Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is an old version of this entry, which may differ significantly from the current revision.
Aging is a complex biological process that is influenced by both intrinsic and extrinsic factors. Recently, it has been discovered that reactive oxygen species can accelerate the aging process, leading to an increased incidence of age-related diseases that are characteristic of aging.
- Mushrooms
- anti-aging
- age-related disease
- cellular mechanisms
- bioactive compounds
1. Introduction
The global population is currently experiencing a significant expansion of aging populations compared to previous years. This trend is reflected in the increase in average life expectancy at birth, which has risen by 6.2 years from 65.3 years in 1990, to 71.5 years in 2013. Additionally, individuals who reach the age of 60 can now expect to live for another 22 years on average [1]. By the year 2040, it is projected that the average life expectancy will increase by 4.4 years for both men and women. Men can expect to live an average of 74.3 years, while women can expect to live an average of 79.7 years. However, these numbers may vary depending on individual health conditions [2]. As the population ages, there has been a noticeable increase in the prevalence of chronic degenerative diseases such as neurodegenerative and cardiovascular diseases, diabetes, and cancer. These diseases contribute to up to 70% of global mortality each year, including premature deaths occurring between the ages of 30 and 70 [1]. It is important to note that, while aging is often accompanied by deteriorative changes and an increased risk of functional declines or diseases, aging itself is not considered a disease. The focus of anti-aging strategies is not to reverse or halt the aging process, but rather to promote healthy aging and reduce the incidence of age-related diseases. The World Health Organization recommends adopting healthy dietary habits, engaging in regular physical activity, and controlling tobacco use as effective measures to alleviate or prevent the incidence of chronic diseases. By following these guidelines, the risk of developing age-related diseases can be reduced [3].
There is growing evidence to suggest that healthy aging can be promoted by consuming nutraceuticals and following various dietary patterns, such as caloric restriction, intermittent fasting, a Mediterranean diet, an Okinawan diet, and a Nordic diet. These dietary patterns have been evaluated for their negative correlation with aging and age-related conditions and diseases [4][5], which has led to a search for anti-aging components from food sources and an investigation of the underlying mechanisms of anti-aging pathways. Bioactive compounds derived from plant sources, including fruits and vegetables, roots, seeds, and edible flowers, have been suggested to exert anti-aging effects. These compounds include certain polysaccharides, phenolic compounds, and peptides [6][7]. In recent years, mushrooms—filamentous fungi with fruiting bodies—have also been shown to possess enormous pharmacological attributes that are valuable for healthy aging. These attributes include anti-oxidant, immunomodulatory, neuroprotective, anti-inflammatory, and anti-cancer properties [8][9][10][11].
Mushrooms are nutritious foods that are rich in carbohydrates and proteins, with a lower content of lipids [12]. In addition to their nutritional value, mushrooms contain various bioactive compounds, such as β-glucans, lectins, and linolenic acids, which can be isolated through different extraction methods. These compounds confer a variety of pharmacological activities and may enhance the immune system and strengthen the biological function of the body [13]. Regular intake of mushrooms or their extracts may help alleviate age-related diseases.
2. Aging
2.1. Aging and Age-Related Diseases
Aging is a complex process that involves the time-dependent accumulation of diverse deleterious changes in cells, tissues, organs, or systems that increase vulnerability to chronic illness and death [14][15]. Nine candidate hallmarks of aging have been identified and classified, including primary hallmarks (genomic instability, telomere attrition, epigenetic alterations, and loss of proteostasis), antagonistic hallmarks (deregulated nutrient sensing, mitochondrial dysfunction, and cellular senescence), and integrative hallmarks (stem cell exhaustion and altered intercellular communication), all of which are correlated with each other [16]. The antagonistic hallmarks exert positive effects at low levels but negatively affect the organism at high levels [16]. For example, reactive oxygen species (ROS) are important signaling molecules that play a role in regulating cellular functions, but excessive levels can lead to oxidative damage and contribute to aging. The primary hallmarks are the contributors to molecular damage during aging, while the integrative hallmarks are signs of failure of cellular homeostasis and metabolism mechanisms to ameliorate the damage. These hallmarks are interconnected with each other and could serve as a guidance to decipher the mechanistic molecular basis for prolonging health span and development of strategies for longevity, such as stem-cell-based therapies, epigenetic drugs, anti-inflammatory drugs, and dietary restrictions [16].
The free radical theory of aging, proposed in 1956 by Denham Harman [17], is a widely accepted theory of aging. The theory postulates that the aging process is triggered by the initiation of free radical reactions, leading to increased generation of free radicals by damaged mitochondria with increasing age [18]. Major sources of free radical reactions in mammals include non-enzymatic reaction of oxygen, ionizing radiation, cytochrome P-450 system, respiratory chain, phagocytosis, and prostaglandin synthesis, which lead to the accumulation of oxidative damage and may shorten the lifespan. Several defenses that alleviate the damage of the reactions include DNA repair mechanisms, superoxide dismutase, glutathione peroxidase, and anti-oxidants (e.g., carotenes and vitamin E) [15][19].
ROS are byproducts of oxidative metabolism that can induce cellular defense mechanisms against oxidative invasion at low doses, potentially prolonging health span and lifespan. However, long-term excessive exposure to ROS can lead to the oxidation of nucleic acids, proteins, and lipids, causing damage to macromolecules and mitochondrial dysfunction. This can disrupt cell homeostasis and result in cellular death [20]. ROS production is driven by progressive mitochondrial dysfunction with increasing age, creating a positive feedback loop of ROS generation and oxidative damage accumulation [18]. Concurrently, oxidative stress arises due to excessive ROS levels and limited anti-oxidant defense capability, leading to cellular senescence and a shortened lifespan. The accumulation of oxidative damage to macromolecules and mitochondria contributes to detrimental consequences, such as pathophysiological changes, functional decline, and accelerated aging, which are associated with age-related conditions such as inflammation, cardiovascular diseases, neurodegenerative diseases, autoimmune diseases, and cancer [21].
It is important to note that aging itself is not a disease. Age-related diseases can be considered “symptoms” of aging, initiated by minor disturbances that are intensified via vicious positive feedback loops, destabilizing the physiology of an organism and potentially leading to destruction (i.e., mortality) if no negative feedback loops are in place [22]. For example, low-grade inflammation can intensify in chronic inflammation, leading to decreased muscle mass, decreased physical activity, and excess fat deposition. This can further contribute to obesity, diabetes, and cardiovascular problems. Eventually, cardiovascular diseases can arise and worsen the physiological status of an individual by triggering chronic inflammation. To minimize cumulative damage to different organs and maintain cell function for healthy aging, interventions that can interrupt or break the vicious cycles of age-related diseases can be implemented, including medications, lifestyle adjustments, and dietary management (Figure 1).
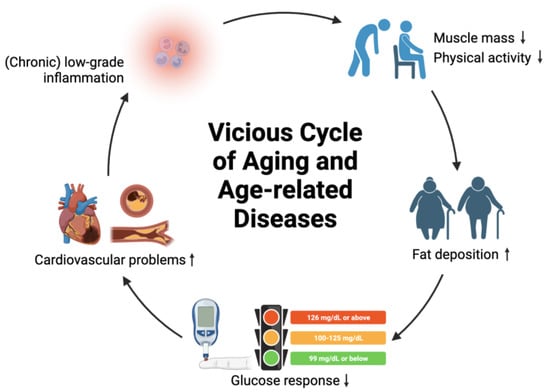
Figure 1. Concept of vicious cycle of aging and age-related diseases. Symbol ↑ denotes increase; symbol ↓ denotes decrease.
2.2. Aging and Dietary Intervention
The lifestyle of an individual is closely linked to their health span and lifespan. One of the main ways to modify lifestyle for better health maintenance and to reduce the incidence of age-related diseases is through dietary management. Unhealthy dietary habits and lifestyle can accelerate the aging process by causing molecular and cellular damage. For example, a sedentary lifestyle, combined with a “Western diet”, that is high in energy but lacking in nutrition, has been associated with reduced lifespan and increased occurrence of age-related conditions such as obesity, type 2 diabetes, and cancer [23]. On the other hand, caloric restriction (CR) has been shown to slow down the rate of aging and extend health span. CR involves reducing total energy intake by 20% to 40% while ensuring optimal nutrition, compared to an ad libitum diet. This approach has been demonstrated to extend lifespan and health span in various experimental models, including yeast, fruit flies, mice, nonhuman primates, and even humans [24][25][26].
According to the theory of aging, CR enhances longevity by reducing oxidative damage and increasing resistance to oxidative stress through specific signaling pathways. The stress caused by CR, such as nutrient deprivation, activates defense mechanisms against oxidative damage, thereby slowing down the aging process [27]. CR also affects physiological pathways that may mediate anti-aging effects, such as the insulin-like growth factor-1 and insulin signaling pathways, the mammalian target of rapamycin (mTOR) pathway, and the sirtuins pathway [24][28][29]. Previous studies have demonstrated the potential of implementing CR as an anti-aging regimen, as adherence to this dietary management reduces biomarkers associated with the development of age-related diseases, including cardiovascular diseases, autoimmune disorders, neurodegenerative diseases, diabetes, and cancer [29][30][31]. Therefore, CR can be considered as the mechanistic foundation for healthy aging strategies involving dietary intervention, which can prolong lifespan and maintain physiological function for an extended health span.
Despite the potential benefits of CR, it can be challenging for individuals to adhere to it in the long term due to various pitfalls and health concerns, such as hypotension, osteoporosis, slower wound healing, depression, and irritability [32]. As a result, scientists have explored alternative diet regimens and studied different dietary patterns that may offer similar benefits to CR but are more feasible for humans to sustain. One such approach is intermittent fasting, which shares the same concept as CR. Intermittent fasting activates cellular pathways that enhance the body’s intrinsic defense against oxidative stress, promotes the removal of damaged molecules, and facilitates tissue repair and growth. It also helps to suppress inflammation and improve stress resistance [33][34].
In addition to dietary modifications, researchers have developed anti-aging drugs that mimic the effects of CR. Examples include rapamycin and metformin, which have shown promising effects in various model organisms and clinical trials. Rapamycin delays aging by inhibiting mTOR, thereby maintaining the normal functioning of mitochondria and stem cells. Metformin, on the other hand, affects telomere length, reduces oxidative damage to DNA, and modulates the synthesis and degradation of age-related proteins [35][36]. However, it is important to note that there are concerns and side effects associated with the use of these drugs. For instance, rapamycin may lead to nephrotoxicity and thrombocytopenia, while metformin may cause vitamin B12 deficiency and lactic acid accumulation [37][38]. Therefore, there is a need to explore naturally occurring compounds that have significant anti-aging effects with minimal side effects.
Nutraceuticals and dietary supplements are also viable alternatives for anti-aging and extending health span. Examples include curcumin, quercetin, ginseng, and medicinal mushrooms, which exhibit anti-inflammatory, immunomodulatory, and antioxidative effects [39][40][41]. A diet rich in fruits and vegetables, which provide a significant number of nutraceuticals and phytochemicals, is crucial for maintaining overall health. Interestingly, mushrooms, although not classified as animals or plants but as part of the fungal kingdom, are often considered as vegetables. They are low in calories, sodium, and fat, while being a valuable source of fiber, phenolic compounds, β-glucans, selenium, glutathione, B vitamins, and vitamin D. These components serve as protective agents against oxidative damage, which accelerates aging [12]. Medicinal mushrooms have also been used for centuries in traditional therapies, like Chinese medicine and Indian Ayurveda medicine, to alleviate symptoms of various diseases [42]. The bioactive compounds found in mushrooms may contribute to their anti-aging effects through various physiological pathways involved in aging and age-related diseases.
2.3. Ageing, Mental Health and Gender
Gender and mental health can significantly impact ageing experiences. Gender influences ageing in various ways, including health outcomes, social roles and expectations, and economic status. Women are more likely to experience depression, anxiety, and stress due to factors such as caregiving responsibilities, hormonal changes, and discrimination [43]. Women also tend to report higher levels of loneliness and social isolation in later life. In contrast, men may experience social isolation and mental health issues due to societal expectations of masculinity, which can lead to reluctance in seeking help for mental health problems [44].
Gender differences in health outcomes are well-documented, with women living longer but experiencing more chronic health conditions than men. Women are more likely to experience osteoporosis, urinary incontinence, and depression than men. Women also experience menopause, which can lead to physical and psychological symptoms [45]. Men, on the other hand, are more likely to experience heart disease, stroke, and certain types of cancer. Biological factors such as sex hormones, genetics, and lifestyle factors like diet, exercise, and smoking influence gender differences in health outcomes [46].
Gender roles and expectations can influence ageing experiences [47]. Women are often expected to take on caregiving roles for children, spouses, or ageing parents, which can lead to stress and impact their own health and well-being. Women may also face ageism and discrimination in the workplace, leading to financial insecurity in later life. Men, on the other hand, may experience pressure to maintain their independence and financial stability, leading to social isolation and mental health issues [43][44][48].
Gender differences in economic status can also impact ageing experiences. Women often earn less than men over their lifetimes, leading to lower retirement savings and financial insecurity in later life. Women are also more likely to work part-time or take career breaks to care for children or ageing parents, which can impact their pension entitlements. This can lead to poverty and social exclusion in later life [47][49]. Mental health issues, such as depression, anxiety, and cognitive impairment, can also impact ageing experiences. Depression is a common mental health issue among older adults and can lead to social isolation, physical illness, and suicide. Anxiety can affect quality of life and daily functioning. Cognitive impairment, including dementia, can result in memory loss, decision-making difficulties, and loss of independence and increased caregiving needs [50].
Studies have shown that gender and mental health can interact to influence ageing experiences [28][36][43][44][51][52]. Women with depression may be more prone to physical disability and cognitive decline in later life compared to men with depression. Similarly, men with higher levels of anxiety may be more likely to experience cognitive decline than women with anxiety [43]. Addressing gender and mental health in ageing policies and practices is crucial to ensure that older adults receive appropriate support and services. This includes promoting gender equity, addressing mental health stigma, and providing accessible and affordable mental health care for older adults [22].
3. Components of Mushrooms and Their Anti-Aging Effects
Mushrooms have long been recognized for their nutritional value and potential health benefits. Edible mushrooms are not only rich in protein, fiber, vitamins, and minerals but also have low levels of fat, making them highly nutritious [53][54]. They contain all the essential amino acids and have a higher protein content compared to most vegetables, making them particularly beneficial for vegetarians. In addition to their nutritional value, edible mushrooms, as fungi, have the ability to produce a wide range of chemical compounds known as mycochemicals. These mycochemicals can act as bioactive substances with various advantages for human health [55]. Mushrooms have been found to contain significant levels of mycochemicals that serve as bioactive compounds, offering a range of health benefits against aging and age-related diseases [53][54].
3.1. Bioactive Compounds in Mushrooms
Bioactive compounds extracted from mushrooms have been extensively studied for their ability to enhance cellular functions and provide health benefits. The following text summarizes four representative categories of bioactive compounds found in mushrooms: carbohydrates, proteins, lipids, and phenolic compounds.
3.1.1. Carbohydrates
Carbohydrates derived from mushrooms have been extensively studied for their anti-tumor, anti-inflammatory, and immunomodulatory activities [56][57]. Numerous monosaccharides found in mushrooms, including arabinose, fructose, fucose, galactose, glucose, mannose, mannitol, rhamnose, trehalose, and xylose, have been identified as exhibiting these activities. They primarily achieve this through the activation of cytokines, such as interferons and interleukins, and involve cellular pathways that include dendritic cells, natural killer cells, neutrophils, and cytotoxic macrophages [57][58]. β-Glucans, the main type of carbohydrates found in mushrooms, have been shown to possess antioxidative, anti-cancer, immunomodulatory, and neuroprotective properties. They are considered potent agents for stimulating the immune system and protecting against carcinogens, pathogens, and toxins [59][60][61][62][63][64]. The biological activity and health benefits of β-glucans isolated from mushrooms, particularly in relation to immune health, are crucial for healthy aging. Supplementation with mushroom carbohydrates, which contain β-glucans, could be an effective strategy for anti-aging. Table 1 provides a list of various mushrooms that contain bioactive carbohydrates.
Table 1. Bioactive carbohydrates in selected mushrooms.
| Mushrooms | Common Names | Bioactive Compounds | Source and Yield | Bioactivities | References |
|---|---|---|---|---|---|
| Agaricus bisporus | Button mushroom | Heteropolysaccharide Abnp1001, Abnp1002, Abap1001, Abap1002 | Concentrated industrial wastewater of A. bisporus; 0.989 mg/g, 1.849 mg/g, 0.128 mg/g, and 0.68 mg/g (Abnp1001, Abnp1002, Abap1001, Abap1002) | Hepatoprotective | [65] |
| Heteropolysaccharide AcAPS, AcAPS-1, AcAPS-2, AcAPS-3, with rhamnose and glucose as major monosaccharide | Dried fruiting body; yield n.s. | Hepatoprotective, nephroprotective, antioxidative | [66] | ||
| Polysaccharide extracts, main components n.s. | Whole mushroom; yield n.s. | Anti-tumor, immunostimulatory | [67] | ||
| Heteropolysaccharide/Mannogalacoglucan mannose, galactose, glucose | Freeze-dried fresh fruiting body; 41.4% yield (w/w dry weight) | Anti-tumor | [68] | ||
| β-glucan | Dried fresh fruiting body; yield n.s. | Immunostimulatory | [69] | ||
| Fructose, mannitol, trehalose | Fresh fruiting body; 5.79% (white mushroom) & 4.27% (brown mushroom) (w/w fresh weight) | n.s. | [70] | ||
| Calocybe indica |
Milky mushroom | Polysaccharide extracts, main components n.s. | Fresh fruiting body; 3.27% (w/w dry weight) | Anti-oxidant, neuroprotective | [71] |
| Flammulina velutipes |
Enoki/Golden needle mushroom | Polysaccharide extracts, main components n.s. | Base of stipe; yield n.s. | Anti-tumor | [72] |
| Polysaccharide extracts, main components n.s. | Fresh whole-mushroom; yield n.s. | Neuroprotective | [73] | ||
| Fructose, mannitol, sucrose, trehalose | Fresh fruiting body; 8.29% (w/w fresh weight) | n.s. | [70] | ||
| Ganoderma lucidum |
Ling Zhi | Polysaccharide extracts, main components n.s. | Mycelia; 71.99% (w/w dry weight) | Anti-inflammation, ameliorating insulin resistance, suppressing lipid accumulation, regulation of gut microbiota | [74] |
| Polysaccharide extracts, main components n.s. | Commercialized spray dried mycelia; 91.48% (w/w dry weight) | Improving intestinal barrier functions | [75] | ||
| Arabinose, galactose, glucose, xylose | Whole mushroom; yield n.s. | Anti-tumor | [76] | ||
| Polysaccharide extracts, main components n.s. | Dried conidial powder; 2% (w/w dry weight, crude extracts) | Promote cognitive function and neural progenitor proliferation | [77] | ||
| Lentinula edodes | Shiitake mushroom | Glucose, galactose, mannose, arabinose | Fruiting body; 1.3% (w/w dry weight, purified polysaccharide cLEP1) | Therapeutic to cervical carcinoma | [78] |
| Rhamnose | Residue/byproduct; yield n.s. | Anti-inflammatory, anti-oxidant | [79] | ||
| Pyranose, β-d-glucans (β-(1→3)-D-glucose as backbone & β-(1→6)-D-glucose as side chains) | Dried fruiting body; 0.76% (w/w dry weight) | Anti-tumor | [80] | ||
| Mannogalactoglucan-type polysaccharides WPLE-N-2, WPLE-A0.5-2 | Fruiting body; yield n.s. | Anti-cancer, immunomodulatory | [81] | ||
| Lentinan (β-(1,3)-glucan with β-(1,6) branches) | Dried fruiting body (commercial product); 2.6% (w/w dry weight) | Anti-tumor | [82] | ||
| Mannitol, trehalose, arabinose | Dried powder; 23.3% (mannitol), 13.2% (trehalose), 1.79% (arabinose) (w/w dry weight) | n.s. | [83] | ||
| Pleurotus eryngii |
King oyster mushroom |
Mannose, glucose, galactose | Fresh whole-mushroom; 5.4% (w/w dry weight) | Anti-tumor | [84] |
| Heteropolysaccharides, novel fractions PEPE-1, PEPE-2, PEPE-3 (mannose, glucose, galactose, xylose) | Fresh mushroom residue; yield n.s. | Anti-tumor | [85] | ||
| Mannose, glucose, galactose | Fresh whole-mushroom; 28.3% (w/w dry weight) | Immunomodulatory | [86] | ||
| Pleurotus ostreatus |
Oyster mushroom | Crude polysaccharide extracts | Fresh whole-mushroom; 61% (w/w) | Alleviation of cognitive impairment | [87] |
| Crude polysaccharide extracts | Fresh whole-mushroom; 63.98% (w/w) | Regulation of dislipidemia | [88] | ||
| Homogeneous polysaccharides, fractions POMP1, POMP2, POMP3 | Mycelia; yield n.s. | Anti-tumor | [89] |
n.s., not specified; Abnp, Agaricus bisporus polysaccharides between 5 kDa and 100 kDa; Abap, Agaricus bisporus polysaccharides under 5 kDa; AcAPS, purified fractions of acidic-extractable polysaccharides; WPLE, mannogalactoglucan-type polysaccharides from Lentinus edodes; POMP, Pleurotus ostreatus mycelium polysaccharide.
3.1.2. Proteins
Compared to other food sources, mushrooms contain higher levels of bioactive proteins such as lectins, ribosome inactivating proteins, fungal immunomodulatory proteins, and laccases, which possess various biological activities (Table 2) including antioxidative, immunomodulatory, anti-inflammatory, and anti-cancer properties [90]. Lectins are non-immune proteins or glycoproteins that bind to specific carbohydrates on cell surfaces, acting as nutraceuticals with immunomodulatory, anti-tumor, and anti-proliferative properties [90]. Other mushroom proteins, such as laccase, fungal immunomodulatory protein, and ribosome inactivating proteins, have distinct bioactive activities. Laccases are considered multicopper oxidases implicated in processes such as pathogenesis, morphogenesis, and immunogenesis of an organism [90]. Fungal immunomodulatory proteins purified from mushrooms, such as Ganoderma lucidum, Ganoderma tsugae, Poria cocos, and Trametes versicolor, have been suggested as potential adjuvants for tumor therapy due to their structural similarity to human antibodies and their ability to suppress tumor metastasis and invasion [91][92][93][94][95].
Table 2. Bioactive proteins in mushrooms.
| Mushrooms | Common Names |
Bioactive Compounds/Substances * |
Bioactivities | References |
|---|---|---|---|---|
| Agaricus bisporus | Button mushroom | Lectin | Immunomodulatory | [96] |
| Cerrena unicolor | Mossy maze polypore | Laccase | Anti-tumor | [97] |
| Coprinus comatus | Shaggy mane/chicken drumstick mushroom | Laccase | Anti-viral | [98] |
| Flammulina velutipes | Enoki/Golden needle mushroom | FIP | Anti-inflammatory | [99] |
| RIP | Anti-viral | [100] | ||
| Ganoderma applanatum | Artist’s conk | Lectin | Anti-tumor | [101] |
| Ganoderma lucidum | Lingzhi | Laccase | Anti-viral | [102] |
| Ganoderma tsugae | Hemlock reishi | FIP | Immunomodulatory | [103] |
| Hypsizygus marmoreus | Jade mushroom | RIPs (hypsin, marmorin) | Anti-fungal, anti-tumor | [104][105] |
| Inonotus baumii | Sanghuang | Laccase | Anti-tumor | [106] |
| Macrolepiota procera | Parasol mushroom | Lectin | Anti-tumor | [107] |
| Pleurotus cornucopiae | Golden oyster | Laccase | Anti-viral, anti-tumor | [108] |
| Pleurotus eryngii | King oyster mushroom | Laccase | Anti-viral | [109] |
| Pleurotus ostreatus | Oyster mushroom | Lectin | Immunomodulatory | [110] |
| Sparassis latifolia | Cauliflower mushroom | Lectin | Anti-fungal, anti-bacteria | [111] |
* Include various categories and sub-categories of proteins. FIP, fungal immunomodulatory protein. RIP, ribosome inactivating protein.
The ribosome inactivating protein family acts as rRNA N-glycosylase, inactivating 60S ribosomal subunits through an N-glycosidic cleavage that eliminates one or more adenosine residues from rRNA to inhibit protein synthesis [112]. Members of the ribosome inactivating protein family, such as trichosanthin, luffin, ricin, and abrin, have been of considerable interest due to their potent activity against viral infections and their potential use as immunotoxins for cancer treatment by conjugating with monoclonal antibodies [113][114][115]. However, it is noteworthy that some mushroom ribosome inactivating proteins may be hazardous and pose adverse effects on health. For instance, hypsin from Hypsizigus mamoreus has been reported to increase in vitro cell death [116]. Therefore, it is important to elucidate the structure-functional properties of mushroom proteins as they may be toxic to humans when consumed. Table 2 lists various bioactive proteins derived from mushrooms.
3.1.3. Lipids
Although mushrooms have a low fat content ranging from 0.1% to 16.3%, they are a good source of high-quality essential fatty acids such as oleic acid (1–60.3% of total fatty acids in 100 g), linoleic acid (0–81.1% of total fatty acids in 100 g), and linolenic acid (0–28.8% of total fatty acids in 100 g) [117]. Table 3 summarizes the lipid profiles of various mushrooms in terms of the content of saturated fatty acids (SFA), monounsaturated fatty acids (MUFA), and polyunsaturated fatty acids (PUFA). Mushrooms are good sources of unsaturated fatty acids, as observed in a study by Günç Ergönül et al. [118] who investigated the fatty acid compositions of six wild edible mushroom species and found that unsaturated fatty acids predominated over saturated ones. In most nutritional characterization studies, mushroom fatty acids are commonly determined using gas-liquid chromatography coupled with a flame ionization detector. However, the sample extraction method used prior to measurement may impact the final outcome of lipid profiles. For instance, a study by Sinanoglou et al. [119] investigated the lipid profiles of Laetiporus sulphureus using different combinations of extraction methods and two individual solvents and found variations among the four combinations [119]. Ergosterol, the major sterol found in mushrooms, accounts for the major lipid component of fungal extracellular vesicles as well [120]. Ergosterol extracted from medicinal mushroom Ganoderma lucidum has been shown to exert anti-oxidant effects and reduce the risk of cardiovascular diseases while extending lifespan [55][121][122]. Compared to lipids from animal sources, edible mushrooms are advantageous due to their high levels of polyunsaturated fatty acids, which may regulate various physiological functions in age-related diseases, such as decreasing blood pressure and triglyceride levels, and reducing the risks of age-related cardiovascular diseases, arthritis, and neurodegenerative diseases [64][123]. Therefore, mushrooms may play a significant role in human nutrition and anti-aging regimens based on their fatty acid profiles.
Table 3. Lipid profiles in mushrooms.
| Mushrooms | Common Name | Total SFA (% of Total FA) |
Total MUFA (% of Total FA) |
Total PUFA (% of Total FA) |
Measurement Techniques | References |
|---|---|---|---|---|---|---|
| Agaricus blazei | Almond mushroom | 24.4 | 2.0 | 73.6 | GC-FID | [83] |
| Agaricus bisporus | White button mushroom | 20.3 | 1.4 | 78.3 | Capillary GLC-FID | [70] |
| Brown button mushroom | 18.4 | 1.8 | 79.8 | |||
| Agrocybe cylindracea | Poplar mushroom | 28.1 | 2.83 | 69.1 | Capillary GLC-FID | [124] |
| Boletus reticulatus | Summer cep | 21.1 | 40.3 | 38.4 | GLC-FID | [118] |
| Coprinus comatus | Shaggy mane/Lawyer’s cap | 23.8 | 11.4 | 64.8 | Capillary GLC-FID | [124] |
| Flammulina velutipes | Enoki/Golden needle mushroom | 18.5 | 7.2 | 74.3 | Capillary GLC-FID | [70] |
| 20.7 | 18.6 | 60.7 | GLC-FID | [118] | ||
| Lactarius deliciocus | Saffron milkcap | 20.8 | 42.0 | 37.3 | Capillary GLC-FID | [124] |
| Lactarius salmonicolor | Salmon milkcap | 19.0 | 19.6 | 61.6 | GLC-FID | [118] |
| Lentinus edodes | Shiitake mushroom | 16.7 | 3.5 | 79.8 | GC-FID | [83] |
| 15.1 | 2.9 | 82.0 | Capillary GLC-FID | [70] | ||
| Pleurotus eryngii | King oyster mushroom | 17.4 | 13.1 | 69.4 | Capillary GLC-FID | [70] |
| Pleurotus ostreatus | Oyster mushroom | 17.0 | 13.6 | 69.4 | Capillary GLC-FID | [70] |
| 21.8 | 11.4 | 66.5 | GLC-FID | [118] | ||
| Polyporus squamosus | Dryad’s saddle | 25.2 | 34.3 | 40.6 | GLC-FID | [118] |
| Russula anthracina | - | 23.7 | 53.3 | 22.9 | GLC-FID | [118] |
| Laetiporus sulphureus | Sulphur polypore | 21.6 | 17.6 | 60.8 | GC-FID, TLC-FID |
[119] |
| Suillus collinitus | - | 17.5 | 34.4 | 47.4 | Capillary GLC-FID | [124] |
| Tricholoma myomyces | Grey knight mushroom | 15.8 | 46.3 | 37.8 | Capillary GLC-FID | [124] |
SFA, saturated fatty acid. MUFA, monosaturated fatty acid. PUFA, polysaturated fatty acid. GC-FID, gas chromatography coupled with flame ionization detector. GLC-FID, gas-liquid chromatography coupled with flame ionization detection. TLC-FID, thin layer chromatography–flame ionization detection.
3.1.4. Phenolic Compounds
Phenolic compounds found in mushrooms are typically considered secondary metabolites. The most prominent phenolic compounds in mushrooms include heteroglycans, lectins, phenolic acids (such as ferulic, gallic, and cinnamic acids), flavonoids (including hesperetin, quercetin, kaempferol, and naringenin), steroids, alkaloids, tannins, chitinous substances, terpenoids, and tocopherols. These compounds exhibit various biological activities, including anti-oxidant, anti-tumor, anti-inflammatory, anti-hyperglycemic, anti-osteoporotic, anti-tyrosinase, and anti-microbial effects, primarily due to their strong antioxidative properties [125][126][127][128]. Some of the preferred mushroom species for extracting phenolic compounds include Agaricus brasiliensis (almond mushroom), Cantharellus cibarius (chanterelle), Lactarius indigo (indigo milk cap), Inonotus obliquus (chaga mushroom), and Melanoleuca cognate [126][129][130][131]. Table 4 provides a summary of representative phenolic compounds extracted from various mushroom species.
Table 4. Extractable phenolic compounds in mushrooms.
| Phenolic Compound Categories |
Phenolic Compounds |
Mushroom Sources | References |
|---|---|---|---|
| Phenolic acids | Ferulic acid | Agaricus brasiliensis, Agrocybe aegerita, Calocybe indica, Cantharellus cibarius | [126][127][132][133][134] |
| Gallic acid | Agaricus brasiliensis, Agrocybe aegerita, Calocybe indica, Cantharellus cibarius, Ganoderma lucidum, Pleurotus citrinopileatus, Pleurotus pulmonarius, Russula aurora | [126][130][132][133][134][135][136][137][138] | |
| Cinnamic acid | Amanita crocea, Ganoderma lucidum, Pleurotus ostreatus, Suilus belinii | [135][139][140][141] | |
| Caffeic acid | Calocybe indica, Cantharellus cibarius, Hyphodontia paradoxa, Inonotus obliquus, Pleurotus citrinopileatus, Pleurotus pulmonarius, | [127][130][133][134][142][143] | |
| p-Coumaric acid | Agaricus brasiliensis, Agaricus subrufescens, Amanita crocea, Hyphodontia paradoxa, Laccaria amethystea, Melanoleuca cognate, Pleurotus ostreatus | [56][126][129][139][140][142][144] | |
| p-Hydroxybenzoic acid | Agaricus brasilensis, Amanita crocea, Cantharellus cibarius, Lactarius indigo, Lentinus edodes, Melanoleuca cognate, Suillus belinii | [126][129][134][138][139][141] | |
| Fumaric acid | Agaricus brasiliensis | [126] | |
| Vanillic acid | Morchella esculenta (L.) Pers., Russula emetic | [136][137] | |
| Syringic acid | Hyphodontia paradoxa, Morchella esculenta (L.) Pers. | [129][130][136][142] | |
| Protocatechuic acid | Agrocybe aegerita, Calocybe indica, Cantharellus cibarius, Hyphodontia paradoxa, Inonotus obliquus, Melanoleuca, Morchella esculenta (L.) Pers., Suillus belinii, Russula emetic | [129][130][132][133][134][136][137][141][142] | |
| Rosmarinic acid | Hyphodontia paradoxa, Russula aurora, Russula emetic | [137][142][145] | |
| Flavonoids | Quercetin | Ganoderma lucidum, Laccaria amethystea, Pleurotus citrinopileatus, | [135][143] |
| Kaempferol | Ganoderma lucidum, Lactarius indigo | [135][146] | |
| Hesperetin | Calocybe indica, Ganoderma lucidum | [133][135] | |
| Naringenin | Calocybe indica, Ganoderma lucidum | [133][135] | |
| Catechin | Laccaria amethystea, Russula emetic | [137][144] | |
| Myricetin | Cantharellus cibarius, Lactarius indigo | [134][146] | |
| Procyanidin | Lactarius indigo | [146] | |
| Rutin | Pleurotus citrinopileatus, Russula emetic | [137][143] | |
| Tannins | Tannic acid | Agaricus silvaticus, Hydnum rufescens, Meripilus giganteus, Pleurotus citrinopileatus, Pleurotus ostreatus, Pleurotus tuber-regium(fries) | [147][148][149] |
| Tocopherols | α-Tocopherol | Agaricus bisporus, Boletus badius, Lepista inversa, Pleurotus ostreatus, Russula delica | [150][151] |
| β-Tocopherol | Laccaria laccata | [150] | |
| γ-Tocopherol | Clitocybe alexandri | [150] | |
| δ-Tocopherol | Lepista inversa | [150] |
This entry is adapted from the peer-reviewed paper 10.3390/jof10030215
References
- Murray, C.; Barber, R.M.; Foreman, K.J.; Ozgoren, A.A.; Abdallah, F.; Abera, S.F.; Aboyans, V.; Abraham, J.P.; Abubakar, I.; Aburaddad, L.J. Global, regional, and national disability-adjusted life years (DALYs) for 306 diseases and injuries and healthy life expectancy (HALE) for 188 countries, 1990–2013: Quantifying the epidemiological transition. Lancet 2015, 386, 2145–2191.
- Foreman, K.J.; Marquez, N.; Dolgert, A.; Fukutaki, K.; Fullman, N.; McGaughey, M.; Pletcher, M.A.; Smith, A.E.; Tang, K.; Yuan, C.W.; et al. Forecasting life expectancy, years of life lost, and all-cause and cause-specific mortality for 250 causes of death: Reference and alternative scenarios for 2016-40 for 195 countries and territories. Lancet 2018, 392, 2052–2090.
- World Health Organization. Preventing chronic diseases: A vital investment. Prev. Chronic Dis. A Vital Invest. 2008, 126, 95.
- de Cabo, R.; Mattson, M.P. Effects of intermittent fasting on health, aging, and disease. N. Engl. J. Med. 2019, 381, 2541–2551.
- Dominguez, L.J.; Veronese, N.; Baiamonte, E.; Guarrera, M.; Parisi, A.; Ruffolo, C.; Tagliaferri, F.; Barbagallo, M. Healthy aging and dietary patterns. Nutrients 2022, 14, 889.
- Gao, Y.; Wei, Y.; Wang, Y.; Gao, F.; Chen, Z. Lycium barbarum: A traditional Chinese herb and a promising anti-aging agent. Aging Dis. 2017, 8, 778–791.
- Kunugi, H.; Mohammed Ali, A. Royal jelly and its components promote healthy aging and longevity: From animal models to humans. Int. J. Mol. Sci. 2019, 20, 4662.
- Hetland, G.; Johnson, E.; Lyberg, T.; Bernardshaw, S.; Tryggestad, A.M.A.; Grinde, B. Effects of the medicinal mushroom Agaricus blazei Murill on immunity, infection and cancer. Scand. J. Immunol. 2008, 68, 157015.
- Im, K.H.; Nguyen, T.K.; Choi, J.; Lee, T.S. In vitro antioxidant, anti-diabetes, anti-dementia, and inflammation inhibitory effect of Trametes pubescens fruiting body extracts. Molecules 2016, 21, 639.
- Lee, J.E.; Lee, I.S.; Kim, K.C.; Yoo, I.D.; Yang, H.M. ROS scavenging and anti-wrinkle effects of clitocybin A isolated from the mycelium of the mushroom Clitocybe aurantiaca. J. Microbiol. Biotechnol. 2017, 27, 933–938.
- Yuan, F.; Gao, Z.; Liu, W.; Li, H.; Zhang, Y.; Feng, Y.; Song, X.; Wang, W.; Zhang, J.; Huang, C.; et al. Characterization, antioxidant, anti-aging and organ protective effects of sulfated polysaccharides from Flammulina velutipes. Molecules 2019, 24, 3517.
- Jo Feeney, M.; Miller, A.M.; Roupas, P. Mushrooms-biologically distinct and nutritionally unique: Exploring a “Third Food Kingdom”. Nutr. Today 2014, 49, 301–307.
- Elkhateeb, W.A. What medicinal mushroom can do? J. Chem. Res. 2020, 5, 106–118.
- Franceschi, C.; Garagnani, P.; Morsiani, C.; Conte, M.; Santoro, A.; Grignolio, A.; Monti, D.; Capri, M.; Salvioli, S. The continuum of aging and age-related diseases: Common mechanisms but different rates. Front. Med. 2018, 5, 61.
- Harman, D. The aging process. Proc. Natl. Acad. Sci. USA 1981, 78, 7124–7128.
- Lopez-Otin, C.; Blasco, M.A.; Partridge, L.; Serrano, M.; Kroemer, G. The hallmarks of aging. Cell 2013, 153, 1194–1217.
- Harman, D. Aging: A theory based on free radical and radiation chemistry. J. Gerontol. 1956, 11, 298–300.
- Harman, D. Free radical theory of aging: Dietary implications. Am. J. Clin. Nutr. 1972, 25, 839–843.
- Van Remmen, H.; Ikeno, Y.; Hamilton, M.; Pahlavani, M.; Wolf, N.; Thorpe, S.R.; Alderson, N.L.; Baynes, J.W.; Epstein, C.J.; Huang, T.T.; et al. Life-long reduction in MnSOD activity results in increased DNA damage and higher incidence of cancer but does not accelerate aging. Physiol. Genom. 2003, 16, 29–37.
- Chen, Q.; Xu, B.J.; Huang, W.S.; Amrouche, A.T.; Maurizio, B.; Simal-Gandara, J.; Tundis, R.; Xiao, J.B.; Zou, L.; Lu, B.Y. Edible flowers as functional raw materials: A review on anti-aging properties. Trends Food Sci. Technol. 2020, 106, 30–47.
- Liguori, I.; Russo, G.; Curcio, F.; Bulli, G.; Aran, L.; Della-Morte, D.; Gargiulo, G.; Testa, G.; Cacciatore, F.; Bonaduce, D.; et al. Oxidative stress, aging, and diseases. Clin. Interv. Aging 2018, 13, 757–772.
- Belikov, A.V. Age-related diseases as vicious cycles. Ageing Res. Rev. 2019, 49, 11–26.
- Lopez-Otin, C.; Galluzzi, L.; Freije, J.M.P.; Madeo, F.; Kroemer, G. Metabolic control of longevity. Cell 2016, 166, 802–821.
- Fontana, L.; Partridge, L.; Longo, V.D. Extending healthy life span—From yeast to humans. Science 2010, 328, 321–326.
- Lee, S.H.; Min, K.J. Caloric restriction and its mimetics. BMB Rep. 2013, 46, 181–187.
- McCay, C.M.; Crowell, M.F.; Maynard, L.A. The effect of retarded growth upon the length of life span and upon the ultimate body size. 1935. Nutrition 1989, 5, 155–171, discussion 172.
- Martins, I.; Galluzzi, L.; Kroemer, G. Hormesis, cell death and aging. Aging 2011, 3, 821–828.
- Kenyon, C.J. The genetics of ageing. Nature 2010, 464, 504–512.
- Miller, R.A. Cell stress and aging: New emphasis on multiplex resistance mechanisms. J. Gerontol. A Biol. Sci. Med. Sci. 2009, 64, 179–182.
- Cava, E.; Fontana, L. Will calorie restriction work in humans? Aging 2013, 5, 507–514.
- de Magalhaes, J.P. The scientific quest for lasting youth: Prospects for curing aging. Rejuvenation Res. 2014, 17, 458–467.
- Dirks, A.J.; Leeuwenburgh, C. Caloric restriction in humans: Potential pitfalls and health concerns. Mech. Ageing Dev. 2006, 127, 1–7.
- Di Francesco, A.; Di Germanio, C.; Bernier, M.; de Cabo, R. A time to fast. Science 2018, 362, 770–775.
- Mattson, M.P.; Moehl, K.; Ghena, N.; Schmaedick, M.; Cheng, A. Intermittent metabolic switching, neuroplasticity and brain health. Nat. Rev. Neurosci. 2018, 19, 63–80.
- Hu, D.; Xie, F.; Xiao, Y.; Lu, C.; Zhong, J.; Huang, D.; Chen, J.; Wei, J.; Jiang, Y.; Zhong, T. Metformin: A potential candidate for targeting aging mechanisms. Aging Dis. 2021, 12, 480–493.
- Zhang, Y.; Zhang, J.; Wang, S. The role of rapamycin in healthspan extension via the delay of organ aging. Ageing Res. Rev. 2021, 70, 101376.
- Li, J.; Kim, S.G.; Blenis, J. Rapamycin: One drug, many effects. Cell Metab. 2014, 19, 373–379.
- Soukas, A.A.; Hao, H.; Wu, L. Metformin as anti-aging therapy: Is it for everyone? Trends Endocrinol. Metab. 2019, 30, 745–755.
- Martel, J.; Ko, Y.F.; Liau, J.C.; Lee, C.S.; Ojcius, D.M.; Lai, H.C.; Young, J.D. Myths and realities surrounding the mysterious caterpillar fungus. Trends Biotechnol. 2017, 35, 1017–1021.
- Martel, J.; Ko, Y.F.; Ojcius, D.M.; Lu, C.C.; Chang, C.J.; Lin, C.S.; Lai, H.C.; Young, J.D. Immunomodulatory properties of plants and mushrooms. Trends Pharmacol. Sci. 2017, 38, 967–981.
- Martel, J.; Ojcius, D.M.; Chang, C.J.; Lin, C.S.; Lu, C.C.; Ko, Y.F.; Tseng, S.F.; Lai, H.C.; Young, J.D. Anti-obesogenic and antidiabetic effects of plants and mushrooms. Nat. Rev. Endocrinol. 2017, 13, 149–160.
- Petrovska, B.B. Historical review of medicinal plants’ usage. Pharmacogn. Rev. 2012, 6, 1–5.
- Kiely, K.M.; Brady, B.; Byles, J. Gender, mental health and ageing. Maturitas 2019, 129, 76–84.
- Cedrone, F.; Catalini, A.; Stacchini, L.; Berselli, N.; Caminiti, M.; Mazza, C.; Cosma, C.; Minutolo, G.; Di Martino, G. The role of gender in the association between mental health and potentially preventable hospitalizations: A single-center retrospective observational study. Int. J. Environ. Res. Public Health 2022, 19, 14691.
- Rinsky-Halivni, L.; Brammli-Greenberg, S.; Christiani, D.C. Ageing workers’ mental health during COVID-19: A multilevel observational study on the association with the work environment, perceived workplace safety and individual factors. BMJ Open 2022, 12, e064590.
- Bockting, W.; Coleman, E.; Deutsch, M.B.; Guillamon, A.; Meyer, I.; Meyer, W., 3rd; Reisner, S.; Sevelius, J.; Ettner, R. Adult development and quality of life of transgender and gender nonconforming people. Curr. Opin. Endocrinol. Diabetes Obes. 2016, 23, 188–197.
- Thomas Tobin, C.S.; Erving, C.L.; Hargrove, T.W.; Satcher, L.A. Is the Black-White mental health paradox consistent across age, gender, and psychiatric disorders? Aging Ment. Health 2022, 26, 196–204.
- Zhang, X.; Yan, Y.; Ye, Z.; Xie, J. Descriptive analysis of depression among adolescents in Huangshi, China. BMC Psychiatry 2023, 23, 176.
- Ulep, V.G.T.; Uy, J.; Casas, L.D. What explains the large disparity in child stunting in the Philippines? A decomposition analysis. Public Health Nutr. 2022, 25, 2995–3007.
- Xu, C.; Ganesan, K.; Liu, X.; Ye, Q.; Cheung, Y.; Liu, D.; Zhong, S.; Chen, J. Prognostic value of negative emotions on the incidence of breast cancer: A systematic review and meta-analysis of 129,621 patients with breast cancer. Cancers 2022, 14, 475.
- Lorenzo, E.C.; Kuchel, G.A.; Kuo, C.L.; Moffitt, T.E.; Diniz, B.S. Major depression and the biological hallmarks of aging. Ageing Res. Rev. 2023, 83, 101805.
- Yeap, B.B. Hormonal changes and their impact on cognition and mental health of ageing men. Maturitas 2014, 79, 227–235.
- Barros, L.; Correia, D.M.; Ferreira, I.C.; Baptista, P.; Santos-Buelga, C. Optimization of the determination of tocopherols in Agaricus sp. edible mushrooms by a normal phase liquid chromatographic method. Food Chem. 2008, 110, 1046–1050.
- Mattila, P.; Konko, K.; Eurola, M.; Pihlava, J.M.; Astola, J.; Vahteristo, L.; Hietaniemi, V.; Kumpulainen, J.; Valtonen, M.; Piironen, V. Contents of vitamins, mineral elements, and some phenolic compounds in cultivated mushrooms. J. Agric. Food Chem. 2001, 49, 2343–2348.
- Kalac, P. A review of chemical composition and nutritional value of wild-growing and cultivated mushrooms. J. Sci. Food Agric. 2013, 93, 209–218.
- Ferreira, I.C.; Barros, L.; Abreu, R.M. Antioxidants in wild mushrooms. Curr. Med. Chem. 2009, 16, 1543–1560.
- Wasser, S.P. Current findings, future trends, and unsolved problems in studies of medicinal mushrooms. Appl. Microbiol. Biotechnol. 2011, 89, 1323–1332.
- Valverde, M.E.; Hernandez-Perez, T.; Paredes-Lopez, O. Edible mushrooms: Improving human health and promoting quality life. Int. J. Microbiol. 2015, 2015, 376387.
- Falch, B.H.; Espevik, T.; Ryan, L.; Stokke, B.T. The cytokine stimulating activity of (1→3)-beta-D-glucans is dependent on the triple helix conformation. Carbohydr. Res. 2000, 329, 587–596.
- Kataoka, K.; Muta, T.; Yamazaki, S.; Takeshige, K. Activation of macrophages by linear (1→3)-beta-D-glucans. Impliations for the recognition of fungi by innate immunity. J. Biol. Chem. 2002, 277, 36825–36831.
- Khan, M.A.; Tania, M.; Liu, R.; Rahman, M.M. Hericium erinaceus: An edible mushroom with medicinal values. J. Complement. Integr. Med. 2013, 10, 253–258.
- Vetvicka, V.; Yvin, J.C. Effects of marine beta-1,3 glucan on immune reactions. Int. Immunopharmacol. 2004, 4, 721–730.
- Zaidman, B.Z.; Yassin, M.; Mahajna, J.; Wasser, S.P. Medicinal mushroom modulators of molecular targets as cancer therapeutics. Appl. Microbiol. Biotechnol. 2005, 67, 453–468.
- Heleno, S.A.; Barros, L.; Martins, A.; Queiroz, M.J.; Santos-Buelga, C.; Ferreira, I.C. Phenolic, polysaccharidic, and lipidic fractions of mushrooms from northeastern Portugal: Chemical compounds with antioxidant properties. J. Agric. Food Chem. 2012, 60, 4634–4640.
- Huang, J.; Ou, Y.; Yew, T.W.; Liu, J.; Leng, B.; Lin, Z.; Su, Y.; Zhuang, Y.; Lin, J.; Li, X.; et al. Hepatoprotective effects of polysaccharide isolated from Agaricus bisporus industrial wastewater against CCl₄-induced hepatic injury in mice. Int. J. Biol. Macromol. 2016, 82, 678–686.
- Li, S.; Liu, H.; Wang, W.; Wang, X.; Zhang, C.; Zhang, J.; Jing, H.; Ren, Z.; Gao, Z.; Song, X.; et al. Antioxidant and anti-aging effects of acidic-extractable polysaccharides by Agaricus bisporus. Int. J. Biol. Macromol. 2018, 106, 1297–1306.
- Zhang, Y.; Ma, G.; Fang, L.; Wang, L.; Xie, J. The immunostimulatory and anti-tumor activities of polysaccharide from Agaricus bisporus (brown). J. Food Nutr. Res. 2014, 2, 122–126.
- Pires, A.; Ruthes, A.C.; Cadena, S.; Iacomini, M. Cytotoxic effect of a mannogalactoglucan extracted from Agaricus bisporus on HepG2 cells. Carbohydr. Polym. 2017, 170, 33–42.
- Smiderle, F.R.; Alquini, G.; Tadra-Sfeir, M.Z.; Iacomini, M.; Wichers, H.J.; Van Griensven, L.J. Agaricus bisporus and Agaricus brasiliensis (1→6)-β-D-glucans show immunostimulatory activity on human THP-1 derived macrophages. Carbohydr. Polym. 2013, 94, 91–99.
- Reis, F.S.; Barros, L.; Martins, A.; Ferreira, I.C. Chemical composition and nutritional value of the most widely appreciated cultivated mushrooms: An inter-species comparative study. Food Chem. Toxicol. 2012, 50, 191–197.
- Govindan, S.; Johnson, E.E.; Christopher, J.; Shanmugam, J.; Thirumalairaj, V.; Gopalan, J. Antioxidant and anti-aging activities of polysaccharides from Calocybe indica var. APK2. Exp. Toxicol. Pathol. 2016, 68, 329–334.
- Chen, G.T.; Fu, Y.X.; Yang, W.J.; Hu, Q.H.; Zhao, L.Y. Effects of polysaccharides from the base of Flammulina velutipes stipe on growth of murine RAW264.7, B16F10 and L929 cells. Int. J. Biol. Macromol. 2018, 107, 2150–2156.
- Yang, W.; Yu, J.; Zhao, L.; Ma, N.; Fang, Y.; Pei, F.; Mariga, A.M.; Hu, Q. Polysaccharides from Flammulina velutipes improve scopolamine-induced impairment of learning and memory of rats. J. Funct. Foods 2015, 18, 411–422.
- Xu, S.; Dou, Y.; Ye, B.; Wu, Q.; Wang, Y.; Hu, M.; Ma, F.; Rong, X.; Guo, J. Ganoderma lucidum polysaccharides improve insulin sensitivity by regulating inflammatory cytokines and gut microbiota composition in mice. J. Funct. Foods 2017, 38, 545–552.
- Jin, M.; Zhu, Y.; Shao, D.; Zhao, K.; Xu, C.; Li, Q.; Yang, H.; Huang, Q.; Shi, J. Effects of polysaccharide from mycelia of Ganoderma lucidum on intestinal barrier functions of rats. Int. J. Biol. Macromol. 2017, 94, 1–9.
- Yang, G.; Yang, L.; Zhuang, Y.; Qian, X.; Shen, Y. Ganoderma lucidum polysaccharide exerts anti-tumor activity via MAPK pathways in HL-60 acute leukemia cells. J. Recept. Signal Transduct. Res. 2016, 36, 6–13.
- Huang, S.; Mao, J.; Ding, K.; Zhou, Y.; Zeng, X.; Yang, W.; Wang, P.; Zhao, C.; Yao, J.; Xia, P.; et al. Polysaccharides from Ganoderma lucidum promote cognitive function and neural progenitor proliferation in mouse model of Alzheimer’s disease. Stem Cell Rep. 2017, 8, 84–94.
- Ya, G. A Lentinus edodes polysaccharide induces mitochondrial-mediated apoptosis in human cervical carcinoma HeLa cells. Int. J. Biol. Macromol. 2017, 103, 676–682.
- Ren, Z.; Li, J.; Song, X.; Zhang, J.; Wang, W.; Wang, X.; Gao, Z.; Jing, H.; Li, S.; Jia, L. The regulation of inflammation and oxidative status against lung injury of residue polysaccharides by Lentinula edodes. Int. J. Biol. Macromol. 2018, 106, 185–192.
- Wang, J.; Li, W.; Huang, X.; Liu, Y.; Li, Q.; Zheng, Z.; Wang, K. A polysaccharide from Lentinus edodes inhibits human colon cancer cell proliferation and suppresses tumor growth in athymic nude mice. Oncotarget 2017, 8, 610–623.
- Jeff, I.B.; Fan, E.; Tian, M.; Song, C.; Yan, J.; Zhou, Y. In vivo anticancer and immunomodulating activities of mannogalactoglucan-type polysaccharides from Lentinus edodes (Berkeley) Singer. Cent. Eur. J. Immunol. 2016, 41, 47–53.
- Xu, H.; Zou, S.; Xu, X.; Zhang, L. Anti-tumor effect of β-glucan from Lentinus edodes and the underlying mechanism. Sci. Rep. 2016, 6, 288–302.
- Carneiro, A.A.; Ferreira, I.C.; Dueñas, M.; Barros, L.; da Silva, R.; Gomes, E.; Santos-Buelga, C. Chemical composition and antioxidant activity of dried powder formulations of Agaricus blazei and Lentinus edodes. Food Chem. 2013, 138, 2168–2173.
- Ren, D.; Wang, N.; Guo, J.; Yuan, L.; Yang, X. Chemical characterization of Pleurotus eryngii polysaccharide and its tumor-inhibitory effects against human hepatoblastoma HepG-2 cells. Carbohydr. Polym. 2016, 138, 123–133.
- Ma, G.; Yang, W.; Mariga, A.M.; Fang, Y.; Ma, N.; Pei, F.; Hu, Q. Purification, characterization and antitumor activity of polysaccharides from Pleurotus eryngii residue. Carbohydr. Polym. 2014, 114, 297–305.
- Xu, D.; Wang, H.; Zheng, W.; Gao, Y.; Wang, M.; Zhang, Y.; Gao, Q. Charaterization and immunomodulatory activities of polysaccharide isolated from Pleurotus eryngii. Int. J. Biol. Macromol. 2016, 92, 30–36.
- Zhang, Y.; Yang, X.; Jin, G.; Yang, X.; Zhang, Y. Polysaccharides from Pleurotus ostreatus alleviate cognitive impairment in a rat model of Alzheimer’s disease. Int. J. Biol. Macromol. 2016, 92, 935–941.
- Zhang, Y.; Wang, Z.; Jin, G.; Yang, X.; Zhou, H. Regulating dyslipidemia effect of polysaccharides from Pleurotus ostreatus on fat-emulsion-induced hyperlipidemia rats. Int. J. Biol. Macromol. 2017, 101, 107–116.
- Cao, X.Y.; Liu, J.L.; Yang, W.; Hou, X.; Li, Q.J. Antitumor activity of polysaccharide extracted from Pleurotus ostreatus mycelia against gastric cancer in vitro and in vivo. Mol. Med. Rep. 2015, 12, 2383–2389.
- Xu, X.; Yan, H.; Chen, J.; Zhang, X. Bioactive proteins from mushrooms. Biotechnol. Adv. 2011, 29, 667–674.
- Chang, H.H.; Sheu, F. Anti-tumor mechanisms of orally administered a fungal immunomodulatory protein from Flammulina velutipes in mice. FASEB J. 2006, 20, 297–306.
- Lin, C.H.; Sheu, G.T.; Lin, Y.W.; Yeh, C.S.; Huang, Y.H.; Lai, Y.C.; Chang, J.G.; Ko, J.L. A new immunomodulatory protein from Ganoderma microsporum inhibits epidermal growth factor mediated migration and invasion in A549 lung cancer cells. Process Biochem. 2010, 45, 1537–1542.
- Peek, H.W.; Halkes, S.B.A.; Tomassen, M.M.M.; Mes, J.J.; Landman, W.J.M. In vivo screening of five phytochemicals/extracts and a fungal immunomodulatory protein against colibacillosis in broilers. Avian Pathol. 2013, 42, 235–247.
- Lin, W.H.; Hung, C.H.; Hsu, C.-N.; Lin, J.Y. Dimerization of the N-terminal amphipathic helix domain of the fungal immunomodulatory protein from Ganoderma tsugae defined by a yeast two-hybrid system and site-directed mutagenesis. J. Biol. Chem. 1997, 272, 20044–20048.
- Chang, H.H.; Yeh, C.H.; Sheu, F. A novel immunomodulatory protein from Poria cocos induces toll-like receptor 4-dependent activation within mouse peritoneal macrophages. J. Agric. Food Chem. 2009, 57, 6129–6139.
- Ditamo, Y.; Rupil, L.L.; Sendra, V.G.; Nores, G.A.; Roth, G.A.; Irazoqui, F.J. In vivo immunomodulatory effect of the lectin from edible mushroom Agaricus bisporus. Food Funct. 2016, 7, 262–279.
- Matuszewska, A.; Karp, M.; Jaszek, M.; Janusz, G.; Osińska-Jaroszuk, M.; Sulej, J.; Stefaniuk, D.; Tomczak, W.; Giannopoulos, K. Laccase purified from Cerrena unicolor exerts antitumor activity against leukemic cells. Oncol. Lett. 2016, 11, 2009–2018.
- Zhao, S.; Rong, C.B.; Kong, C.; Liu, Y.; Xu, F.; Miao, Q.J.; Wang, S.X.; Wang, H.X.; Zhang, G.Q. A novel laccase with potent antiproliferative and HIV-1 reverse transcriptase inhibitory activities from mycelia of mushroom Coprinus comatus. Biomed. Res. Int. 2014, 2014, 417461.
- Chu, P.Y.; Sun, H.L.; Ko, J.L.; Ku, M.S.; Lin, L.J.; Lee, Y.T.; Liao, P.F.; Pan, H.H.; Lu, H.L.; Lue, K.H. Oral fungal immunomodulatory protein-Flammulina velutipes has influence on pulmonary inflammatory process and potential treatment for allergic airway disease: A mouse model. J. Microbiol. Immunol. Infect. 2017, 50, 297–306.
- Wang, H.; Ng, T.B. Isolation and characterization of velutin, a novel low-molecular-weight ribosome-inactivating protein from winter mushroom (Flammulina velutipes) fruiting bodies. Life Sci. 2001, 68, 2151–2168.
- Kumaran, S.; Pandurangan, A.K.; Shenbhagaraman, R.; Esa, N.M. Isolation and characterization of lectin from the Artist’s Conk medicinal mushroom, Ganoderma applanatum (Agaricomycetes), and evaluation of its antiproliferative activity in HT-29 colon cancer cells. Int. J. Med. Mushrooms 2017, 19, 675–684.
- Wang, H.X.; Ng, T.B. A laccase from the medicinal mushroom Ganoderma lucidum. Appl. Microbiol. Biotechnol. 2006, 72, 508–513.
- Hsin, I.L.; Wang, S.C.; Li, J.R.; Ciou, T.C.; Wu, C.H.; Wu, H.M.; Ko, J.L. Immunomodulatory proteins FIP-gts and chloroquine induce caspase-independent cell death via autophagy for resensitizing cisplatin-resistant urothelial cancer cells. Phytomedicine 2016, 23, 1566–1573.
- Lam, S.K.; Ng, T.B. Hypsin, a novel thermostable ribosome-inactivating protein with antifungal and antiproliferative activities from fruiting bodies of the edible mushroom Hypsizigus marmoreus. Biochem. Biophys. Res. Commun. 2001, 285, 1071–1085.
- Wong, J.H.; Wang, H.X.; Ng, T.B. Marmorin, a new ribosome inactivating protein with antiproliferative and HIV-1 reverse transcriptase inhibitory activities from the mushroom Hypsizigus marmoreus. Appl. Microbiol. Biotechnol. 2008, 81, 669–674.
- Sun, J.; Chen, Q.J.; Zhu, M.-U.; Wang, H.X.; Zhang, G.Q. An extracellular laccase with antiproliferative activity from the sanghuang mushroom Inonotus baumii. J. Mol. Catal. B Enzym. 2014, 99, 20–25.
- Žurga, S.; Nanut, M.P.; Kos, J.; Sabotič, J. Fungal lectin MpL enables entry of protein drugs into cancer cells and their subcellular targeting. Oncotarget 2017, 8, 26896–26910.
- Wu, X.; Huang, C.; Chen, Q.; Wang, H.; Zhang, J. A novel laccase with inhibitory activity towards HIV-I reverse transcriptase and antiproliferative effects on tumor cells from the fermentation broth of mushroom Pleurotus cornucopiae. Biomed. Chromatogr. 2014, 28, 548–553.
- Wang, H.X.; Ng, T.B. Purification of a laccase from fruiting bodies of the mushroom Pleurotus eryngii. Appl. Microbiol. Biotechnol. 2006, 69, 521–535.
- He, M.; Su, D.; Liu, Q.; Gao, W.; Kang, Y. Mushroom lectin overcomes hepatitis B virus tolerance via TLR6 signaling. Sci. Rep. 2017, 7, 5814.
- Chandrasekaran, G.; Lee, Y.C.; Park, H.; Wu, Y.; Shin, H.J. Antibacterial and antifungal activities of lectin extracted from fruiting bodies of the Korean cauliflower medicinal mushroom, Sparassis latifolia (Agaricomycetes). Int. J. Med. Mushrooms 2016, 18, 291–309.
- May, M.J.; Hartley, M.R.; Roberts, L.M.; Krieg, P.A.; Osborn, R.W.; Lord, J.M. Ribosome inactivation by ricin A chain: A sensitive method to assess the activity of wild-type and mutant polypeptides. EMBO J. 1989, 8, 301–308.
- Zhou, K.; Fu, Z.; Chen, M.; Lin, Y.; Pan, K. Structure of trichosanthin at 1.88 Å resolution. Proteins Struct. Funct. Bioinform. 1994, 19, 4–13.
- Zhu, F.; Zhou, Y.K.; Ji, Z.L.; Chen, X.R. The plant ribosome-inactivating proteins play important roles in defense against pathogens and insect pest attacks. Front. Plant Sci. 2018, 9, 146.
- Domashevskiy, A.V.; Goss, D.J. Pokeweed antiviral protein, a ribosome inactivating protein: Activity, inhibition and prospects. Toxins 2015, 7, 274–298.
- Ng, T.B.; Lam, J.S.; Wong, J.H.; Lam, S.K.; Ngai, P.H.; Wang, H.X.; Chu, K.T.; Chan, W.Y. Differential abilities of the mushroom ribosome-inactivating proteins hypsin and velutin to perturb normal development of cultured mouse embryos. Toxicol. In Vitro 2010, 24, 1250–1257.
- Sande, D.; Oliveira, G.P.d.; Moura, M.A.F.E.; Martins, B.d.A.; Lima, M.T.N.S.; Takahashi, J.A. Edible mushrooms as a ubiquitous source of essential fatty acids. Food Res. Int. 2019, 125, 108524.
- Günç Ergönül, P.; Akata, I.; Kalyoncu, F.; Ergönül, B. Fatty acid compositions of six wild edible mushroom species. Sci. World J. 2013, 2013, 163964.
- Sinanoglou, V.J.; Zoumpoulakis, P.; Heropoulos, G.; Proestos, C.; Ćirić, A.; Petrovic, J.; Glamoclija, J.; Sokovic, M. Lipid and fatty acid profile of the edible fungus Laetiporus sulphurous. Antifungal and antibacterial properties. J. Food Sci. Technol. 2015, 52, 3264–3272.
- Rodrigues, M.L.; Nimrichter, L.; Oliveira, D.L.; Frases, S.; Miranda, K.; Zaragoza, O.; Alvarez, M.; Nakouzi, A.; Feldmesser, M.; Casadevall, A. Vesicular polysaccharide export in Cryptococcus neoformans is a eukaryotic solution to the problem of fungal trans-cell wall transport. Eukaryot. Cell 2007, 6, 48–59.
- Guillamón, E.; García-Lafuente, A.; Lozano, M.; D’Arrigo, M.; Martínez, J. Edible mushrooms: Role in the prevention of cardiovascular diseases. Fitoterapia 2010, 81, 715–723.
- Weng, Y.; Xiang, L.; Matsuura, A.; Zhang, Y.; Huang, Q.; Qi, J. Ganodermasides A and B, two novel anti-aging ergosterols from spores of a medicinal mushroom Ganoderma lucidum on yeast via UTH1 gene. Bioorg. Med. Chem. 2010, 18, 999–1002.
- Saiki, P.; Kawano, Y.; Griensven, L.; Miyazaki, K. The anti-inflammatory effect of Agaricus brasiliensis is partly due to its linoleic acid content. Food Funct. 2017, 8, 4150–4158.
- Ergon, P.; Ergonul, B.; Kalyoncu, F.; Akata, I. Fatty acid compositions of five wild edible mushroom species collected from Turkey. Int. J. Pharmacol. 2012, 8, 463–466.
- Abdelshafy, A.M.; Belwal, T.; Liang, Z.; Wang, L.; Li, D.; Luo, Z.; Li, L. A comprehensive review on phenolic compounds from edible mushrooms: Occurrence, biological activity, application and future prospective. Critic. Rev. Food Sci. Nutr. 2022, 62, 6204–6224.
- Bach, F.; Zielinski, A.A.F.; Helm, C.V.; Maciel, G.M.; Pedro, A.C.; Stafussa, A.P.; Ávila, S.; Haminiuk, C.W.I. Bio compounds of edible mushrooms: In vitro antioxidant and antimicrobial activities. LWT 2019, 107, 214–220.
- Contato, A.G.; Inácio, F.D.; de Araújo, C.A.V.; Brugnari, T.; Maciel, G.M.; Haminiuk, C.W.I.; Bracht, A.; Peralta, R.M.; de Souza, C.G.M. Comparison between the aqueous extracts of mycelium and basidioma of the edible mushroom Pleurotus pulmonarius: Chemical composition and antioxidant analysis. J. Food Meas. Charact. 2020, 14, 830–837.
- Mutukwa, I.B.; Hall, C.A., III; Cihacek, L.; Lee, C.W. Evaluation of drying method and pretreatment effects on the nutritional and antioxidant properties of oyster mushroom (Pleurotus ostreatus). J. Food Process. Preserv. 2019, 43, e13910.
- Bahadori, M.B.; Sarikurkcu, C.; Yalcin, O.U.; Cengiz, M.; Gungor, H. Metal concentration, phenolics profiling, and antioxidant activity of two wild edible Melanoleuca mushrooms (M. cognata and M. stridula). Microchem. J. 2019, 150, 104172.
- Hwang, A.Y.; Yang, S.C.; Kim, J.; Lim, T.; Cho, H.; Hwang, K.T. Effects of non-traditional extraction methods on extracting bioactive compounds from chaga mushroom (Inonotus obliquus) compared with hot water extraction. LWT 2019, 110, 80–84.
- Stojkovic, D.; Smiljkovic, M.; Ciric, A.; Glamoclija, J.; Griensven, L.V.; Ferreira, I.; Sokovic, M. An insight into antidiabetic properties of six medicinal and edible mushrooms: Inhibition of α-amylase and α-glucosidase linked to type-2 diabetes. S. Afr. J. Bot. 2019, 120, 100–103.
- Lin, S.; Ching, L.T.; Lam, K.; Cheung, P.C.K. Anti-angiogenic effect of water extract from the fruiting body of Agrocybe aegerita. LWT 2017, 75, 155–163.
- Alam, N.; Sikder, M.M.; Karim, M.A.; Amin, S. Antioxidant and antityrosinase activities of milky white mushroom. Bangladesh J. Bot. 2019, 48, 1065–1073.
- Palacios, I.; Lozano, M.; Moro, C.; D’Arrigo, M.; Rostagno, M.A.; Martínez, J.A.; García-Lafuente, A.; Guillamón, E.; Villares, A. Antioxidant properties of phenolic compounds occurring in edible mushrooms. Food Chem. 2011, 128, 674–678.
- Veljović, S.; Veljović, M.; Nikićević, N.; Despotović, S.; Radulović, S.; Nikšić, M.; Filipović, L. Chemical composition, antiproliferative and antioxidant activity of differently processed Ganoderma lucidum ethanol extracts. J. Food Sci. Technol. 2017, 54, 1312–1320.
- Gąsecka, M.; Siwulski, M.; Mleczek, M. Evaluation of bioactive compounds content and antioxidant properties of soil-growing and wood-growing edible mushrooms. J. Food Process. Preserv. 2018, 42, e13386.
- Kaewnarin, K.; Suwannarach, N.; Kumla, J.; Lumyong, S. Phenolic profile of various wild edible mushroom extracts from Thailand and their antioxidant properties, anti-tyrosinase and hyperglycaemic inhibitory activities. J. Funct. Foods 2016, 27, 352–364.
- Islam, T.; Yu, X.; Xu, B. Phenolic profiles, antioxidant capacities and metal chelating ability of edible mushrooms commonly consumed in China. LWT 2016, 72, 423–431.
- Alkan, S.; Uysal, A.; Kasik, G.; Vlaisavljevic, S.; Berežni, S.; Zengin, G. Chemical characterization, antioxidant, enzyme inhibition and antimutagenic properties of eight mushroom species: A comparative study. J. Fungi 2020, 6, 166.
- de Souza Campos Junior, F.A.; Petrarca, M.H.; Meinhart, A.D.; de Jesus Filho, M.; Godoy, H.T. Multivariate optimization of extraction and validation of phenolic acids in edible mushrooms by capillary electrophoresis. Food Res. Int. 2019, 126, 108685.
- Souilem, F.; Fernandes, Â.; Calhelha, R.C.; Barreira, J.C.M.; Barros, L.; Skhiri, F.; Martins, A.; Ferreira, I.C.F.R. Wild mushrooms and their mycelia as sources of bioactive compounds: Antioxidant, anti-inflammatory and cytotoxic properties. Food Chem. 2017, 230, 40–48.
- Nowacka-Jechalke, N.; Olech, M.; Nowak, R. Chapter 11—Mushroom polyphenols as chemopreventive agents. In Polyphenols: Prevention and Treatment of Human Disease, 2nd ed.; Watson, R.R., Preedy, V.R., Zibadi, S., Eds.; Academic Press: Cambridge, MA, USA, 2018; pp. 137–150.
- Gogoi, P.; Chutia, P.; Singh, P.; Mahanta, C.L. Effect of optimized ultrasound-assisted aqueous and ethanolic extraction of Pleurotus citrinopileatus mushroom on total phenol, flavonoids and antioxidant properties. J. Food Proc. Engin. 2019, 42, e13172.
- Liu, Y.T.; Sun, J.; Luo, Z.Y.; Rao, S.Q.; Su, Y.J.; Xu, R.R.; Yang, Y.J. Chemical composition of five wild edible mushrooms collected from Southwest China and their antihyperglycemic and antioxidant activity. Food Chem. Toxicol. 2012, 50, 1238–1244.
- Çayan, F.; Deveci, E.; Tel-Çayan, G.; Duru, M.E. Identification and quantification of phenolic acid compounds of twenty-six mushrooms by HPLC–DAD. J. Food Measu. Charact. 2020, 14, 1690–1698.
- Yahia, E.M.; Gutiérrez-Orozco, F.; Moreno-Pérez, M.A. Identification of phenolic compounds by liquid chromatography-mass spectrometry in seventeen species of wild mushrooms in Central Mexico and determination of their antioxidant activity and bioactive compounds. Food Chem. 2017, 226, 14–22.
- Akindahunsi, A.A.; Oyetayo, F.L. Nutrient and antinutrient distribution of edible mushroom, Pleurotus tuber-regium (fries) singer. LWT 2006, 39, 548–553.
- Garrab, M.; Edziri, H.; El Mokni, R.; Mastouri, M.; Mabrouk, H.; Douki, W. Phenolic composition, antioxidant and anticholinesterase properties of the three mushrooms Agaricus silvaticus Schaeff., Hydnum rufescens Pers. and Meripilus giganteus (Pers.) Karst. in Tunisia. S. Afr. J. Bot. 2019, 124, 359–363.
- Pavithra, M.; Sridhar, K.R.; Greeshma, A.A.; Tomita-Yokotani, K. Bioactive potential of the wild mushroom Astraeus hygrometricus in South-west India. Mycology 2016, 7, 191–202.
- Heleno, S.A.; Barros, L.; Sousa, M.J.; Martins, A.; Ferreira, I.C.F.R. Tocopherols composition of Portuguese wild mushrooms with antioxidant capacity. Food Chem. 2010, 119, 1443–1450.
- Jayakumar, T.; Thomas, P.A.; Geraldine, P. In-vitro antioxidant activities of an ethanolic extract of the oyster mushroom, Pleurotus ostreatus. Innov. Food Sci. Emerg. Technol. 2009, 10, 228–234.
This entry is offline, you can click here to edit this entry!
