Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is an old version of this entry, which may differ significantly from the current revision.
BioCholine Powder is a polyherbal feed additive composed of Achyrantes aspera, Trachyspermum ammi, Azadirachta indica, and Citrullus colocynthis.
- nutraceuticals
- polyherbals
- plant feed additives
- choline
- meta-analyses
1. Polyherbal (BioCholine) Characteristics
BioCholine Powder Polyherbal is a polyherbal mix made from Indian species, such as Achyrantes aspera, Trachyspermum ammi, Azadirachta indica, and Citrullus colocynthis, and Andrographis. It is a standardized feed plant additive produced by Indian Herbs Specialties Pvt Ltd. (Uttar Pradesh, India), with certifications including ISO 9001 [1], GMP, and GMP Plus. It is marketed in some European countries and almost all Latin American countries by Nuproxa Switzerland Ltd., (Etoy, Switzerland) [2].
It differs from synthetic protected choline products in that it provides Ptdcho and not choline chloride; besides being phytobiotic, it offers secondary metabolites with different nutraceutical properties [3]. It is not an herb spice extract, so it preserves other active molecules (some with greater predominance) and nutrients (or precursors) that explain the biological effects observed in supplemented animals [4].
Some secondary metabolites found in BioCholine have antimicrobial effects [2][5][6] that may give it resistance to ruminal degradation of choline conjugates. Mendoza et al. [2] analyzed the volatile compounds of the mixture by flash gas chromatography electronic nose. These authors reported 15 relevant compounds, including aromas, alcohols, phenolics, and aldehydes, with compounds that could reduce microbial activity, such as aldehyde β-pins, Trans-2-Undecenal, and 1-propanol. Roque-Jiménez et al. [5] characterized BioCholine Powder using gas chromatography coupled with mass spectrophotometry and reported 19 organic compounds. Some of these compounds belong to the methyl groups hexadecenoic acid methyl ester (C16:0) and octadecenoic acid methyl ester (C18:1 cis 9, 12; C18:1 cis 9; C18:1 cis 8). Orzuna-Orzuna et al. (unpublished data from a doctoral dissertation at Universidad Autónoma Chapingo Postgraduate in Animal Production; Supplementary File S1) used a gas chromatography–mass selective detector and confirmed the predominance of C18:1 cis 9, 12 (58.94%) and the presence of C16:0 (16.24%) and some C18:1 cis 9 (0.98%). These have been associated with fatty acid methyl esters with DNA methyl groups; another important compound is thymol, which can prevent acetylcholine reduction and increase recycling. Some components, such as thymol [7][8][9], Phenol, 4-methoxy-2,3,6-trimethyl [10], and 9.17-Octadecadienal (Z) [11], have antimicrobial activity.
The supplier of the polyherbal reports content of choline conjugates between 1.76 and 1.8%, a higher value than that reported by Leal et al. [12] of 0.978% analyzed with High-performance Thin Layer Chromatography, with total phospholipids of 1.68%. Dazuk et al. [6], using high-performance thin-layer chromatography, reported phosphatidylethanolamine, phosphatidylinositol, phosphatidylcholine, lysophosphatidylcholine, and 16.1% PTCho natural choline conjugates. The polyherbal has other metabolites that can affect the rumen microbiota and the metabolism of ruminants. Roque-Jiménez et al. [5] detected the presence of compounds containing methyl groups, such as hexadecenoic acid methyl ester (C16:0) and octadecenoic acid methyl ester (C18:1 cis-9; C18:1 cis-8), related to the methylation status of DNA. BioCholine also contains tannins and flavonoids [6].
2. Gene Expression
Nutrigenomic studies show the effects of BioCholine expressed at the cellular level, allowing people to explain changes in productive variables, health, and antioxidant status. In the calf experiment [13], changes in leukocyte expression were evaluated by microarrays from blood samples collected on day 60 of the experiment, when the calves consumed 4.95 ± 0.686 kg of starter concentrate per day and the milk had been suspended for 25 days, so the rumen was adapted to eating solids, and the response could be interpreted as calves with a developed rumen (BW 93.21 ± 9.53). Microarray results showed that 1093 genes were upregulated and 1349 were downregulated with BioCholine, with marked changes in 13 genes related to lipid metabolism, 9 genes related to carbohydrate metabolism, and 9 genes related to oxidation–reduction.
The review of expression change in specific genes allows people to explain some beneficial effects of BioCholine; among the genes that were overexpressed were PPARα (peroxisome proliferator-activated receptor Alpha, +2.82), Acoxl (acyl-coenzyme A oxidase-like, +4.0), Gck (glucokinase, +2.06), TGFβ-1 (transforming growth factor, beta 1, +3.40), and Defa 14 (defensin, alpha 14, +3.0), and among those that were under-regulated were G6pc3 (Glucose 6 phosphatase catalytic 3, −3.80), G6pc3 (glucose 6-phosphatase catalytic 3, −3.76), and Ugdh (UDP-glucose dehydrogenase −2.01) [13].
The PPARα gene has implications for the oxidation of fatty acids in ketogenesis, and its effects result in lower blood triglyceride levels [14]. In contrast, the genes related to glucose metabolism are involved in the homeostatic regulation of blood glucose levels [15]. The Acoxl gene is involved in peroxisomal fatty acid beta-oxidation [16] in the metabolism of very long-chain fatty acids [17]. Genes of the TGF-β family have essential roles in tissue development, cell activity, and bone metabolism, as well as in the control of the immunological response, healing process, and inflammatory response [18]. The Defa 14 gene is an antimicrobial peptide found in the skin, intestinal mucosal, and respiratory tract and has been related to antimicrobial and antiviral properties [19]. Ontological analysis and specific genes make it possible to explain the improvements in health and immune response observed in the experiments in which these indicators were evaluated [13][20][21].
Other nutrigenomic evidence derived from a growth-fattening experiment with Pelibuey lambs showed changes detected by microarrays in liver samples, which are indirect indicators of the presence of bypass metabolites (Orzuna-Orzuna et al., unpublished data from the doctoral dissertation). In these lambs, BioCholine differentially modified the expression of 2312 genes, of which 1135 were downregulated and 1177 were upregulated by at least 1.5 times compared to the control lambs. The ontological analysis showed that BioCholine stimulated several pathways, including folate biosynthesis, nucleotide excision repair, oxidative phosphorylation, endocrine resistance, platelet activation, thermogenesis, and chemokine signaling pathway, and reduced others, such as the TGF-beta signaling pathway and fluid shear stress. These pathways provide insights into cellular processes that affect metabolism and health influenced by the polyherbal.
The expression of specific genes provides additional information that helps explain the benefits of the polyherbal. The genes oxidative phosphorylation NADH–ubiquinone oxidoreductase subunit and NADH–ubiquinone oxidoreductase subunit A1 are overexpressed 2.66 and 2.47 times, respectively, and these genes are involved in mitochondrial integrity and the cell’s antioxidant defense systems against cellular reactive oxygen species (ROS) [22]. The polyherbal also promotes higher expression of the SOS Ras/Rho guanine nucleotide exchange factor 2 (+2.5) gene, which is involved in cell signaling conditions [23]. Another gene stimulated +1.99 times is glutathione S-transferase, mu 2, which has hepatoprotective effects [24]. Therefore, the antioxidant and detoxifying capacities were improved with BioCholine.
The leptin gene is overexpressed 1.59 times and is an essential hormone in energy metabolism and intake [25], with important physiological effects. Other genes are under-expressed, such as transforming activin receptor IIA growth factor (−2.78 times), beta receptor II (−2.5 times), latent transforming growth factor beta binding protein 1 (−2.33 times), and transforming growth factor, beta 1 (−2.09), which participate in the TGF-beta signaling pathway regulated through feedback mechanisms that control the magnitude of its signals [26].
The higher methylation in sheep supplemented with BioCholine can be explained by the contribution of labile-free methyl groups for DNA methylation and the presumable saving of choline by thymol, which promotes acetylcholine recycling by inhibiting acetylcholinesterase activity (Figure 2) [27]. Other studies [28][29] provide evidence that a few compounds reported in BioCholine serve as methyl donors and modify the methylation status of DNA, e.g., hexadecenoic acid methyl ester (C16:0), octadecenoic acid methyl ester (C18:1 cis-9; C18:1 cis-8), and thymol. The effects on ewes and their offspring indirectly indicate that some BioCholine metabolites resist rumen degradation.
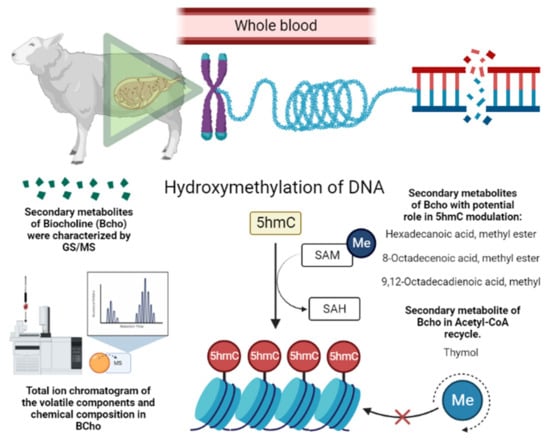
Figure 2. Presumable epigenetic mechanism of BioCholine over DNA methylation in ewes. SAM: S-Adenosyl methionine; SAH: S-Adenosyl homocysteine; Me: methionine; 5hmC: 5-Hydroxymethylcytosine.
3. Rumen Fermentation
One experiment with ewes evaluated the effects of BioCholine on rumen fermentation [12], where no changes in rumen pH, acetate, and total VFA were reported. Compared to the control, supplementation with 4 and 8 g/d of BioCholine reduced the ruminal propionate concentration by 24.0 and 17.3% in lambs sampled on days 15 and 45 of the experimental period. Butyrate was reduced by 30.5% in the samples from the 8 g/d group. Nunes et al. [30] also evaluated ruminal fermentation in dairy cows fed increasing levels of BioCholine (0, 7, 14, and 21 g/d) and did not observe differences in ruminal VFA concentration and molar proportions, NH3-N, nitrogen balance, and digestibility (DM, crude protein, NDF, and ether extract); however, ruminal pH and OM digestion showed a quadratic reduction (p < 0.10), with the lowest values at 7 g/d, which coincided with the quadratic response in milk production (p < 0.05).
The statistical differences observed by Leal et al. [12] in butyrate and propionate have little impact on fermentation efficiency since a minimal CO2 reduction (1.45% of the control) and a marginal methane increase (2.67%) are expected when including a polyherbal. The indicator of microbial activity (methylene blue test) showed a significant boost with herbal supplementation (24.1 and 50.2% for doses 4 and 8 g/d) [12]. The in vitro gas production of BioCholine samples shows that herbal choline has three ruminal degradable fractions, one with fast ruminal degradation (31.8%), another with a medium rate of degradation (40.7%), and one with a slow rate of degradation (27.5%), indicating that an important fraction of the BioCholine Powder can reach the lower tract [31]. Further studies of ruminal fermentation and changes in rumen protozoa and bacteria with metagenomics are required to determine other changes due to the secondary metabolites of BioCholine.
4. Meta-Analyses
The meta-analysis included studies conducted in two countries, México (66.7%) and Brazil (33.3%), and experiments were conducted on commercial farms (37.5%) and experimental facilities (62.5%). The experimental doses of BioCholine in small ruminants ranged from 1.5 to 15 g/d and 7 to 40 g/d in dairy cattle. The experimental periods ranged from 42 to 59 days for growing assays, 20 to 183 days in lactating small ruminants, and 60 to 1095 days in dairy cows.
Dietary inclusion of BioCholine increased (p < 0.01) the average daily gain (ADG), final BW, and milk production. However, dietary supplementation of BioCholine did not affect (p > 0.05) the dry matter intake (DMI) or feed conversion ratio. The I2 values for DMI, milk yield, feed conversion, and final BW indicated high heterogeneity between studies [32][33], whereas ADG had the lowest heterogeneity.
Ruminant blood tests have been reported in 12 experiments [12][13][31][34][35][36][37][38][39][40][41][42]; unfortunately, the blood concentration of Ptdcho was evaluated (evidence of exceeding the metabolites of Ptdcho) only in one experiment [34], whereby increasing the dose of BioCholine caused a linear increase in blood Ptdcho. The other metabolites shown are indicators of effects in the animal due to the polyherbal.
The liver enzymes in the experiments were within the physiological values for sheep, which were healthy. Cross-reactions from some metabolites from BioCholine could be responsible for increasing ALT rather than liver injury [43].
Ruminant types showed some differential responses in blood metabolites. The fattening lambs showed an increase in glucose with BioCholine (p < 0.05), while the rest showed a hypoglycemic effect (ewes, p < 0.10; goats and calves, p < 0.01). The changes observed with glucose metabolism suggest that it will generally have a hypoglycemic effect, but in fattening lambs, energy consumption and the glucose precursor propionate explain the increase [34]. BioCholine contains Azadirachta indica, which has shown hypoglycemic effects [44].
Cholesterol was reduced in lactating goats (p < 0.01) but increased in lambs. Cholesterol increased in four experiments, highlighting the increases in lambs (females and males); in two, a reduction in small lactating ruminants was observed [41][42]. The β-OH-butyrate only increased in two experiments with lactating ewes [39] and decreased in dairy calves [13]. Blood levels could not be related to changes in ruminal butyrate because BioCholine significantly reduced the butyrate concentration [12]. The changes in cholesterol confirm the lipotropic effects of BioCholine and are consistent with the increased expression of PPARα and Acox1 genes [13]. PPARα increases fatty acid oxidation in the liver [45]. Blood cholesterol depends on the mobilization and synthesis of fats and is related to phosphatidylcholine, which participates in synthesizing and exporting triglycerides in very low-density lipoproteins [46].
5. Comparison of BioCholine and RPC in Small Ruminants
Three experiments comparing BioCholine with RPC Reashure have been reported, one in peripartum and postpartum ewes and two in feedlot lambs. In both experiments, the doses were 4 g/sheep/day of BioCholine vs. 4 g/d of RPC. Crosby et al. [47] used 24 Rambouillet ewes supplemented for 30 days before and after calving with a control group, and milk yield differed (p < 0.05) from the control (1.02 kg/d) with both sources: BioCholine (1.57 kg/d) and RPC (1.39 kg/d) (statistically similar). The results showed that the two choline sources improved calving weight and milk production compared to the control, reflected in higher lamb weights at birth and better weight gains at 30 days, showing that BioCholine can replace RPC and obtain similar results [48].
The two experiments comparing BioCholine and RPC in feedlot lambs showed differences, but the substitution effects were not as strong as those of ewes. In the first experiment, 24 Rambouillet lambs (23.4 ± 1.1 kg initial BW) were housed in individual metabolic cages, and treatments were as follows: control (no choline), 4 g/d BioCholine, and 4 g/d RPC in a completely randomized design, used for 42 days [48]. However, no differences were detected in average daily gain (control 222 g/d; BioCholine 250 g/d; and RPC 290 g/d) or feed intake (control 1.06 kg/d; BioCholine 1.07 kg/d; and RPC 1.22 kg/d). However, the final BW improved with the RPC (35.6 kg), followed by BioCholine (33.9 kg) and the control (32.7 kg).
The second experiment was a repetition of the experiment carried out under the same conditions with 24 Rambouillet lambs (23.5 kg ± 3.17 kg initial BW) housed in individual metabolic cages [40] with the same treatments (control group, 4 g/d BioCholine, and 4 g/d RPC) for 42 days. Lambs fed with the polyherbal improved daily gain (320 g/d) and feed intake (1.32 kg/d) compared to the RPC (222 g/d and 1.06 kg/d) and control groups (290 g/d; 1.26 kg/d).
6. Evaluations with Graded Levels of BioCholine in Growing Lambs
Several evaluations with graded levels of BioCholine in sheep have been reported: one with lambs for 52 days [34], three with growing ewes of 75 days [12][37], and two with dairy ewes [39][42], while there is only one reported with RPC in growing lambs [49]. BioCholine increased (p < 0.10) the NEm by 7.15% and the NEg by 9.25% over the control. However, it must be considered that the animal response to high doses of polyherbal is quadratic, and negative reactions can be observed [34]. Still, the data suggest consistent and safe responses with intakes of 4 to 8 g/d of BioCholine in small ruminants.
7. Results of Milk Production in Ewes
In six experiments with ewes [5][35][36][39][42][47], increments in milk production with BioCholine were recorded with different doses and days of supplementation in gestation and lactation, as detected in meta-analyses that included data from ewes and goats. Only one experiment did not show a response in milk production with doses of 0, 4, and 8 g/d BioCholine [42]. The overall SE effect was an increment of +11.35% over the control group (Chi-squared 9.22, p = 0.0024).
The results from the experiment from Roque-Jiménez et al. [5] were reanalyzed by orthogonal contrasts to compare ewes in the control group to those in the BioCholine group (contrast I), and the supply during the last third of gestation vs. BioCholine supplementation throughout gestation (Contrast II) and supplementation with BioCholine showed benefits throughout gestation treatment, in which, after delivery, benefits were observed in the energy balance with an increase (p < 0.01) in milk production of 25.9% with a reduction in weight loss of 25.7%, increasing the weight of their young at birth by 22.4%. These results confirm the effects of choline bypass metabolites and methyl groups that were reflected in greater fetal growth similar to that observed with RPC [50], as well as a better energy balance of the sheep supplemented for longer in gestation with a response effect similar to that observed by Tsiplakou et al. [51] when supplementing RPC (5 g/d) with protected methionine and betaine.
8. Results of Dairy Goats’ Milk Production
The results of two experiments with dairy goats [41][52] of French and Alpine breeds showed surprisingly high responses in milk yield, both conducted at the same experimental campus. Morales et al. [41] orally supplied 0, 4, and 8 g/d, observing a linear response in performance where the 8 g/d doses showed an increase of up to 54%, and in a second experiment where the 8 g/d dose was used for 30 days before delivery and 90 days of lactation, the observed increase (p < 0.05) was 215% [52]. The overall SE effect was +71.84% over the control group (Chi-squared 16.906, p = 0.0001).
Milk yield response in dairy goats was greater than in ewes because dairy goats produce more milk than dairy sheep, which has been associated with differences in energy partitioning and differences in insulin and glucose [53]. Also, there have been reported differences in methylated genes between sheep and goats [54], which could be expressed more by the methylated compounds provided by BioCholine. Some sources of RPC have also been evaluated in goats where one of the experiments did not show a response with 4 g/d for 28 days of lactation in Saanen goats [55]. However, in another study with Etawah-bred goats, the RPC showed a quadratic response with the best increase with 4 g/d of +17% over controls supplemented 52 days before calving and 84 days postpartum [56]. D’Ambrosio et al. [57] observed a response of 14.1% with a supply of 4 g/d supplemented 30 days prepartum and 35 days postpartum to Saanen goats.
9. Response to BioCholine in Dairy Cattle
Four experiments with lactating dairy cattle with supplemental BioCholine have been reported. One was with cows under grazing conditions (Holstein and Jersey) with a mean initial production of 18 kg/d (n = 81) for 90 days, in which milk production increased linearly (p < 0.05) as the dose of polyherbal was increased from 0, 10, to 20 g/d of polyherbal without affecting cows’ BW or milk composition [58]. In another experiment, high-producing Holstein cows supplemented with BioCholine (20 g/d n = 19, 40 g/d n = 17, control n = 20) and other combinations of polyherbals for 90 days since 30 days postpartum showed no response in milk production or composition; however, health indicators were improved, reducing treatment costs (antibiotics, healing, anti-inflammatories, glucogenesis, hormones, intra-mammary treatments, restorative treatments, and vitamins) and being more profitable than the dose of 40 g/d BioCholine. Beneficial residual effects in veterinary expenses and milk production were also detected up to 90 days post-supplementation [21].
Milk production was also evaluated in a Latin square experiment with Holstein cows (163 days in milk and 27.6 kg/d average milk yield) treated with 0, 7, 10, and 21 g/d BioCholine, and a quadratic response (p < 0.05) was observed in milk production with the best response at 7 g/d and a reduction in fat (square effect p < 0.10) without changes in daily intake [30].
There was also a multiannual study in cows (crossbreeding rotational program using Holstein × Montbeliarde × Swedish Red) with an average production of 36 kg/d, where data from 424 control and 442 supplemented cows were analyzed every year for three years in which BioCholine with 0.071% of dry matter of the diet in the entire dairy herd (target dose of 17 g/d for lactating cows) was compared with data from three years without supplement, conducted at a commercial farm, comparing milk production, health status, and replacement data [20]. The supplementation with BioCholine improved fat-corrected milk production by 1.5% (p < 0.001) compared to the average value obtained in the previous years (36.36 vs. 35.80 kg/d) without the polyherbal. Other effects on fertility and health will be discussed later.
10. Responses to Health Indicators by BioCholine
Some of the plants in BioCholine have a wide range of antimicrobial and antioxidant activities through secondary metabolites that stimulate the immune system [59]. As described previously, BioCholine contains volatile metabolites, such as 2-Undecenal, 8-p-menthane diamine, 4-vinylguaiacol, β-pinene, p-cresol [2], and some aldehydes with bacteriostatic and bactericidal effects [60][61][62].
The multiannual evaluation in dairy cattle [20] showed a reduction (p < 0.0001) in abortions (15.65 to 7.29%), clinical (p < 0.005; 12.59 to 6.95%) and subclinical mastitis (p < 0.05; 8.65 to 5.22%), and respiratory disorders (p < 0.10; 12.42 to 8.56%). The experiment with Lacaune ewes with 0 and 5 g/d BioCholine showed a reduction in somatic cell counts of 37.2% in the milk of supplemented ewes (p = 0.07) [35], and the effect was confirmed in another experiment [36] with lactating Lacaune ewes (0, 5, and 10 g/d) in which somatic counts were reduced by 40.42% compared to the control values (samples collected on days 15 and 20 of lactation).
Immunoglobulins (Anti-Clostridium IgG) increased linearly (+12.58%) (p < 0.10) in calves receiving graded levels of BioCholine over the control, and the number of diarrhea events was reduced by 74.71%, pneumonia by 42.29%, and otitis by 49.29%, which resulted in cost reduction in antibiotic doses by 51.03% [13]. In the same experiment, BioCholine supplementation increased gene expression of Klra20 (Killer cell lectin-like receptor subfamily A member 20, +3.9), Pdgfra (platelet-derived growth factor receptor alpha polypeptide, +3.9), Defa14 (Defensin alpha 14, +3.3), Pdgfrl (Platelet-derived growth factor receptor-like, +3.3), Lck (Lymphocyte protein tyrosine kinase, +3.2), and Vpreb3 (Pre-B lymphocyte gene 3, +3.2) and reduced IL10 (Interleukin 10, −2.52), which has an anti-inflammatory capacity. Platelet-activating factor (1-alkyl-2-acetyl-sn-glycero-3-phosphocholine) is a potent immune response activator [63].
In a study by Díaz-Galván et al. [13], arachidonic acid metabolism was one of the main metabolic processes enriched with overexpressed genes due to BioCholine. This fatty acid and linoleic, eicosapentaenoic, or docosahexaenoic acid can be found in the sn2 position of phospholipids [64]. It is important to consider that the types of fatty acids provided in the diet can influence the types of fatty acids present in the phospholipids of blood cells [65], and BioCholine provides phospholipids in the form of phosphatidylcholine and presumably other conjugate choline compounds.
Phospholipids participate in essential cell signaling networks to maintain an effective innate immune response by recognizing molecules derived from their hydrolysis [64]. The contribution of phosphatidylcholine with BioCholine could have increased the expression of the Pla2g2d, Pla2g4e, and Pla2g6 genes, which have cellular expression patterns and indicate greater activity of leukocyte cells and platelets (Figure 3). These genes code for type 2 phospholipases responsible for lysing phospholipids’ sn2 position, such as phosphatidylcholine, and releasing the polyunsaturated fatty acids contained therein [66]. Orzuna-Orzuna (unpublished data from the doctoral dissertation) and Roque-Jiménez et al. [5] measured the fatty acid profile of BioCholine and reported linoleic acid (C18:1 cis 9, 12) as the first long-chain fatty acid with concentrations of 58.94 and 65%, respectively.
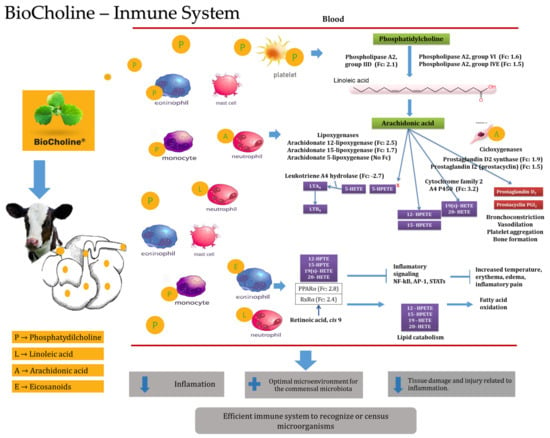
Figure 3. Presumable mode of action of BioCholine over innate immune response in weaning calves. LTA: leukotriene A4, LTB: leukotriene B4, HpETE: hydroperoxyeicosatetraenoic acid, HETE: hydroxyeicosatetraenoic acids, PPARα: peroxisome proliferator-activated receptors, NF-kB: nuclear factor kappa-light-chain-enhancer of activated B cells, AP-1: activator protein.
Linoleic acid acts as a precursor of arachidonic acid [67], which increases the expression of the Alox12 and Alox15 genes that code for lipoxygenase enzymes expressed in eosinophils and platelets (Figure 3) [64]. These enzymes mainly use arachidonic acid (released by phospholipases) to generate monohydroperoxides (HpETEs), although they can also use docosahexaenoic or linoleic acid. This evidence indirectly shows the metabolite’s actions in the animal, suggesting the passing of some through the rumen by increasing the expression of genes in blood cells related to the hydrolysis of phosphatidylcholine. In addition to the genes that code for phospholipases, another example is the expression of the prostaglandin D2 synthase (Ptgds) and prostaglandin I2 (Ptgis) genes, which participate in pathways mediated by cyclooxygenases in immune cells from the oxidation of arachidonic acid (Figure 3) [64].
An increase in the flux of phosphatidylcholine into the blood increases the activity of innate immune cells. These identify the presence of phospholipids as an indicator of injuries (bleeding or trauma) activating homeostatic enzymatic mechanisms that will lead to the production of oxidized lipids that will start biological responses related to the inflammation process to prevent bacterial invasions and initiate the process of inflammation, wound healing, and repair [68].
However, the products derived from the action of phospholipases, lipoxygenases, monooxygenases, and cyclooxygenases act as agonists for activating the transcription factor of peroxisome proliferation alpha (PPARα). PPARα exhibits a potential anti-inflammatory capacity with a substantial impact on the physiology of the immune system by interfering with major inflammatory transcription factors and stimulating the catabolism of inflammatory precursors through fatty acid oxidation [69].
11. Antioxidants
Variables that evaluate the polyherbal’s antioxidant properties have been registered in a few experiments. Alba et al. [35] reported improved antioxidant activity in milk (GPx 59.86% and GST 58.06%) over the control with Lacaune sheep supplemented with 5 g/d BioCholine. In addition, the serum GPx increased by 31.3% (p = 0.07) in samples collected between 7 and 45 days of lactation and reduced Thiobarbituric acid reactive substances assay (TBARS) by 6.25%, which are indicators of better antioxidant status. Alba et al. [36] confirmed the antioxidant effects (p < 0.01) in lactating Lacaune ewes at doses of 5 and 10 g/d. The authors observed a significant reduction in lipid peroxidation (LPO) and reactive oxygen species (ROS) in milk and serum sampled on days 15 and 20 with dietary BioCholine.
The variables that indicate a greater antioxidant condition can be explained by the presence of several metabolites with antioxidant properties in BioCholine, such as 4-vinylguaiacol [2] and hexadecenoic acid methyl ester (C16:0) [5]. 4-vinylguaiacol has demonstrated an antioxidant effect [61], and C17:0 can protect DNA from oxidative damage induced by dietary fats [70].
Another hypothesis of antioxidant capacity would be synthesizing antioxidant molecules from metabolites with rumen escape capacity provided by the polyherbal, such as phosphatidylserine. Transcriptome analysis of hair lamb liver cells (Orzuna-Orzuna unpublished data from the doctoral dissertation) supplemented with BioCholine shows that serine could be used in the liver to synthesize cystathione by increasing the expression of the Cbs (+2.23) gene (which codes for cystathione β-synthetase). This effect would stimulate the expression of the Gss gene (+1.72) that codes for the glutathione synthetase enzyme responsible for synthesizing reduced glutathione via transsulfuration (Figure 4). This non-enzymatic molecule is one of the first lines of defense against oxidative damage [71].
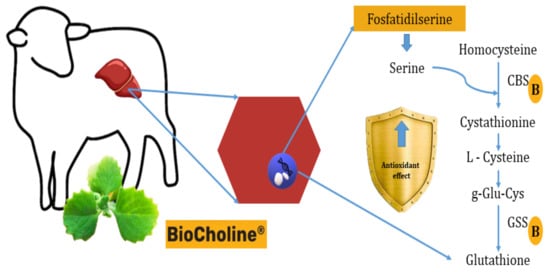
Figure 4. Presumable mode of action of BioCholine over antioxidant capacity in lamb hepatocytes by increasing the synthesis of reduced glutathione from serine and homocysteine. B: BioCholine®, CBS: cystathionine beta synthase, GSS: glutathione synthetase.
In addition, BioCholine reinforces its antioxidant capacity by reducing the production of ROS by increasing the expression of the genes Ndufc1: 2.66, Ndufa1: 2.47, Ndufb4: 1.9, Ndufv2: 1.85, Ndufa3: 1.77 (complex I—NADH: ubiquinone oxidoreductase), Uqcr10: 1.99, Uqcrfs1: 1.87, Uqcrq: 1.78 (complex III—Cytochrome c reductase), and Cox7a2l: 1.55, Cox6b1: 1.51 (complex IV—Cytochrome c oxidase) (Figure 5). The increase in the expression of these genes involved in the electron transport chain would increase their oxidative efficiency in de-electronizing substrates, reducing the loss of electrons and diminishing their interaction with oxygen before it is reduced to water [72][73]. Accordingly, Cavaliere et al. [74] reported a close link between inflammation, redox status, and hepatic mitochondrial respiratory capacity, this last link due to an increased activity of carnitine palmitoyltransferase (CPT), the rate-limiting enzyme for fatty acid entry into the mitochondria.
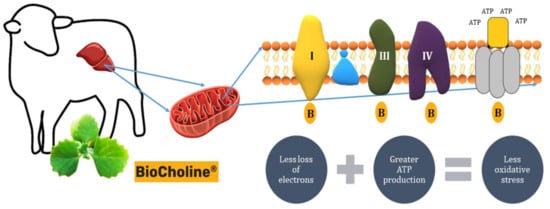
Figure 5. Presumable mode of action of BioCholine on the efficiency of the electron transport chain in hepatic mitochondria, increasing ATP synthesis and reducing the production of reactive oxygen species. B: BioCholine.
These data indicate lower oxidative stress and agree with the genes downregulated by BioCholine: Gsta3 (−2.86), Gsta4 (−2.56), Gstm3 (−2.51), and Gstt4 (−1.57), together with Aldh3a1 (−2.63) and Adh4 (−2.15), which are related to lower glutathione conjugation with toxins in a xenobiotic detoxification process as part of drug metabolism—cytochrome P450 [75][76].
12. Fertility
Little attention has been paid to changes in reproductive variables when using BioCholine. However, in addition to the genes referred to with energy metabolism, Díaz-Galvan et al. [13] showed the overexpression of some genes related to processes that affect reproduction, such as the PELP1 pathway—a novel estrogen receptor-interacting Protein (+4.0), the thyroid hormone signaling pathway X (+2.0), arachidonic acid metabolism (+2.9), and the ribosome pathway (+2.0) [13]. The multiyear evaluation with BioCholine showed that the feed plant additive improved fertility in cows during the first lactation (p < 0.01; 45.33% BioCholine vs. 37.0% control) [20].
This increase in the fertility observed in dairy cattle supplemented with BioCholine [20] could be related to the increase in the expression of genes that code for proteins responsible for synthesizing glutathione. Glutathione in males is an essential endogenous antioxidant responsible for the uptake of ROS in sperm and seminal plasma, protecting it from oxidative damage [77]. Zou et al. [78] added glutathione to the semen extender of Guanzhong dairy goats and observed improvements in sperm fertilization ability by reducing ROS levels.
In females, glutathione protects the ova from damage caused by oxidative stress during folliculogenesis; oocytes with higher levels of intracellular glutathione produce healthier and stronger embryos [79]. Adeoye et al. [80] pointed out that glutathione maintained the biological value of germ cells and implicated it in fertilization and the embryo’s early development.
In dairy cattle, high levels of oxidative stress can lead to dysregulation of reduced glutathione synthesis, manifesting in decreased milk production and reproductive disorders [81]. Given this scenario, the gene changes modulated by BioCholine can improve reproductive efficiency by reducing oxidative stress.
This entry is adapted from the peer-reviewed paper 10.3390/ani14050667
References
- ISO 9001. Available online: https://quality-one.com/iso-9001/ (accessed on 15 May 2023).
- Mendoza, G.D.; Oviedo, M.F.; Pinos, J.M.; Lee-Rangel, H.A.; Vázquez, A.; Flores, R.; Pérez, F.; Roque, A.; Cifuentes, O. Milk production in dairy cows supplemented with herbal choline and methionine. Rev. Fac. Cienc. Agrar. UNCuyo 2020, 52, 332–343.
- Mendoza-Martínez, G.D.; Hernández-García, P.A.; Plata-Pérez, F.X.; Martínez-García, J.A.; Lizarazo-Chaparro, A.C.; Martínez-Cortes, I.; Campillo-Navarro, M.; Lee-Rangel, H.A.; De la Torre-Hernández, M.E.; Gloria-Trujillo, A. Influence of a Polyherbal Choline Source in Dogs: Body Weight Changes, Blood Metabolites, and Gene Expression. Animals 2022, 12, 1313.
- FrankIč, T.; Voljč, M.; Salobir, J.; Rezar, V. Use of herbs and spices and their extracts in animal nutrition. Acta Argic. Slov. 2009, 94, 95–102.
- Roque-Jiménez, J.A.; Mendoza-Martínez, G.D.; Vázquez-Valladolid, A.; Guerrero-González, M.L.; Flores-Ramírez, R.; Pinos-Rodriguez, J.M.; Loor, J.J.; Relling, A.E.; Lee-Rangel, H.A. Supplemental Herbal Choline Increases 5-hmC DNA on Whole Blood from Pregnant Ewes and Offspring. Animals 2020, 10, 1277.
- Dazuk, V.; Tarasconi, L.; Molosse, V.L.; Cécere, B.G.O.; Deolindo, G.L.; Strapazzon, J.V.; Bottari, N.B.; Bissacotti, B.F.; Schetinger, M.R.C.; Sareta, L.; et al. Can the Inclusion of a Vegetable Biocholine Additive in Pig Feed Contaminated with Aflatoxin Reduce Toxicological Impacts on Animal Health and Performance? Animals 2023, 13, 3010.
- Rhayour, K.; Bouchikhi, T.; Tantaoui-Elaraki, A.; Sendide, K.; Remmal, A. The mechanism of bactericidal action of oregano and clove essential oils and of their phenolic major components on Escherichia coli and Bacillus subtilis. J. Essent. Oil Res. 2003, 15, 356.
- Michiels, J.; Missotten, J.; Fremaut, D.; De Smet, S.; Dierick, N. In vitro dose–response of carvacrol, thymol, eugenol and trans-cinnamaldehyde and interaction of combinations for the antimicrobial activity against the pig gut flora. Livest. Sci. 2007, 109, 157–160.
- Gutiérrez-Larraínzar, M.; Rúa, J.; Caro, I.; de Castro, C.; de Arriaga, D.; García-Armesto, M.R.; del Valle, P. Evaluation of antimicrobial and antioxidant activities of natural phenolic compounds against foodborne pathogens and spoilage bacteria. Food Control 2012, 26, 555–563.
- Kim, D.; Kuppusamy, P.; Jung, J.S.; Kim, K.H.; Choi, K.C. Microbial Dynamics and In Vitro Degradation of Plant Secondary Metabolites in Hanwoo Steer Rumen Fluids. Animals 2021, 11, 2350.
- Yuan, J.; Gan, T.; Liu, Y.; Gao, H.; Xu, W.; Zhang, T.; Tan, R.; Cai, Z.; Jiang, H. Composition and antimicrobial activity of the essential oil from the branches of Jacaranda cuspidifolia Mart. growing in Sichuan, China. Nat. Prod. Res. 2018, 32, 1451–1454.
- Leal, K.W.; Alba, D.F.; Cunha, M.G.; Marcon, H.; Oliveira, F.C.; Wagner, R.; Silva, A.D.; Lopes, T.F.; de Jesus, L.S.B.; Schetinger, M.R.C.; et al. Effects of biocholine powder supplementation in ewe lambs: Growth, rumen fermentation, antioxidant status, and metabolism. Biotechnol. Rep. 2021, 29, e00580.
- Díaz-Galván, C.; Méndez-Olvera, E.T.; Martínez-Gómez, D.a.; Gloria-Trujillo, A.; Hernández-García, P.A.; Espinosa Ayala, E.; Palacios-Martínez, M.; Lara-Bueno, A.; Mendoza-Martínez, G.D.; Velázquez-Cruz, L.A. Influence of a Polyherbal Mixture in Dairy Calves: Growth Performance and Gene Expression. Front. Vet. Sci. 2021, 7, 1217.
- Wang, L.; Waltenberger, B.; Pferschy-Wenzig, E.-M.; Blunder, M.; Liu, X.; Malainer, C.; Blazevic, T.; Schwaiger, S.; Rollinger, J.M.; Heiss, E.H.; et al. Natural product agonists of peroxisome proliferator-activated receptor gamma (PPARγ): A review. Biochem. Pharmacol. 2014, 92, 73–89.
- Adeva-Andany, M.M.; Pérez-Felpete, N.; Fernández-Fernández, C.; Donapetry-García, C.; Pazos-García, C. Liver glucose metabolism in humans. Biosci. Rep. 2016, 36, e00416.
- Koller, A.; Spong, A.P.; Lüers, G.H.; Subramani, S. Analysis of the peroxisomal acyl-CoA oxidase gene product from Pichia pastoris and determination of its targeting signal. Yeast 1999, 15, 1035–1044.
- Watkins, P.A.; McGuinness, M.C.; Raymond, G.V.; Hicks, B.A.; Sisk, J.M.; Moser, A.B.; Moser, H.W. Distinction between peroxisomal bifunctional enzyme and acyl-CoA oxidase deficiencies. Ann. Neurol. 1995, 38, 472–477.
- Poniatowski, Ł.A.; Wojdasiewicz, P.; Gasik, R.; Szukiewicz, D. Transforming Growth Factor Beta Family: Insight into the Role of Growth Factors in Regulation of Fracture Healing Biology and Potential Clinical Applications. Mediat. Inflamm. 2015, 2015, 137823.
- Lehrer, R.I.; Lu, W. α-Defensins in human innate immunity. Immunol. Rev. 2012, 245, 84–112.
- Gutiérrez, A.; Gutiérrez, A.; Sánchez, C.; Mendoza, G.D. Effect of including herbal choline in the diet of a dairy herd a multiyear evaluation. Emir. J. Food Agric. 2019, 31, 477–481.
- Ortega-Alvarado, N.I.; Mendoza-Martínez, G.D.; Bárcena-Gama, R.; Hernández-García, P.A.; Espinosa-Ayala, E.; Martínez-García, J.A.; Gloria-Trujillo, A. Economic impact of polyherbal mixtures containing choline, lysine and methionine on milk production and health of dairy cows. Emir. J. Food Agric. 2020, 32, 864–870.
- Koopman, W.J.; Verkaart, S.; Visch, H.J.; van Emst-de Vries, S.; Nijtmans, L.G.; Smeitink, J.A.; Willems, P.H. Human NADH:ubiquinone oxidoreductase deficiency: Radical changes in mitochondrial morphology? Am. J. Physiol. Cell Physiol. 2007, 293, C22–C29.
- Quilliam, L.A.; Rebhun, J.F.; Castro, A.F. A growing family of guanine nucleotide exchange factors is responsible for activation of ras-family GTPases. In Progress in Nucleic Acid Research and Molecular Biology; Academic Press: Cambridge, MA, USA, 2002; Volume 71, pp. 391–444.
- Jin, Y.; Tan, Y.; Zhao, P.; Guo, Y.; Chen, S.; Wu, J.; Ren, Z. Glutathione S-transferase Mu 2 inhibits hepatic steatosis via ASK1 suppression. Commun. Biol. 2022, 5, 326.
- Chilliard, Y.; Delavaud, C.; Bonnet, M. Leptin expression in ruminants: Nutritional and physiological regulations in relation with energy metabolism. Domest. Anim. Endocrinol. 2005, 29, 3–22.
- Miyazono, K. Positive and negative regulation of TGF-β signaling. J. Cell Sci. 2000, 113, 1101–1109.
- Jukic, M.; Politeo, O.; Maksimovic, M.; Milos, M.; Milos, M. In Vitro acetylcholinesterase inhibitory properties of thymol, carvacrol and their derivatives thymoquinone and thymohydroquinone. Phytother. Res. 2007, 21, 259–261.
- Mahmoud, A.M.; Ali, M.M. Methyl Donor Micronutrients that Modify DNA Methylation and Cancer Outcome. Nutrients 2019, 11, 608.
- Anderson, O.S.; Sant, K.E.; Dolinoy, D.C. Nutrition and epigenetics: An interplay of dietary methyl donors, one-carbon metabolism and DNA methylation. J. Nutr. Biochem. 2012, 23, 853–859.
- Nunes, A.T.; Takiya, C.S.; da Silva, G.G.; Ghizzi, L.G.; Grigoletto, N.T.S.; Dias, M.S.S.; Silva, T.B.P.; Junior, P.C.V.; Chesini, R.G.; Curti, P.F.; et al. Increasing doses of biocholine on apparent digestibility, ruminal fermentation, and performance in dairy cows. Livest. Sci. 2022, 260, 104927.
- Rodríguez-Guerrero, V.; Lizarazo, A.C.; Ferraro, S.; Miranda, L.A.; Mendoza, G.D.; Suárez, N. Effect of herbal choline and rumen-protected methionine on lamb performance and blood metabolites. S. Afr. J. Anim. Sci. 2018, 48, 427–434.
- Higgins, J.P.T.; Thompson, S.G.; Deeks, J.J.; Altman, D.G. Measuring inconsistency in meta-analyses. BMJ 2003, 327, 557–560.
- Orzuna-Orzuna, J.F.; Dorantes-Iturbide, G.; Lara-Bueno, A.; Mendoza-Martínez, G.D.; Miranda-Romero, L.A.; Hernández-García, P.A. Effects of Dietary Tannins’ Supplementation on Growth Performance, Rumen Fermentation, and Enteric Methane Emissions in Beef Cattle: A Meta-Analysis. Sustainability 2021, 13, 7410.
- Martínez-Aispuro, J.A.; Mendoza, G.D.; Cordero-Mora, J.L.; Ayala-Monter, M.A.; Sánchez-Torres, M.T.; Figueroa-Velasco, J.L.; Vázquez-Silva, G.; Gloria-Trujillo, A. Evaluation of an herbal choline feed plant additive in lamb feedlot rations. Rev. Bras. Zootec. 2019, 48, e20190020.
- Alba, D.F.; Favaretto, J.A.; Marcon, H.; Saldanha, T.F.; Leal, K.W.; Campigoto, G.; Souza, C.F.; Baldissera, M.D.; Bianchi, A.E.; Vedovatto, M.; et al. Vegetable biocholine supplementation in pre- and postpartum Lacaune sheep: Effects on animal health, milk production and quality. Small Rumin. Res. 2020, 190, 106165.
- Alba, D.F.; Leal, K.; Cunha, M.H.; da Rosa, G.; Marcon, H.; Souza, C.F.; Baldissera, M.D.; Zotti, C.A.; Kavalek, R.L.; Kempka, A.P. Positive effects of biocholine powder dietary supplementation on milk production and quality, and antioxidant responses in lactating ewes: A new nutritional tool. Heliyon 2021, 7, e06732.
- Ayala-Monter, M.A.; Martínez-Aispuro, J.A.; Mendoza-Martínez, G.D.; Sánchez-Torres, M.T.; Cordero-Mora, J.L.; Martínez-Cruz, I. Evaluation of herbal choline in productive performance and blood metabolites of ewes. Agro Productividad 2022, 15, 109.
- Bárcena-Gama, J.R.; Martínez-Aispuro, J.A.; Mendoza-Martínez, G.D.; Cordero Mora, J.L.; Sánchez-Torres, M.T.; Figueroa Velasco, J.L.; Ayala-Monter, M.A. Evaluation of polyherbal methionine and choline in feedlot rations for lambs. S. Afr. J. Anim. Sci. 2020, 50, 731–737.
- Estrada, M. Efecto de la Biocolina a Diferentes Dosis en Ovinos Productores de Leche en Pastoreo. Master’s Thesis, Animal Production and Health, Universidad Nacional Autónoma de México, Mexico City, Mexico, 2020.
- Martínez-García, J.; Garcia-Lopez, J.; Hernández-García, P.; Mendoza-Martínez, G.; Vázquez-Valladolid, A.; Mejia-Delgadillo, M.; Lee-Rangel, H. Changes in Productive Performance, Blood Metabolites and Hematological Parameters of Growing Lambs Supplemented with Two Sources of Choline. Indian J. Anim. Res. 2021, 1, 6.
- Morales, L.A.; Mendoza, M.G.D.; Gutiérrez, M.J.; Lizarazo, C.A.C.; Martínez, G.J.A. Desempeño productivo de cabras lecheras suplementadas con colina herbal durante el periodo de transición. In Proceedings of the XIX Congreso Bienal AMENA, Puerto Vallarta, México, 15–19 October 2019.
- Suárez-Suárez, N.E. Evaluación de Biocolina Sobre Parámetros Productivos en el Periodo de Transición de Ovejas East Friesian. Master’s Thesis, Universiada Autónoma de San Luis Potosí, San Luis Potosí, Mexico, 2018.
- Dufour, D.R.; Lott, J.A.; Nolte, F.S.; Gretch, D.R.; Koff, R.S.; Seeff, L.B. Diagnosis and Monitoring of Hepatic Injury. I. Performance Characteristics of Laboratory Tests. Clin. Chem. 2000, 46, 2027–2049.
- Khosla, P.; Bhanwra, S.; Singh, J.; Seth, S.; Srivastava, R.K. A study of hypoglycaemic effects of Azadirachta indica (Neem) in normaland alloxan diabetic rabbits. Indian J. Physiol. Pharmacol. 2000, 44, 69–74.
- Naeem, A.; Drackley, J.K.; Stamey, J.; Loor, J.J. Role of metabolic and cellular proliferation genes in ruminal development in response to enhanced plane of nutrition in neonatal Holstein calves. J. Dairy Sci. 2012, 95, 1807–1820.
- Zeisel, S.H. Choline: Critical Role During Fetal Development and Dietary Requirements in Adults. Annu. Rev. Nutr. 2006, 26, 229–250.
- Crosby, M.; Mendoza-Martinez, G.; Relling, A.; Vazquez, V.; Lee-Rangel, H.; Martinez, J.; Oviedo, M. Influence of supplemental choline on milk yield, fatty acid profile, and postpartum weight changes in suckling ewes. J. Dairy Sci. 2017, 100, 30813–30814.
- Godinez-Cruz, J.; Cifuentes-López, O.; Cayetano, J.; Lee-Rangel, H.; Mendoza, G.; Vázquez, A.; Roque, A. Effect of choline inclusion on lamb performance and meat characteristics. J. Anim. Sci. 2015, 93, 766.
- Li, H.; Wang, H.; Yu, L.; Wang, M.; Liu, S.; Sun, L.; Chen, Q. Effects of supplementation of rumen-protected choline on growth performance, meat quality and gene expression in longissimus dorsi muscle of lambs. Arch. Anim. Nutr. 2015, 69, 340–350.
- Birch, S.M.; Lenox, M.W.; Kornegay, J.N.; Paniagua, B.; Styner, M.A.; Goodlett, C.R.; Cudd, T.A.; Washburn, S.E. Maternal choline supplementation in a sheep model of first trimester binge alcohol fails to protect against brain volume reductions in peripubertal lambs. Alcohol 2016, 55, 1–8.
- Tsiplakou, E.; Mavrommatis, A.; Kalogeropoulos, T.; Chatzikonstantinou, M.; Koutsouli, P.; Sotirakoglou, K.; Labrou, N.; Zervas, G. The effect of dietary supplementation with rumen-protected methionine alone or in combination with rumen-protected choline and betaine on sheep milk and antioxidant capacity. J. Anim. Physiol. Anim. Nutr. 2017, 101, 1004–1013.
- Bello-Cabrera, D.; Arriaga-Avilés, Y.; Ruiz-Romero, R. Comparación de la ganancia diaria de peso en dos grupos de cabritas alimentadas con leche entera o leche entera suplementada con biocolina. Abanico Vet. 2022, 12, e2022-12.
- Lunesu, M.F.; Bomboi, G.C.; Marzano, A.; Comin, A.; Prandi, A.; Sechi, P.; Nicolussi, P.S.; Decandia, M.; Manca, C.; Atzori, A.S.; et al. Metabolic and hormonal control of energy utilization and partitioning from early to mid lactation in Sarda ewes and Saanen goats. J. Dairy Sci. 2021, 104, 3617–3631.
- Denoyelle, L.; de Villemereuil, P.; Boyer, F.; Khelifi, M.; Gaffet, C.; Alberto, F.; Benjelloun, B.; Pompanon, F. Genetic Variations and Differential DNA Methylation to Face Contrasted Climates in Small Ruminants: An Analysis on Traditionally-Managed Sheep and Goats. Front. Genet. 2021, 12, 745284.
- Baldi, A.; Bruckmaier, R.; Campagnoli, A.; D’Ambrosio, F.; Pecorini, C.; Pinotti, L.; Rebucci, R. Rumen-protected choline supplementation in periparturient dairy goats: Effects on liver and mammary gland. J. Agric. Sci. 2011, 149, 655–661.
- Supriyati; Budiarsana, I.G.M.; Praharani, L.; Krisnan, R.; Sutama, I.K. Effect of choline chloride supplementation on milk production and milk composition of Etawah grade goats. J. Anim. Sci. Technol. 2016, 58, 30.
- D’Ambrosio, F.; Campagnoli, A.; Susca, F.; Fusi, E.; Rebucci, R.; Agazzi, A.; Pinotti, L.; Baldi, A. Effects of rumen-protected choline supplementation in periparturient dairy goats. Vet. Res. Commun. 2007, 31, 393.
- Cañada, L.M.G.; Meráz, R.E.; Mendoza, M.G.D.; Villagrán, B.V.; Castillo, M.D.A. Efecto del nivel de colina herbal en la producción y composición de leche en ganado lechero en pastoreo. Arch. Latinoam. Prod. Anim. 2018, 26, 82.
- Upadhaya, S.D.; Kim, I.H. Efficacy of Phytogenic Feed Additive on Performance, Production and Health Status of Monogastric Animals—A Review. Ann. Anim. Sci. 2017, 17, 929–948.
- Widhalm, B.; Ters, T.; Srebotnik, E.; Rieder-Gradinger, C. Reduction of aldehydes and terpenes within pine wood by microbial activity. Holzforschung 2016, 70, 895–900.
- Esatbeyoglu, T.; Ulbrich, K.; Rehberg, C.; Rohn, S.; Rimbach, G. Thermal stability, antioxidant, and anti-inflammatory activity of curcumin and its degradation product 4-vinyl guaiacol. Food Funct. 2015, 6, 887–893.
- Zhu, S.; Yang, X.; Xu, S.; Zhao, Z.; Jiang, J. Synthesis and antibacterial activity of bis-alkanoyl-amines heterocyclic derivatives of cis-1, 8-p-Menthane Diamine. Chem. Ind. For. Prod. 2017, 37, 79–84.
- Prescott, S.M.; Zimmerman, G.A.; Stafforini, D.M.; McIntyre, T.M. Platelet-Activating Factor and Related Lipid Mediators. Annu. Rev. Biochem. 2000, 69, 419–445.
- O’Donnell, V.B.; Rossjohn, J.; Wakelam, M.J.O. Phospholipid signaling in innate immune cells. J. Clin. Investig. 2019, 128, 2670–2679.
- Haban, P.; Zidekova, E.; Klvanova, J. Supplementation with long-chain n-3 fatty acids in non-insulin-dependent diabetes mellitus (NIDDM) patients leads to the lowering of oleic acid content in serum phospholipids. Eur. J. Nutr. 2000, 39, 201–206.
- Leslie, C.C. Cytosolic phospholipase A2: Physiological function and role in disease. J. Lipid Res. 2015, 56, 1386–1402.
- Adam, O.; Wolfram, G.; Zöllner, N. Influence of Dietary Linoleic Acid Intake with Different Fat Intakes on Arachidonic Acid Concentrations in Plasma and Platelet Lipids and Eicosanoid Biosynthesis in Female Volunteers. Ann. Nutr. Metab. 2003, 47, 31–36.
- Alberts, B.; Johnson, A.; Lewis, J.; Raff, M.; Roberts, K.; Walter, P. Innate immunity. In Molecular Biology of the Cell, 4th ed.; Garland Science: New York, NY, USA, 2002.
- Grabacka, M.; Pierzchalska, M.; Płonka, P.M.; Pierzchalski, P. The Role of PPAR Alpha in the Modulation of Innate Immunity. Int. J. Mol. Sci. 2021, 22, 10545.
- Beeharry, N.; Lowe, J.E.; Hernandez, A.R.; Chambers, J.A.; Fucassi, F.; Cragg, P.J.; Green, M.H.L.; Green, I.C. Linoleic acid and antioxidants protect against DNA damage and apoptosis induced by palmitic acid. Mutat. Res. Fundam. Mol. Mech. Mutagen. 2003, 530, 27–33.
- Martínez-Sámano, J.; Torres-Durán, P.V.; Juárez-Oropeza, M.A. El glutatión y su asociación con las enfermedades neurodegenerativas, la esquizofrenia, el envejecimiento y la isquemia cerebral. Rev. Educ. Bioquímica 2011, 30, 56–67.
- Weiss, H.; Friedrich, T.; Hofhaus, G.; Preis, D. The respiratory-chain NADH dehydrogenase (complex I) of mitochondria. In EJB Reviews 1991; Springer: Berlin/Heidelberg, Germany, 1992; pp. 55–68.
- Bazil, J.N.; Pannala, V.R.; Dash, R.K.; Beard, D.A. Determining the origins of superoxide and hydrogen peroxide in the mammalian NADH: Ubiquinone oxidoreductase. Free. Radic. Biol. Med. 2014, 77, 121–129.
- Cavaliere, G.; Trinchese, G.; Musco, N.; Infascelli, F.; De Filippo, C.; Mastellone, V.; Morittu, V.M.; Lombardi, P.; Tudisco, R.; Grossi, M.; et al. Milk from cows fed a diet with a high forage: Concentrate ratio improves inflammatory state, oxidative stress, and mitochondrial function in rats. J. Dairy Sci. 2018, 101, 1843–1851.
- Hill, B.G.; Bhatnagar, A. Beyond reactive oxygen species: Aldehydes as arbitrators of alarm and adaptation. Circ. Res. 2009, 105, 1044–1046.
- Cao, X.; Kong, X.; Zhou, Y.; Lan, L.; Luo, L.; Yin, Z. Glutathione S-transferase P1 suppresses iNOS protein stability in RAW264.7 macrophage-like cells after LPS stimulation. Free. Radic. Res. 2015, 49, 1438–1448.
- Abdullah, F.; Khan Nor-Ashikin, M.N.; Agarwal, R.; Kamsani, Y.S.; Abd Malek, M.; Bakar, N.S.; Mohammad Kamal, A.A.; Sarbandi, M.S.; Abdul Rahman, N.S.; Musa, N.H. Glutathione (GSH) improves sperm quality and testicular morphology in streptozotocin-induced diabetic mice. Asian J. Androl. 2021, 23, 281–287.
- Zou, J.; Wei, L.; Li, D.; Zhang, Y.; Wang, G.; Zhang, L.; Cao, P.; Yang, S.; Li, G. Effect of Glutathione on Sperm Quality in Guanzhong Dairy Goat Sperm During Cryopreservation. Front. Vet. Sci. 2021, 8, 771440.
- Mukherjee, A.; Malik, H.; Saha, A.P.; Dubey, A.; Singhal, D.K.; Boateng, S.; Saugandhika, S.; Kumar, S.; De, S.; Guha, S.K.; et al. Resveratrol treatment during goat oocytes maturation enhances developmental competence of parthenogenetic and hand-made cloned blastocysts by modulating intracellular glutathione level and embryonic gene expression. J. Assist. Reprod. Genet. 2014, 31, 229–239.
- Adeoye, O.; Olawumi, J.; Opeyemi, A.; Christiania, O. Review on the role of glutathione on oxidative stress and infertility. JBRA Assist. Reprod. 2018, 22, 61–66.
- Necasova, A.; Pechova, A.; Bodor, R.; Masar, M. Evaluation of the glutathione concentration in whole blood of dairy Holstein cows. Vet. Med. 2021, 66, 179–188.
This entry is offline, you can click here to edit this entry!
