Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is an old version of this entry, which may differ significantly from the current revision.
Since CO2 is an important component of gas emissions, its removal from gas streams is of the utmost importance to fulfill various environmental requirements. The technologies used to accomplish this removal are based mainly on absorption, as well as adsorption and membrane processing. Among the materials used in the above separation processes, materials in nano forms offer a potential alternative to other commonly used macromaterials.
- CO2
- absorption
- adsorption
- membranes
1. Introduction
One of the main global concerns is related to the increasing concentration of CO2 in the atmosphere, a consequence of the use of fossil fuels, boosting the greenhouse impact. There is no doubt that greenhouse gas emissions produce climate problems, resulting in challenges in reaching sustainable development. Greenhouse gas emissions are mainly a result of energy production (nearly 70% of global emissions). Many countries have considered the importance of reducing CO2 emissions to fulfill environmental policies related to zero-carbon discharge, helping to reduce the presence of carbon in a number of sectors, with special interest in the energy sector. However, the time to achieve the transition from today’s polluting energy production to a future of zero-emissions technology still seems long, and this is because there is a constant flux of information and developments to improve the capture of contaminant gases (CO2, H2S, CH4, etc.) from gas streams in order to clean them and contribute to a better environment.
Among the technologies employed for this capture, and in the case of CO2 specifically, absorption, adsorption, and membrane technologies are the most widely proposed. Several recent reviews on the utilization of these technologies have been published, including the use of membranes [1]; MXene-based membranes [2]; composite membranes [3][4]; microporous membranes composed of nanopores [5]; nanomaterials [6][7][8][9][10][11], and more specifically, graphene and its 2D nanomaterial derivatives [12]; nanomaterials derived with support from artificial intelligence (AI) [13]; kaolinite-based nanomaterials [14]; MXene nanoderivatives [15]; azobenzene-based supramolecular materials [16]; carbon-bearing nanomaterials [17]; nanobiotechnology using microalgae [18]; nanomaterials for catalyst-assisted solvent regeneration in absorption processes using amine [19]; and finally, some general reviews on technologies for CO2 removal from gas streams [20][21][22][23].
2. Nanomaterials and CO2 Absorption
With quick kinetics, absorption techniques produce remarkable CO2 removal (90%) from gases. The most-used absorbents to achieve this removal include amines, ammonia–water solutions, and alkali compounds. Both pre- and post-combustion processes can integrate these technologies into their general procedure. Besides physical absorption, chemical-driven absorption processes have three components: absorber, solvent, and stripper. Different pieces of equipment were used to capture CO2, i.e., spray columns, packed beds, rotating packed beds, bubble columns, and tray tower absorber layouts.
The absorption of CO2 on Al2O3/MeOH [24] was investigated, and the addition of a porous nickel metal foam increased CO2 capture compared to pure MeOH; this increase was attributable to the forced bubble-breaking mechanism and the hydrodynamics in relation to the process. Moreover, there was a direct relation with the CO2 absorption and the pressure and temperature used in the CO2 capture process (Figure 1). For a constant pressure, increasing the temperature decreased the gas capture, whereas for a constant temperature, increasing the pressure produced a greater removal of CO2 from the gas stream. Also, an increase (0.01–0.1 wt%) in the Al2O3 content is accompanied by an increase in the capture of CO2 with the alcohol.
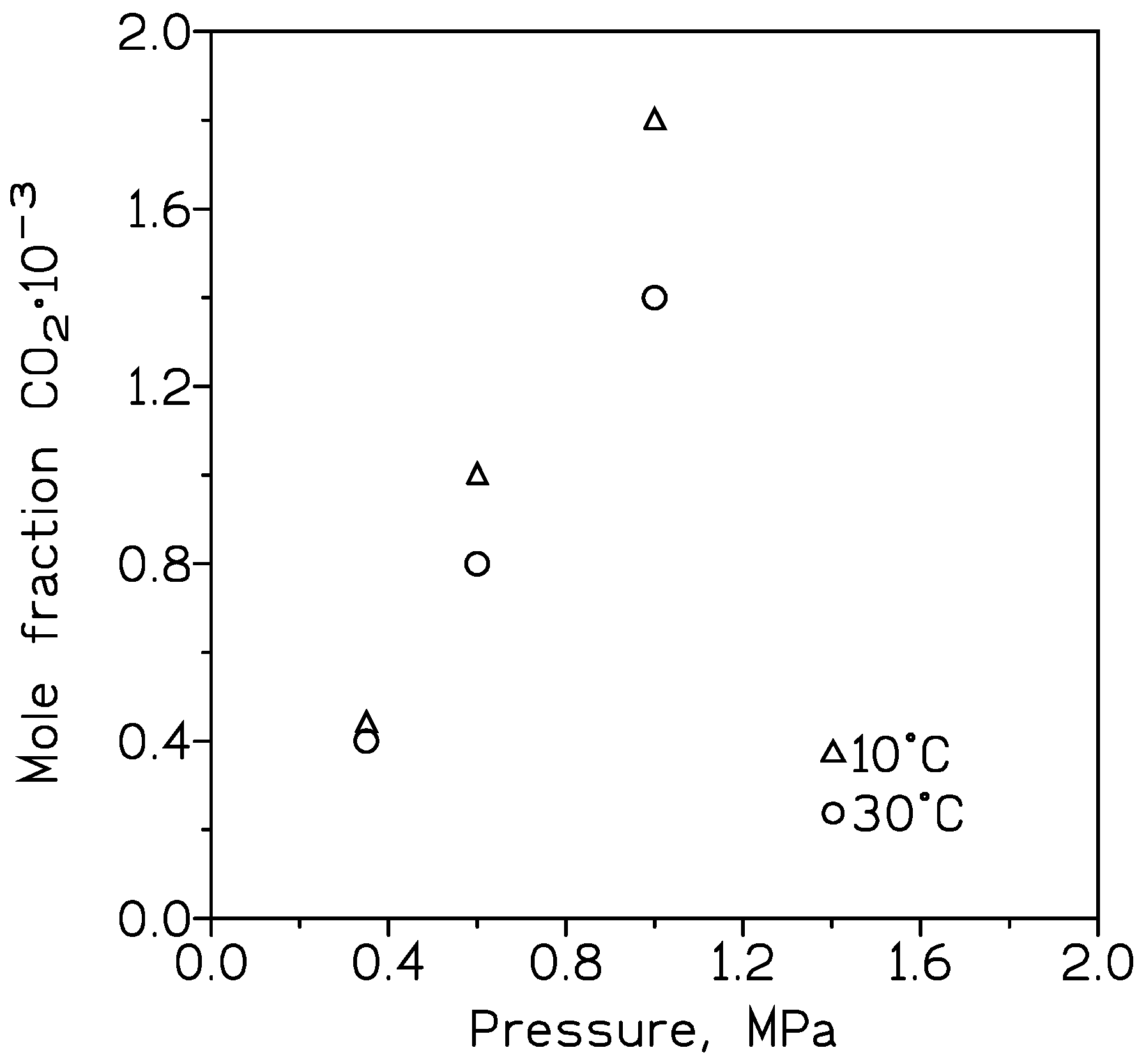
Figure 1. Approximate CO2 removal in MeOH at various temperatures and pressures.
CO2 capture by means of the addition of tetra-n-butyl ammonium bromide (TBAB) semiclathrate to graphite, forming a TBAB+graphite nanofluid, was investigated [25]. The TBAB+graphite nanofluid system increased CO2 removal with respect to the use of the TBAB or TBAB+sodium dodecyl sulfate (SDS) solutions, with 0.2 wt% graphite nanoparticles being the best concentration for hydrate growth in the TBAB+graphite nanofluid.
A metal oxide solid acid catalyst for the catalytic regeneration of a CO2-rich 5 M monoethanolamine (MEA) solution at 90 °C was used [26]. After the adsorption process and the formation of MEACOO−, carbamate decomposition occurred:
This was followed by protonated amine deprotonation and the formation of MEA, H3O+, and H2CO3. The utilization of SO42−/ZrTiOx allowed for a 99% increase in the CO2 desorption rate and a 43% enhancement after continuous cycles.
The performance of single-walled, carbon-deficient silicon carbide nanotubes (Si12C2−X; X = 1; 2) in CO2 capture was investigated [27]. The investigation concluded that Si12C11:Vc1 and Si12C10:Vc2 performed well for CO2 removal and storage, surpassing the CO2 capture efficiency of pristine SWSiCNT. The absorbent properties were due to the C vacancy effect of photoabsorption.
5,6-Dimethylbenzimidazole replaced 2-methylimidazole on the surface of ZIF-8 crystals via the shell–ligand exchange reaction (SLER) [28], improving the thermal stability of ZIF-8. At 40 °C, CO2 uptake using ZIF-8-SLER-PLs increased 30% with respect to that of ZIF-8-PLs. CO2 was loaded onto the modified absorbent via a physical absorption process.
It was demonstrated [29] that the use of nano-SiO2, with varying particle size, in glycerol solution had little effect on CO2 removal; this capture increased with the increase in solid mass in the 0.05–0.15 wt% range, which was attributed to a better gas–liquid mass transfer area. The use of SiO2 increased CO2 desorption as consequence of the presence of more nucleation sites and the heating rate of the base fluid, generating bubbles.
MFCs (MgFe2O4@ZIF-62) containing various magnetic nanoparticle dosages (1–6 wt% of magnetic nanoparticles to ZIF-62 mass) were used in conjunction with a compatible non-penetrating solvent to form a magnetic porous liquid [30]. The use of this absorbent material allowed continuous CO2 capture and release for up to three cycles.
Due to certain operational and economic considerations, there is a relatively urgent necessity to find alternatives to the use of amines for CO2 capture [31]; amino acid salts can be one green alternative to the use of these amines. Thus, potassium L-cysteine for CO2 capture from natural gas was investigated. Its physicochemical properties were measured at different temperatures (25–60 °C) and salt concentrations (5–30 wt%). Experimental results, at 40 °C and 20 bar, showed an important increment of CO2 loading, from 7 to 15 mmol CO2/g amino acid, with the increase of 10–30 wt% in the solvent concentration. CO2 loading was attributable to the following reaction:
while carbonate formation occurred via the following reaction:
By the use of 9.01 wt% tetra butyl ammonium bromide (TBAB) mixed with water-soluble hydroxylated multiwalled carbon nanotube (MWCNTol) material, the formation of CO2 hydrate was investigated [32]. It was concluded that MWCNTols had negligible influence on the CO2 hydrate generation. The use of nanoparticles such as graphene nanoribbons and MWCNTols reduced the induction time, whereas addition of various nanoparticle dosages to the TBAB solution increased the final gas consumption, with a maximum increase of 10.44% in the 9.01 wt% TBAB + 0.08 wt% GN system.
Absorption and conversion of CO2 by an amino-acid-based nanotechnology was described [33]. CO2 was captured as bicarbonate nanomaterials, whereas the amino acid structure governed the formation of bicarbonate nanomaterials. Amino acids presented higher CO2 absorption capacity and faster kinetics compared to the use of 30 wt% monoethanolamine.
Gold nanoparticles were decorated with 1,5,7-triazabicyclo [4.4.0] dec-5-ene and dispersed into methanol in order to capture CO2 [34]. The photocatalyst consisted of two parts: (i) an organic shell responsible for CO2 capture, and (ii) a plasmon-active metal nanoparticle core for activation of captured CO2 and its involvement in the cycloaddition reaction. Results showed the efficiency of the procedure even at the temperature of −40 °C.
The mixture formed by NaP zeolite nanocrystals and 1-dodecyl-3-methylimidazolium chloride ([C12mim][Cl]) ionic liquid was used for CO2 removal in an isothermal high-pressure cell equipped with magnetic stirring [35]. Under various experimental conditions, it was found that 0.02 wt% of zeolite nanoparticles, 0.4 wt% of [C12mim][Cl] ionic liquid, and 0.05 wt% of sodium dodecyl benzene sulfonate in nanofluids resulted in the highest CO2 removal compared to other conditions. This CO2 removal increased by increasing ionic liquid and surfactant concentration up to a limiting value near the critical micelle concentration.
Methyl-diethanolamine-based Fe3O4 improved CO2 absorption compared to methyl-diethanolamine-based CuO, ZnO, and SiO2, whereas CuO nanoparticles presented higher efficiency for CO2 removal from gas-loaded absorbent [36].
Using 2-methylimidazole zinc salt (ZIF-8) modified by tetraethylenepentamine (TEPA), which provided pores, and 1-ethyl-3-methylimidazolium bis(trifluoro-methanesulfonyl)-imide ([EMlm][NTf2]) ionic liquid, used as a sterically hindered diluent, an amine-functionalized type III porous liquid was formed [37]. It was found that with 30TEPA@ZIF-8 nanoparticles, the best CO2 absorption capacity was obtained. Moreover, the CO2 absorption loading of 0.124 mmol/g presented by 5-30TEPA@ZIF-8/[EMlm][NTf2] was 4.43 times higher than the value obtained by the use of 5-0TEPA@ZIF-8/[EMlm][NTf2], whereas a 745% increase of the absorption rate was reached.
A mixture of 26 mol% CO2 and 74 mol% CH4 was used to investigate the separation of both gases [38]. Silica nanoparticles, in KOH medium, modifying the surface of (3-aminopropyl) teriethoxysilane (APTES) were utilized as additives. The best results were derived with silica and KOH-bearing nanofluids; with these components, improvements of 36% (gas consumption), 29% (separation factor), and 38% (recovery factor) resulted with respect to the use of pure water.
CO2 geological sequestration by the use of silica aerogel nanofluid was investigated [39]. Using this material, non-dissolved CO2 molecules were captured in the nanopores of the silica aerogel nanoparticles, increasing the solubility of CO2 in the aqueous phase. Aerogel nanoparticles adsorbed at the CO2–brine interface reduced the interfacial tension.
3. Nanomaterials and CO2 Adsorption
Adsorption processing involves the use of a solid material on which CO2 (and other gases and solutes) is captured by means of physical or chemical processes or a combination of both. Key parameters to yield the best adsorptive properties of the materials are: porosity, pore size, operational stability, presence of reactive groups towards CO2 adsorption, etc., whereas the equipment used is usually described as a packed or fluidized bed.
The fabrication of carbon nanofibers (CNFs) via biaxial electrospinning was investigated [40]. Polymethylmethacrylate (PMMA) and polyacrylonitrile (PAN) were used as core and shell precursors, respectively. Further, Co3O4 nanoparticles were included in the PAN shell, increasing its roughness and surface area. The uniform distribution of Co3O4 resulted in a better flexibility of the hollow carbon nanofiber material (HCNF-Co), providing more vacant oxygen sites to increase CO2 adsorption loading. HCNF-Co nanofibers exhibited CO2 capture uptake of 3.28 mmol/g at 25 °C. Experimental results indicated that HCNF-Co had remarkable CO2 selectivity (S = 26) over N2.
Heterojunctions of Co3O4 with different morphologies and modified carbon nitride (CN) were investigated in order to optimize their properties to degrade CO2 under UV–visible irradiation [41]. A solvothermal synthesis was used to fabricate the cobalt oxide from metal–organic framework structures, yielding ultrathin 2D Co3O4 nanosheets (Co3O4-NS). These nanosheets presented improved photocatalytic properties compared to those of the bulk Co3O4/CN composites. CO2 reduction was improved due to (i) the match of the planar surface of CN and the 2D structure of Co3O4-NS, which resulted in a larger interface, and (ii) improvement in charge carrier lifetime.
The authors of [42] described the utilization of 2D nanomaterial MXenes and activated carbon (AC) to form sandwich-type materials and nanocomposites for CO2 adsorption using a fixed-bed column. These investigations included CO2 breakthrough measurements at a fixed 15% CO2 concentration, with an inlet flow rate at 200 mL/min and temperatures in the 25–55 °C range. The highest CO2 adsorption load (near 9 mg/g) was yielded with AC/MXene sandwich adsorbent at 25 °C, which was nearly a 37% improvement in CO2 adsorption capacity over the use of pristine AC. AC/MXene sandwich-type nanomaterials can be used, with a small loss of their CO2 adsorption uptake, under various cyclic experiments.
ZIF-8 hollow nanospheres, for selective CO2 separation and storage, were developed [43]. The optimum hollow ZIF-8 nanosphere material, with a uniform size distribution (Figure 2), had a CO2 adsorption uptake of 2.24 mmol/g at 0 °C and 1.75 bar, selective (12.15) CO2/N2 separation, 1.5–1.75 wt% CO2 storage capacity, and a reasonable stability, up to four CO2 adsorption/desorption cycles, at 25 °C.
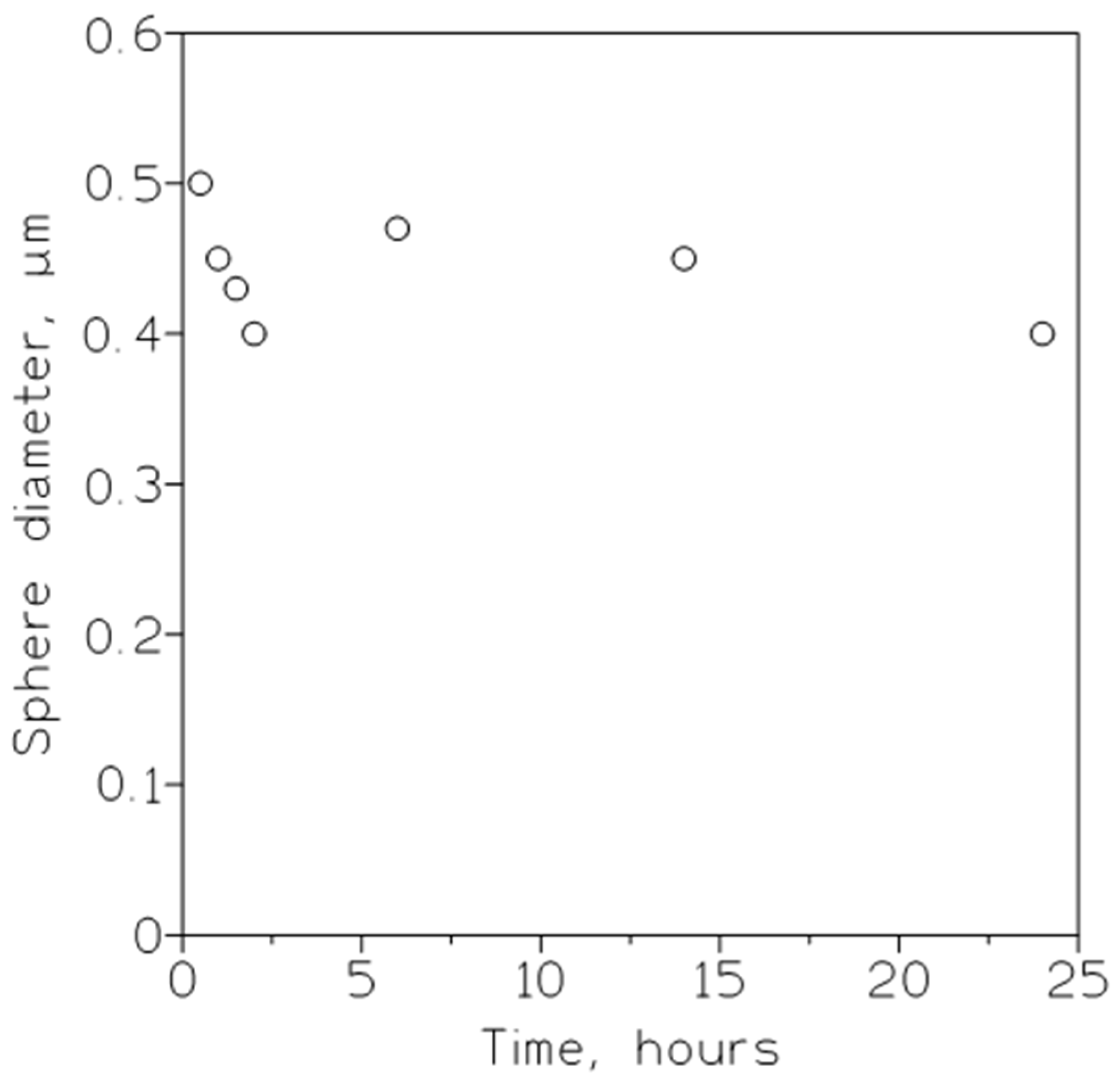
Figure 2. Influence of synthesis time on the average diameter of soft template hollow ZIF-8 nanospheres. Surfactant/oil ratio: 75 g/L.
A heterogeneous catalyst comprising silver nanoparticles and a porous N-heterocyclic carbene polymer (Ag@POP-NL-3) was developed [44]. This nanomaterial has a regular distribution of silver nanoparticles and nitrogen activation groups. The catalyst presented good properties for the selective adsorption and activation of CO2, allowing the conversion, under mild conditions, of low CO2 (30 vol%) concentrations, as presented in lime kiln waste gas, into cyclic carbonate. CO2 was loaded onto the adsorbent by carboxylative cyclization of the gas with propargylic alcohols also present in the system.
The adsorption uptake of CO2 on NaY@polyacrylate matrix was increased by 17.9% while H2O adsorption uptake decreased by 36.6% compared to pristine NaY [45]. In addition, H2O adsorption was reduced by 54.8% after adding ZIF into composites.
The authors of [46] described a maximum CO2 loading (0.75 mmol/g) on triethylamine-doped rice husk silica nanoparticles, with an average increase in CO2 adsorption with the increase (1 to 5 wt%) in the amine loading on the surface modifiers. Amine loadings greater than 5 wt% produced agglomeration of the particles which is detrimental with respect to CO2 capture. CO2 uptake corresponded to the Langmuir isotherm model.
The use of Zn-N pillar MOFs resulted in: CO2 capture of 3.82 mmol/g (25 °C and 101 kPa), a selectivity CO2/N2 factor of 132, and stable structure (no change after exposure to 1000% RH environment for seven days) [47].
The performance of graphene oxide (GO)-coated zinc tetraphenylporphyrin (ZnTPP/GO) nanocomposites in the photocatalytic degradation of CO2 was investigated [48]. The encapsulation of GO in ZnTPP nanocrystals promotes CO2 adsorption, interfacial reaction, and stability and accelerates the separation of photoinduced carriers on ZnTPP (0.1 ps vs. 425.9 ps), the transportation from ZnTPP to GO (2.3 ps vs. 83.6 ps), and their final enrichment on GO.
A porous ZIF-11@ZIF-8 core–shell composite structure metal–organic framework was fabricated using the solvent-assisted linker exchange (SALE) procedure [49]. Adsorptions at 25 °C and equilibrium pressures up to 4 bar showed an increase (near 100%) in CO2 adsorption uptake of ZIF-11@ZIF-8 nanoparticles (8.21 mmol/g) compared to the pristine ZIF-11 (4.35 mmol/g). Experimental results on gas uptake fitted well with the Langmuir isotherm equation. CO2/N2 and CO2/CH4 selectivities also increased by 131% and 92%, respectively.
Activated carbon (AC) was synthesized from date fruit seeds and chemically activated with KOH to improve CO2 loading [50]. From thermogravimetric analyses, 94% and 67% higher average CO2 capture loads were measured for KOH-promoted ACs compared to the original adsorbents. The activated carbon improved its fluidization by the use of hydrophobic silica nanoparticles (NPs). The SiO2-decorated (2.5 wt%) modified ACs had a 45% higher bed expansion ratio, which was associated with the absence of bubbles and a homogeneous fluidized regime.
Mesoporous CeO2, ZrO2, and Ce-Zr composite nanoparticles with a large surface area were fabricated using the hydrothermal template-assisted synthesis procedure, and CO2 adsorption properties of these materials were investigated under equilibrium and dynamic operations [51]. Better CO2 adsorption was yielded for Ce-Zr nanomaterial due to the presence of strong O2− base sites and many surface oxygen species. After five adsorption/desorption cycles, the composites presented a reasonable stability with a slight decrease in CO2 adsorption uptakes in dry flow and in the presence of water vapor.
Treatment via surface N2 plasma of zinc porphyrin (ZnTCPP) ultrathin nanosheets induced nitrogen vacancies (NVs) and resulted in a material with photocatalytic CO2 reduction activity and selectivity [52]. It was shown that the photocatalytic activity of NVs-ZnTCPP can be attributed to nitrogen-vacancy-induced spin polarization by reducing the reaction barriers and inhibiting the recombination of photoexcited carriers.
It was reported [53] that CO2 uptake (11.8 mmol/g (78% total adsorption)) after four cycles on a MOF-derived nano-CaO (average size of 100 nm) was due to high stability produced in the final material by the change in the original fiber-bundle-like MOF structure to nanosheets, and further to regular CaO spheres. CO2 uptake onto the adsorbent corresponded to the following equation:
Lewis base and dual hydrogen bond donor (HBD) units were integrated into an organosilicon precursor, and triazine and hydrazo site co-modified periodic mesoporous organosilicas (THPMOs) were prepared via a hydrothermal self-assembly method [54]. The THPMOs had BET surface areas in the 699–876 m2/g range and low-pressure CO2 adsorption loadings at 0 °C. If combined with tetrabutylammonium iodide (TBAI) ionic liquid, the mixture promoted the model cycloaddition of CO2 in an effective form, with the gas fixed to epoxides.
In [55], nano-TiO2 was added to cement pastes to investigate its performance regarding the CO2 uptake rate. Prismatic samples with dimensions of 16 × 4 × 4 cm of 0.5 water/binder cement paste with and without nano-TiO2 particles were used. CO2 uptakes showed that nano-TiO2 addition improves the CO2 uptake rate of cement pastes, changing the pore structure and allowing the removal of more CO2 at lower gas concentrations. CO2 loaded similarly to Equation (4), but Ca(OH)2 reacted with the gas to form CaCO3 and water.
MIL-101(Cr)-NH2 has higher CO2 adsorption capacity than MIL-101(Fe)-NH2 [56], whereas the adsorption of methane and nitrogen by MIL-101(Cr)-NH2 is lower than the adsorption of these gases by MIL-101(Fe)-NH2, leading to a higher selectivity of CO2 over the two gases for MIL-101(Cr)-NH2. At elevated temperature and pressure, the chemisorption mechanism is predominant, which is attributable to the performance of amines, which adsorbed more CO2 at these higher temperatures and pressure. Gas adsorption was explained by the use of a hybrid equation between Langmuir and Khan models:
where qs, Q, and n represent the parameters of the model, with P being the pressure of the vapor phase at the equilibrium.
This entry is adapted from the peer-reviewed paper 10.3390/separations11010001
References
- Yuan, Z.; Tang, J.; Chen, D.; Li, Y.; Hong, Z.; He, X. Membranes for hydrogen rainbow toward industrial decarbonization: Status, challenges and perspectives from materials to processes. Chem. Eng. J. 2023, 470, 144328.
- Ahmad, I.; Alayande, A.B.; Jee, H.; Wang, Z.; Park, Y.-J.; Im, K.S.; Nam, S.Y. Recent progress of MXene-based membranes for high-performance and efficient gas separation. Diam. Relat. Mater. 2023, 135, 109883.
- Dai, Y.; Niu, Z.; Luo, W.; Wang, Y.; Mu, P.; Li, J. A review on the recent advances in composite membranes for CO2 capture processes. Sep. Purif. Technol. 2023, 307, 122752.
- Luo, W.; Li, F.; Li, H.; Zhang, Z.; Zhang, X.; Liang, Y.; Huang, G. From 0D to 3D nanomaterial-based composite membranes for CO2 capture: Recent advances and perspectives. J. Environ. Chem. Eng. 2023, 11, 110657.
- Dai, Y.; Niu, Z.; Wang, Y.; Zhong, S.; Mu, P.; Li, J. Recent advances and prospect of emerging microporous membranes for high-performance CO2 capture. Sep. Purif. Technol. 2023, 318, 123992.
- Alli, Y.A.; Oladoye, P.O.; Ejeromedoghene, O.; Bankole, O.M.; Alimi, O.A.; Omotola, E.O.; Olanrewaju, C.A.; Philippot, K.; Adeleye, A.S.; Ogunlaja, A.S. Nanomaterials as catalysts for CO2 transformation into value-added products: A review. Sci. Total Environ. 2023, 868, 161547.
- Baena-Moreno, F.M.; Leventaki, E.; Riddell, A.; Wojtasz-Mucha, J.; Bernin, D. Effluents and residues from industrial sites for carbon dioxide capture: A review. Environ. Chem. Lett. 2023, 21, 319–337.
- Hanifa, M.; Agarwal, R.; Sharma, U.; Thapliyal, P.C.; Singh, L.P. A review on CO2 capture and sequestration in the construction industry: Emerging approaches and commercialised technologies. J. CO2 Util. 2023, 67, 102292.
- Li, H. CO2 capture by various nanoparticles: Recent development and prospective. J. Clean. Prod. 2023, 414, 137679.
- Segneri, V.; Trinca, A.; Libardi, N.; Colelli, L.; Micciancio, M.; Vilardi, G. Nanoparticles used for CO2 capture by adsorption: A review. Chem. Eng. Trans. 2023, 101, 133–138.
- Youns, Y.T.; Manshad, A.K.; Ali, J.A. Sustainable aspects behind the application of nanotechnology in CO2 sequestration. Fuel 2023, 349, 128680.
- Azni Farhana Mazri, N.; Arifutzzaman, A.; Kheireddine Aroua, M.; Ekhlasur Rahman, M.; Ali Mazari, S. Graphene and its tailoring as emerging 2D nanomaterials in efficient CO2 absorption: A state-of-the-art interpretative review. Alex. Eng. J. 2023, 77, 479–502.
- Chen, H.; Zheng, Y.; Li, J.; Li, L.; Wang, X. AI for nanomaterials development in clean energy and carbon capture, utilization and storage (CCUS). ACS Nano 2023, 17, 9763–9792.
- Chen, M.; Yang, T.; Han, J.; Zhang, Y.; Zhao, L.; Zhao, J.; Li, R.; Huang, Y.; Gu, Z.; Wu, J. The application of mineral kaolinite for environment decontamination: A review. Catalysts 2023, 13, 123.
- Cui, Y.; Zhu, J.; Tong, H.; Zou, R. Advanced perspectives on MXene composite nanomaterials: Types synthetic methods, thermal energy utilization and 3D-printed techniques. iScience 2023, 26, 105824.
- Younis, M.; Ahmad, S.; Atiq, A.; Farooq, M.A.; Huang, M.; Abbas, M. Recent progress in azobenzene-based supramolecular materials and applications. Chem. Rec. 2023, 23, e202300126.
- Zhu, W.; Yue, Y.; Wang, H.; Zhang, B.; Hou, R.; Xiao, J.; Huang, X.; Ishag, A.; Sun, Y. Recent advances on energy and environmental application of graphitic carbón nitride (g-C3N4)-based photocatalysts: A review. J. Environ. Chem. Eng. 2023, 12, 110164.
- de Morais, M.G.; Vargas, B.P.; da Silva Vaz, B.; Cardias, B.B.; Costa, J.A.V. Advances in the synthesis and applications of nanomaterials to increase CO2 biofixation in microalgal cultivation. Clean Technol. Environ. Policy 2023, 25, 617–632.
- Waseem, M.; Al-Marzouqi, M.; Ghasem, N. A review of catalytically enhanced CO2-rich amine solutions regeneration. J. Environ. Chem. Eng. 2023, 11, 110188.
- Chowdhury, S.; Kumar, Y.; Shrivastava, S.; Patel, S.K.; Sangwai, J.S. A review on the recent scientific and commercial progress on the direct air capture technology to manage atmospheric CO2 concentrations and future erspectives. Energy Fuels 2023, 37, 10733–10757.
- Jaiswar, G.; Dabas, N.; Chaudhary, S.; Jain, V.P. Progress in absorption of environmental carbon dioxide using nanoparticles and membrane technology. Int. J. Environ. Sci. Technol. 2023, 20, 10385–10404.
- Peu, S.D.; Das, A.; Hossain, M.S.; Akanda, M.A.; Akanda, M.M.; Rahman, M.; Miah, M.N.; Das, B.K.; Islam, A.R.; Salah, M.M. A comprehensive review on recent advancements in absorption-based post combustion carbon capture technologies to obtain a sustainable energy sector with clean environment. Sustainability 2023, 15, 5827.
- Zhang, C.; Zhang, X.; Su, T.; Zhang, Y.; Wang, L.; Zhu, X. Modification schemes of efficient sorbents for trace CO2 capture. Renew. Sustain. Energy Rev. 2023, 18, 113473.
- Boldoo, T.; Ham, J.; Cho, H. Evaluation of CO2 absorption characteristics of low cost Al2O3/MeOH nanoabsorbent using porous nickel foam for high efficiency CO2 absorption system. J. Clean. Prod. 2023, 384, 135624.
- Ge, B.-B.; Yan, J.; Zhong, D.-L.; Lu, Y.-Y.; Li, X.-Y. CO2 capture enhancement by forming tetra-n-butyl ammonium bromide semiclathrate in graphite nanofluids. Can. J. Chem. Eng. 2023, 101, 4128–4137.
- Geng, Z.; Yang, Y.; Wang, Y.; Zhu, T.; Xu, W. Catalytic regeneration of amine-based absorbents for CO2 capture: The effect of acidic sites and accessibility. Sep. Purif. Technol. 2023, 327, 124889.
- Itas, Y.S.; Razali, R.; Tata, S.; Kolo, M.; Lawal, A.; Alrub, S.A.; El Ghoul, J.; Khandaker, M.U. DFT studies on the effects of C vacancy on the CO2 capture mechanism of silicon carbide anotubes photocatalyst (Si12C12-X; X = 1; 2). Silicon 2023, 1–11.
- Jin, G.; Wang, H.; Zhang, K.; Zhang, H.; Fan, J.; Wang, J.; Guo, D.; Wang, Z. ZIF-8 based porous liquids with high hydrothermal stability for carbon capture. Mater. Today Commun. 2023, 36, 106820.
- Li, Y.; Lu, H.; Liu, Y.; Wu, K.; Zhu, Y.; Liang, B. CO2 absorption and desorption enhancement by nano-SiO2 in DBU-glycerol solution with high viscosity. Sep. Purif. Technol. 2023, 309, 122983.
- Mahdavi, H.; Sadiq, M.M.; Smith, S.J.D.; Mulet, X.; Hill, M.R. Underlying potential evaluation of the real-process applications of magnetic porous liquids. J. Mater. Chem. A 2023, 11, 16846–16853.
- Tengku Hassan, T.N.A.; Mohd Shariff, A.; Abd Aziz, N.F.; Mustafa, N.F.A.; Tan, L.S.; Abdul Halim, H.N.; Mohamed, M.; Hermansyah, H. Aqueous potassium salt of L-cysteine as potential CO2 removal solvent: An investigation on physicochemical properties and CO2 loading capacity. Sustainability 2023, 15, 11558.
- Wang, S.-L.; Xiao, Y.-Y.; Zhou, S.-D.; Jiang, K.; Yu, Y.-S.; Rao, Y.-C. Synergistic effect of water-soluble hydroxylated multi-wall carbon nanotubes and graphene nanoribbons coupled with tetra butyl ammonium bromide on kinetics of carbon dioxide hydrate formation. Energies 2023, 16, 5831.
- Wang, X.; Bao, Z.; Akhmedov, N.G.; Hopkinson, D.; Hoffman, J.; Duan, Y.; Egbebi, A.; Resnik, K.; Li, B. Unique biological amino acids turn CO2 emission into novel nanomaterials with three switchable product pathways. Environ. Technol. Innov. 2023, 32, 103279.
- Zabelina, A.; Dedek, J.; Guselnikova, O.; Zabelin, D.; Trelin, A.; Miliutina, E.; Kolska, Z.; Siegel, J.; Svorcik, V.; Vana, J.; et al. Photoinduced CO2 conversion under Arctic conditions─the high potential of plasmon chemistry under low temperature. ACS Catal. 2023, 13, 3830–3840.
- Zare, A.; Darvishi, P.; Lashanizadegan, A.; Zerafat, M. Theoretical and experimental investigation of CO2 solubility in nanofluids containing NaP zeolite nanocrystals and ionic liquid. Can. J. Chem. Eng. 2023, 101, 3925–3936.
- Zarei, F.; Keshavarz, P. Intensification of CO2 absorption and desorption by metal/non-metal oxide nanoparticles in bubble columns. Environ. Sci. Pollut. Res. 2023, 30, 19278–19291.
- Zhao, X.; Ding, Y.; Ma, L.; Zhu, X.; Wang, H.; Cheng, M.; Liao, Q. An amine-functionalized strategy to enhance the CO2 absorption of type III porous liquids. Energy 2023, 279, 127975.
- Khanmohammadian, E.; Mohammadi, M.; Hashemi, R.; Eslami, S.; Reza Ehsani, M. Improvement of gas hydrate-based CO2 capture from CH4/CO2 mixture using silica and modified silica nanoparticles in the presence of potassium hydroxide. Fuel 2023, 334, 126458.
- Lu, T.; Li, Z.; Du, L. Enhanced CO2 geological sequestration using silica aerogel nanofluid: Experimental and molecular dynamics insights. Chem. J. 2023, 474, 145566.
- Ali, N.; Babar, A.A.; Wang, X.; Yu, J.; Ding, B. Hollow, porous, and flexible Co3O4-doped carbon nanofibers for efficient CO2 capture. Adv. Eng. Mater. 2023, 25, 2201335.
- Anagnostopoulou, M.; Zindrou, A.; Cottineau, T.; Kafizas, A.; Marchal, C.; Deligiannakis, Y.; Keller, V.; Christoforidis, K.C. MOF-derived defective Co3O4 nanosheets in carbon nitride nanocomposites for CO2 photoreduction and H2 production. ACS Appl. Mater. Interfaces 2023, 15, 6817–6830.
- Arifutzzaman, A.; Musa, I.N.; Aroua, M.K.; Saidur, R. MXene based activated carbon novel nano-sandwich for efficient CO2 adsorption in fixed-bed column. J. CO2 Util. 2023, 68, 102353.
- Butt, F.S.; Lewis, A.; Rea, R.; Mazlan, N.A.; Chen, T.; Radacsi, N.; Mangano, E.; Fan, X.; Yang, Y.; Yang, S.; et al. Highly-controlled soft-templating synthesis of hollow ZIF-8 nanospheres for selective CO2 separation and storage. ACS Appl. Mater. Interfaces 2023, 15, 31740–317545.
- Chen, P.-B.; Yang, J.-W.; Rao, Z.-X.; Wang, Q.; Tang, H.-T.; Pan, Y.-M.; Liang, Y. Efficient in-situ conversion of low-concentration carbon dioxide in exhaust gas using silver nanoparticles in N-heterocyclic carbene polymer. J. Colloid Interface Sci. 2023, 652, 866–877.
- Chi, S.; Ye, Y.; Zhao, X.; Liu, J.; Jin, J.; Du, L.; Mi, J. Porous molecular sieve polymer composite with high CO2 adsorption efficiency and hydrophobicity. Sep. Purif. Technol. 2023, 307, 122738.
- Giraldo, L.J.; Medina, O.E.; Ortiz-Perez, V.; Franco, C.A.; Cortes, F.B. Enhanced carbon storage process from flue gas streams using rice husk silica nanoparticles: An approach in shallow coal bed methane reservoirs. Energy Fuels 2023, 37, 2945–2959.
- Gu, Y.-M.; Wang, Y.-H.; Zhao, S.-S.; Fan, H.-J.; Liu, X.-W.; Lai, Z.; Wang, S.-D. N-donating and water-resistant Zn-carboxylate frameworks for humid carbon dioxide capture from flue gas. Fuel 2023, 336, 126793.
- He, Y.; Wang, Z.; Cao, A.; Xu, X.; Li, J.; Zhang, B.; Kang, L. Construction of graphene oxide-coated zinc tetraphenyporphyrin nanostructures for photocatalytic CO2 reduction to highly selective CH4 product. J. Colloid Interface Sci. 2023, 638, 123–134.
- Hosseini, S.R.; Omidkhah, M.; Mehri Lihgyan, Z.; Norouzbahari, S.; Ghadimi, A. Synthesis, characterization, and gas adsorption performance of an efficient hierarchical ZIF-11@ZIF-8 core–shell metal–organic framework (MOF). Sep. Purif. Technol. 2023, 307, 122679.
- Iranvandi, M.; Tahmasebpoor, M.; Azimi, B.; Heidari, M.; Pevida, C. The novel SiO2-decorated highly robust waste-derived activated carbon with homogeneous fluidity for the CO2 capture process. Sep. Purif. Technol. 2023, 306, 122625.
- Issa, G.; Kormunda, M.; Tumurbaatar, O.; Szegedi, A.; Kovacheva, D.; Karashanova, D.; Popova, M. Impact of Ce/Zr ratio in the nanostructured ceria and zirconia composites on the selective CO2 adsorption. Nanomaterials 2023, 13, 2428.
- Jin, Z.; Zhang, J.; Qiu, J.; Hu, Y.; Di, T.; Wang, T. Nitrogen vacancy-induced spin polarization of ultrathin zinc porphyrin nanosheets for efficient photocatalytic CO2 reduction. J. Colloid Interface Sci. 2023, 652, 122–131.
- Liu, Z.; Lu, Y.; Wang, C.; Zhang, Y.; Jin, X.; Wu, J.; Wang, Y.; Zeng, J.; Yan, Z.; Sun, H.; et al. MOF-derived nano CaO for highly efficient CO2 fast adsorption. Fuel 2023, 340, 127476.
- Liu, M.; Ma, C.; Cheng, X.; Gao, K.; Zhang, G.; Wang, D.; Liu, F. New insight into multiple hydrogen-bond networks of functional organosilicas system for collaborative transformation of CO2 under mild conditions. Sep. Purif. Technol. 2023, 317, 123937.
- Lopez-Arias, M.; Moro, C.; Francioso, V.; Elgaali, H.H.; Velay-Lizancos, M. Effect of nanomodification of cement pastes on the CO2 uptake rate. Constr. Build. Mater. 2023, 404, 133165.
- Mahdipoor, H.R.; Ebrahimi, R.; Ganji Babakhani, E.; Halladj, R.; Safari, N.; Ganji, H. Investigating the selective adsorption of CO2 by MIL-101(Cr)-NH2 and modeling the equilibrium data using a new three-parameter isotherm. Colloids Surf. A Physicochem. Eng. Asp. 2023, 675, 131971.
This entry is offline, you can click here to edit this entry!
