Listeria monocytogenes is a Gram-positive pathogenic bacterium which can be found in soil or water. Infection with the microorganism can occur after ingestion of contaminated food products. Small and large outbreaks of listeriosis have been described in the past. L. monocytogenes can cause a number of different clinical syndromes, most frequently sepsis, meningitis, and rhombencephalitis, particularly in immunocompromised hosts. L. monocytogenes systemic infections can develop following tissue penetration across the gastrointestinal tract or to hematogenous spread to sterile sites, possibly evolving towards bacteremia. L. monocytogenes only rarely causes bone or joint infections, usually in the context of prosthetic material that can provide a site for bacterial seeding.
- L. monocytogenes
- imaging
- microbiological diagnosis
- surgical approach
- antibiotic treatment
- infections
1. Introduction
| Dairy Products | Fruits and Vegetables | Meat Products | Fish Products |
|---|---|---|---|
| Pasteurized whole milk Chocolate milk Soft cheese (different types) Hard cheese Mexican-style cheese Goat cheese Ice cream Fresh cream |
Coleslaw (cabbage) Lettuce Corn Rice salad Salted mushrooms Sprouts Strawberries Nectarines Apples Cantaloupes Blueberries Stone fruit |
Delicatessen foods (deli meats) Pâté Foie gras Uncooked hot dogs “Rillettes” Pork tongue in aspic Pork pie Beef Turkey franks Jellied pork Cooked ham Ox tongue Undercooked chicken |
Shrimp salad Tuna salad Smoked fish |
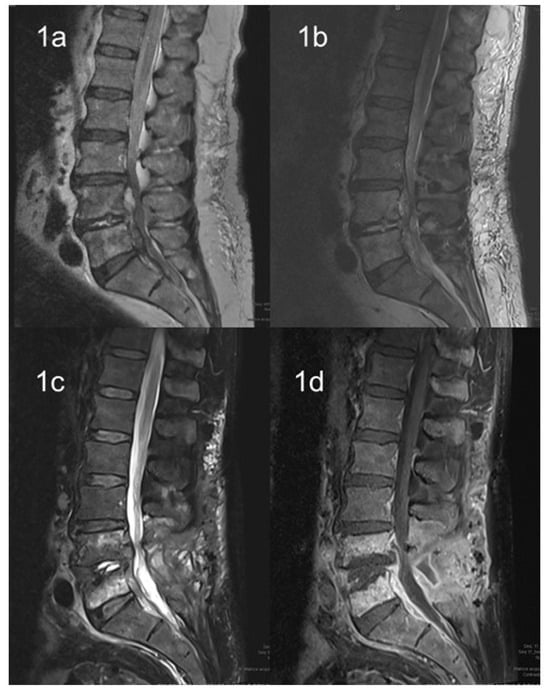
2. Imaging Techniques
3. Microbiological Diagnosis
4. Surgical Approach
5. Antibiotic Treatment of Bone and Vertebral L. monocytogenes Infections
| Author | Age | Gender | Co-Morbidities | Clinical Symptoms | Duration of Symptoms | Antibiotic Treatment and Duration | Surgery |
|---|---|---|---|---|---|---|---|
| Adebolu et al. [47] | 60 | M | Polymyalgia rheumatica | Back pain | 12 months | Ampicillin, IV, 6 weeks Gentamicin, IV, 2 weeks |
Yes |
| Khan et al. [48] | 69 | M | Prior spinal laminectomy | Back pain | 5 months | Ampicillin, IV * Gentamicin, IV * |
Yes |
| Camp et al. [49] | 67 | M | DM, prior lumbar surgery | Back pain | Unknown | Oxacillin, IV * Streptomycin, IV * |
Yes |
| Chirgwin et al. [50] | 57 | M | DM, asthma | Fever, back pain | 3 weeks | Ampicillin, IV, 6 weeks Tobramycin, IV, 6 weeks |
Yes |
| Aubin et al. [51] | 92 | M | DM, heart failure, hip arthroplasty | Fever | 1 week | Amoxicillin, IV, 6 days Gentamicin, IV, 4 days Trimethoprim-sulfamethoxazole, oral, 12 weeks |
Yes |
| Hasan et al. [52] | 63 | M | DM, aortic valve replacement | Fever, back pain | 2 days | Benzyl penicillin, IV, 6 weeks Rifampicin, oral, 4 weeks Amoxicillin, oral, 18 weeks |
Yes |
| Duarte et al. [53] | 65 | M | DM | Fever | 5 days | Ampicillin, IV, 2 weeks Amoxicillin, oral, 12 weeks |
Yes |
| Al Ohaly et al. [54] | 79 | M | Hypertension, carotid bypass, repair of AAA | Back pain | 3 weeks | Ampicillin, IV, 6 weeks | No |
This entry is adapted from the peer-reviewed paper 10.3390/microorganisms12010178
References
- Welshimer, H.J.; Donker-Voet, J. Listeria monocytogenes in nature. Appl. Microbiol. 1971, 21, 516–519.
- Linke, K.; Ruckerl, I.; Brugger, K.; Karpiskova, R.; Walland, J.; Muri-Klinger, S.; Tichy, A.; Wagner, M.; Stessl, B. Reservoirs of Listeria species in three environmental ecosystems. Appl. Environ. Micro. 2014, 80, 5583–5592.
- Law, J.W.; Mutalib, N.S.; Chan, K.G.; Lee, L.H. An insight into the isolation, enumeration, and molecular detection of Listeria monocytogenes in food. Front. Microbiol. 2015, 6, 1227–1242.
- Graves, L.M.; Swaminathan, B.; Ajello, G.W.; Malcolm, G.D.; Weaver, R.E.; Ransom, R.; Dever, K.; Plikaytis, B.D.; Schuchat, A.; Wenger, J.D.; et al. Comparison of three selective enrichment methods for the isolation of Listeria monocytogenes from naturally contaminated foods. J. Food Prot. 1992, 55, 952–959.
- Bres, V.; Yang, H.; Hsu, E.; Ren, Y.; Cheng, Y.; Wisniewski, M.; Hanhan, M.; Zaslavsky, P.; Noll, N.; Weaver, B.; et al. Listeria monocytogenes LmG2 detection assay using transcription mediated amplification to detect Listeria monocytogenes in selected foods and stainless steel surfaces. J. AOAC Int. 2014, 97, 1343–1358.
- Reissbrodt, R. New chromogenic plating media for detection and enumeration of pathogenic Listeria spp.: An overview. Int. J. Food Microbiol. 2004, 95, 1–9.
- Grif, K.; Hein, I.; Wagner, M.; Brandl, E.; Mpamugo, O.; McLauchlin, J.; Dierich, M.P.; Allerberger, F. Prevalence and characterization of Listeria monocytogenes in the feces of healthy Austrians. Wien. Klin. Wochenschr. 2001, 113, 737–742.
- Gahan, C.G.; Hill, C. Listeria monocytogenes: Survival and adaptation in the gastrointestinal tract. Front. Cell Infect. Microbiol. 2014, 4, 9–16.
- Travier, L.; Guadagnini, S.; Gouin, E.; Dufour, A.; Chenal-Francisque, V.; Cossart, P.; Olivo-Marin, J.C.; Ghigo, J.M.; Disson, O.; Lecuit, M. ActA promotes Listeria monocytogenes aggregation, intestinal colonization and carriage. PLoS Pathog. 2013, 9, e1003131.
- Low, J.C.; Renton, C.P. Septicaemia, encephalitis and abortions in a housed flock of sheep caused by Listeria monocytogenes type 1/2. Vet. Rec. 1985, 116, 147–150.
- Schlech, W.F., III; Lavigne, P.M.; Bortolussi, R.A.; Allen, A.C.; Haldane, E.V.; Wort, A.J.; Hightower, A.W.; Johnson, S.E.; King, S.H.; Nicholls, E.S.; et al. Epidemic listeriosis: Evidence for transmission by food. N. Engl. J. Med. 1983, 308, 203–206.
- Büla, C.J.; Bille, J.; Glauser, M.P. An epidemic of food-borne listeriosis in western Switzerland: Description of 57 cases involving adults. Clin. Infect. Dis. 1995, 20, 66–72.
- Centers for Disease Control and Prevention (CDC). Outbreak of listeriosis associated with homemade Mexican-style cheese: North Carolina, October 2000–January 2001. MMWR Morb. Mortal. Wkly. Rep. 2001, 50, 560–562.
- Fretz, R.; Pichler, J.; Sagel, U.; Much, P.; Ruppitsch, W.; Pietzka, A.T.; Stoger, A.; Huhulescu, S.; Heuberger, S.; Appl, G.; et al. Update: Multinational listeriosis outbreak due to ‘quargel’, a sour milk curd cheese, caused by two different L. monocytogenesserotype 1/2a strains, 2009–2010. Euro Surveill. 2010, 22, 19543.
- Carrique-Mas, J.J.; Hökeberg, I.; Andersson, Y.; Arneborn, M.; Tham, W.; Danielsson-Tham, M.L.; Osterman, B.; Leffler, M.; Steen, M.; Eriksson, E.; et al. Febrile gastroenteritis after eating on-farm manufactured fresh cheese: An outbreak of listeriosis? Epidemiol. Infect. 2003, 130, 79–86.
- Linnan, M.J.; Mascola, L.; Lou, X.D.; Goulet, V.; May, S.; Salminen, C.; Hird, D.W.; Yonekura, M.L.; Hayes, P.; Weaver, R.; et al. Epidemic listeriosis associated with Mexican-style cheese. N. Engl. J. Med. 1988, 319, 823–828.
- Choi, M.J.; Jackson, K.A.; Medus, C.; Beal, J.; Rigdon, C.E.; Cloyd, T.C.; Forstner, M.J.; Ball, J.; Bosch, S.; Bottichio, L.; et al. Multistate outbreak of listeriosis linked to soft-ripened cheese: United States, 2013. MMWR Morb. Mortal. Wkly. Rep. 2014, 63, 294–295.
- Jackson, K.A.; Biggerstaff, M.; Tobin-D’Angelo, M.; Sweat, D.; Klos, R.; Nosari, J.; Garrison, O.; Boothe, E.; Saathoff-Huber, L.; Hainstock, L.; et al. Multistate outbreak of Listeria monocytogenes associated with Mexican-style cheese made from pasteurized milk among pregnant, Hispanic women. J. Food Prot. 2011, 74, 949–953.
- Koch, J.; Dworak, R.; Prager, R.; Becker, B.; Brockmann, S.; Wicke, A.; Wichmann-Schauer, H.; Hof, H.; Werber, D.; Stark, K. Large listeriosis outbreak linked to cheese made from pasteurized milk, Germany, 2006–2007. Foodborne Pathog. Dis. 2010, 7, 1581–1584.
- Heiman, K.E.; Garalde, V.B.; Gronostaj, M.; Jackson, K.A.; Beam, S.; Joseph, L.; Saupe, A.; Ricotta, E.; Waechter, H.; Wellman, A.; et al. Multistate outbreak of listeriosis caused by imported cheese and evidence of cross-contamination of other cheeses, USA, 2012. Epidemiol. Infect. 2016, 144, 2698–2708.
- Fleming, D.W.; Cochi, S.L.; MacDonald, K.L.; Brondum, J.; Hayes, P.S.; Plikaytis, B.D.; Holmes, M.B.; Audurier, A.; Broome, C.V.; Reingold, A.L. Pasteurized milk as a vehicle of infection in an outbreak of listeriosis. N. Engl. J. Med. 1985, 312, 404–407.
- Dalton, C.B.; Austin, C.C.; Sobel, J.; Hayes, P.S.; Bibb, W.F.; Graves, L.M.; Swaminathan, B.; Proctor, M.E.; Griffin, P.M. An outbreak of gastroenteritis and fever due to Listeria monocytogenes in milk. N. Engl. J. Med. 1997, 336, 100–105.
- Lyytikäinen, O.; Autio, T.; Maijala, R.; Ruutu, P.; Honkanen-Buzalski, T.; Miettinen, M.; Hatakka, M.; Mikkola, J.; Anttila, V.J.; Johansson, T.; et al. An outbreak of Listeria monocytogenes serotype 3a infections from butter in Finland. J. Infect. Dis. 2000, 181, 1838–1841.
- MMWR. Public health dispatch: Outbreak of listeriosis: Northeastern United States. MMWR Morb. Mortal. Wkly. Rep. 2002, 51, 950–951.
- de Valk, H.; Vaillant, V.; Jacquet, C.; Rocourt, J.; Le Querrec, F.; Stainer, F.; Quelquejeu, N.; Pierre, O.; Pierre, V.; Desenclos, J.C.; et al. Two consecutive nationwide outbreaks of listeriosis in France, October 1999–February 2000. Am. J. Epidemiol. 2001, 154, 944–950.
- Hachler, H.; Marti, G.; Giannini, P.; Lehner, A.; Jost, M.; Beck, J.; Weiss, F.; Bally, B.; Jermini, M.; Stephan, R.; et al. Outbreak of listeriosis due to imported cooked ham. Euro Surveill. 2013, 18, 20469.
- Smith, B.; Larsson, J.T.; Lisby, M.; Müller, L.; Madsen, S.B.; Engberg, J.; Bangsborg, J.; Ethelberg, S.; Kemp, M. Outbreak of listeriosis caused by infected beef meat from a meals-on-wheels delivery in Denmark 2009. Clin. Microbiol. Infect. 2011, 17, 50–52.
- Currie, A.; Farber, J.M.; Nadon, C.; Sharma, D.; Whitfield, Y.; Gaulin, C.; Galanis, E.; Bekal, S.; Flint, J.; Tschetter, L.; et al. Multi-province listeriosis outbreak linked to contaminated deli meat consumed primarily in institutional settings, Canada, 2008. Foodborne Pathog. Dis. 2015, 12, 645–652.
- National Institute for Communicable Diseases. Situation Report on Listeriosis Outbreak, South Africa, 2017. 4 December 2017; pp. 1–3. Available online: https://s3-us-west-2.amazonaws.com/listeriaclassactioncoza-images/pdfs/NICD-Situation-Report-20-December-2017.pdf (accessed on 11 December 2023).
- Self, J.L.; Conrad, A.; Stroika, S.; Jackson, A.; Burnworth, L.; Beal, J.; Wellman, A.; Jackson, K.A.; Bidol, S.; Gerhardt, T.; et al. Outbreak of listeriosis associated with consumption of packaged salad: United States and Canada. MMWR Morb. Mortal. Wkly. Rep. 2016, 65, 879–881.
- Angelo, K.M.; Conrad, A.R.; Saupe, A.; Dragoo, H.; West, N.; Sorenson, A.; Barnes, A.; Doyle, M.; Beal, J.; Jackson, K.A.; et al. Multistate outbreak of Listeria monocytogenes infections linked to whole apples used in commercially produced, prepackaged caramel apples: United States, 2014–2015. Epidemiol. Infect. 2017, 145, 848–856.
- Jackson, B.R.; Salter, M.; Tarr, C.; Conrad, A.; Harvey, E.; Steinbock, L.; Saupe, A.; Sorenson, A.; Katz, L.; Stroika, S.; et al. Listeriosis associated with stone fruit: United States, 2014. MMWR Morb. Mortal. Wkly. Rep. 2014, 64, 282–283.
- Gaul, L.K.; Farag, N.H.; Shim, T.; Kingsley, M.A.; Silk, B.J.; Hyytia-Trees, E. Hospital-acquired listeriosis outbreak caused by contaminated diced celery: Texas, 2010. Clin. Infect. Dis. 2013, 56, 20–26.
- McCollum, J.T.; Cronquist, A.B.; Silk, B.J.; Jackson, K.A.; O’Connor, K.A.; Cosgrove, S.; Gossack, J.P.; Parachini, S.S.; Jain, N.S.; Ettestad, P.; et al. Multistate outbreak of listeriosis associated with cantaloupe. N. Engl. J. Med. 2013, 369, 944–953.
- Silk, B.J.; McCoy, M.H.; Iwamoto, M.; Griffin, P.M. Foodborne listeriosis acquired in hospitals. Clin. Infect. Dis. 2014, 59, 532–540.
- Datta, A.R.; Burall, L.S. Serotype to genotype: The changing landscape of listeriosis outbreak investigations. Food Microbiol. 2017, 75, 18–27.
- Kwong, J.C.; Mercoulia, K.; Tomita, T.; Easton, M.; Li, H.Y.; Bulach, D.M.; Stinear, T.P.; Seemann, T.; Howden, B.P. Prospective whole genome sequencing enhances national surveillance of Listeria monocytogenes. J. Clin. Microbiol. 2016, 54, 333–342.
- De Lucas, E.M.; Mandly, A.G.; Gutiérrez, A.; Pellón, R.; Martín-Cuesta, L.; Izquierdo, J.; Sánchez, E.; Ruiz, E.; Quintana, F. CT-guided fine-needle aspiration in vertebral osteomyelitis: True usefulness of a common practice. Clin. Rheumatol. 2009, 28, 315–320.
- McNamara, A.L.; Dickerson, E.C.; Gomez-Hassan, D.M.; Cinti, S.K.; Srinivasan, A. Yield of Image-Guided Needle Biopsy for Infectious Discitis: A Systematic Review and Meta-Analysis. Am. J. Neuroradiol. 2017, 38, 2021–2027.
- Trampuz, A.; Piper, K.E.; Jacobson, M.J.; Hanssen, A.D.; Unni, K.K.; Osmon, D.R.; Mandrekar, J.N.; Cockerill, F.R.; Steckelberg, J.M.; Greenleaf, J.F.; et al. Sonication of removed hip and knee prostheses for diagnosis of infection. N. Engl. J. Med. 2007, 357, 654–663.
- Marschall, J.; Bhavan, K.P.; Olsen, M.A.; Fraser, V.G.; Wright, N.M.; Warren, D.K. The impact of prebiopsy antibiotics on pathogen recovery in hematogenous vertebral osteomyelitis. Clin. Infect. Dis. 2011, 52, 867–872.
- Charlier, C.; Perrodeau, E.; Leclercq, A.; Cazenave, B.; Pilmis, B.; Henry, B.; Lopes, A.; Maury, M.M.; Moura, A.; Lortholary, O.; et al. Clinical features and prognostic factors of listeriosis: The MONALISA national prospective cohort study. Lancet Infect. Dis. 2017, 17, 510–519.
- Merle-Melet, M.; Dossou-Gbete, L.; Maurer, P.; Meyer, P.; Lozniewski, A.; Kuntzburger, O.; Weber, M.; Gerard, A. Is amoxicillin-cotrimoxazole the most appropriate antibiotic regimen for listeria meningoencephalitis? Review of 22 cases and the literature. J. Infect. 1996, 33, 79–85.
- Louthrenoo, W.; Schumacher, H.R., Jr. Listeria monocytogenes osteomyelitis complicating leukemia: Report and literature review of Listeria osteoarticular infections. J. Rheumatol. 1990, 17, 107–110.
- Del Pozo, J.L.; de la Garza, R.G.; de Rada, P.D.; Ornilla, E.; Yuste, J.R. Listeria monocytogenes septic arthritis in a patient treated with mycophenolate mofetil for polyarteritis nodosa: A case report and review of the literature. Int. J. Infect. Dis. 2013, 17, e132–e133.
- Kubota, T.; Mori, Y.; Yamada, G.; Cammack, I.; Shinohara, T.; Matsuzaka, S.; Hoshi, T. Listeria monocytogenes Ankle Osteomyelitis in a Patient with Rheumatoid Arthritis on Adalimumab: A Report and Literature Review of Listeria monocytogenes Osteomyelitis. Intern. Med. 2021, 60, 3171–3176.
- Adebolu, O.I.; Sommer, J.; Idowu, A.B.; Lao, N.; Riaz, T. Vertebral osteomyelitis and epidural abscess due to Listeria monocytogenes—Case report and review of literature. J. Bone Jt. Infect. 2022, 7, 75–79.
- Khan, K.M.; Pao, W.; Kendler, J. Epidural abscess and vertebral osteomyelitis caused by Listeria monocytogenes: Case report and literature review. Scand. J. Infect. Dis. 2001, 33, 714–716.
- Camp, C.; Luft, W.C. Listeria monocytogenes osteomyelitis. Guthrie Bull. 1973, 43, 32–38.
- Chirgwin, K.; Gleich, S. Listeria monocytogenes osteomyelitis. Arch. Intern. Med. 1989, 149, 931–932.
- Aubin, G.G.; Boutoille, D.; Bourcier, R.; Caillon, J.; Lepelletier, D.; Bémer, P.; Corvec, S. Unusual Case of Spondylodiscitis due to Listeria monocytogenes. J. Bone Jt. Infect. 2016, 1, 7–9.
- Hasan, T.; Chik, W.; Chen, S.; Kok, J. Successful treatment of Listeria monocytogenes prosthetic valve endocarditis using rifampicin and benzylpenicillin in combination with valve replacement. JMM Case Rep. 2017, 4, e005085.
- Duarte, F.; Moreira Pinto, S.; Trigo, A.C.; Guimaraes, F.; Pereira, R.; Neno, M.; Correia de Abreu, R.; Neves, I. A rare presentation of Listeria monocytogenes infection: Perianal abscess associated with lumbar spine osteitis. IDCases 2019, 15, e00488.
- Al Ohaly, R.; Ranganath, N.; Saffie, M.G.; Shroff, A. Listeria spondylodiscitis: An uncommon etiology of a common condition; a case report. BMC Infect. Dis. 2020, 20, 559.
- Bongiovanni, M.; Barda, B.; Martinetti Lucchini, G.; Gaia, V.; Merlani, G.; Bernasconi, E. Invasive Listeriosis in Southern Switzerland: A Local Problem That Is Actually Global. Clin. Infect. Dis. 2023, 77, 161–162.
- Berbari, E.F.; Kanj, S.S.; Kowalski, T.J.; Darouiche, R.O.; Widmer, A.F.; Schmitt, S.K.; Hendershot, E.F.; Holtom, P.D.; Huddleston, P.M., 3rd; Petermann, G.W.; et al. Infectious Diseases Society of America (IDSA) Clinical Practice Guidelines for the Diagnosis and Treatment of Native Vertebral Osteomyelitis in Adults. Clin. Infect. Dis. 2015, 61, e26–e46.
