential strategy for enhancing drug loading during the soaking step and tuning the release kinetics. Cyclodextrins present a hydrophobic cavity that is suitable for accommodating hydrophobic drugs. They have been successfully co-polymerized with the lens backbone material to control drug delivery over time [25,50], or mixed to the drug solution prior to lens soaking in order to enhance the apparent aqueous solubility of hydrophobic drugs [51]. Vitamin E is considered to be a promising molecule for providing a hydrophobic diffusion barrier for the release of hydrophilic drugs [52–56]. When hydrophobic drugs are involved, drug molecules diffuse through the highly viscous vitamin E agglomerates, resulting in a slower release kinetic [57]. Because of the hydrophobic nature of vitamin E, it can be easily incorporated into the lenses by soaking in a vitamin E-ethanol solution. After ethanol evaporation, vitamin agglomerates remain trapped into the polymer network. However, a loss in oxygen permeability, protein adsorption, and changes in the mechanical properties can be associated with the use of this functional molecule in therapeutic lenses [22]. Aggregates that are composed of long-chain surfactants and oppositely charged ionic drugs can be added to the lens pre-polymer mixture to extend drug release over time by entrapment of the drug molecules [58]. The suitability of the lens polymer network to load and release drugs in a sustained fashion can also be improved by co-polymerization with functional monomers presenting a stronger affinity with the target drugs [59–61] or by the modification of the lens charge [62,63].
1.3. Coating
Several methods have been suggested to produce coatings on drug-eluting biomedical devices (e.g., layer-by-layer deposition, spray coating, dip coating, plasma-assisted grafting) with the purpose of implementing drug-eluting reservoirs [64–73]. Besides, these coatings may be used in order to modify the hydrophilicity of the device surface [74] and, also, as diffusion barriers to drug release [75–77]. The main concern with the design of coatings to be applied onto CLs or IOLs is the preservation of the optical properties of the original lenses. In the case of coatings with biodegradable polymers, the degradation products must be biocompatible with the surrounding tissues. Furthermore, the coating adhesion to the lens material should be sufficient for avoiding the presence of floating debris on the cornea or in the anterior chamber, which results from the coating detachment from CLs or IOLs, respectively.
1.4. Molecular Imprinting
Molecular imprinting can be obtained by two different methods, which involve either covalent or noncovalent bonds (e.g., ionic, hydrophobic, hydrogen bonds) between the template drug and functional monomers. The latter method is the most widely used in drug delivery, as it is associated to an easier and faster drug dissociation kinetics. Stable noncovalent drug-monomer complexes are established by solubilizing both the template drug and the functional monomers in the prepolymer mixture [78]. With the subsequent polymerization step, the functional monomers are covalently bonded to the polymer backbone, but not the drug, due to the presence of aromatic rings or other highly stable molecular structures that are less prone to react. The drug is removed during the washing cycles to which the newly polymerized biomaterials are subjected to eliminate potentially toxic unreacted monomers [79]. After drug removal, tailored memory sites remain imprinted in the polymer and, by mimicking the drug’s natural receptors, they create an oriented functional material with high affinity to the template drug [80]. The drug is then re-loaded in the polymer by soaking and interacting with the memory sites. This process can lead to a higher drug loading and a slower release profile [81], and it was successfully applied to therapeutic CLs and IOLs [82–88]. The use of miscible combinations of drugs and monomers is generally preferable over non-miscible ones in order to avoid the use of solvents, since their presence could prevent the orientation of monomers around the drug molecules and result in a less effective imprinting [89]. The drug affinity and the release kinetics are determined by the type of functional monomers and their concentration in the matrix: the ratio between functional monomers and drug molecules must be optimized for each combination of drug and monomers [22,89–91]. This ratio has to be sufficiently high to create enough interactions and retard drug release; however, an excessive monomer concentration could interfere with drug diffusion during the loading phase and could also impede an organized orientation of molecules, thus hindering any effect on the template drug.
1.5. Supercritical Impregnation
Supercritical fluids are characterized by a high density, low viscosity, high diffusivity, and low interfacial tension [92]. These features are favorable for the diffusion of such fluids in the polymer matrices of CLs or IOLs [26,60,92–97]. Impregnation with a supercritical fluid consists in the dissolution of the drug in the solvent (generally supercritical CO2), followed by the interaction with the target material [22]. Supercritical CO2 is a better solvent for drugs than water, and also a better plasticizer for polymer networks: therefore, drug loading is enhanced when compared to traditional techniques like soaking [46]. This method is considered a ‘green’ alternative, as it forces the impregnation of the drug in the lens without using organic solvents. Moreover, CO2 is spontaneously released from the lens after impregnation, avoiding the purification steps that are usually associated to the use of solvents [92]. An optimized loading and release can be obtained by tuning the processing parameters, like pressure, temperature, presence of a co-solvent, duration of impregnation, and depressurization rate [22,46]. The choice of suitable parameters is fundamental: a too rapid depressurization, for example, can damage the lens material (foaming phenomenon [26]) and, therefore, compromise the optical functionality of the device. However, a too slow depressurization limits the efficacy of impregnation, as it reduces drug deposition into the polymer network [98]. The complex set-up is a limitation in the use of supercritical impregnation, as compared to other drug-loading techniques, and it can hinder its incorporation into the manufacturing process [89]. When applied to IOLs, supercritical impregnation allowed obtaining a sustained drug release over several weeks with various types of drugs (e.g., dexamethasone sodium and ciprofloxacin [26,92,95], methotrexate [97], cefuroxime sodium, and timolol maleate [98]). However, when the technology was applied to the thinner CLs, an initial burst release was commonly detected, followed by a sustained release for a few hours [93,94,99]. In order to overcome this issue, the combination of molecular imprinting and impregnation demonstrated to be an interesting alternative for the achievement of a gradual release, even in the first hours of CL wearing [100].
1.6. Incorporation of Nanocarriers
Colloidal nanocarriers entrapping active substances (e.g., polymeric biodegradable or non-degradable nanoparticles [74,101–108], liposomes, microemulsions, micelles) can be easily incorporated into the polymeric matrix of the lens [22]. Most of the nanoparticles for drug-delivery (homogeneous nanospheres or heterogeneous nanocapsules) are biodegradable polymers, which can release the drugs by degradation. Other mechanisms, such as light-induced release, can be implemented on non-degradable carriers [109]. Changes in pH and temperature, drug diffusion through the particle core/shell and matrix swelling also influence the drug delivery profile [110]. Liposomes have a structure that is similar to the biological membranes, in which lipophilic drugs can be loaded into the phospholipid bilayer and hydrophilic drugs into the aqueous core [111]. Polymeric micelles are spontaneously formed structures with promising applications in the delivery of hydrophobic drugs [112,113]. Microemulsions increase the drug-loading capacity during soaking and they present good thermodynamic stability, while preserving the ease of fabrication of the therapeutic lenses [114–116]. Despite the advantages of these innovative systems, significant problems need to be overcome during their design as drug-carriers for ophthalmic applications. In fact, changes in the mechanical properties and water content can be associated to their use in hydrogels [22]. Moreover, their natural tendency to aggregation must be hampered in order avoid a decrease in transparency [10].
1.7. Drug Reservoirs
Drug-eluting ocular implants have long been used as drug delivery platforms. However, their eventual migration after injection in the eye is one possible complication. The physical link of these implants to IOLs may overcome the problem and guarantee their correct positioning in the anterior chamber of the eye. Despite being more commonly applied on IOLs [23,27,117–119], examples of incorporated reservoirs were also implemented on CLs [120–126]. Drug reservoirs are usually constituted of biodegradable polymers: the material must be biocompatible, and its degradation products should not cause chronic inflammation or toxicity in the ocular tissues [47]. Reservoirs guarantee a prolonged (usually months) and controlled release over time. Moreover, the optical properties of the lens are not compromised, as they can be attached to the haptics of IOLs or incorporated into the peripheral part of CLs [12]. On the other hand, the design and fabrication of reservoirs can be complex and the physical link with the lens might be difficult to manage [46]. In the case of IOLs, the presence of these reservoirs can also raise problems while loading the lens in the injector system prior to surgery or in the ejection process [47].
2. Lenses for Ocular Diseases
2.1. Glaucoma
Glaucoma is the leading cause of irreversible blindness worldwide, and it is characterized by a progressive visual field loss due to the degeneration of retinal ganglion cells and optic nerve changes [5,127]. Although the relationship between diabetes and the development of glaucoma is still controversial and further investigation is needed, recent studies [128,129] have shown a significantly higher intraocular pressure (IOP) in diabetic patients when compared to non-diabetics, possibly due to an impaired autonomic function [129] and the progression of microvascular injury that is associated with chronic hyperglycemia [130]. An elevated IOP is considered to be a major risk factor for glaucoma, as it is associated to retinal ischemia and mechanical stress, as well as to an impaired ocular blood flow to retinal neurons [128]. The current treatment of glaucoma involves the lowering of IOP by pharmacological administration, laser, or surgical procedures [131]. The balance between the aqueous humor production and outflow from the anterior chamber of the eye regulates the IOP [128]. Therefore, current drug treatments are targeted at the reduction of aqueous humor production and/or the increase of the outflow facility. This can be achieved by use of beta-adrenergic antagonists (e.g., timolol, betaxolol, puerarin), sympathomimetic agents (e.g., epinephrine), parasympathomimetic miotic agents (e.g., pilocarpine), carbonic anhydrase inhibitors (e.g., dorzolamide), or prostaglandin analogues (e.g., latanoprost, bimatoprost) [132].
Many examples of therapeutic CLs for the treatment of glaucoma are described in the literature. Several strategies were followed in order to improve drug loading: optimized soaking, the incorporation of functional molecules, coating, molecular imprinting, supercritical impregnation, incorporation of nanocarriers, and reservoir attachment. A prolonged release over days was obtained even by soaking in a drug solution, which constitutes the easiest drug loading method. By the incorporation of drug eluting reservoirs, drug release was extended over weeks. Despite the wide research on the topic, only a few examples of in vivo tests on rabbits, dogs, and monkeys were reported, with a lack of preclinical and clinical tests on glaucoma patients, which are indispensable for the future commercial application of the designed devices.
2.2. Cataract
A cataract is defined as the opacification of the natural crystalline lens with a consequent decrease in the quality of vision [140]. It is the leading cause of vision loss in elderly patients, causing visual impairment in about 30% of the population over 65 years old, but its incidence is also increasing in the younger population due to the exposition of UV radiation, smoke, use of steroids, increased incidence of diabetes and malnutrition [46,141]. Approximately 20% of cataract surgeries in the western population are performed on diabetic patients [142]. Diabetic crystalline lenses are characterized by an increased level of free radicals and impaired antioxidant capacity, which leads to an increased susceptibility to oxidative stress. Cataract development occurs at an earlier age in presence of diabetes, but an appropriate metabolic control can contribute to the lens recovery in young patients [143]. The administration of aldose-reductase inhibitors (ARIs) and antioxidants was also demonstrated to be beneficial in the delay of diabetic cataract in the early stage of the pathology and it could be useful for its prevention in patients at risk. The use of CLs that are loaded with aldose-reductase inhibitors or antioxidants for the prevention of cataract in diabetic patients, especially in the young population, demonstrated, in in vitro studies, to be a promising approach.
Cataract surgery currently remains the standard treatment of severe cataractous eyes. The procedure consists in the removal of the damaged lens and a subsequent implant of an IOL. Despite the advances in the technique and the evolution in the different types of IOLs made this procedure one of the most cost-effective in the current healthcare [46], some post-operative complications can still occur and cause patient discomfort and visual impairment, as well as a prolonged recovery time [145]. An inflammatory response due to the physical trauma that is associated to surgery is considered to be physiological and it is generally treated with topical non-steroidal anti-inflammatory drugs (NSAIDs) or corticosteroids. If untreated, persistent inflammation can lead to pseudophakic cystoid macular edema (PCME), uveitis, iritis, glaucoma, and an increase of IOP [145,146]. Diabetic patients are at increased risk of developing these diseases. A recent clinical trial by McCafferty et al. [146] on 662 patients evidenced that PCME was the most common complication after cataract surgery. Because of the higher sensitivity of the vascular bed in diabetic patients, approximately 4–12% of patients affected by diabetes mellitus [142,147] and up to 56% of patients with diabetic retinopathy [148] are expected to develop PCME after IOL implant. Ocular infection, often linked to endophthalmitis, is another possible adverse condition, and it is mostly caused by bacterial migration from the lid and conjunctiva to the intraocular space. In this case, prevention and treatment involve the use of topical corticosteroids and antibiotics [145]. The migration and proliferation of epithelial cells to the posterior surface of the IOL can also lead to visual impairment after surgery; the phenomenon is known as posterior capsule opacification (PCO), and it is commonly treated by a laser procedure [149]. Postoperative endophthalmitis and early PCO are both reported to develop at a higher incidence rate in diabetic patients [150,151].
The current prophylaxis after cataract surgery consists in the administration of antibiotics and anti-inflammatory drugs (NSAIDs and/or corticosteroids) in the form of eye drops for two to four weeks after IOL implantation. The Food and Drug Administration approved a new intracameral drug-release device (DEXYCU, Icon Bioscience Inc., Newark, CA) in 2018 for the treatment of postoperative inflammation associated to cataract surgery [152]. The device is constituted by an injectable dexamethasone suspension, which is able to guarantee a 21-days long therapeutic level of drug release. The suspension is injected at the end of cataract surgery, forming a surface tension-based sphere positioned aside the IOL and behind the iris. The use of CLs as drug depots for antibiotics and anti-inflammatories has been proposed as a less-invasive alternative [21,111]. However, in the case of cataract surgery, postoperative prophylaxis could be more efficiently achieved by use of drug-loaded IOLs, in order to overcome the issues that are related to patient compliance and poor drug permeability through the cornea, to which both eye drops administration and drug-eluting CLs are subjected [47]. The possibility of a dual loading of both antibiotics and anti-inflammatory drugs on IOLs constitutes an interesting solution for addressing both post-surgical infection and inflammation. Drug release for 15 days or longer can be obtained with various drugs, thus confirming the suitability of double loaded IOLs for the substitution of the current eye drops prophylaxis, usually administered for two weeks after surgery. Examples of drug eluting IOLs for the prevention of PCO were also reported and, interestingly, long-term effects were observed in rabbits, even in the case of short-term drug release. As previously stated for drug-eluting CLs, extended in vivo tests and subsequent pre-clinical tests are also missing for drug-eluting IOLs, despite the promising results that were obtained in rabbit models.
2.3. Corneal Diseases
Keratoconjunctivitis sicca, or dry eye syndrome, is a common condition of the anterior segment of the eye, which can be associated to discomfort, burning sensation, external body sensation, ocular pain, light sensitivity, and intermittent blurred vision [159]. The presence of a normal tear film is fundamental for corneal health and immune protection [160] and the prevention of corneal injuries. Ocular lubrication can be compromised by reduced tear production and/or instability of the tear film, leading to a rapid evaporation from the eye surface. Various causes have been identified, including environmental factors (low ambient humidity, excessive wind or dust, temperature extremes, air conditioning), ageing, allergies, a prolonged computer use, metabolic conditions and nutritional deficiencies, contact lens use, and the prolonged administration of systemic or topical drugs (e.g., antiglaucoma medications or preservative-containing eye drops) [161]. Patients that are affected by neuropathic disorders, Sjögren’s syndrome, lupus, blepharitis, or congenital abnormalities of the lid are more prone to developing dry eye symptoms [162].
Other common corneal diseases are keratitis, a corneal inflammation that is usually treated by topical or systemic administration of antibiotics, biocides, antifungals, or antivirals, depending on the type of infection [163], and exposure keratopathy, often caused by an inadequate eyelid closure and treated by the use of lubricating drops or by surgical procedure [164]. Bullous keratopathy and Fuch’s dystrophy, on the contrary, are caused by an impairment of the corneal endothelium which loses the ability to drain fluid out of the cornea. Hypertonic topical solutions are usually prescribed to reduce the resulting corneal edema [165], and the concomitant wearing of bandage contact lenses is suggested to increase the residence time of eyedrops on the corneal surface [166,167]. Inherited corneal dystrophies, such as keratoconus, lattice dystrophy, and map-dot-fingerprint dystrophy, can lead to a major visual impairment at their advanced stage [168].
Although often overlooked, complications in the anterior segment of the eye are also common in diabetic patients [169]. It is estimated that diabetic keratopathy affects approximately 47–64% of diabetic patients and, if not treated, it can lead to major visual impairment [170]. Keratopathy is associated with an increased risk of corneal epithelial defects, recurrent erosions, delayed epithelial wound healing, tear film alteration, edema, and neurotropic corneal ulcers [160,169,170]. In severe cases, hyperglycemia and microvascular damage cause corneal neurotrophic lesions and a progressive decrease in corneal nerve fiber density, which lead to a loss of sensitivity, an impairment of the epithelial healing process, and a lack of feedback control over tear secretion [169,171]. An abnormal regulation of the healing mechanism can cause corneal opacity and blindness [172]. Despite the suggested causality between peripheral neuropathy and diabetic keratopathy, direct alterations in the corneal epithelium were also observed in less severe conditions, without signs of neuropathy [160]. In the early stage of the pathology, diabetic patients frequently experience dry eye symptoms [171]. Hyperglycemia, in fact, can cause a microvascular damage to the lacrimal gland, also contributing to the low tear secretion [169]. Alterations in the tear film were also associated to the presence of inflammation, oxidative stress, and the accumulation of advanced glycation end products (AGEs) in the lacrimal gland [160]. Tear film changes, such as a reduced lipid layer quality and film stability, were also registered in diabetics [173].
The first-line treatment of the cornea is based on the use of artificial tears to maintain a lubricated ocular surface, but a few examples of CLs releasing moisturizing agents are already present on the market. The current research is focused on the optimization of the release profiles of those agents, alone or in association with drugs for pathological conditions. In this latter case, the administration of eye drops is the standard of care, but many therapeutic CLs eluting corticosteroids and NSAIDs were reported in literature. More recently, specific drugs addressed to the treatment of keratopathy were suggested (i.e., naltrexone, growth factors, aldose reductase inhibitors, and antioxidants), and a few examples reported their sustained release by drug-eluting CLs. Preliminary clinical tests were conducted with therapeutic CLs releasing epidermal growth factors with encouraging results. However, extended experimentation is required due to the limited number of patients involved. Interestingly, the use of therapeutic CLs eluting aldose reductase inhibitors or antioxidants could have a double effect on both corneal issues and cataract prevention in the diabetic eye. In fact, the possibility of simultaneous treatment of different diseases could be an appealing objective in the design of therapeutic lenses that are addressed to diabetic patients due to the relationship between several ocular pathologies and chronic hyperglycemia.
2.4. Posterior Segment Diseases
Macular degeneration, diabetic retinopathy and diabetic macular edema are the most common vision-impairing diseases of the back of the eye [207]. AMD is the leading cause of elderly blindness in the United States, affecting 9.2% of individuals over 50 years old [208,209]. The pathology can be manifested under two forms, namely dry and wet AMD. Although dry AMD is more frequent, with an incidence above 85% in AMD patients, wet AMD is responsible for 90% of severe vision loss cases [209]. In the case of dry AMD, a thinning of the retinal pigment epithelium is observed, leading to blurred central vision [209]. Wet AMD is associated to retinal hemorrhage and fibrovascular tissue formation, due to an abnormal neovascularization, with the accumulation of subretinal or intraretinal fluid [210]. Cardiovascular diseases, which are linked to a higher hydrostatic pressure in the eye vessels, constitute a risk factor for the development of wet AMD [208], as well as smoking, obesity, and hereditary factors. Most of the pathologies of the back of the eye in diabetic patients, such as DR and DME, are caused by the long term and chronic progression of microvascular damage in the retina associated with persistent hyperglycemia [211]. In its non-proliferative stage, DR is associated to an abnormal vessel permeability or the presence of microaneurysms in the capillaries. Consequently, the leaking of fluid and its accumulation in the surrounding tissue can lead to the progression of macular edema [6] and, if the subsequent swelling or thickening of the retina occurs in the fovea, DME can significantly affect vision [211]. DME can be diagnosed at any stage of DR, and its incidence increases with the progression of DR and with the duration of diabetes. Poorly controlled blood pressure, smoking, a high cholesterol level, and a reduced kidney function are also considered to be risk factors for the development of the pathology [211]. At its proliferative stage, DR can be directly related to vision impairment. In fact, neovascularization is promoted on the retinal surface due to the occlusion of capillaries, but the fragility of the newly formed capillaries leads to frequent hemorrhages. The accumulation of blood in the vitreous affects vision, while the formation of fibrotic tissue can lead to traction retinal detachment and permanent blindness [6,211]. Approximately one-third of diabetic patients are expected to develop DR, and one-tenth of these are associated to sight-threatening states [212]. Even if strict blood glucose and blood pressure control have a protective effect on the development and progression of retinopathy, they are not associated to the complete elimination of the threat.
The treatment of disorders of the posterior segment of the eye is a challenging topic in ocular drug delivery. The standard of care consists in laser treatments or invasive intravitreal injections. The possibility of using therapeutic lenses for the treatment of the posterior segment of the eye is almost unexplored. Despite this, a few in vivo studies detected drug amounts in the retina after drug delivery through therapeutic CLs. Based on the reported cases, further research on the ocular drug delivery pathway to the posterior segment and the therapeutic efficacy of ophthalmic lenses to target tissues in the back of the eye is suggested. In fact, future in vivo studies could evidence the suitability of previously developed devices for drug delivery to the retina. The incorporation of innovative drug delivery methods, such as drug-eluting nanoparticles, into ophthalmic lenses could also constitute an interesting future approach for a minimally invasive sustained drug delivery to the back of the eye.
3. Concluding Remarks and Future Perspectives
Surprisingly, the impressive efforts that were made by researchers around the world on the optimization of drug-eluting ophthalmic lenses did not yet result in the commercialization of these devices. As recently described by Lanier et al. [215], several reasons may be pointed out for the apparent lack of interest of the pharmaceutical industry to invest in those systems. One common limitation of all innovative methods referred above is the need for the optimization of each specific system drug/lens. In fact, it is not possible to extrapolate the results that were obtained with a so-called model drug, because the drug release behavior and eventual alterations of the physical properties of the lenses after loading depend on the specific interactions between the drug and the polymeric matrix. For example, when using the molecular imprinting technique, the optimization of each combination polymer/monomer/crosslinker/template must be done. The control of the drug release by coating the lens strongly depends on the characteristics of the drug molecule: a very efficient coating for one drug may be inefficient for other similar drugs. Besides choosing the ideal combinations of components, it is also necessary to determine the adequate amounts of drug loaded: it has to be sufficient to ensure clinically relevant therapeutic release, but it cannot affect key aspects of the lens, namely transparency, Young modulus, ionic and oxygen permeability, wettability, and water content. In the case of the addition of other agents capable of sustaining the drug release, such as the functional monomers in the imprinting technique, vitamin E or surfactants, the preservation of the lens properties has to be ensured. The optimization of the combination of materials and loading conditions may be still more demanding when multiple drugs are needed for the treatment. Some of the methods of preparation of drug-loaded lenses involve complex manufacturing, namely the incorporation of nanoparticles or drug reservoirs, the LbL coating, supercritical impregnation, which may be a drawback for scaling up production. Important issues, like the lack of drug stability during processing, the prevention of burst release, protein adherence, sterilization, and storage conditions, have been addressed, but need more intense investigation. In general, several innovative drug-eluting lenses have been submitted to in vivo studies, which demonstrated promising results; however, further studies involving the assessment of long-term safety are missing as well as extended clinical tests. Fortunately, there are solutions for many of the technical problems described above. The minimization of burst release and protein adherence may be achieved with adequate coatings and/or optimized formulations. The storage conditions may involve immersion of the lenses in drug solutions, due to the equilibrium loading method, or keeping the lenses in dry state. Sterilization methods, which have no detrimental effects, have been proposed. Thus, the last step before commercialization needs a positive evaluation of the costs and benefits. The benefits seem to be huge, when considering that drug-eluting CLs may decrease the risks that are associated with their usual wear (keratitis, corneal erosion, dry eye syndrome, conjunctivitis) and avoid the frequent administration of eye drops for the treatment of ocular diseases, while drug-loaded IOLs may substitute the invasive intracameral and intravitreal injections. Thus, the commercialization of drug-loaded ophthalmic lenses is probable in the near future, and ongoing research on this subject continues to be relevant.
This entry is adapted from the peer-reviewed paper 10.3390/pharmaceutics13010036
References
- Shatz, W.; Aaronson, J.; Yohe, S.; Kelley, R.F.; Kalia, Y.N. Strategies for modifying drug residence time and ocular bioavailability to decrease treatment frequency for back of the eye diseases. Expert Opin. Drug Deliv. 2019, 16, 43–57, doi:10.1080/17425247.2019.1553953.
- Khalaj, M.; Barikani, A.; Ghasemi, H. Eye disorders in old people. Glob. J. Health Sci. 2013, 5, 79–86, doi:10.5539/gjhs.v5n1p79.
- Balyeat, R.M.; Bowen, R. Allergic conjunctivitis. Community Eye Health J. 2017, 29, S7–S10, doi:10.1097/00007611-193511000-00009.
- Priyavarshini, R.; Amit, B.P. Recent development in the artificial treatment for dry eye disease. Int. J. Res. Pharm. Sci. 2020, 11, 1902–1907.
- Song, B.J.; Aiello, L.P.; Pasquale, L.R. Presence and Risk Factors for Glaucoma in Patients with Diabetes. Curr. Diab. Rep. 2016, 16, 124, doi:10.1007/s11892-016-0815-6.
- Williams, R.; Airey, M.; Baxter, H.; Forrester, J.; Kennedy-Martin, T.; Girach, A. Epidemiology of diabetic retinopathy and macular oedema: A systematic review. Eye 2004, 18, 963–983, doi:10.1038/sj.eye.6701476.
- Shah, T.J.; Conway, M.D.; Peyman, G.A. Intracameral dexamethasone injection in the treatment of cataract surgery induced inflammation: Design, development, and place in therapy. Clin. Ophthalmol. 2018, 12, 2223–2235, doi:10.2147/OPTH.S165722.
- Tyson, S.L.; Bailey, R.; Roman, J.S.; Zhan, T.; Hark, L.A.; Haller, J.A. Clinical outcomes after injection of a compounded pharmaceutical for prophylaxis after cataract surgery: A large-scale review. Curr. Opin. Ophthalmol. 2017, 28, 73–80, doi:10.1097/ICU.0000000000000329.
- Hermann, M.M.; Stündag, C.Ü.; Diestelhorst, M. Electronic compliance monitoring of topical treatment after ophthalmic surgery. Int. Ophthalmol. 2010, 30, 385–390, doi:10.1007/s10792-010-9362-3.
- Xu, J.; Xue, Y.; Hu, G.; Lin, T.; Gou, J.; Yin, T.; He, H.; Zhang, Y.; Tang, X. A comprehensive review on contact lens for ophthalmic drug delivery. J. Control. Release 2018, 281, 97–118, doi:10.1016/j.jconrel.2018.05.020.
- An, J.A.; Kasner, O.; Samek, D.A.; Lévesque, V. Evaluation of eyedrop administration by inexperienced patients after cataract surgery. J. Cataract Refract. Surg. 2014, 40, 1857–1861, doi:10.1016/j.jcrs.2014.02.037.
- Cooper, R.C.; Yang, H. Hydrogel-based ocular drug delivery systems: Emerging fabrication strategies, applications, and bench-to-bedside manufacturing considerations. J. Control. Release 2019, 306, 29–39, doi:10.1016/j.jconrel.2019.05.034.
- Jünemann, A.G.M.; Chorągiewicz, T.; Ozimek, M.; Grieb, P.; Rejdak, R. Drug bioavailability from topically applied ocular drops. Does drop size matter? Ophthalmol. J. 2016, 1, 29–35, doi:10.5603/oj.2016.0005.
- Ramsay, E.; del Amo, E.M.; Toropainen, E.; Tengvall-Unadike, U.; Ranta, V.P.; Urtti, A.; Ruponen, M. Corneal and conjunctival drug permeability: Systematic comparison and pharmacokinetic impact in the eye. Eur.. J. Pharm. Sci. 2018, 119, 83–89, doi:10.1016/j.ejps.2018.03.034.
- Kim, Y.C.; Chiang, B.; Wu, X.; Prausnitz, M.R. Ocular delivery of macromolecules. J. Control. Release 2014, 190, 172–181, doi:10.1016/j.jconrel.2014.06.043.
- Nagai, N.; Nakazawa, Y.; Ito, Y.; Kanai, K.; Okamoto, N.; Shimomura, Y. A nanoparticle-based ophthalmic formulation of dexamethasone enhances corneal permeability of the drug and prolongs its corneal residence time. Biol. Pharm. Bull. 2017, 40, 1055–1062, doi:10.1248/bpb.b17-00137.
- Chang, D.T.W.; Herceg, M.C.; Bilonick, R.A.; Camejo, L.; Schuman, J.S.; Noecker, R.J. Intracameral dexamethasone reduces inflammation on the first postoperative day after cataract surgery in eyes with and without glaucoma. Clin. Ophthalmol. 2009, 3, 345–355, doi:10.2147/opth.s5730.
- Kiernan, D.F.; Hariprasad, S.M. Controversies in the management of Irvine-Gass syndrome. Ophthalmic Surg. Lasers Imaging 2013, 44, 522–527, doi:10.3928/23258160-20131105-01.
- Grzybowski, A.; Kanclerz, P. The Role of Steroids and NSAIDs in Prevention and Treatment of Postsurgical Cystoid Macular Edema. Curr. Pharm. Des. 2019, 24, 4896–4902, doi:10.2174/1381612825666190206104524.
- Nayak, K.; Misra, M. A review on recent drug delivery systems for posterior segment of eye. Biomed. Pharmacother. 2018, 107, 1564–1582, doi:10.1016/j.biopha.2018.08.138.
- Hui, A. Contact lenses for ophthalmic drug delivery. Clin. Exp. Optom. 2017, 100, 494–512, doi:10.1111/cxo.12592.
- Maulvi, F.A.; Soni, T.G.; Shah, D.O. A review on therapeutic contact lenses for ocular drug delivery. Drug Deliv. 2016, 23, 3017–3026, doi:10.3109/10717544.2016.1138342.
- Garty, S.; Shirakawa, R.; Warsen, A.; Anderson, E.M.; Noble, M.L.; Bryers, J.D.; Ratner, B.D.; Shen, T.T. Sustained antibiotic release from an intraocular lens-hydrogel assembly for cataract surgery. Investig. Ophthalmol. Vis. Sci. 2011, 52, 6109–6116, doi:10.1167/iovs.10-6071.
- Topete, A.; Serro, A.P.; Saramago, B. Dual drug delivery from intraocular lens material for prophylaxis of endophthalmitis in cataract surgery. Int. J. Pharm. 2019, 558, 43–52, doi:10.1016/j.ijpharm.2018.12.028.
- Li, X.; Zhao, Y.; Wang, K.; Wang, L.; Yang, X.; Zhu, S. Cyclodextrin-containing hydrogels as an intraocular lens for sustained drug release. PLoS ONE 2017, 12, e0189778, doi:10.1371/journal.pone.0189778.
- Bouledjouidja, A.; Masmoudi, Y.; Sergent, M.; Trivedi, V.; Meniai, A.; Badens, E. Drug loading of foldable commercial intraocular lenses using supercritical impregnation. Int. J. Pharm. 2016, 500, 85–99, doi:10.1016/j.ijpharm.2016.01.016.
- Eperon, S.; Bossy-Nobs, L.; Petropoulos, I.K.; Gurny, R.; Guex-Crosier, Y. A biodegradable drug delivery system for the treatment of postoperative inflammation. Int. J. Pharm. 2008, 352, 240–247, doi:10.1016/j.ijpharm.2007.10.054.
- Fonn, D.; Sweeney, D. The Benefits of Silicone Hydrogel Daily Disposable Lenses: A look at why silicone hydrogel daily disposable lenses are our preferred option for daily wear. Contact Lens Spectr. 2015, 30, 42–45.
- Moreddu, R.; Vigolo, D.; Yetisen, A.K. Contact Lens Technology: From Fundamentals to Applications. Adv. Healthc. Mater. 2019, 8, 1900368, doi:0.1002/adhm.201900368.
- Mohammadi, S.; Jones, L.; Gorbet, M. Extended latanoprost release from commercial contact lenses: In vitro studies using corneal models. PLoS ONE 2014, 9, e106653, doi:10.1371/journal.pone.0106653.
- Schultz, C.; Breaux, J.; Schentag, J.; Morck, D. Drug delivery to the posterior segment of the eye through hydrogel contact lenses. Clin. Exp. Optom. 2011, 94, 212–218, doi:10.1111/j.1444-0938.2010.00553.x.
- Davis, J.L.; Yi, N.Y.; Salmon, J.H.; Charlton, A.N.; Colitz, C.M.H.; Gilger, B.C. Sustained-release celecoxib from incubated acrylic intraocular lenses suppresses lens epithelial cell growth in an ex vivo model of posterior capsule opacity. J. Ocul. Pharmacol. Ther. 2012, 28, 359–368, doi:10.1089/jop.2011.0196.
- Wertheimer, C.; Brandlhuber, U.; Kook, D.; Mayer, W.J.; Laubichler, P.; Wolf, A.; Kampik, A.; Eibl-Lindner, K. Erufosine, a phosphoinositide-3-kinase inhibitor, to mitigate posterior capsule opacification in the human capsular bag model. J. Cataract Refract. Surg. 2015, 41, 1484–1489, doi:10.1016/j.jcrs.2015.02.034.
- Wertheimer, C.; Kueres, A.; Siedlecki, J.; Braun, C.; Kassumeh, S.; Wolf, A.; Mayer, W.; Priglinger, C.; Priglinger, S.; Eibl-Lindner, K. The intraocular lens as a drug delivery device for an epidermal growth factor–Receptor inhibitor for prophylaxis of posterior capsule opacification. Acta Ophthalmol. 2018, 96, e874–e882, doi:10.1111/aos.13759.
- Kassumeh, S.; Kueres, A.; Hillenmayer, A.; von Studnitz, A.; Elhardt, C.; Ohlmann, A.; Priglinger, S.G.; Wertheimer, C.M. Development of a drug-eluting intraocular lens to deliver epidermal growth factor receptor inhibitor gefitinib for posterior capsule opacification prophylaxis. Eur. J. Ophthalmol. 2019, doi:10.1177/1120672119891042.
- Wertheimer, C.; Kassumeh, S.; Piravej, N.P.; Nilmayer, O.; Braun, C.; Priglinger, C.; Luft, N.; Wolf, A.; Mayer, W.J.; Priglinger, S.G.; et al. The intraocular lens as a drug delivery device: In vitro screening of pharmacologic substances for the prophylaxis of posterior capsule opacification. Investig. Ophthalmol. Vis. Sci. 2017, 58, 6408–6418, doi:10.1167/iovs.17-22555.
- Atabek Yigit, E.; Ercal, N. Release of N-acetylcysteine and N-acetylcysteine amide from contact lenses. Eye Contact Lens 2013, 39, 335–340, doi:10.1097/ICL.0b013e3182a2f8bc.
- Horne, R.R.; Rich, J.T.; Bradley, M.W.; Pitt, W.G. Latanoprost uptake and release from commercial contact lenses. J. Biomater. Sci. Polym. Ed. 2020, 31, 1–19, doi:10.1080/09205063.2019.1669126.
- Lipnitzki, I.; Bronshtein, R.; Ben Eliahu, S.; Marcovich, A.L.; Kleinmann, G. Hydrophilic acrylic intraocular lens as a drug delivery system: Influence of the presoaking time and comparison to intracameral injection. J. Ocul. Pharmacol. Ther. 2013, 29, 414–418, doi:10.1089/jop.2012.0062.
- Topete, A.; Oliveira, A.S.; Fernandes, A.; Nunes, T.G.; Serro, A.P.; Saramago, B. Improving sustained drug delivery from ophthalmic lens materials through the control of temperature and time of loading. Eur. J. Pharm. Sci. 2018, 117, 107–117, doi:10.1016/j.ejps.2018.02.017.
- Filipe, H.P.; Bozukova, D.; Pimenta, A.; Vieira, A.P.; Oliveira, A.S.; Galante, R.; Topete, A.; Masson, M.; Alves, P.; Coimbra, P.; et al. Moxifloxacin-loaded acrylic intraocular lenses: In vitro and in vivo performance. J. Cataract Refract. Surg. 2019, 45, 1808–1817, doi:10.1016/j.jcrs.2019.07.016.
- Pimenta, A.F.R.; Serro, A.P.; Colaço, R.; Chauhan, A. Optimization of intraocular lens hydrogels for dual drug release: Experimentation and modelling. Eur. J. Pharm. Biopharm. 2019, 141, 51–57, doi:10.1016/j.ejpb.2019.05.016.
- Schultz, C.L.; Morck, D.W. Contact lenses as a drug delivery device for epidermal growth factor in the treatment of ocular wounds. Clin. Exp. Optom. 2010, 93, 61–65, doi:10.1111/j.1444-0938.2010.00459.x.
- Pastori, V.; Tavazzi, S.; Lecchi, M. Lactoferrin-loaded contact lenses: Eye protection against oxidative stress. Cornea 2015, 34, 693–697, doi:10.1097/ICO.0000000000000435.
- Pastori, V.; Tavazzi, S.; Lecchi, M. Lactoferrin-loaded contact lenses counteract cytotoxicity caused in vitro by keratoconic tears. Contact Lens Anterior Eye 2019, 42, 253–257, doi:10.1016/j.clae.2018.12.004.
- González-Chomón, C.; Concheiro, A.; Alvarez-Lorenzo, C. Drug-eluting intraocular lenses. Materials (Basel) 2011, 4, 1927–1940, doi:10.3390/ma4111927.
- Liu, Y.C.; Wong, T.T.; Mehta, J.S. Intraocular lens as a drug delivery reservoir. Curr. Opin. Ophthalmol. 2013, 24, 53–59, doi:10.1097/ICU.0b013e32835a93fc.
- Madni, A.; Rahem, M.A.; Tahir, N.; Sarfraz, M.; Jabar, A.; Rehman, M.; Kashif, P.M.; Badshah, S.F.; Khan, K.U.; Santos, H.A. Non-invasive strategies for targeting the posterior segment of eye. Int. J. Pharm. 2017, 530, 326–345, doi:10.1016/j.ijpharm.2017.07.065.
- Dixon, P.; Chauhan, A. Effect of the surface layer on drug release from delefilcon-A (Dailies Total1®) contact lenses. Int. J. Pharm. 2017, 529, 89–101, doi:10.1016/j.ijpharm.2017.06.036.
- Xu, J.; Li, X.; Sun, F. Cyclodextrin-containing hydrogels for contact lenses as a platform for drug incorporation and release. Acta Biomater. 2010, 6, 486–493, doi:10.1016/j.actbio.2009.07.021.
- García-Fernández, M.J.; Tabary, N.; Martel, B.; Cazaux, F.; Oliva, A.; Taboada, P.; Concheiro, A.; Alvarez-Lorenzo, C. Poly-(cyclo)dextrins as ethoxzolamide carriers in ophthalmic solutions and in contact lenses. Carbohydr. Polym. 2013, 98, 1343–1352, doi:10.1016/j.carbpol.2013.08.003.
- Hsu, K.H.; Carbia, B.E.; Plummer, C.; Chauhan, A. Dual drug delivery from vitamin e loaded contact lenses for glaucoma therapy. Eur. J. Pharm. Biopharm. 2015, 94, 312–321, doi:10.1016/j.ejpb.2015.06.001.
- Sekar, P.; Chauhan, A. Effect of vitamin-E integration on delivery of prostaglandin analogs from therapeutic lenses. J. Colloid Interface Sci. 2019, 539, 457–467, doi:10.1016/j.jcis.2018.12.036.
- Lee, D.; Cho, S.; Park, H.S.; Kwon, I. Ocular drug delivery through pHEMA-Hydrogel contact lenses Co-loaded with lipophilic vitamins. Sci. Rep. 2016, 6, 34194, doi:10.1038/srep34194.
- Peng, C.C.; Ben-Shlomo, A.; MacKay, E.O.; Plummer, C.E.; Chauhan, A. Drug delivery by contact lens in spontaneously glaucomatous dogs. Curr. Eye Res. 2012, 37, 204–211, doi:10.3109/02713683.2011.630154.
- Peng, C.C.; Burke, M.T.; Carbia, B.E.; Plummer, C.; Chauhan, A. Extended drug delivery by contact lenses for glaucoma therapy. J. Control. Release 2012, 162, 152–158, doi:10.1016/j.jconrel.2012.06.017.
- Kim, J.; Peng, C.C.; Chauhan, A. Extended release of dexamethasone from silicone-hydrogel contact lenses containing vitamin E. J. Control. Release 2010, 148, 110–116, doi:10.1016/j.jconrel.2010.07.119.
- Torres-Luna, C.; Koolivand, A.; Fan, X.; Agrawal, N.R.; Hu, N.; Zhu, Y.; Domszy, R.; Briber, R.M.; Wang, N.S.; Yang, A. Formation of drug-participating catanionic aggregates for extended delivery of non-steroidal anti-inflammatory drugs from contact lenses. Biomolecules 2019, 9, 593, doi:10.3390/biom9100593.
- Andrade-Vivero, P.; Fernandez-Gabriel, E.; Alvarez-Lorenzo, C.; Concheiro, A. Improving the Loading and Release of NSAIDs frompHEMA Hydrogels by Copolymerization withFunctionalized Monomers. J. Pharm. Sci. 2007, 96, 802–813.
- González-Chomón, C.; Braga, M.E.M.; De Sousa, H.C.; Concheiro, A.; Alvarez-Lorenzo, C. Antifouling foldable acrylic IOLs loaded with norfloxacin by aqueous soaking and by supercritical carbon dioxide technology. Eur. J. Pharm. Biopharm. 2012, 82, 383–391, doi:10.1016/j.ejpb.2012.07.007.
- Xu, J.; Li, X.; Sun, F. Preparation and evaluation of a contact lens vehicle for puerarin delivery. J. Biomater. Sci. Polym. Ed. 2010, 21, 271–288, doi:10.1163/156856209X415774.
- Kakisu, K.; Matsunaga, T.; Kobayakawa, S.; Sato, T.; Tochikubo, T. Development and efficacy of a drug-releasing soft contact lens. Investig. Ophthalmol. Vis. Sci. 2013, 54, 2551–2561, doi:10.1167/iovs.12-10614.
- Soluri, A.; Hui, A.; Jones, L. Delivery of ketotifen fumarate by commercial contact lens materials. Optom. Vis. Sci. 2012, 89, 1140–1149, doi:10.1097/OPX.0b013e3182639dc8.
- Kassumeh, S.A.; Wertheimer, C.M.; von Studnitz, A.; Hillenmayer, A.; Priglinger, C.; Wolf, A.; Mayer, W.J.; Teupser, D.; Holdt, L.M.; Priglinger, S.G.; et al. Poly(lactic-co-glycolic) Acid as a Slow-Release Drug-Carrying Matrix for Methotrexate Coated onto Intraocular Lenses to Conquer Posterior Capsule Opacification. Curr. Eye Res. 2018, 43, 702–708, doi:10.1080/02713683.2018.1437455.
- Manju, S.; Kunnatheeri, S. Layer-by-Layer modification of poly (methyl methacrylate) intra ocular lens: Drug delivery applications. Pharm. Dev. Technol. 2010, 15, 379–385, doi:10.3109/10837450903262025.
- Pimenta, A.F.R.; Vieira, A.P.; Colaço, R.; Saramago, B.; Gil, M.H.; Coimbra, P.; Alves, P.; Bozukova, D.; Correia, T.R.; Correia, I.J.; et al. Controlled release of moxifloxacin from intraocular lenses modified by Ar plasma-assisted grafting with AMPS or SBMA: An in vitro study. Colloids Surf. B Biointerfaces 2017, 156, 95–103, doi:10.1016/j.colsurfb.2017.04.060.
- Mehta, P.; Al-Kinani, A.A.; Haj-Ahmad, R.; Arshad, M.S.; Chang, M.W.; Alany, R.G.; Ahmad, Z. Electrically atomised formulations of timolol maleate for direct and on-demand ocular lens coatings. Eur. J. Pharm. Biopharm. 2017, 119, 170–184, doi:10.1016/j.ejpb.2017.06.016.
- Mehta, P.; Al-Kinani, A.A.; Arshad, M.S.; Chang, M.W.; Alany, R.G.; Ahmad, Z. Development and characterisation of electrospun timolol maleate-loaded polymeric contact lens coatings containing various permeation enhancers. Int. J. Pharm. 2017, 532, 408–420, doi:10.1016/j.ijpharm.2017.09.029.
- Mehta, P.; Al-Kinani, A.A.; Arshad, M.S.; Singh, N.; van der Merwe, S.M.; Chang, M.W.; Alany, R.G.; Ahmad, Z. Engineering and Development of Chitosan-Based Nanocoatings for Ocular Contact Lenses. J. Pharm. Sci. 2019, 108, 1540–1551, doi:10.1016/j.xphs.2018.11.036.
- Silva, D.; Pinto, L.F.V.; Bozukova, D.; Santos, L.F.; Serro, A.P.; Saramago, B. Chitosan/alginate based multilayers to control drug release from ophthalmic lens. Colloids Surf. B Biointerfaces 2016, 147, 81–89, doi:10.1016/j.colsurfb.2016.07.047.
- Vieira, A.P.; Pimenta, A.F.R.; Silva, D.; Gil, M.H.; Alves, P.; Coimbra, P.; Mata, J.L.G.C.; Bozukova, D.; Correia, T.R.; Correia, I.J.; et al. Surface modification of an intraocular lens material by plasma-assisted grafting with 2-hydroxyethyl methacrylate (HEMA), for controlled release of moxifloxacin. Eur. J. Pharm. Biopharm. 2017, 120, 52–62, doi:10.1016/j.ejpb.2017.08.006.
- Liu, H.; Zhang, Y.; Ma, H.; Zhang, C.; Fu, S. Comparison of posterior capsule opacification in rabbit eyes receiving different administrations of rapamycin. Graefe’s Arch. Clin. Exp. Ophthalmol. 2014, 252, 1111–1118, doi:10.1007/s00417-014-2656-9.
- Lin, L.; Lin, Q.; Li, J.; Han, Y.; Chang, P.; Lu, F.; Zhao, Y.E. ROCK inhibitor modified intraocular lens as an approach for inhibiting the proliferation and migration of lens epithelial cells and posterior capsule opacification. Biomater. Sci. 2019, 7, 4208–4217, doi:10.1039/c9bm00787c.
- Han, Y.; Tang, J.; Xia, J.; Wang, R.; Qin, C.; Liu, S.; Zhao, X.; Chen, H.; Lin, Q. Anti-adhesive and antiproliferative synergistic surface modification of intraocular lens for reduced posterior capsular opacification. Int. J. Nanomed. 2019, 14, 9047–9061, doi:10.2147/IJN.S215802.
- Silva, D.; Sousa, H.C.D.; Gil, M.H.; Santos, L.F.; Moutinho, G.M.; Serro, A.P.; Saramago, B. Antibacterial layer-by-layer coatings to control drug release from soft contact lenses material. Int. J. Pharm. 2018, 553, 186–200, doi:10.1016/j.ijpharm.2018.10.041.
- Paradiso, P.; Colaço, R.; Mata, J.L.G.; Krastev, R.; Saramago, B.; Serro, A.P. Drug release from liposome coated hydrogels for soft contact lenses: The blinking and temperature effect. J. Biomed. Mater. Res. Part. B Appl. Biomater. 2017, 105, 1799–1807, doi:10.1002/jbm.b.33715.
- Zhang, Z.; Huang, W.; Lei, M.; He, Y.; Yan, M.; Zhang, X.; Zhao, C. Laser-triggered intraocular implant to induce photodynamic therapy for posterior capsule opacification prevention. Int. J. Pharm. 2016, 498, 1–11, doi:10.1016/j.ijpharm.2015.10.006.
- Alvarez-Lorenzo, C.; Concheiro, A. Molecularly imprinted materials as advanced excipients for drug delivery systems. Biotechnol. Annu. Rev. 2006, 12, 225–268, doi:10.1016/S1387-2656(06)12007-4.
- Alvarez-Lorenzo, C.; Yañez, F.; Concheiro, A. Ocular drug delivery from molecularly-imprinted contact lenses. J. Drug Deliv. Sci. Technol. 2010, 20, 237–248, doi:10.1016/S1773-2247(10)50041-8.
- Alvarez-Lorenzo, C.; Anguiano-Igea, S.; Varela-García, A.; Vivero-Lopez, M.; Concheiro, A. Bioinspired hydrogels for drug-eluting contact lenses. Acta Biomater. 2019, 84, 49–62, doi:10.1016/j.actbio.2018.11.020.
- White, C.J.; McBride, M.K.; Pate, K.M.; Tieppo, A.; Byrne, M.E. Extended release of high molecular weight hydroxypropyl methylcellulose from molecularly imprinted, extended wear silicone hydrogel contact lenses. Biomaterials 2011, 32, 5698–5705, doi:10.1016/j.biomaterials.2011.04.044.
- Anirudhan, T.S.; Nair, A.S.; Parvathy, J. Extended wear therapeutic contact lens fabricated from timolol imprinted carboxymethyl chitosan-g-hydroxy ethyl methacrylate-g-poly acrylamide as a onetime medication for glaucoma. Eur. J. Pharm. Biopharm. 2016, 109, 61–71, doi:10.1016/j.ejpb.2016.09.010.
- Yan, F.; Liu, Y.; Han, S.; Zhao, Q.; Liu, N. Bimatoprost Imprinted Silicone Contact Lens to Treat Glaucoma. AAPS Pharmscitech 2020, 21, 63, doi:10.1208/s12249-020-1622-6.
- Ribeiro, A.; Veiga, F.; Santos, D.; Torres-Labandeira, J.J.; Concheiro, A.; Alvarez-Lorenzo, C. Bioinspired Imprinted PHEMA-Hydrogels for ocular delivery of carbonic anhydrase inhibitor drugs. Biomacromolecules 2011, 12, 701–709, doi:10.1021/bm101562v.
- Ribeiro, A.; Veiga, F.; Santos, D.; Torres-Labandeira, J.J.; Concheiro, A.; Alvarez-Lorenzo, C. Receptor-based biomimetic NVP/DMA contact lenses for loading/eluting carbonic anhydrase inhibitors. J. Memb. Sci. 2011, 383, 60–69, doi:10.1016/j.memsci.2011.08.030.
- Deng, J.; Chen, S.; Chen, J.; Ding, H.; Deng, D.; Xie, Z. Self-Reporting Colorimetric Analysis of Drug Release by Molecular Imprinted Structural Color Contact Lens. ACS Appl. Mater. Interfaces 2018, 10, 34611–34617, doi:10.1021/acsami.8b11655.
- Alvarez-Rivera, F.; Serro, A.P.; Silva, D.; Concheiro, A.; Alvarez-Lorenzo, C. Hydrogels for diabetic eyes: Naltrexone loading, release profiles and cornea penetration. Mater. Sci. Eng. C 2019, 105, 10092, doi:10.1016/j.msec.2019.110092.
- Alvarez-Rivera, F.; Concheiro, A.; Alvarez-Lorenzo, C. Epalrestat-loaded silicone hydrogels as contact lenses to address diabetic-eye complications. Eur. J. Pharm. Biopharm. 2018, 122, 126–136, doi:10.1016/j.ejpb.2017.10.016.
- White, C.J.; Tieppo, A.; Byrne, M.E. Controlled drug release from contact lenses: A comprehensive review from 1965-present. J. Drug Deliv. Sci. Technol. 2011, 21, 369–384, doi:10.1016/S1773-2247(11)50062-0.
- Zaidi, S.A. Latest trends in molecular imprinted polymer based drug delivery systems. RSC Adv. 2016, 6, 88807–88819, doi:10.1039/c6ra18911c.
- Hiratani, H.; Mizutani, Y.; Alvarez-Lorenzo, C. Controlling drug release from imprinted hydrogels by modifying the characteristics of the imprinted cavities. Macromol. Biosci. 2005, 5, 728–733, doi:10.1002/mabi.200500065.
- Bouledjouidja, A.; Masmoudi, Y.; Sergent, M.; Badens, E. Effect of operational conditions on the supercritical carbon dioxide impregnation of anti-inflammatory and antibiotic drugs in rigid commercial intraocular lenses. J. Supercrit. Fluids 2017, 130, 63–75, doi:10.1016/j.supflu.2017.07.015.
- Costa, V.P.; Braga, M.E.M.; Guerra, J.P.; Duarte, A.R.C.; Duarte, C.M.M.; Leite, E.O.B.; Gil, M.H.; de Sousa, H.C. Development of therapeutic contact lenses using a supercritical solvent impregnation method. J. Supercrit. Fluids 2010, 52, 306–316, doi:10.1016/j.supflu.2010.02.001.
- Costa, V.P.; Braga, M.E.M.; Duarte, C.M.M.; Alvarez-Lorenzo, C.; Concheiro, A.; Gil, M.H.; de Sousa, H.C. Anti-glaucoma drug-loaded contact lenses prepared using supercritical solvent impregnation. J. Supercrit. Fluids 2010, 53, 165–173, doi:10.1016/j.supflu.2010.02.007.
- Bouledjouidja, A.; Masmoudi, Y.; Li, Y.; He, W.; Badens, E. Supercritical impregnation and optical characterization of loaded foldable intraocular lenses using supercritical fluids. J. Cataract Refract. Surg. 2017, 43, 1343–1349, doi:10.1016/j.jcrs.2017.07.033.
- Ongkasin, K.; Masmoudi, Y.; Tassaing, T.; Le-Bourdon, G.; Badens, E. Supercritical loading of gatifloxacin into hydrophobic foldable intraocular lenses—Process control and optimization by following in situ CO2 sorption and polymer swelling. Int. J. Pharm. 2020, 581, 119247, doi:10.1016/j.ijpharm.2020.119247.
- Ongkasin, K.; Masmoudi, Y.; Wertheimer, C.M.; Hillenmayer, A.; Eibl-Lindner, K.H.; Badens, E. Supercritical fluid technology for the development of innovative ophthalmic medical devices: Drug loaded intraocular lenses to mitigate posterior capsule opacification. Eur. J. Pharm. Biopharm. 2020, 149, 248–256, doi:10.1016/j.ejpb.2020.02.011.
- Masmoudi, Y.; Ben Azzouk, L.; Forzano, O.; Andre, J.M.; Badens, E. Supercritical impregnation of intraocular lenses. J. Supercrit. Fluids 2011, 60, 98–105, doi:10.1016/j.supflu.2011.08.014.
- Yokozaki, Y.; Sakebe, J.; Ng, B.; Shimoyama, Y. Effect of temperature, pressure and depressurization rate on release profile of salicylic acid from contact lenses prepared by supercritical carbon dioxide impregnation. Chem. Eng. Res. Des. 2015, 100, 89–94.
- Yañez, F.; Martikainen, L.; Braga, M.E.M.; Alvarez-Lorenzo, C.; Concheiro, A.; Duarte, C.M.M.; Gil, M.H.; De Sousa, H.C. Supercritical fluid-assisted preparation of imprinted contact lenses for drug delivery. Acta Biomater. 2011, 7, 1019–1030, doi:10.1016/j.actbio.2010.10.003.
- Jung, H.J.; Abou-Jaoude, M.; Carbia, B.E.; Plummer, C.; Chauhan, A. Glaucoma therapy by extended release of timolol from nanoparticle loaded silicone-hydrogel contact lenses. J. Control. Release 2013, 165, 82–89, doi:10.1016/j.jconrel.2012.10.010.
- Jung, H.J.; Chauhan, A. Temperature sensitive contact lenses for triggered ophthalmic drug delivery. Biomaterials 2012, 33, 2289–2300, doi:10.1016/j.biomaterials.2011.10.076.
- Kim, H.J.; Zhang, K.; Moore, L.; Ho, D. Diamond nanogel-embedded contact lenses mediate lysozyme-dependent therapeutic release. ACS Nano 2014, 8, 2998–3005, doi:10.1021/nn5002968.
- Lee, S.H.; Kim, H.J.; Kim, D.H.; Chang, W.S.; Vales, T.P.; Kim, J.W.; Kim, K.H.; Kim, J.K. Thermo-sensitive nanogel-laden bicontinuous microemulsion drug-eluting contact lenses. J. Biomed. Mater. Res. Part. B Appl. Biomater. 2019, 107, 1159–1169, doi:10.1002/jbm.b.34209.
- Han, Y.; Xu, X.; Wang, Y.; Liu, S.; Zhao, X.; Chen, H.; Lin, Q. Drug Eluting Intraocular Lens Surface Modification for PCO Prevention. Colloids Interface Sci. Commun. 2018, 24, 40–44, doi:10.1016/j.colcom.2018.03.007.
- Huang, X.; Wang, Y.; Cai, J.P.; Ma, X.Y.; Li, Y.; Cheng, J.W.; Wei, R.L. Sustained release of 5-fluorouracil from chitosan nanoparticles surface modified intra ocular lens to prevent posterior capsule opacification: An in vitro and in vivo study. J. Ocul. Pharmacol Ther. 2013, 29, 208–215, doi:10.1089/jop.2012.0184.
- Maulvi, F.A.; Patil, R.J.; Desai, A.R.; Shukla, M.R.; Vaidya, R.J.; Ranch, K.M.; Vyas, B.A.; Shah, S.A.; Shah, D.O. Effect of gold nanoparticles on timolol uptake and its release kinetics from contact lenses: In vitro and in vivo evaluation. Acta Biomater. 2019, 86, 350–362, doi:10.1016/j.actbio.2019.01.004.
- Sharma, M.; Bhowmick, R.; Gappa-Fahlenkamp, H. Drug-Loaded Nanoparticles Embedded in a Biomembrane Provide a Dual-Release Mechanism for Drug Delivery to the Eye. J. Ocul. Pharmacol Ther. 2016, 32, 565–573, doi:10.1089/jop.2016.0050.
- De Jong, W.H.; Borm, P.J.A. Drug delivery and nanoparticles: Applications and hazards. Int. J. Nanomed. 2008, 3, 133–149, doi:10.2147/ijn.s596.
- Rizvi, S.A.A.; Saleh, A.M. Applications of nanoparticle systems in drug delivery technology. Saudi Pharm. J. 2018, 26, 64–70, doi:10.1016/j.jsps.2017.10.012.
- Dubald, M.; Bourgeois, S.; Andrieu, V.; Fessi, H. Ophthalmic drug delivery systems for antibiotherapy- A review. Pharmaceutics 2018, 10, 10, doi:10.3390/pharmaceutics10010010.
- Mun, J.; Mok, J.W.; Jeong, S.; Cho, S.; Joo, C.K.; Hahn, S.K. Drug-eluting contact lens containing cyclosporine-loaded cholesterol-hyaluronate micelles for dry eye syndrome. RSC Adv. 2019, 9, 16578–16585, doi:10.1039/c9ra02858g.
- Xu, J.; Ge, Y.; Bu, R.; Zhang, A.; Feng, S.; Wang, J.; Gou, J.; Yin, T.; He, H.; Zhang, Y.; et al. Co-delivery of latanoprost and timolol from micelles-laden contact lenses for the treatment of glaucoma. J. Control. Release 2019, 305, 18–28, doi:10.1016/j.jconrel.2019.05.025.
- Maulvi, F.A.; Desai, A.R.; Choksi, H.H.; Patil, R.J.; Ranch, K.M.; Vyas, B.A.; Shah, D.O. Effect of surfactant chain length on drug release kinetics from microemulsion-laden contact lenses. Int. J. Pharm. 2017, 524, 193–204, doi:10.1016/j.ijpharm.2017.03.083.
- Xu, W.; Jiao, W.; Li, S.; Tao, X.; Mu, G. Bimatoprost loaded microemulsion laden contact lens to treat glaucoma. J. Drug Deliv. Sci. Technol. 2019, 54, 101330, doi:10.1016/j.jddst.2019.101330.
- Wei, N.; Dang, H.; Huang, C.; Sheng, Y. Timolol loaded microemulsion laden silicone contact lens to manage glaucoma: In vitro and in vivo studies. J. Dispers. Sci. Technol. 2020, 1–9, doi:10.1080/01932691.2019.1710183.
- Siqueira, R.C.; Ribeiro Filho, E.; Fialho, S.L.; Lucena, L.R.; Maia Filho, A.; Haddad, A.; Jorge, R.; Scott, I.U.; Da Silva Cunha, A. . Pharmacokinetic and toxicity investigations of a new intraocular lens with a dexamethasone drug delivery system: A pilot study. Ophthalmologica 2006, 220, 338–342, doi:10.1159/000094626.
- Tan, D.W.N.; Lim, S.G.; Wong, T.T.; Venkatraman, S.S. Sustained antibiotic-eluting intra-ocular lenses: A new approach. PLoS ONE 2016, 11, e0163857, doi:10.1371/journal.pone.0163857.
- Eperon, S.; Rodriguez-Aller, M.; Balaskas, K.; Gurny, R.; Guex-Crosier, Y. A new drug delivery system inhibits uveitis in an animal model after cataract surgery. Int. J. Pharm. 2013, 443, 254–261, doi:10.1016/j.ijpharm.2012.12.033.
- Ross, A.E.; Bengani, L.C.; Tulsan, R.; Maidana, D.E.; Salvador-Culla, B.; Kobashi, H.; Kolovou, P.E.; Zhai, H.; Taghizadeh, K.; Kuang, L.; et al. Topical sustained drug delivery to the retina with a drug-eluting contact lens. Biomaterials 2019, 217, 119285, doi:10.1016/j.biomaterials.2019.119285.
- Maulvi, F.A.; Lakdawala, D.H.; Shaikh, A.A.; Desai, A.R.; Choksi, H.H.; Vaidya, R.J.; Ranch, K.M.; Koli, A.R.; Vyas, B.A.; Shah, D.O. vitro and in vivo evaluation of novel implantation technology in hydrogel contact lenses for controlled drug delivery. J. Control. Release 2016, 226, 47–56, doi:10.1016/j.jconrel.2016.02.012.
- Desai, A.R.; Maulvi, F.A.; Pandya, M.M.; Ranch, K.M.; Vyas, B.A.; Shah, S.A.; Shah, D.O. Co-delivery of timolol and hyaluronic acid from semi-circular ring-implanted contact lenses for the treatment of glaucoma: In vitro and in vivo evaluation. Biomater. Sci. 2018, 6, 1580–1591, doi:10.1039/c8bm00212f.
- Desai, A.R.; Maulvi, F.A.; Desai, D.M.; Shukla, M.R.; Ranch, K.M.; Vyas, B.A.; Shah, S.A.; Sandeman, S.; Shah, D.O. Multiple drug delivery from the drug-implants-laden silicone contact lens: Addressing the issue of burst drug release. Mater. Sci. Eng. C 2020, 112, 110885, doi:10.1016/j.msec.2020.110885.
- Ciolino, J.B.; Stefanescu, C.F.; Ross, A.E.; Salvador-Culla, B.; Cortez, P.; Ford, E.M.; Wymbs, K.A.; Sprague, S.L.; Mascoop, D.R.; Rudina, S.S.; et al. In vivo performance of a drug-eluting contact lens to treat glaucoma for a month. Biomaterials 2014, 35, 432–439, doi:10.1016/j.biomaterials.2013.09.032.
- Ciolino, J.B.; Ross, A.E.; Tulsan, R.; Watts, A.C.; Wang, R.F.; Zurakowski, D.; Serle, J.B.; Kohane, D.S. Latanoprost-Eluting Contact Lenses in Glaucomatous Monkeys. Ophthalmology 2016, 123, 2085–2092, doi:10.1016/j.ophtha.2016.06.038.
- Song, C.; Ben-Shlomo, G.; Que, L. A Multifunctional Smart Soft Contact Lens Device Enabled by Nanopore Thin Film for Glaucoma Diagnostics and in Situ Drug Delivery. J. Microelectromech. Syst. 2019, 28, 810–816, doi:10.1109/JMEMS.2019.2927232.
- Weinreb, R.N.; Aung, T.; Medeiros, F.A. The Pathophysiology and Treatment of Glaucoma: A Review. JAMA 2014, 311, 1901–1911, doi:10.1001/jama.2014.3192.
- Wong, V.H.Y.; Bui, B.V.; Vingrys, A.J. Clinical and experimental links between diabetes and glaucoma. Clin. Exp. Optom. 2011, 94, 4–23, doi:10.1111/j.1444-0938.2010.00546.x.
- Hanyuda, A.; Sawada, N.; Yuki, K.; Uchino, M.; Ozawa, Y.; Sasaki, M.; Yamagishi, K.; Iso, H.; Tsubota, K.; Tsugane, S. Relationships of diabetes and hyperglycaemia with intraocular pressure in a Japanese population: The JPHC-NEXT Eye Study. Sci. Rep. 2020, 10, 5355, doi:10.1038/s41598-020-62135-3.
- Gerber, A.L.; Harris, A.; Siesky, B.; Lee, E.; Schaab, T.J.; Huck, A.; Amireskandari, A. Vascular Dysfunction in Diabetes and Glaucoma: A Complex Relationship Reviewed. J. Glaucoma 2015, 24, 474–479, doi:10.1097/IJG.0000000000000137.
- Capitena Young, C.E.; Kahook, M.Y.; Seibold, L.K. Novel Drug Delivery Systems for the Treatment of Glaucoma. Curr. Ophthalmol. Rep. 2019, 7, 143–149, doi:10.1007/s40135-019-00210-3.
- Carvalho, I.M.; Marques, C.S.; Oliveira, R.S.; Coelho, P.B.; Costa, P.C.; Ferreira, D.C. Sustained drug release by contact lenses for glaucoma treatment—A review. J. Control. Release 2015, 202, 76–82, doi:10.1016/j.jconrel.2015.01.023.
- Szigiato, A.A.; Podbielski, D.W.; Ahmed, I.I.K. Sustained drug delivery for the management of glaucoma. Expert Rev. Ophthalmol. 2017, 12, 173–186, doi:10.1080/17469899.2017.1280393.
- Sihota, R.; Angmo, D.; Ramaswamy, D.; Dada, T. Simplifying “target” intraocular pressure for different stages of primary open-angle glaucoma and primary angle-closure glaucoma. Indian J. Ophthalmol. 2018, 66, 495–505, doi:10.4103/ijo.IJO_1130_17.
- Maulvi, F.A.; Soni, T.G.; Shah, D.O. Extended Release of Timolol from Ethyl Cellulose Microparticles Laden Hydrogel Contact Lenses. Open Pharm. Sci. J. 2015, 2, 1–12, doi:10.2174/1874844901502010001.
- Mu, C.; Shi, M.; Liu, P.; Chen, L.; Marriott, G. Daylight-Mediated, Passive, and Sustained Release of the Glaucoma Drug Timolol from a Contact Lens. ACS Cent. Sci. 2018, 4, 1677–1687, doi:10.1021/acscentsci.8b00641.
- Lee, S.H.; Shin, K.S.; Kim, J.W.; Kang, J.Y.; Kim, J.K. Stimulus-responsive contact lens for IOP measurement or temperature-triggered drug release. Transl Vis. Sci. Technol. 2020, 9, 1–10, doi:10.1167/tvst.9.4.1.
- Zhu, Q.; Cheng, H.; Huo, Y.; Mao, S. Sustained ophthalmic delivery of highly soluble drug using pH-triggered inner layer-embedded contact lens. Int. J. Pharm. 2018, 544, 100–111, doi:10.1016/j.ijpharm.2018.04.004.
- Peng, C.C.; Burke, M.T.; Chauhan, A. Transport of topical anesthetics in vitamin e loaded silicone hydrogel contact lenses. Langmuir 2012, 28, 1478–1487, doi:10.1021/la203606z.
- Gupta, V.; Rajagopala, M.; Ravishankar, B. Etiopathogenesis of cataract: An appraisal. Indian J. Ophthalmol. 2014, 62, 103–110, doi:10.4103/0301-4738.121141.
- McCarty, C.A. Cataract in the 21st century: Lessons from previous epidemiologic research. Clin. Exp. Optom. 2002, 85, 91–96, doi:10.1111/j.1444-0938.2002.tb03014.x.
- Laursen, S.B.; Erichsen, J.H.; Holm, L.M.; Kessel, L. Prevention of macular edema in patients with diabetes after cataract surgery. J. Cataract Refract. Surg. 2019, 45, 854–869, doi:10.1016/j.jcrs.2019.04.025.
- Pollreisz, A.; Schmidt-Erfurth, U. Diabetic Cataract—Pathogenesis, Epidemiology and Treatment. J. Ophthalmol. 2010, 2010, 608751, doi:10.1155/2010/608751.
- Varela-Garcia, A.; Concheiro, A.; Alvarez-Lorenzo, C. Cytosine-functionalized bioinspired hydrogels for ocular delivery of antioxidant transferulic acid. Biomater. Sci. 2020, 8, 1171–1180, doi:10.1039/c9bm01582e.
- Dua, H.S.; Attre, R. Treatment of Post-operative Inflammation following Cataract Surgery–A Review. Eur. Ophthalmic Rev. 2012, 6, 98, doi:10.17925/eor.2012.06.02.98.
- McCafferty, S.; Harris, A.; Kew, C.; Kassm, T.; Lane, L.; Levine, J.; Raven, M. Pseudophakic Pseudophakic cystoid macular edema prevention and risk factors; prospective study with adjunctive once daily topical nepafenac 0.3% versus placebo. BMC Ophthalmol. 2017, 17, 16, doi:10.1186/s12886-017-0405-7.
- Eriksson, U.; Alm, A.; Bjärnhall, G.; Granstam, E.; Matsson, A.W. Macular edema and visual outcome following cataract surgery in patients with diabetic retinopathy and controls. Graefe’s Arch. Clin. Exp. Ophthalmol. 2011, 249, 349–359, doi:10.1007/s00417-010-1484-9.
- Wielders, L.H.P.; Lambermont, V.A.; Schouten, J.S.A.G.; Van Den Biggelaar, F.J.H.M.; Worthy, G.; Simons, R.W.P.; Winkens, B.; Nuijts, R.M.M.A. Prevention of cystoid macular edema after cataract surgery in nondiabetic and diabetic patients: A systematic review and meta-analysis. Am. J. Ophthalmol. 2015, 160, 968-981.e33, doi:10.1016/j.ajo.2015.07.032.
- Khambhiphant, B.; Liumsirijarern, C.; Saehout, P. The effect of Nd: YAG laser treatment of posterior capsule opacification on anterior chamber depth and refraction in pseudophakic eyes. Clin. Ophthalmol. 2015, 9, 557–561, doi:10.2147/OPTH.S80220.
- Kiziltoprak, H.; Tekin, K.; Inanc, M.; Goker, Y.S. Cataract in diabetes mellitus. World J. Diabetes 2019, 10, 140–153, doi:10.4239/wjd.v10.i3.140.
- Praveen, M.R.; Vasavada, A.R.; Shah, G.D.; Shah, A.R.; Khamar, B.M.; Dave, K.H. A prospective evaluation of posterior capsule opacification in eyes with diabetes mellitus: A case-control study. Eye 2014, 28, 720–727, doi:10.1038/eye.2014.60.
- Donnenfeld, E.; Holland, E. Dexamethasone Intracameral Drug-Delivery Suspension for Inflammation Associated with Cataract Surgery: A Randomized, Placebo-Controlled, Phase III Trial. Ophthalmology 2018, 125, 799–806, doi:10.1016/j.ophtha.2017.12.029.
- Pimenta, A.F.R.; Serro, A.P.; Colaço, R.; Chauhan, A. Drug delivery to the eye anterior chamber by intraocular lenses: An in vivo concentration estimation model. Eur. J. Pharm. Biopharm. 2018, 133, 63–69, doi:10.1016/j.ejpb.2018.10.004.
- Morarescu, D.; West-Mays, J.A.; Sheardown, H.D. Effect of delivery of MMP inhibitors from PDMS as a model IOL material on PCO markers. Biomaterials 2010, 31, 2399–2407, doi:10.1016/j.biomaterials.2009.11.108.
- Yu, N.; Fang, F.; Wu, B.; Zeng, L.; Cheng, Y. State of the art of intraocular lens manufacturing. Int. J. Adv. Manuf. Technol. 2018, 98, 1103–1130, doi:10.1007/s00170-018-2274-5.
- Tan, X.; Zhan, J.; Zhu, Y.; Cao, J.; Wang, L.; Liu, S.; Wang, Y.; Liu, Z.; Qin, Y.; Wu, M.; et al. Improvement of Uveal and Capsular Biocompatibility of Hydrophobic Acrylic Intraocular Lens by Surface Grafting with 2-Methacryloyloxyethyl Phosphorylcholine-Methacrylic Acid Copolymer. Sci. Rep. 2017, 7, 40462, doi:10.1038/srep40462.
- Huang, Y.S.; Bertrand, V.; Bozukova, D.; Pagnoulle, C.; Labrugère, C.; De Pauw, E.; De Pauw-Gillet, M.C.; Durrieu, M.C. RGD surface functionalization of the hydrophilic acrylic intraocular lens material to control posterior capsular opacification. PLoS ONE 2014, 9, e114973, doi:10.1371/journal.pone.0114973.
- Tetz, M.; Jorgensen, M.R. New Hydrophobic IOL Materials and Understanding the Science of Glistenings. Curr. Eye Res. 2015, 40, 969–981, doi:10.3109/02713683.2014.978476.
- Molokhia, S.A.; Thomas, S.C.; Garff, K.J.; Mandell, K.J.; Wirostko, B.M. Anterior eye segment drug delivery systems: Current treatments and future challenges. J. Ocul. Pharmacol. Ther. 2013, 29, 92–105, doi:10.1089/jop.2012.0241.
- Ljubimov, A V. Diabetic complications in the cornea. Vision Res. 2017, 139, 138–152, doi:10.1016/j.visres.2017.03.002.
- Clayton, J.A. Dry eye. N. Engl. J. Med. 2018, 378, 2212–2223, doi:10.1056/NEJMra1407936.
- Findlay, Q.; Reid, K. Dry eye disease: When to treat and when to refer. Aust. Prescr. 2018, 41, 160–163, doi:10.18773/AUSTPRESCR.2018.048.
- Karsten, E. Diversity of Microbial Species Implicated in Keratitis: A Review. Open Ophthalmol. J. 2012, 6, 110–124, doi:10.2174/1874364101206010110.
- Kuruvilla, S.; Peter, J.; David, S.; Premkumar, P.S.; Ramakrishna, K.; Thomas, L.; Vedakumar, M.; Peter, J.V. Incidence and risk factor evaluation of exposure keratopathy in critically ill patients: A cohort study. J. Crit Care 2015, 30, 400–404, doi:10.1016/j.jcrc.2014.10.009.
- Dutescu, R.M.; Panfil, C.; Schrage, N. Osmolarity of prevalent eye drops, side effects, and therapeutic approaches. Cornea 2015, 34, 560–566, doi:10.1097/ICO.0000000000000368.
- Daphna, O.; Mimouni, M.; Keshet, Y.; Ben Ishai, M.; Barequet, I.S.; Knyazer, B.; Mrukwa-Kominek, E.; Zarnowski, T.; Chen-Zion, M.; Marcovich, A. Therapeutic HL-Contact Lens versus Standard Bandage Contact Lens for Corneal Edema: A Prospective, Multicenter, Randomized, Crossover Study. J. Ophthalmol. 2020, 2020, 8410920, doi:10.1155/2020/8410920.
- Siu, G.D.J.Y.; Young, A.L.; Jhanji, V. Alternatives to corneal transplantation for the management of bullous keratopathy. Curr. Opin. Ophthalmol. 2014, 25, 347–352, doi:10.1097/ICU.0000000000000062.
- Soh, Y.Q.; Kocaba, V.; Weiss, J.S.; Jurkunas, U.V.; Kinoshita, S.; Aldave, A.J.; Mehta, J.S. Corneal dystrophies. Nat. Rev. Dis. Prim. 2020, 6, 46.
- Han, S.B.; Yang, H.K.; Hyon, J.Y. Influence of diabetes mellitus on anterior segment of the eye. Clin. Interv. Aging 2019, 14, 53–63, doi:10.2147/CIA.S190713.
- Abdelkader, H.; Patel, D.V.; Mcghee, C.N.; Alany, R.G. New therapeutic approaches in the treatment of diabetic keratopathy: A review. Clin. Exp. Ophthalmol. 2011, 39, 259–270, doi:10.1111/j.1442-9071.2010.02435.x.
- de Alves, M. C.; Carvalheira, J.B.; Módulo, C.M.; Rocha, E.M. Tear film and ocular surface changes in diabetes mellitus. Arq. Bras. Oftalmol. 2008, 71, 96–103, doi:10.1590/s0004-27492008000700018.
- Zidan, G.; Rupenthal, I.D.; Greene, C.; Seyfoddin, A. Medicated ocular bandages and corneal health: Potential excipients and active pharmaceutical ingredients. Pharm. Dev. Technol. 2018, 23, 255–260, doi:10.1080/10837450.2017.1377232.
- Misra, S.L.; Patel, D.V.; McGhee, C.N.J.; Pradhan, M.; Kilfoyle, D.; Braatvedt, G.D.; Craig, J.P. Peripheral neuropathy and tear film dysfunction in type 1 diabetes mellitus. J. Diabetes Res. 2014, 2014, 848659, doi:10.1155/2014/848659.
- Shih, K.C.; Lam, K.L.; Tong, L. A systematic review on the impact of diabetes mellitus on the ocular surface. Nutr. Diabetes 2017, 7, e251, doi:10.1038/nutd.2017.4.
- Ali, M.; Byrne, M.E. Controlled release of high molecular weight hyaluronic acid from molecularly imprinted hydrogel contact lenses. Pharm. Res. 2009, 26, 714–726.
- Maulvi, F.A.; Soni, T.G.; Shah, D.O. Extended release of hyaluronic acid from hydrogel contact lenses for dry eye syndrome. J. Biomater. Sci. Polym. Ed. 2015, 26, 1035–1050, doi:10.1080/09205063.2015.1072902.
- Maulvi, F.A.; Shaikh, A.A.; Lakdawala, D.H.; Desai, A.R.; Pandya, M.M.; Singhania, S.S.; Vaidya, R.J.; Ranch, K.M.; Vyas, B.A.; Shah, D.O. Design and optimization of a novel implantation technology in contact lenses for the treatment of dry eye syndrome: In vitro and in vivo evaluation. Acta Biomater. 2017, 53, 211–221, doi:10.1016/j.actbio.2017.01.063.
- White, C.J.; Dipasquale, S.A.; Byrne, M.E. Controlled Release of Multiple Therapeutics from Silicone Hydrogel Contact Lenses. Optom. Vis. Sci. 2016, 93, 377–386.
- Kim, G.; Kim, H.J.; Noh, H. pH sensitive soft contact lens for selective drug-delivery. Macromol. Res. 2018, 26, 278–283, doi:10.1007/s13233-018-6029-9.
- Pitt, W.G.; Jack, D.R.; Zhao, Y.; Nelson, J.L.; Pruitt, J.D. Loading and release of a phospholipid from contact lenses. Optom. Vis. Sci. 2011, 88, 502–506, doi:10.1097/OPX.0b013e31820e9ff8.
- Pitt, W.G.; Jack, D.R.; Zhao, Y.; Nelson, J.L.; Pruitt, J.D. Transport of phospholipid in silicone hydrogel contact lenses. J. Biomater. Sci. Polym. Ed. 2012, 23, 527–541, doi:10.1163/092050611X554174.
- Pitt, W.G.; Zhao, Y.; Jack, D.R.; Perez, K.X.; Jones, P.W.; Marelli, R.; Nelson, J.L.; Pruitt, J.D. Extended elution of phospholipid from silicone hydrogel contact lenses. J. Biomater. Sci. Polym. Ed. 2015, 26, 224–234, doi:10.1080/09205063.2014.994947.
- Chen, W.; Zhang, X.; Li, J.; Wang, Y.; Chen, Q.; Hou, C.; Al, E. Efficacy of osmoprotectants on prevention and treatment of murine dry eye. Investig. Ophth Vis. Sci. 2013, 54, 6287–6297.
- Hsu, K.H.; De La Jara, P.L.; Ariyavidana, A.; Watling, J.; Holden, B.; Garrett, Q.; Chauhan, A. Release of betaine and dexpanthenol from vitamin E modified silicone-hydrogel contact lenses. Curr. Eye Res. 2015, 40, 267–273, doi:10.3109/02713683.2014.917192.
- Zhang, X.; Zhao, L.; Deng, S.; Sun, X.; Wang, N. Dry Eye Syndrome in Patients with Diabetes Mellitus: Prevalence, Etiology, and Clinical Characteristics. J. Ophthalmol. 2016, 2016, 1–7, doi:10.1155/2016/8201053.
- Guzman-Aranguez, A.; Fonseca, B.; Carracedo, G.; Martin-Gil, A.; Martinez-Aguila, A.; Pintor, J. Dry Eye Treatment Based on Contact Lens Drug Delivery: A Review. Eye Contact Lens 2016, 42, 280–288, doi:10.1097/ICL.0000000000000184.
- Phan, C.M.; Subbaraman, L.; Jones, L. Contact lenses for antifungal ocular drug delivery: A review. Expert Opin. Drug Deliv. 2014, 11, 537–546, doi:10.1517/17425247.2014.882315.
- Garg, P.; Venuganti, V.V.K.; Roy, A.; Roy, G. Novel drug delivery methods for the treatment of keratitis: Moving away from surgical intervention. Expert Opin. Drug Deliv. 2019, 16, 1381–1391, doi:10.1080/17425247.2019.1690451.
- Liu, X.; Chen, J.; Qu, C.; Bo, G.; Jiang, L.; Zhao, H.; Zhang, J.; Lin, Y.; Hua, Y.; Yang, P.; et al. A Mussel-Inspired Facile Method to Prepare Multilayer-AgNP-Loaded Contact Lens for Early Treatment of Bacterial and Fungal Keratitis. ACS Biomater. Sci. Eng. 2018, 4, 1568–1579, doi:10.1021/acsbiomaterials.7b00977.
- Aveyard, J.; Deller, R.C.; Lace, R.; Williams, R.L.; Kaye, S.B.; Kolegraff, K.N.; Curran, J.M.; D’Sa, R.A. Antimicrobial Nitric Oxide Releasing Contact Lens Gels for the Treatment of Microbial Keratitis. ACS Appl. Mater. Interfaces 2019, 11, 37491–37501, doi:10.1021/acsami.9b13958.
- Hewitt, M.G.; Morrison, P.W.J.; Boostrom, H.M.; Morgan, S.R.; Fallon, M.; Lewis, P.N.; Whitaker, D.; Brancale, A.; Varricchio, C.; Quantock, A.J.; et al. In Vitro Topical Delivery of Chlorhexidine to the Cornea: Enhancement Using Drug-Loaded Contact Lenses and β-Cyclodextrin Complexation, and the Importance of Simulating Tear Irrigation. Mol. Pharm. 2020, 17, 1428–1441, doi:10.1021/acs.molpharmaceut.0c00140.
- Huang, J.F.; Zhong, J.; Chen, G.P.; Lin, Z.T.; Deng, Y.; Liu, Y.L.; Cao, P.Y.; Wang, B.; Wei, Y.; Wu, T.; et al. A Hydrogel-Based Hybrid Theranostic Contact Lens for Fungal Keratitis. ACS Nano 2016, 10, 6464–6473, doi:10.1021/acsnano.6b00601.
- Gallagher, A.G.; McLean, K.; Stewart, R.M.K.; Wellings, D.A.; Allison, H.E.; Williams, R.L. Development of a poly-ε-lysine contact lens as a drug delivery device for the treatment of fungal keratitis. Investig. Ophthalmol. Vis. Sci. 2017, 58, 4499–4505, doi:10.1167/iovs.17-22301.
- Phan, C.M.; Bajgrowicz, M.; Gao, H.; Subbaraman, L.N.; Jones, L.W. Release of fluconazole from contact lenses using a novel in vitro eye model. Optom Vis. Sci. 2016, 93, 387–394, doi:10.1097/OPX.0000000000000760.
- Jaishankar, D.; Buhrman, J.S.; Valyi-Nagy, T.; Gemeinhart, R.A.; Shukla, D. Extended release of an anti–heparan sulfate peptide from a contact lens suppresses corneal herpes simplex virus-1 infection. Investig. Ophthalmol. Vis. Sci. 2016, 57, 169–180, doi:10.1167/iovs.15-18365.
- Varela-Garcia, A.; Gomez-Amoza, J.L.; Concheiro, A.; Alvarez-Lorenzo, C. Imprinted contact lenses for ocular administration of antiviral drugs. Polymers 2020, 12, 2026, doi:10.3390/POLYM12092026.
- Jacob, S.; Kumar, D.A.; Agarwal, A.; Basu, S.; Sinha, P.; Agarwal, A. Contact lens-assisted collagen cross-linking (CACXL): A new technique for cross-linking thin corneas. J. Refract. Surg. 2014, 30, 366–372, doi:10.3928/1081597X-20140523-01.
- Kubrak-Kisza, M.; Kisza, K.J.; Misiuk-Hojło, M. Corneal Cross-Linking: An Example of Photoinduced Polymerization as a Treatment Modality in Keratoconus. Polim. Med. 2016, 46, 89–94, doi:10.17219/pim/65010.
- Malhotra, C.; Jain, A.K.; Gupta, A.; Ram, J.; Ramatchandirane, B.; Dhingra, D.; Sachdeva, K.; Kumar, A. Demarcation line depth after contact lens–assisted corneal crosslinking for progressive keratoconus: Comparison of dextran-based and hydroxypropyl methylcellulose–based riboflavin solutions. J. Cataract Refract. Surg. 2017, 43, 1263–1270, doi:10.1016/j.jcrs.2017.07.032.
- Han, S.B.; Liu, Y.-C.; Mohamed-Noriega, K.; Mehta, J.S. Application of Novel Drugs for Corneal Cell Regeneration. J. Ophthalmol. 2018, 2018, 1–9, doi:10.1155/2018/1215868.
- Wollensak, G.; Spörl, E.; Herbst, H. Biomechanical efficacy of contact lens-assisted collagen cross-linking in porcine eyes. Acta Ophthalmol. 2019, 97, e84–e90, doi:10.1111/aos.13828.
- Holland, S.; Morck, D.; Schultz, C. Treatment of corneal defects with delayed re-epithelization with a medical device/drug delivery system for epidermal growth factor. Clin. Exp. Ophthalmol. 2012, 40, 662–668, doi:10.1111/j.1442-9071.2012.02795.x.
- Sandri, G.; Bonferoni, M.C.; Rossi, S.; Delfino, A.; Riva, F.; Icaro Cornaglia, A.; Marrubini, G.; Musitelli, G.; Del Fante, C.; Perotti, C.; et al. Platelet lysate and chondroitin sulfate loaded contact lenses to heal corneal lesions. Int. J. Pharm. 2016, 509, 188–196, doi:10.1016/j.ijpharm.2016.05.045.
- Nakahara, M.; Miyata, K.; Otani, S.; Miyai, T.; Nejima, R.; Yamagami, S.; Amano, S. A randomised, placebo controlled clinical trial of the aldose reductase inhibitor CT-112 as management of corneal epithelial disorders in diabetic patients. Br. J. Ophthalmol. 2005, 89, 266–268, doi:10.1136/bjo.2004.049841.
- Dogru, M.; Kojima, T.; Simsek, C.; Tsubotav, K. Potential role of oxidative stress in ocular surface inflammation and dry eye disease. Investig. Ophthalmol. Vis. Sci. 2018, 59, DES163–DES168, doi:10.1167/iovs.17-23402.
- Shoham, A.; Hadziahmetovic, M.; Dunaief, J.L.; Mydlarski, M.B.; Schipper, H.M. Oxidative stress in diseases of the human cornea. Free Radic. Biol. Med. 2008, 45, 1047–1055, doi:10.1016/j.freeradbiomed.2008.07.021.
- Agrahari, V.; Agrahari, V.; Mandal, A.; Pal, D.; Mitra, A.K. How are we improving the delivery to back of the eye? Advances and challenges of novel therapeutic approaches. Expert Opin. Drug Deliv. 2017, 14, 1145–1162, doi:10.1080/17425247.2017.1272569.
- Rastogi, N.; Smith, R.T. Association of age-related macular degeneration and reticular macular disease with cardiovascular disease. Surv Ophthalmol. 2016, 61, 422–433, doi:10.1016/j.survophthal.2015.10.003.
- Mehta, S. Age-Related Macular Degeneration. Prim. Care-Clin. Off. Pract. 2015, 42, 377–391, doi:10.1016/j.pop.2015.05.009.
- Dedania, V.S.; Grob, S.; Zhang, K.; Bakri, S.J. Pharmacogenomics of response to anti-vegf therapy in exudative age-related macular degeneration. Retina 2015, 35, 381–391, doi:10.1142/9789814663076_0004.
- Davidson, J.A.; Ciulla, T.A.; McGill, J.B.; Kles, K.A.; Anderson, P.W. How the diabetic eye loses vision. Endocrine 2007, 32, 107–116, doi:10.1007/s12020-007-0040-9.
- Simó, R.; Hernández, C. Neurodegeneration in the diabetic eye: New insights and therapeutic perspectives. Trends Endocrinol. Metab. 2014, 25, 23–33, doi:10.1016/j.tem.2013.09.005.
- Christopher, K.; Chauhan, A. Contact Lens Based Drug Delivery to the Posterior Segment Via Iontophoresis in Cadaver Rabbit Eyes. Pharm. Res. 2019, 36, 87.
- Molokhia, S.A.; Sant, H.; Simonis, J.; Bishop, C.J.; Burr, R.M.; Gale, B.K.; Ambati, B.K. The capsule drug device: Novel approach for drug delivery to the eye. Vis. Res. 2010, 50, 680–685, doi:10.1016/j.visres.2009.10.013.
- Lanier, O.L.; Christopher, K.G.; Macoon, R.M.; Yifan Yu, P.; Sekar, P.; Chauhan, A. Commercialization challenges for drug eluting contact lenses. Expert Opin Drug Deliv. 2020, 17, 1133–1149, doi:10.1080/17425247.2020.1787983.