Mitochondria are double-membrane organelles within eukaryotic cells that act as cellular power houses owing to their ability to efficiently generate the ATP required to sustain normal cell function. Also, they represent a “hub” for the regulation of a plethora of processes, including cellular homeostasis, metabolism, the defense against oxidative stress, and cell death. Mitochondrial dysfunctions are associated with a wide range of human diseases with complex pathologies, including metabolic diseases, neurodegenerative disorders, and cancer. Therefore, regulating dysfunctional mitochondria represents a pivotal therapeutic opportunity in biomedicine. Marine ecosystems are biologically very diversified and harbor a broad range of organisms, providing both novel bioactive substances and molecules with meaningful biomedical and pharmacological applications. Many mitochondria-targeting marine-derived molecules have been described to regulate mitochondrial biology, thus exerting therapeutic effects by inhibiting mitochondrial abnormalities, both in vitro and in vivo, through different mechanisms of action.
- mitochondria
- disease
- therapy
- marine natural products
- marine organisms
1. Introduction
| Compound(s) | Marine Organism |
Mechanism of Action Regarding Mitochondria |
Cell Line or Model of Disease Used in Preclinical Studies |
|---|---|---|---|
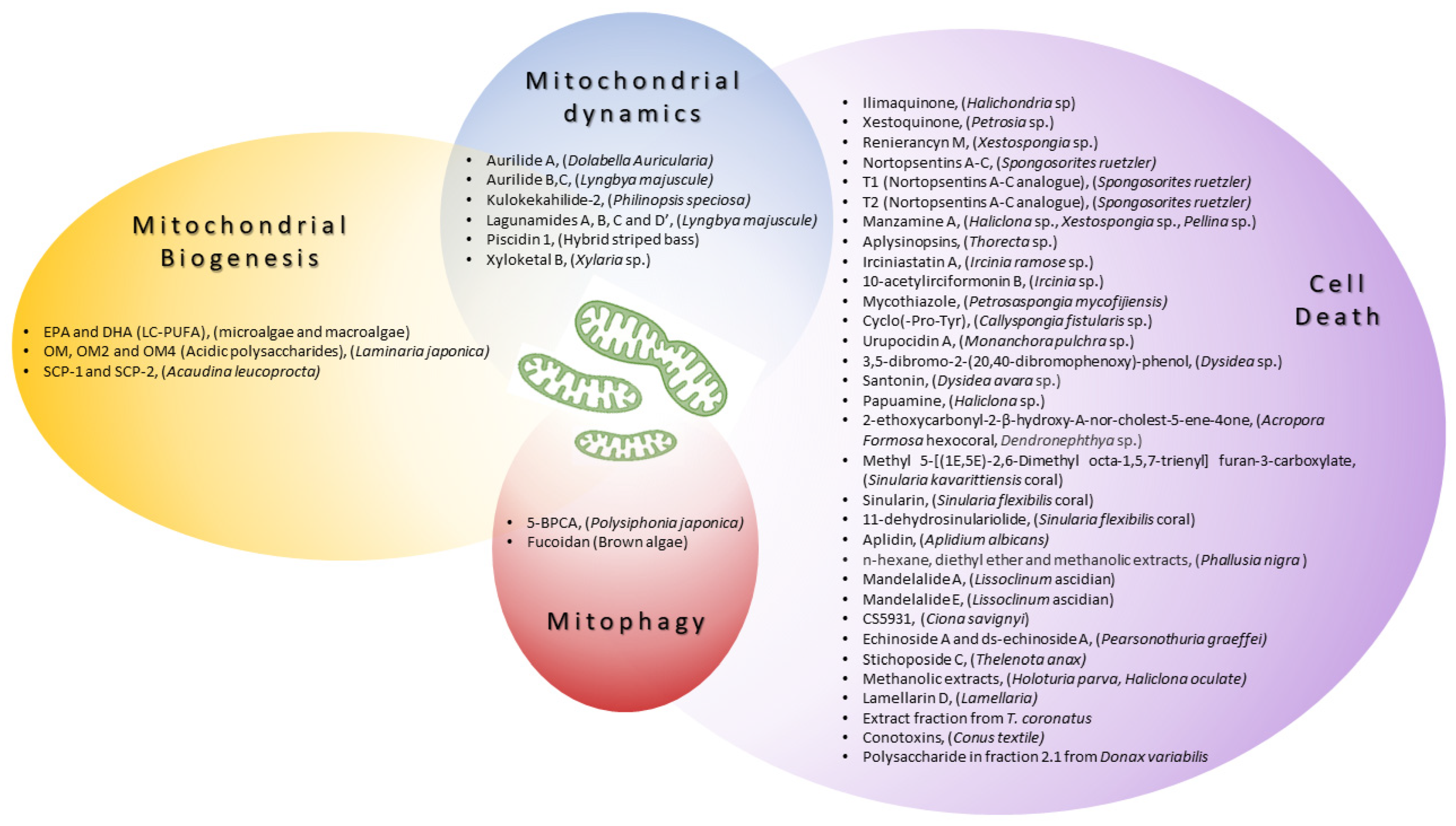
| Mitochondrial Biogenesis | |||
| n-3 PUFA eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA) | Microalgae and macroalgae | ↑PGC1-α, ↑NRF1, ↑mitochondrial biogenesis | C57BL/6J epididymal fat [1] |
| oligomannuronate (OM) and OM-chromium (III) complexes (OM2 and OM4) | Laminaria japonica | ↑PGC1-α, ↑mitochondrial function, ↑mitochondrial biogenesis | C2C12, 3T3-L1 [2] |
| SCP-1 and SCP-2 | Acaudina leucoprocta | ↑AMPK/PGC1-α, ↑NRF2, ↑mitochondrial biogenesis, ↓oxidative stress | Fatigue test in ICR mice [3] |
| Mitochondrial dynamics | |||
| Aurilide A | Dolabella auricularia | Accelerate OPA-1 processing, mitochondrial fragmentation, and the release of CytC [4] | HeLa S3, NCI60 panel [5] |
| Aurilide B, C | Lyngbya majuscule | HCT-8, P388, A549, SK-OV-3, PC-3 [6] | |
| Kulokekahilide-2 | Philinopsis speciosa | P388, SK-OV-3, MDA-MB-435 [7] | |
| Lagunamides A, B, C and D | L. majuscule | P388, A549, PC3, HCT8, SK-OV3, HCT8, MCF7 [8][9] | |
| Piscidin-1 | Hybrid striped bass | ↓MFN1, ↓MFN2, ↓OPA1, ↓OXPHOS, ↑DRP1, ↑FIS1, ↑mtROS, mitochondrial dysfunction, apoptosis | MG63 [10][11] |
| Xyloketal B | Xylaria sp. | ↑Drp1, ↓mitochondrial fregmentation, ↓mitochondrial superoxide production |
In vitro model of ischemic stroke in PC12 [12] |
| Mitophagy | |||
| 5-BPCA | Polysiphonia japonica | The preservation of PARKIN expression and stabilization of mitochondrial morphology | Model of palmitate (PA)-induced lipotoxicity in a rat pancreatic β-cell line (Ins-1 cells) [13] |
| Fucoidan: treatment with Fucoidan nanoparticles loaded with proanthocyanidins | Brown algae | ↑PINK1, ↑PARKIN, ↓mtDNA release | A model of cisplatin-induced damage in vitro (HK-2 cells) and in vivo (Kunming mice) [14] |
| Compound(s) | Marine Organism | In Vitro/In Vivo Models | Mechanism of Action Regarding Mitochondria | Disease Area |
|---|---|---|---|---|
| Ilimaquinone | Halichondria sp. | MCF-7, MDA-MB-231 | caspase activation, ↑ROS, ↓Δψm | Breast cancer [15] |
| Xestoquinone | Petrosia sp. | Molt-4, K562, Sup-T1 | ↑ROS, ↓HSP90 | Leukemia [16] |
| Renieranycin M | Xestospongia sp. | H460 | ↑BAX, ↓MCL1, ↓BCL2, caspase activation | Lung cancer [17][18] |
| Nortopsentins A-C | Spongosorites ruetzler | P388 cells | caspase activation | Leukemia [19] |
| T1 (Nortopsentins A-C analogue) |
Spongosorites ruetzleri | HCT-116 colorectal cancer cells | caspase activation, ↑mitochondrial trans-membrane potential | Colon cancer [20] |
| T2 (Nortopsentins A-C analogue) |
Spongosorites ruetzleri | HCT-116 colorectal cancer cells | caspase activation, ↑mitochondrial trans-membrane potential | Colon cancer [19] |
| Manzamine A | Haliclona sp., Xestospongia sp., Pellina sp. |
HCT116 cells | ↓BCL2, Δψm loss, ↑caspase activation, CytC release, | Colon cancer [21] |
| Aplysinopsins | Thorecta sp. | K562 cells | ↓BCL2, Δψm loss | Leukemia [22] |
| Irciniastatin A | Ircinia ramose sp. | Jurkat cells | ↑ROS, ↑JNK, ↑p38, apoptosis | Leukemia [23] |
| 10-acetylirciformonin B | Ircinia sp. | HL 60 cells | ↓BCL2, ↓Bcl-xL, ↑BAX, ↑ROS, CytC release, apoptosis | Leukemia [23] |
| Mycothiazole | Petrosaspongia mycofijiensis | T47D cells | ↓HIF-1 signaling, ↓mitochondrial function |
Breast tumor [24] |
| Cyclo(-Pro-Tyr) | Callyspongia fistularis sp. | HepG2 cell | ↓BCL2, ↑BAX, ↑ROS, apoptosis | Hepatocellular carcinoma [25] |
| Urupocidin A | Monanchora pulchra sp. | PCa cells | Δψm loss, ↑ROS, CytC release, apoptosis | Prostate cancer [26] |
| 3,5-dibromo-2-(20,40-dibromophenoxy)-phenol | Dysidea sp. | PANC-1 | Complex II inhibition | Pancreatic carcinoma [27] |
| Santonin | Dysidea avara sp. | ALL B-lymphocytes | ↓Δψm, ↑ROS, CytC release, apoptosis | Acute lymphoblastic leukemia [28] |
| Papuamine | Haliclona sp. | MCF-7 | mitochondrial damage and JNK activation | Breast cancer [29] |
| 2-ethoxycarbonyl-2-β-hydroxy-A-nor-cholest-5-ene-4one | Acropora Formosa hexocoral, Dendronephthya sp. | A549 | ↓ TNF-α, ↓IL-8, ↓Bcl2, ↓MMP2, ↓MMP9, ↑ROS, ↑ BAX, ↑p21, CytC release | Lung cancer [30] |
| Methyl 5-[(1E,5E)-2,6-Dimethyl octa-1,5,7-trienyl] furan-3-carboxylate | Sinularia kavarittiensis coral | THP-1 | ↓Bcl-xL, ↑BAX, ↑ROS, ↓Δψm, CytC release, apoptosis |
Leukemia [31] |
| Sinularin | Sinularia flexibilis coral | SK-HEP-1 | ↑ROS, ↓Δψm,↓OXPHOS, apoptosis |
Liver cancer [32][33] |
| 11-dehydro-sinulariolide | Sinularia flexibilis coral | Ca9-22 | ∆Ψm loss, ↑caspase-3/-9 ↑Bax, ↓Bcl-2/Bcl-Xl, CytC release, apoptosis |
Melanoma [32] |
| Aplidin | Aplidium albicans | MOLT-4, NIH3T3 | ↑ROS, ↓Δψm, ↓ATP, apoptosis | Leukemia, Lymphoma [34][35] |
| n-hexane, diethyl ether and methanolic extracts | Phallusia nigra | Isolated mitochondria from skin tissue of melanoma induced albino/Wistar rats | mitochondrial swelling, ↑ROS, ↓Δψm, CytC release, apoptosis | Melanoma [36] |
| Mandelalide A | Lissoclinum ascidian | NCI-H460, Neuro-2A, HeLa cells | complex V inhibition, apoptosis | Lung cancer, Neuroblastoma [37] |
| Mandelalide E | Lissoclinum ascidian | NCI-H460, HeLa, U87-MG, HCT116 | apoptosis | Lung cancer, Glioblastoma [38] |
| CS5931 | Ciona savignyi | HCT-8 | ↑caspase-3, ↑caspase-9, ↑Bax, ↓Δψm, CytC release, apoptosis | Colon cancer [39] |
| Echinoside A and ds-echinoside A | Pearsonothuria graeffei | HepG2, mice | apoptosis | Hepatocarcinoma [40] |
| Stichoposide C | Thelenota anax | HL-60, K562, THP-1, NB4, SNU-C4, HT-29, CT-26; mouse CT-26 subcutaneous tumor and HL-60 leukemia xenograft models | ↑Fas, ↑caspase-3, ↑caspase-8, cleavage of Bid, mitochondrial damage, apoptosis | Leukemia, Colorectal cancer [41] |
| Methanolic extracts | Holoturia parva, Haliclona oculate sp. | Mitochondria isolated from a rat model of hepatocellular carcinoma | ↑ROS, ↓Δψm, CytC release, ↑caspase-3, apoptosis | Hepatocellular carcinoma [42] |
| Lamellarin D | Lamellaria | p388 | ↓Bcl-2, ↓Δψm, ↑caspase-3, ↑caspase-9, apoptosis | Leukemia [43][44] |
| Extract fraction of T. coronatus | Turbo coronatus | EOC cells | ↑ROS, ↓Δψm, CytC release, mitochondrial swelling, apoptosis and necrosis | Epithelial ovarian cancer [45] |
| Conotoxins | Conus textile | U87MG | ↑ROS, ↓Δψm, CytC release, ↑caspase-3, ↑caspase-9, ↑Bax/Bcl-2 | Glioma [46] |
| Polysaccharide in fraction 2.1 | Donax variabilis | A549 | Mitochondrial disfunction, ↓Δψm, CytC release, ↑caspase-3, ↑caspase-9, ↑Bax/Bcl-2, apoptosis | Lung cancer cells [47] |
2. Challenges and Future Perspectives
This entry is adapted from the peer-reviewed paper 10.3390/ijms25020834
References
- Popov, L.D. Mitochondrial biogenesis: An update. J. Cell. Mol. Med. 2020, 24, 4892–4899.
- Halling, J.F.; Pilegaard, H. PGC-1α-mediated regulation of mitochondrial function and physiological implications. Appl. Physiol. Nutr. Metab. 2020, 45, 927–936.
- Chen, Q.; Ruan, D.; Shi, J.; Du, D.; Bian, C. The multifaceted roles of natural products in mitochondrial dysfunction. Front. Pharmacol. 2023, 14, 1093038.
- Eze, C.N.; Onyejiaka, C.K.; Ihim, S.A.; Ayoka, T.O.; Aduba, C.C.; Ndukwe, J.K.; Nwaiwu, O.; Onyeaka, H. Bioactive compounds by microalgae and potentials for the management of some human disease conditions. AIMS Microbiol. 2023, 9, 55–74.
- Furse, S.; Virtue, S.; Snowden, S.G.; Vidal-Puig, A.; Stevenson, P.C.; Chiarugi, D.; Koulman, A. Dietary PUFAs drive diverse system-level changes in lipid metabolism. Mol. Metab. 2022, 59, 101457.
- Flachs, P.; Horakova, O.; Brauner, P.; Rossmeisl, M.; Pecina, P.; Franssen-Van Hal, N.; Ruzickova, J.; Sponarova, J.; Drahota, Z.; Vlcek, C.; et al. Polyunsaturated fatty acids of marine origin upregulate mitochondrial biogenesis and induce beta-oxidation in white fat. Diabetologia 2005, 48, 2365–2375.
- Xie, X.T.; Cheong, K.L. Recent advances in marine algae oligosaccharides: Structure, analysis, and potential prebiotic activities. Crit. Rev. Food Sci. Nutr. 2022, 62, 7703–7717.
- Aziz, E.; Batool, R.; Khan, M.U.; Rauf, A.; Akhtar, W.; Heydari, M.; Rehman, S.; Shahzad, T.; Malik, A.; Mosavat, S.H.; et al. An overview on red algae bioactive compounds and their pharmaceutical applications. J. Complement. Integr. Med. 2020, 17.
- Hao, J.; Hao, C.; Zhang, L.; Liu, X.; Zhou, X.; Dun, Y.; Li, H.; Li, G.; Zhao, X.; An, Y.; et al. OM2, a Novel Oligomannuronate-Chromium(III) Complex, Promotes Mitochondrial Biogenesis and Lipid Metabolism in 3T3-L1 Adipocytes via the AMPK-PGC1α Pathway. PLoS ONE 2015, 10, e0131930.
- Yu, Y.; Wu, G.; Jiang, Y.; Li, B.; Feng, C.; Ge, Y.; Le, H.; Jiang, L.; Liu, H.; Shi, Y.; et al. Sea Cucumber Peptides Improved the Mitochondrial Capacity of Mice: A Potential Mechanism to Enhance Gluconeogenesis and Fat Catabolism during Exercise for Improved Antifatigue Property. Oxid. Med. Cell. Longev. 2020, 2020, 4604387.
- Wang, Q.; Shi, J.; Zhong, H.; Abdullah; Zhuang, J.; Zhang, J.; Wang, J.; Zhang, X.; Feng, F. High-degree hydrolysis sea cucumber peptides improve exercise performance and exert antifatigue effect via activating the NRF2 and AMPK signaling pathways in mice. J. Funct. Foods 2021, 86, 104677.
- Yang, D.; Ying, J.; Wang, X.; Zhao, T.; Yoon, S.; Fang, Y.; Zheng, Q.; Liu, X.; Yu, W.; Hua, F. Mitochondrial Dynamics: A Key Role in Neurodegeneration and a Potential Target for Neurodegenerative Disease. Front. Neurosci. 2021, 15, 654785.
- Marin, W.; Marin, D.; Ao, X.; Liu, Y. Mitochondria as a therapeutic target for cardiac ischemia-reperfusion injury (Review). Int. J. Mol. Med. 2021, 47, 485–499.
- Suenaga, K.; Mutou, T.; Shibata, T.; Itoh, T.; Fujita, T.; Takada, N.; Hayamizu, K.; Takagi, M.; Irifune, T.; Kigoshi, H.; et al. Aurilide, a cytotoxic depsipeptide from the sea hare Dolabella auricularia: Isolation, structure determination, synthesis, and biological activity. Tetrahedron 2004, 60, 8509–8527.
- Suenaga, K.; Mutou, T.; Shibata, T.; Itoh, T.; Kigoshi, H.; Yamada, K. Isolation and stereostructure of aurilide, a novel cyclodepsipeptide from the Japanese sea hare Dolabella auricularia. Tetrahedron Lett. 1996, 37, 6771–6774.
- Han, B.; Gross, H.; Goeger, D.E.; Mooberry, S.L.; Gerwick, W.H. Aurilides B and C, cancer cell toxins from a Papua New Guinea collection of the marine cyanobacterium Lyngbya majuscula. J. Nat. Prod. 2006, 69, 572–575.
- Nakao, Y.; Yoshida, W.Y.; Takada, Y.; Kimura, J.; Yang, L.; Mooberry, S.L.; Scheuer, P.J. Kulokekahilide-2, a cytotoxic depsipeptide from a cephalaspidean mollusk Philinopsis speciosa. J. Nat. Prod. 2004, 67, 1332–1340.
- Tripathi, A.; Puddick, J.; Prinsep, M.R.; Rottmann, M.; Chan, K.P.; Chen, D.Y.K.; Tan, L.T. Lagunamide C, a cytotoxic cyclodepsipeptide from the marine cyanobacterium Lyngbya majuscula. Phytochemistry 2011, 72, 2369–2375.
- Tripathi, A.; Puddick, J.; Prinsep, M.R.; Rottmann, M.; Tan, L.T. Lagunamides A and B: Cytotoxic and antimalarial cyclodepsipeptides from the marine cyanobacterium Lyngbya majuscula. J. Nat. Prod. 2010, 73, 1810–1814.
- Luo, D.; Putra, M.Y.; Ye, T.; Paul, V.J.; Luesch, H. Isolation, Structure Elucidation and Biological Evaluation of Lagunamide D: A New Cytotoxic Macrocyclic Depsipeptide from Marine Cyanobacteria. Mar. Drugs 2019, 17, 83.
- Kaneda, M.; Kawaguchi, S.; Fujii, N.; Ohno, H.; Oishi, S. Structure-Activity Relationship Study on Odoamide: Insights into the Bioactivities of Aurilide-Family Hybrid Peptide-Polyketides. ACS Med. Chem. Lett. 2018, 9, 365–369.
- Tripathi, A.; Fang, W.; Leong, D.T.; Tan, L.T. Biochemical studies of the lagunamides, potent cytotoxic cyclic depsipeptides from the marine cyanobacterium Lyngbya majuscula. Mar. Drugs 2012, 10, 1126–1137.
- Sato, S.I.; Murata, A.; Orihara, T.; Shirakawa, T.; Suenaga, K.; Kigoshi, H.; Uesugi, M. Marine natural product aurilide activates the OPA1-mediated apoptosis by binding to prohibitin. Chem. Biol. 2011, 18, 131–139.
- Semenzato, M.; Cogliati, S.; Scorrano, L. Prohibitin(g) cancer: Aurilide and killing by Opa1-dependent cristae remodeling. Chem. Biol. 2011, 18, 8–9.
- Wasilewski, M.; Scorrano, L. The changing shape of mitochondrial apoptosis. Trends Endocrinol. Metab. 2009, 20, 287–294.
- Michon, S.; Cavelier, F.; Salom-Roig, X.J. Synthesis and Biological Activities of Cyclodepsipeptides of Aurilide Family from Marine Origin. Mar. Drugs 2021, 19, 55.
- Lin, H.J.; Huang, T.C.; Muthusamy, S.; Lee, J.F.; Duann, Y.F.; Lin, C.H. Piscidin-1, an antimicrobial peptide from fish (hybrid striped bass morone saxatilis x M. chrysops), induces apoptotic and necrotic activity in HT1080 cells. Zoolog. Sci. 2012, 29, 327–332.
- Cheng, M.H.; Pan, C.Y.; Chen, N.F.; Yang, S.N.; Hsieh, S.; Wen, Z.H.; Chen, W.F.; Wang, J.W.; Lu, W.H.; Kuo, H.M. Piscidin-1 Induces Apoptosis via Mitochondrial Reactive Oxygen Species-Regulated Mitochondrial Dysfunction in Human Osteosarcoma Cells. Sci. Rep. 2020, 10, 5045.
- Zhao, J.; Li, L.; Ling, C.; Li, J.; Pang, J.Y.; Lin, Y.C.; Liu, J.; Huang, R.; Wang, G.L.; Pei, Z.; et al. Marine compound Xyloketal B protects PC12 cells against OGD-induced cell damage. Brain Res. 2009, 1302, 240–247.
- Nigam, M.; Suleria, H.A.R.; Farzaei, M.H.; Mishra, A.P. Marine anticancer drugs and their relevant targets: A treasure from the ocean. Daru 2019, 27, 491–515.
- Cha, S.H.; Zhang, C.; Heo, S.J.; Jun, H.S. 5-Bromoprotocatechualdehyde Combats against Palmitate Toxicity by Inhibiting Parkin Degradation and Reducing ROS-Induced Mitochondrial Damage in Pancreatic β-Cells. Antioxidants 2021, 10, 264.
- Barbosa, A.I.; Coutinho, A.J.; Costa Lima, S.A.; Reis, S. Marine Polysaccharides in Pharmaceutical Applications: Fucoidan and Chitosan as Key Players in the Drug Delivery Match Field. Mar. Drugs 2019, 17, 654.
- Zhang, X.; Wei, Z.; Xue, C. Physicochemical properties of fucoidan and its applications as building blocks of nutraceutical delivery systems. Crit. Rev. Food Sci. Nutr. 2022, 62, 8935–8953.
- Gao, X.; Yin, Y.; Liu, S.; Dong, K.; Wang, J.; Guo, C. Fucoidan-proanthocyanidins nanoparticles protect against cisplatin-induced acute kidney injury by activating mitophagy and inhibiting mtDNA-cGAS/STING signaling pathway. Int. J. Biol. Macromol. 2023, 245, 125541.
- Zuo, W.; Kwok, H.F. Development of Marine-Derived Compounds for Cancer Therapy. Mar. Drugs 2021, 19, 342.
- Nova, P.; Gomes, A.M.; Costa-Pinto, A.R. It comes from the sea: Macroalgae-derived bioactive compounds with anti-cancer potential. Crit. Rev. Biotechnol. 2023.
- Fakhri, S.; Abdian, S.; Moradi, S.Z.; Delgadillo, B.E.; Fimognari, C.; Bishayee, A. Marine Compounds, Mitochondria, and Malignancy: A Therapeutic Nexus. Mar. Drugs 2022, 20, 625.
- Wang, E.; Sorolla, M.A.; Krishnan, P.D.G.; Sorolla, A. From Seabed to Bedside: A Review on Promising Marine Anticancer Compounds. Biomolecules 2020, 10, 248.
- Ruiz-Torres, V.; Encinar, J.A.; Herranz-López, M.; Pérez-Sánchez, A.; Galiano, V.; Barrajón-Catalán, E.; Micol, V. An Updated Review on Marine Anticancer Compounds: The Use of Virtual Screening for the Discovery of Small-Molecule Cancer Drugs. Molecules 2017, 22, 1037.
- Bai, L.Y.; Su, J.H.; Chiu, C.F.; Lin, W.Y.; Hu, J.L.; Feng, C.H.; Shu, C.W.; Weng, J.R. Antitumor Effects of a Sesquiterpene Derivative from Marine Sponge in Human Breast Cancer Cells. Mar. Drugs 2021, 19, 244.
- Wang, K.C.; Lu, M.C.; Hsu, K.C.; El-Shazly, M.; Shih, S.P.; Lien, S.T.; Kuo, F.W.; Yang, S.C.; Chen, C.L.; Yang, Y.C.S.H. The Antileukemic Effect of Xestoquinone, A Marine-Derived Polycyclic Quinone-Type Metabolite, Is Mediated through ROS-Induced Inhibition of HSP-90. Molecules 2021, 26, 7037.
- Sirimangkalakitti, N.; Chamni, S.; Suwanborirux, K.; Chanvorachote, P. Renieramycin M Attenuates Cancer Stem Cell-like Phenotypes in H460 Lung Cancer Cells. Anticancer Res. 2017, 37, 615–622.
- Pinkhien, T.; Maiuthed, A.; Chamni, S.; Suwanborirux, K.; Saito, N.; Chanvorachote, P. Bishydroquinone Renieramycin M Induces Apoptosis of Human Lung Cancer Cells Through a Mitochondria-dependent Pathway. Anticancer Res. 2016, 36, 6327–6333.
- Carbone, A.; Pennati, M.; Barraja, P.; Montalbano, A.; Parrino, B.; Spano, V.; Lopergolo, A.; Sbarra, S.; Doldi, V.; Zaffaroni, N.; et al. Synthesis and antiproliferative activity of substituted 3-1H-pyrrolopyridines, marine alkaloid nortopsentin analogues. Curr. Med. Chem. 2014, 21, 1654–1666.
- Diana, P.; Carbone, A.; Barraja, P.; Martorana, A.; Gia, O.; DallaVia, L.; Cirrincione, G. 3,5-bis(3′-indolyl)pyrazoles, analogues of marine alkaloid nortopsentin: Synthesis and antitumor properties. Bioorg. Med. Chem. Lett. 2007, 17, 6134–6137.
- Lin, L.C.; Kuo, T.T.; Chang, H.Y.; Liu, W.S.; Hsia, S.M.; Huang, T.C. Manzamine A Exerts Anticancer Activity against Human Colorectal Cancer Cells. Mar. Drugs 2018, 16, 252.
- Song, S.; Kim, S.; El-Sawy, E.R.; Cerella, C.; Orlikova-Boyer, B.; Kirsch, G.; Christov, C.; Dicato, M.; Diederich, M. Anti-Leukemic Properties of Aplysinopsin Derivative EE-84 Alone and Combined to BH3 Mimetic A-1210477. Mar. Drugs 2021, 19, 285.
- Voultsiadou Eleni, E. Therapeutic properties and uses of marine invertebrates in the ancient Greek world and early Byzantium. J. Ethnopharmacol. 2010, 130, 237–247.
- Chen, W.; Zhao, H.; Li, Y. Mitochondrial dynamics in health and disease: Mechanisms and potential targets. Signal Transduct. Target. Ther. 2023, 8, 333.
- Saide, A.; Lauritano, C.; Ianora, A. A Treasure of Bioactive Compounds from the Deep Sea. Biomedicines 2021, 9, 1556.
- Sikorska, J.; Hau, A.M.; Anklin, C.; Parker-Nance, S.; Davies-Coleman, M.T.; Ishmael, J.E.; McPhail, K.L. Mandelalides A–D, cytotoxic macrolides from a new Lissoclinum species of South African tunicate. J. Org. Chem. 2012, 77, 6066–6075.
- Romano, G.; Almeida, M.; Coelho, A.V.; Cutignano, A.; Gonçalves, L.G.; Hansen, E.; Khnykin, D.; Mass, T.; Ramšak, A.; Rocha, M.S.; et al. Biomaterials and Bioactive Natural Products from Marine Invertebrates: From Basic Research to Innovative Applications. Mar. Drugs 2022, 20, 219.
- García-Poza, S.; Leandro, A.; Cotas, C.; Cotas, J.; Marques, J.C.; Pereira, L.; Gonçalves, A.M.M. The Evolution Road of Seaweed Aquaculture: Cultivation Technologies and the Industry 4.0. Int. J. Environ. Res. Public Health 2020, 17, 6528.
- Ding, Q.; Ye, C. Microbial cell factories based on filamentous bacteria, yeasts, and fungi. Microb. Cell Fact. 2023, 22, 20.
- Fatehullah, A.; Tan, S.H.; Barker, N. Organoids as an in vitro model of human development and disease. Nat. Cell Biol. 2016, 18, 246–254.
